Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Primary neurorrhaphy remains the gold standard in nerve repair; all other methods of repair are judged in comparison to primary neurorrhaphy.
Excessive tension will inhibit nerve regeneration, however a small amount of tension to achieve primary coaptation is acceptable.
In the event of a nerve gap, several options for repair exist including:
Mobilization and primary coaptation
Interpositional nerve autografting
Interpositional nerve allograft or conduit
Nerve transfer
Prognosis following nerve injury is impacted by several factors:
Patient age and comorbidities
Location of injury (proximal versus distal peripheral nerve)
Mechanism of injury (crush versus avulsion versus transection)
Timing of repair
Method of repair (tension, alignment, scarring)
Following any technique of repair, nerve regeneration occurs at a maximum of 1 mm/day, making distance from site of injury to the end-target muscle the most important prognostic factor for recovery.
Access video lecture content for this chapter online at Elsevier eBooks+ ![]()
Nerve injuries present a unique problem in patient management due to the prolonged periods of recovery and the necessity of having viable axons reach the end targets prior to irreversible atrophy and fibrosis ( ![]() ). These patients may suffer significant negative impacts, including incomplete functional recovery, chronic pain, depression, and loss of productivity. While primary nerve repair and grafting have not changed significantly in the last several decades, there have been several paradigm shifts as our understanding of the internal neural topography has improved and new techniques and products have become available. The gold standard in nerve repair remains primary neurorrhaphy. In the setting of a nerve gap, other alternatives are necessary to avoid excessive tension, which causes permanent scarring at the repair site and impairs recovery. Autologous nerve grafting has been the procedure of choice for addressing a nerve gap, however donor sites are limited and inevitably leave deficits. While nerve autografts are considered the “gold standard” for reconstruction of a nerve gap, it should actually be considered a “bronze standard” as functional recovery is never normal. Alternative techniques for nerve gap reconstruction include acellular nerve allografts, conduits, and nerve transfers. In terms of prognosis, time is muscle , with irreversible muscle atrophy occurring as early as one year after denervation. Furthermore, time is pathway , with advancing time leading to a worsening quality of cellular support for nerve regeneration along the pathway. This chapter reviews the various methods of addressing a nerve injury, with emphasis on patient selection, timing, surgical techniques, and prognosis.
). These patients may suffer significant negative impacts, including incomplete functional recovery, chronic pain, depression, and loss of productivity. While primary nerve repair and grafting have not changed significantly in the last several decades, there have been several paradigm shifts as our understanding of the internal neural topography has improved and new techniques and products have become available. The gold standard in nerve repair remains primary neurorrhaphy. In the setting of a nerve gap, other alternatives are necessary to avoid excessive tension, which causes permanent scarring at the repair site and impairs recovery. Autologous nerve grafting has been the procedure of choice for addressing a nerve gap, however donor sites are limited and inevitably leave deficits. While nerve autografts are considered the “gold standard” for reconstruction of a nerve gap, it should actually be considered a “bronze standard” as functional recovery is never normal. Alternative techniques for nerve gap reconstruction include acellular nerve allografts, conduits, and nerve transfers. In terms of prognosis, time is muscle , with irreversible muscle atrophy occurring as early as one year after denervation. Furthermore, time is pathway , with advancing time leading to a worsening quality of cellular support for nerve regeneration along the pathway. This chapter reviews the various methods of addressing a nerve injury, with emphasis on patient selection, timing, surgical techniques, and prognosis.
A peripheral nerve injury can be classified in several ways ( Table 21.1 ). Historically, the first classification system by Sir Herbert Seddon (1943) was based on gross and histologic anatomical changes rather than mechanism of injury. He described three types of nerve injuries:
Neurapraxia: involves a segment of demyelination and/or conduction block at a discrete area along the course of a nerve.
Axonotmesis: implies direct axonal damage.
Neurotmesis: implies transection of the peripheral nerve.
| Electromyography | |||||||
|---|---|---|---|---|---|---|---|
| Sunderland (Seddon) degree of injury | Injury | Recovery | Rate of recovery | Acute nerve injuries | Chronic nerve injuries | ||
| Fibs/PSW | MUPs | Fibs/PSW | MUPs | ||||
| 1st degree (Neurapraxia) | Focal demyelination with conduction block that resolves spontaneously | Favorable Complete |
Fast <12 weeks |
Absent | Normal | Absent | Normal |
| 2nd degree (Axonotmesis) | Axonal rupture without interruption of the basal lamina tubes | Favorable Complete |
Slow 1 mm/day |
Present | Collateral sprouting or nascent MUPs ( ≤ 3 months) |
Absent | Chronic and/or polyphasic (nascent) MUPs |
| 3rd degree (Axonotmesis) | Rupture of both axons and basal lamina tubes, some scar | Variable Incomplete |
Slow 1 mm/day |
Present | Collateral sprouting or nascent MUPs ( ≤ 4 months) |
Absent | Chronic and/or polyphasic (nascent) MUPs |
| 4th degree (Neurotmesis) | Neuroma-in-continuity with complete scar block | Unfavorable None |
None | Present | Absent | Absent | Absent |
| 5th degree (Neurotmesis) | Complete transection | Unfavorable None |
None | Present | Absent | Absent | Absent |
| 6th degree (Mackinnon) | Combination of 1st to 4th degree ± normal fascicles | Variable | Variable | Variable | |||
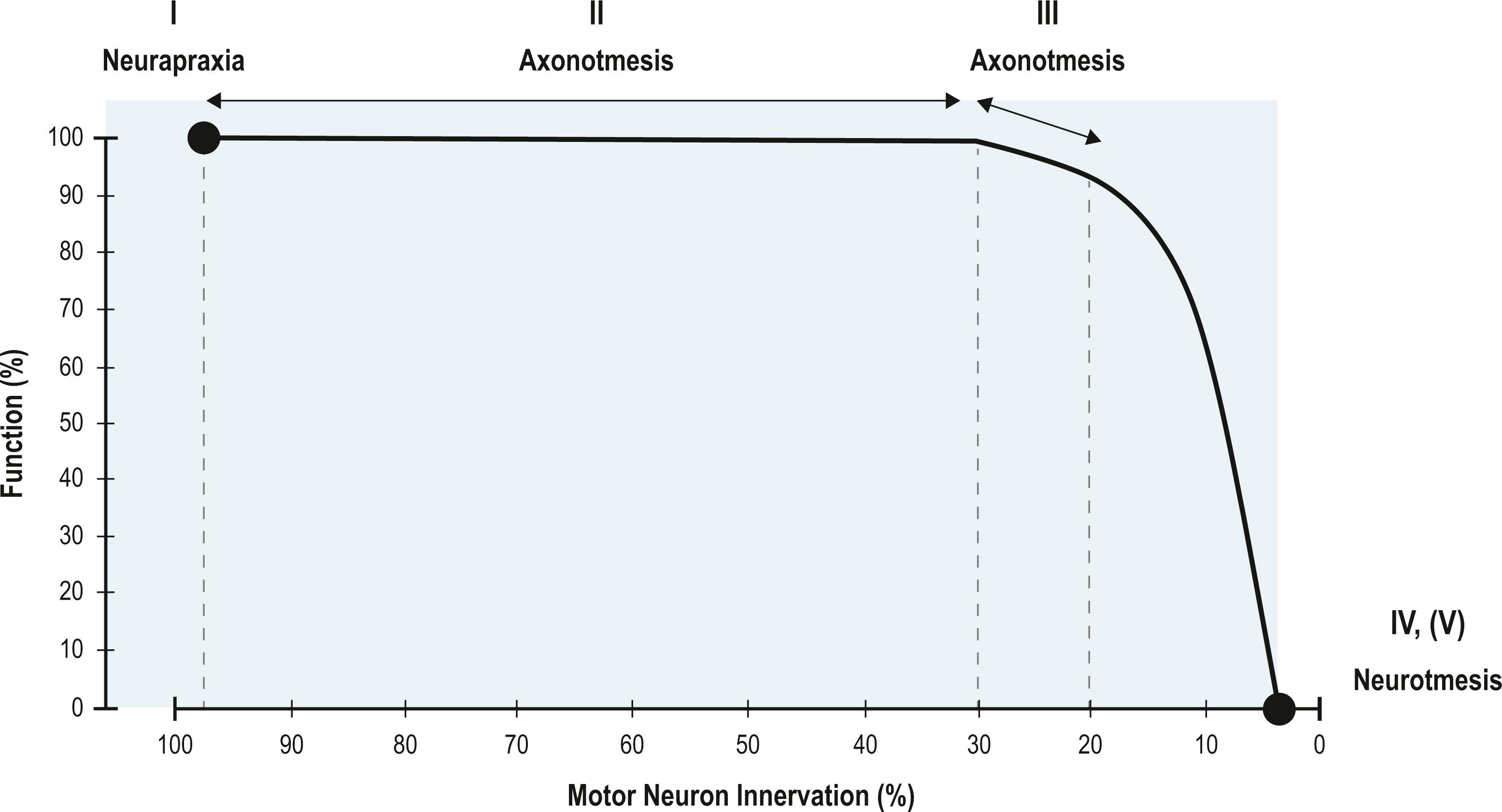
In neurapraxic injuries, Wallerian degeneration does not occur and the prognosis is excellent with anticipated recovery of full strength and function, generally within 3 months of injury. In both axonotmetic and neurotmetic injuries, Wallerian degeneration occurs distal to the site of injury. Recovery is possible within an axonotmetic injury, but Seddon described a degree of scarring and less complete recovery within this type of nerve injury. In neurotmesis, there will be no recovery. Sunderland expanded upon the earlier Seddon classification and emphasized five degrees of nerve injury. Mackinnon later went on to include a sixth degree (see Table 21.1 ). Establishing the degree of injury is critical to select the appropriate treatment and predict recovery. It is anticipated that a first-degree (neurapraxia) injury will make a full and speedy recovery. There is a subset of Sunderland first-degree injuries where an ongoing ischemic conduction block prevents any nerve signal transmission, but once the compression is removed, there is full nearly immediate recovery, which Mackinnon has recently termed a zero-degree injury. Second-degree (axonotmetic) injuries will make a full recovery with nascent axons regenerating across the injury at the classic rate of 1–1.5 mm/day and collateral sprouting occurring from remaining adjacent motor units. Fourth-degree (axonotmetic) injuries, also known as a neuroma-in-continuity, and fifth-degree (neurotmetic) injuries do not recover. Third-degree (axonotmetic) and sixth-degree (multi-level) injuries recover partially and at a slower rate for different reasons. The difficulty with surgical treatment of a sixth-degree injury is limiting the repair to fascicles affected by fourth- and fifth-degree damage, and not damaging adjacent fascicles with potential for spontaneous recovery. It is important for surgeons to recognize that patients may lose up to 70–80% of the motor unit pool before a functional deficit is noted due to the ability of motor units to collaterally sprout and innervate territories up to 5 times their original size ( Fig. 21.1 ).
As seen in Table 21.1 , nerve injuries can be classified based on their degree of damage and the components of the nerve that have been affected. While this method of classification is useful for prognosis and surgical decision-making, the mechanism of injury also provides a useful method of classification that allows for an algorithmic approach to treatment. Nerve injuries can therefore be classified as either open or closed, and then subdivided by mechanism of injury as penetrating, avulsion or crush injuries, or stretch and avulsion injuries.
Penetrating trauma can be a result of sharp or blunt penetration and will often have concomitant injuries to adjacent structures such as blood vessel, tendon, and muscle. A sharp laceration, such as from a knife or a piece of glass, will almost always necessitate exploration if a nerve deficit is present on initial clinical examination. The likelihood that the nerve is partially or completely transected is high. These injuries should be explored within 72 hours if distal stimulation using an intraoperative nerve stimulator is needed. In addition, the further from the time of injury, the more difficult mobilization of the proximal and distal ends to coapt primarily becomes. In the event of a penetrating trauma associated with a vascular injury, immediate exploration is warranted. Unfortunately, in significant proximal injuries necessitating arterial reconstruction with or without underlying fractures, a nerve injury can be overlooked due to the more urgent vascular and orthopedic injuries. In these situations, when a nerve deficit is noticed postoperatively, it is unclear if the nerve injury is from the inciting event, iatrogenic during the repair of the other injuries, or secondary to edema or hematoma. While magnetic resonance imaging or ultrasound may be helpful to evaluate for the latter, internal scarring of the nerve may not always be clearly determined.
Blunt penetrating and blast injuries, such as gunshot wounds or injuries due to an incendiary device, are often managed conservatively similar to closed crush and traction injuries. There is potential for spontaneous recovery. Local tissue edema often causes a neurapraxia, which should resolve within 12 weeks. If recovery is not evident on serial clinical examination and electrodiagnostic studies by four months, the algorithm for a crush injury should be followed ( Algorithm 21.1 ).
Crush injuries are common peripheral nerve injuries to the upper extremity. External crush may be complicated by increased internal pressure from hematomas, fractures, and local tissue edema. When minor, this may cause a temporary neurapraxia, but with greater compression, the likelihood of a permanent injury increases. The most severe consequence of a soft-tissue crush injury is the progression to compartment syndrome, which is a surgical emergency. Often an early sign of impending compartment syndrome is a decrease in vibration sensibility. Later findings include pain, loss of motor function, and loss of distal pulses.
While nerves are fairly resistant to injury given their elasticity, a mixed nerve injury may occur. A more extensive crush injury can cause local tissue damage that contributes more to the loss of function than the original nerve injury. Muscle tissue is the most susceptible to external forces. Even if the injury is not significant enough to cause a compartment syndrome, the local destruction of muscle may lead to muscle necrosis. Tendon and skin are more resistant and can withstand higher compressive forces before irreversible damage to the cells results.
The nerve portion of a crush injury is usually treated conservatively. Exploration is warranted if nerve recovery does not follow the anticipated pattern of recovery based on distance from injury to end-target muscle. Along with avulsion injuries, the treatment algorithm for a crush injury involves serial clinical examination and electrodiagnostic studies as dictated by the anticipated recovery ( Algorithm 21.2 ).
When the strain on the nerve exceeds a certain limit, the internal structure of the nerve becomes injured without any appreciable external evidence of injury. Nerves that are stretched to the point of avulsing from their proximal origin suggest a high-velocity, or high-impact, injury that is often associated with limb or life-threatening injuries that take precedence over the repair of the nerve. The nerves tend to be avulsed around areas of tethering, such as bony foramina or the spinal cord. If the proximal nerve can be accessed, grafting of the nerve may be possible. For areas such as the cranial nerves or the spinal roots, where the portion proximal to the avulsion is not accessible, reconstruction is typically done by either nerve or tendon transfers. The algorithm for treatment of these injuries involves focusing first on the nerve injury, with repair or reconstruction dictated by degree of injury, followed by procedures to augment recovery such as tendon transfers and joint arthrodesis (see Algorithm 21.2 ).
In situations where no distal nerve is accessible, such as avulsions from the neuromuscular junction, implantation of the proximal nerve directly into the muscle to neurotize the muscle is an alternative. Some studies show as good as MRC 4/5 motor recovery 1–2 years after direct nerve to muscle neurotization, however experimental studies do not support these findings; rather, recovery is much less than a nerve coaptation would produce.
Sharp penetrating injuries with an acute nerve deficit should be explored early (within 72 hours) if stimulation of the distal end is needed and also to increase the odds of a tension-free primary neurorrhaphy. Crush and traction injuries should be treated conservatively with serial examination and electrodiagnostic studies at regular intervals based on expected recovery.
A thorough history and physical examination remain the mainstay of diagnosis for a peripheral nerve deficit. A component of experience in recognizing common patterns of nerve injury can facilitate this.
Sensation can be tested using Semmes–Weinstein filaments, two-point static and moving discrimination, or the “ten test”. The quick and easy “ten test” uses the patient's own subjective perception to moving light touch to elicit differences in sensation, using the contralateral digit as a reference point. This technique is particularly useful in young children, who may have difficulty cooperating with two-point discrimination, or with rapid assessment of a patient being rushed to the operating room for an associated vascular injury. The presence of a Tinel’s sign indicates some degree of axonal injury (e.g., axonotmesis or neurotmesis), instead of a conduction block or demyelination (e.g., neurapraxia) and is useful to monitor progression of recovery.
Motor deficits are recognized by focused examination of all muscle groups in the upper extremity, with patterns of weakness or inactivity being used to diagnose the involved peripheral nerve and level of injury. The sensory collapse test (SCT), formerly known as the “scratch collapse test”, is an additional tool that allows evaluators to identify offending point(s) of compression in a multi-level injury.
In patients with an acute deficit, determination of whether the nerve will recover spontaneously, or whether surgical intervention is required, can often be difficult. The mechanism of injury can assist in preoperative evaluation. Any sharp, penetrating injury should be explored within 72 hours. Similarly, any injury where there is a high index of suspicion for nerve transection should be explored and repaired. A key clinical finding to differentiate between a neurotmetic injury and a less severe injury such as a neurapraxia or axonotmesis is the presence or absence of pain. Neurotmetic injuries typically present with significantly more neuropathic pain than injuries that remain in-continuity. The advantage to early repair, and specifically repair within 2 weeks, is that the proximal and distal nerve ends have not retracted and primary neurorrhaphy is often possible. Closed traction injuries, partial avulsion injuries, and crush injuries with an associated nerve palsy are more difficult to evaluate. Waiting 3–4 months with serial clinical and electrodiagnostic examination is advocated.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here