Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
autofluorescence imaging blue-light imaging colitis-associated neoplasia Crohn’s disease Crohn's Disease Endoscopic Index of Severity confidence interval colorectal cancer dye chromoendoscopy confocal laser endomicroscopy the Frankfurt Advanced Chromoendoscopic IBD LEsions flexible spectral imaging color enhancement high definition gastrointestinal inflammatory bowel disease image-enhanced endoscopy ileal pouch-anal anastomosis magnification endoscopy the Mayo Endoscopic Score narrow-band imaging the Narrow-band Imaging International Colorectal Endoscopic (classification) optical coherence tomography odds ratio the Paddington International Virtual Chromoendoscopy primary sclerosing cholangitis the Surveillance for Colorectal Endoscopic Neoplasia Detection and Management in Inflammatory Bowel Disease Patients standard definition ulcerative colitis white-light endoscopy AFI
BLI
CAN
CD
CDEIS
CI
CRC
DCE
CLE
FACILE
FICE
HD
GI
IBD
IEE
IPAA
ME
MES
NBI
NICE
OCT
OR
PICaSSO
PSC
SCENIC
SD
UC
WLE
Endoscopy plays a key role in the diagnosis, differential diagnosis, assessment of response to treatment, and surveillance of colitis-associated neoplasia (CAN). Endoscopy is the most reliable and commonly used modality for the measurement of disease activity. Mucosal healing (MH) has become a treatment goal. MH has been mainly defined with conventional white-light endoscopy (WLE). Therefore the terminology of MH and endoscopic healing has been used interchangeably. The availability of various image-enhanced endoscopy (IEE) technologies may further define MH, one step closer to histology level.
Colorectal cancer (CRC) is one of the leading causes of mortality worldwide with more than 1.8 million new cases diagnosed annually . In the United States, CRC is the second leading cause of cancer death among men and women . Patients with ulcerative colitis (UC) or Crohn’s disease (CD) of the colon are at twofold higher risk of the development of CRC than the general population . Cancer in patients with UC occurs at a younger age and the incidence increases with time, approaching 18% after 30 years of disease . Apart from disease duration, other risk factors for CAN include younger age at diagnosis, increased inflammatory activity, greater extent of chronic inflammation, coexisting primary sclerosing cholangitis (PSC), and a family history of a first-degree relative with CRC . Patients with left-sided or extensive UC and those with CD involving at least one-third of the colon or more than one segment of the colon are the target population for the surveillance program . Colonoscopy plays key role in surveillance of CAN . Guidelines recommend colonoscopy surveillance for CAN 8–10 years after disease diagnosis and continued surveillance every 1–3 years thereafter and the surveillance of UC patients at the time of diagnosis of PSC .
In the prior era of standard-definition (SD) or high-definition (HD) endoscopy, the practice pattern of endoscopic surveillance of dysplasia in patients with UC or CD of the colon used to be random biopsies in four quadrants every 10 cm of throughout the colon and targeted biopsies of any aberrant, raised or strictured areas with a total of at least 32–33 samples. The random biopsy technique is expensive, time-consuming, and invasive . Even with the extensive biopsies, less than 0.1% of the surface area of the colon is surveyed and the detection rate has been low (<2 per 1000 biopsies taken). The multiple random biopsies may still miss dysplastic lesions .
Over the past decade, endoscopic technology has evolved to improve the surveillance of dysplasia in long-standing IBD. HD-WLE provides images with a resolution of more than 1 million pixels, in comparison to SD-WLE with a resolution of 100,000–400,000 pixels. HD-WLE has been instrumental in identifying polyps (especially sessile polyps) that may have been missed by SD-WLE . However, even HD endoscopy is not able to characterize mucosal structure at microscopic level. Efforts, therefore, are being made to develop modalities to guide or even replace standard tissue biopsy. The terminology of optical biopsy has been used .
Corresponding to the evolution from SD and HD endoscopy to magnification endoscopy (ME), other imaging techniques have been developed. Various forms of IEE have been developed to characterize mucosal and submucosal features. IEE enhances surface, tone, and contrast. The modalities include ME, dye chromoendoscopy (DCE), dye-less narrow-band imaging (NBI), blue-light imaging (BLI), confocal laser endomicroscopy (CLE) , endocytoscopy, and optical coherence tomography (OCT). Applications of CLE, OCT, and ME in inflammatory bowel disease (IBD) are detailed in Chapter 20 , Confocal endomicroscopy and other image-enhanced endoscopy in inflammatory bowel disease. The enhanced quality of endoscopic imaging provides better tools to characterize disease activity and CAN. Currently main applications of IEE are the assessment of disease activity and response to treatment, and characterization of CAN.
A variety of instruments have been developed for the measurement of disease activity in CD and UC, for example, the Crohn's Disease Endoscopic Index of Severity (CDEIS) and the Mayo Endoscopic Score (MES). These disease instruments are detailed in Chapter 14 , Endoscopic scores in inflammatory bowel disease. These scoring systems were developed based on WLE. Due to the limitation of WLE, currently available disease activity instruments characterize edema, erythema, vascular pattern, bleeding, erosions, and ulcers. None of these instruments included the specific endoscopic features for the definition of MH.
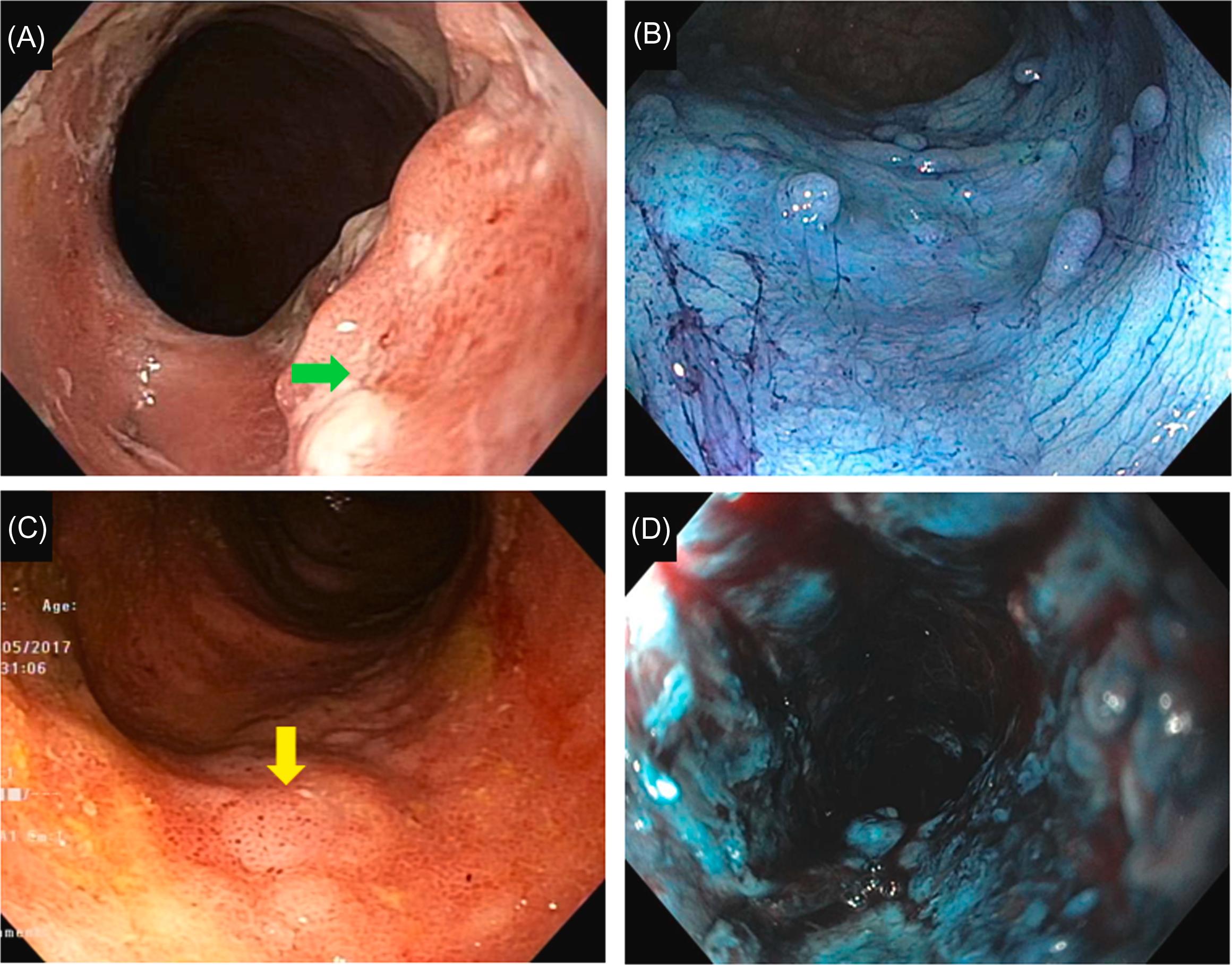
With IEE, attempts have been made to further characterize mucosal (such as scars and drop out of crypts) and superficial vascular structure in IBD, particularly in UC. For example, the Paddington International Virtual Chromoendoscopy (PICaSSO) was proposed to quantify the disease activity in UC . The PICaSSO Classification consists of two categories, mucosal architecture (including erosions, crypt abscess, and erosions) and vascular architecture (including vessel dilation and bleeding), accounting extent and distribution of the abnormalities ( Figs. 19.1 and 19.2 ; Table 19.1 ). The development of IEE-based disease activity scores may help further define MH, endoscopic remission or deep remission.
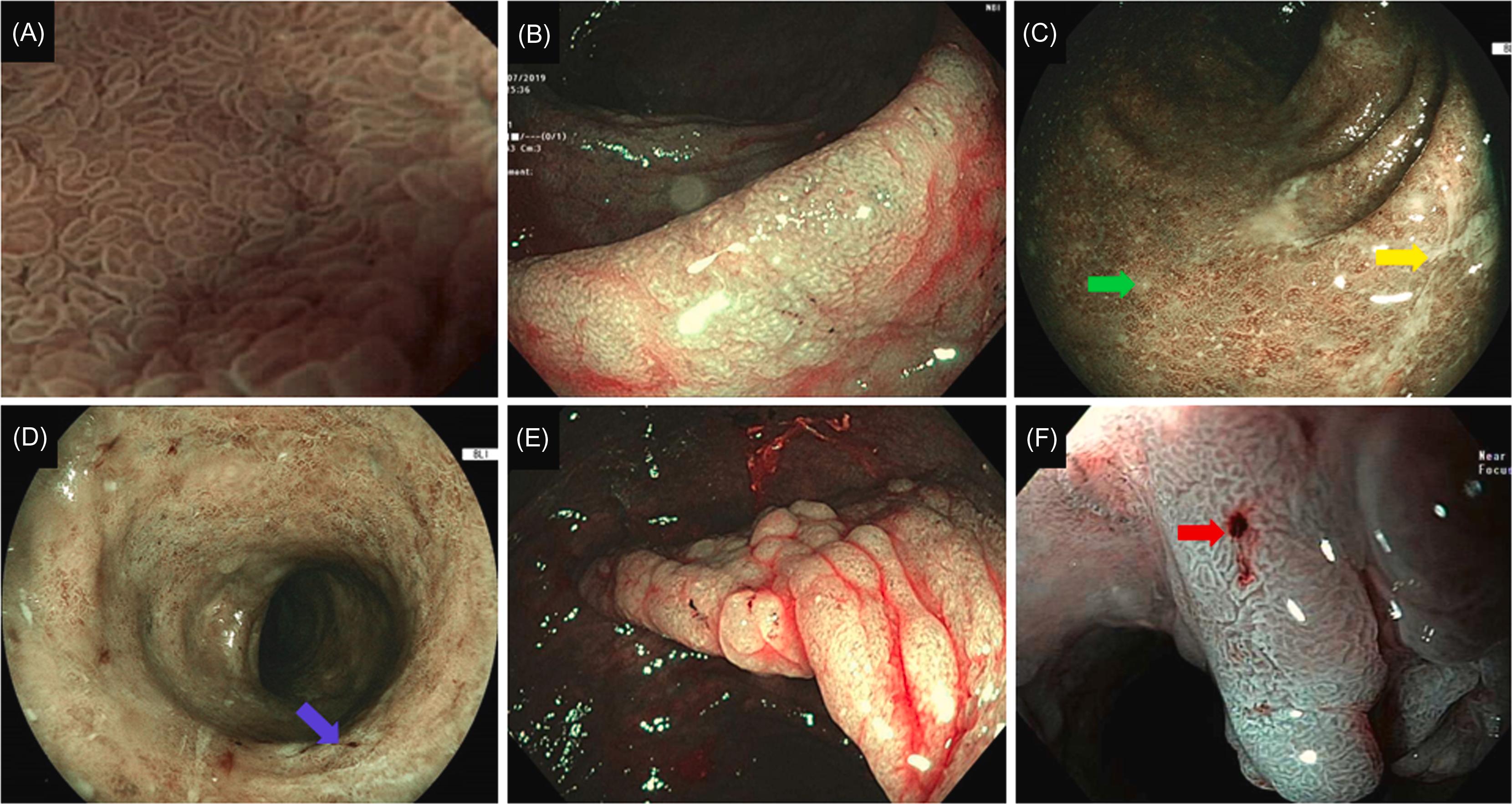
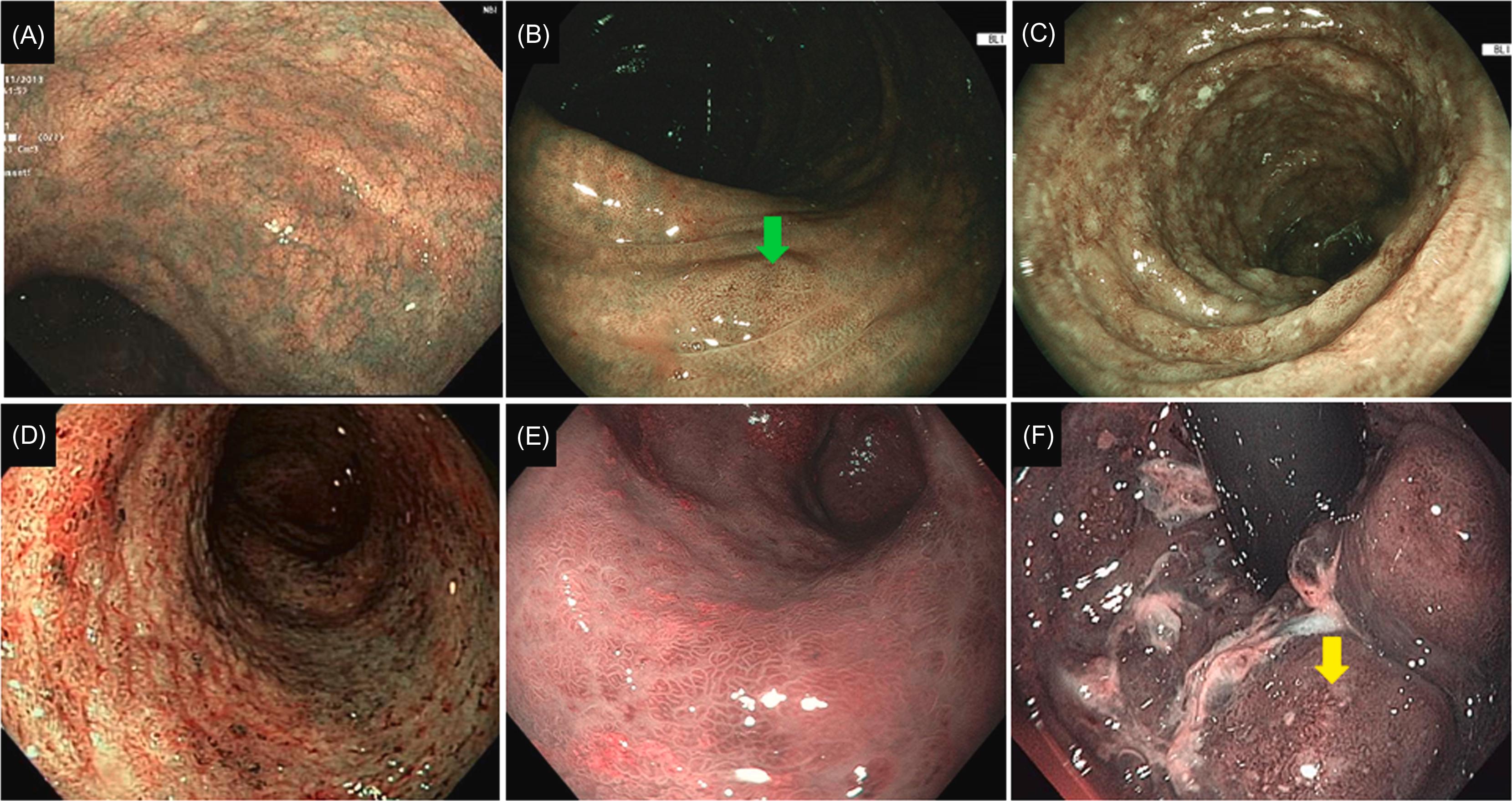
| Category | Subcategory | Description |
|---|---|---|
| PICaSSO mucosal-architecture | 0—No mucosal defect | A: Continuous/regular crypts |
| B: Crypts not visible (scar) | ||
| C: Discontinuous and or dilated/elongated crypts | ||
| I—Microerosion or cryptal abscess | 1: Discrete | |
| 2: Patchy | ||
| 3: Diffuse | ||
| II—Erosions size <5 mm | 1: Discrete | |
| 2: Patchy | ||
| 3: Diffuse | ||
| PICaSSO vascular-architecture | 0—Vessels without dilatation | A: Roundish following crypt architecture |
| B: Vessels not visible (scar) | ||
| C: Sparse (deep) vessels without dilatation | ||
| I—Vessels with dilatation | A: Roundish with dilatation | |
| B: Crowded or tortuous superficial vessels with dilatation | ||
| II—Intramucosal bleeding | A: Roundish with dilatation | |
| B: Crowded or tortuous superficial vessels with dilatation | ||
| III—Luminal bleeding | A: Roundish with dilatation | |
| B: Crowded or tortuous superficial vessels with dilatation |
The main application of IEE has been the surveillance of CAN in patients with UC. The Kudo Classification of the pit pattern of non-IBD colorectal neoplastic lesions was proposed based on ME in 1996. The classification consists of five categories: Type I round pits; Type II stellar or papillary pits; Type III-L large tubular or roundish pits; Type III-S small tubular or roundish pits; Type IV branch-like or gyrus-like pits; and Type V nonstructural pits Kudo pit pattern Types III–V is considered as being dysplastic and Kudo pit pattern Types I–II is considered as being nondysplastic or predictors of dysplasia in patients with long-standing IBD ( Figs. 19.3 and 19.4 ; Table 19.2 ) . The Kudo Classification was initially designed to depict non-IBD colonic neoplasia. The application of the Kudo Classification in IBD can be challenging, as regenerative hyperplastic villous mucosa with inflammation-associated elongated and irregular pits, making the distinction between inflammatory, hyperplastic, and neoplastic changes difficult.
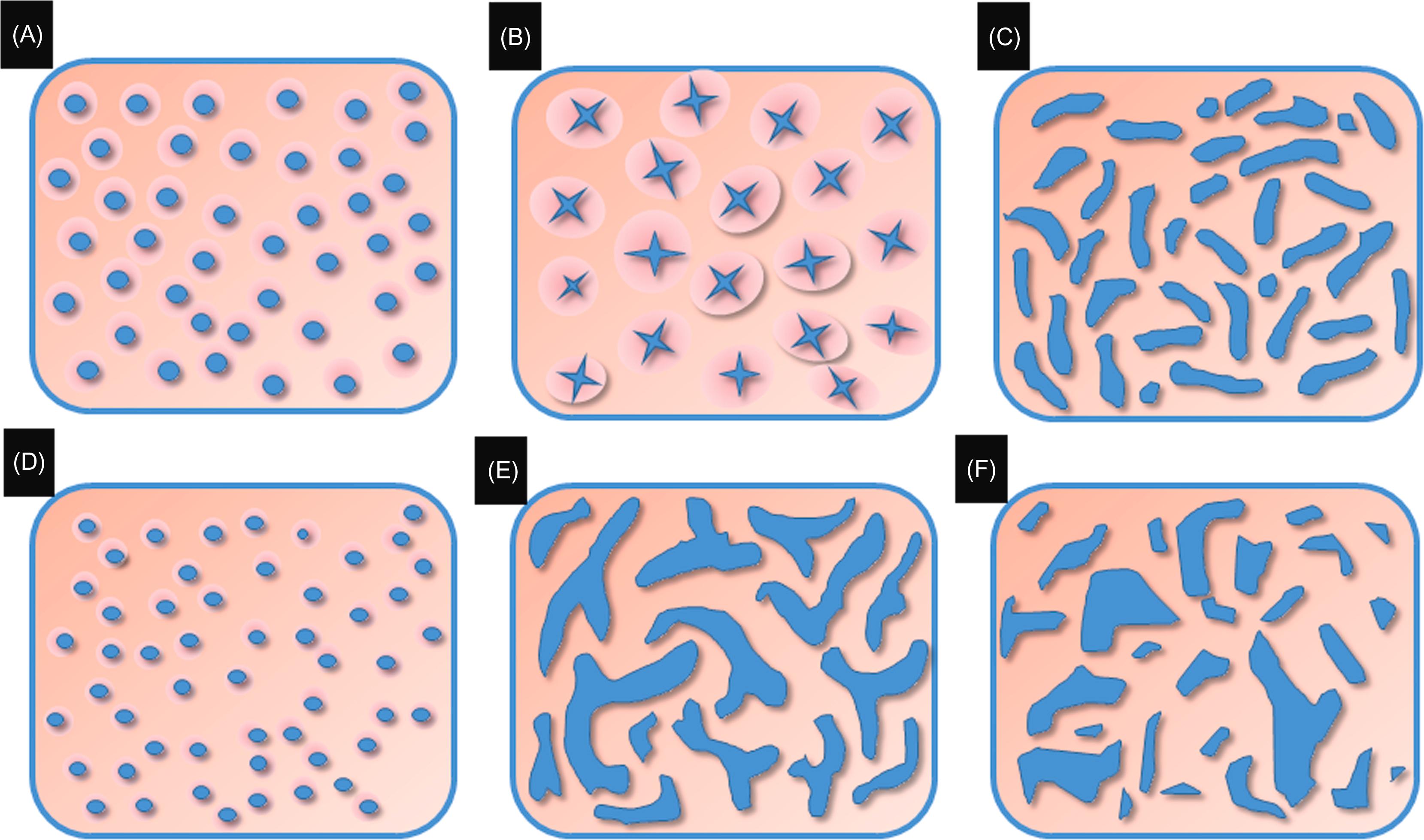
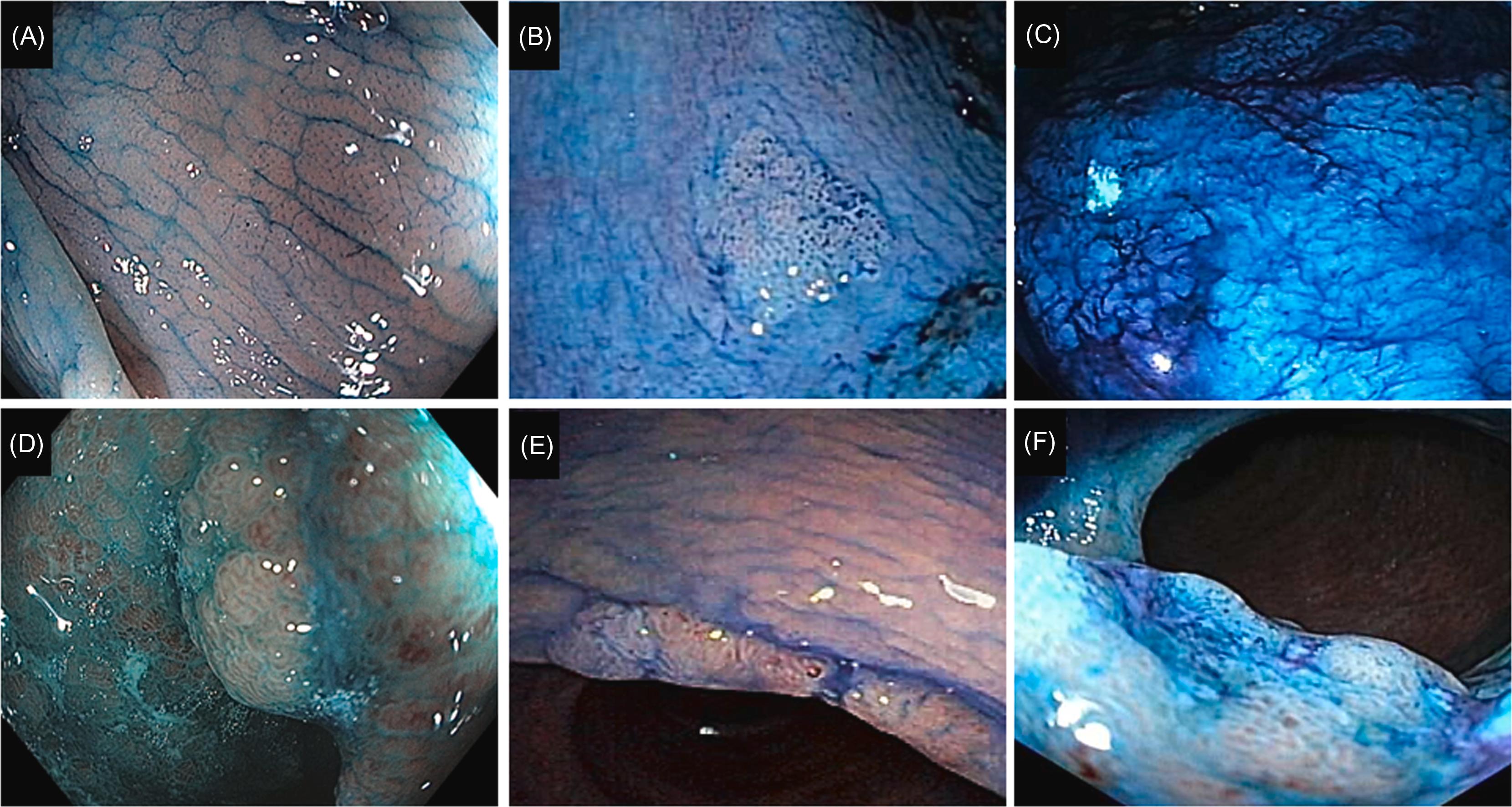
| Description | Explanation | |
|---|---|---|
| Type I | Round pits | Normal pit pattern |
| Type II | Stellar or papillary pits | |
| Type III-L | Large tubular or roundish pits | Larger than normal pit pattern |
| Type III-S | Small tubular or roundish pits | Smaller than normal pit pattern |
| Type IV | Branch-like or gyrus-like pits | |
| Type V | Amorphous or nonstructural pattern |
Other classification systems of colon polyps have been proposed, including the Narrow-band Imaging International Colorectal Endoscopic (NICE) Classification and the Showa Classification . The NICE Classification was proposed to differentiate nonneoplastic from neoplastic lesions, based on lesion color, microvascular architecture, and surface pattern . It classifies colorectal lesions into three categories: Type I as a hyperplastic lesion, Type II as an adenoma, and Type III as invasive cancer.
In addition to the pit pattern, morphology of mucosal structure and depth of CAN lesions may be categorized by the Paris Classification, which was designed for endoscopic classification of superficial neoplastic lesions in the colon as well as esophagus and stomach ( Fig. 19.5 ; Table 19.3 ) . The application of the Kudo Classification and Paris Classification are enhanced with the availability and application of various IEE. The Paris Classification was modified in a statement by the Surveillance for Colorectal Endoscopic Neoplasia Detection and Management in Inflammatory Bowel Disease Patients (SCENIC): International Consensus Recommendations ( Fig. 19.6 ; Table 19.4 ). The SCENIC consensus statement guides colonoscopic surveillance of CAN which was endorsed by the American Society for Gastrointestinal Endoscopy and American Gastroenterological Association. The endoscopic features of the modified Paris Classification listed in the SCENIC statement help clinicians to make decision on endoscopic resection versus surgical intervention.
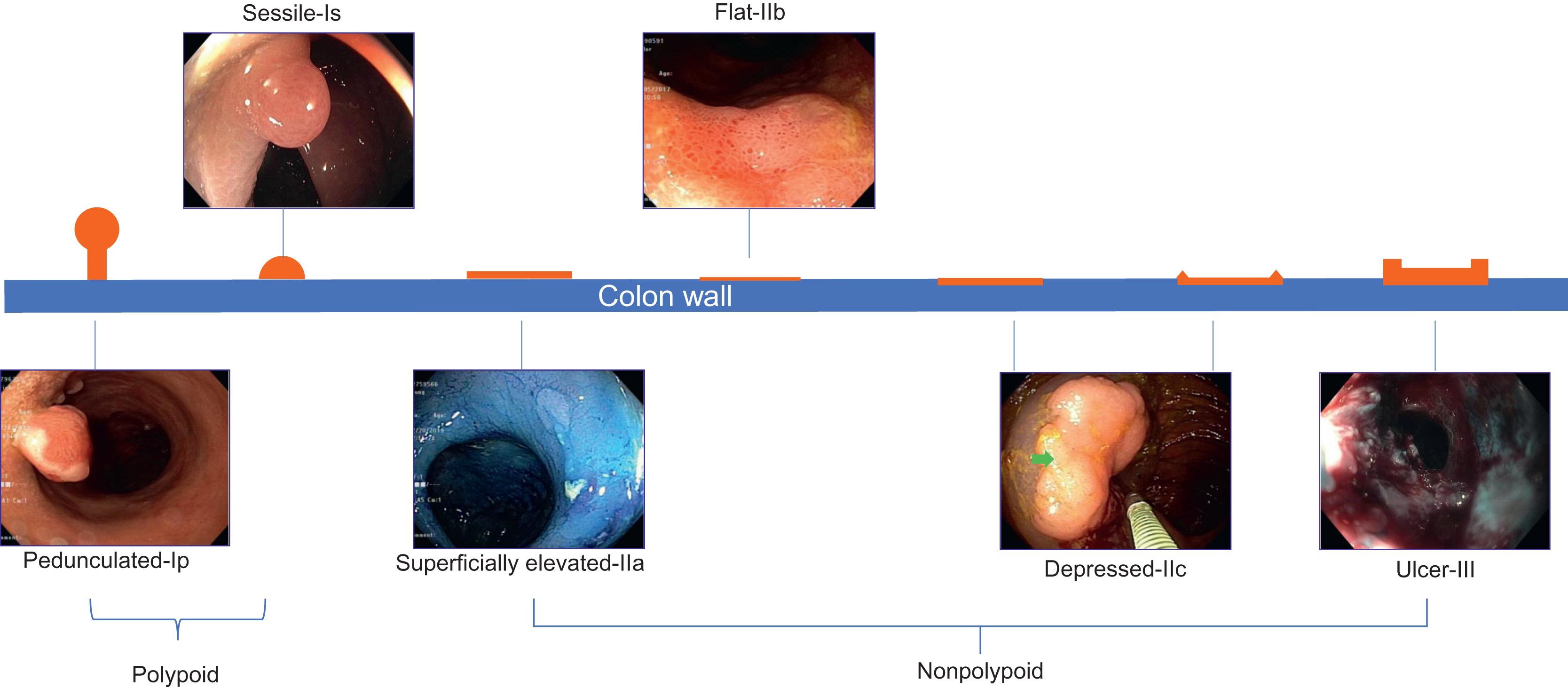
| Morphology | Category | Subcategory | Description | |
|---|---|---|---|---|
| Type 0 | Polypoid | 0-I | 0-Ip | Pedunculated polypoid |
| 0-Is | Sessile polypoid | |||
| Nonpolypoid and nonexcavated | 0-II | 0-IIa | Slightly elevated | |
| 0-IIb | Flat | |||
| 0-IIc | Slightly depressed without ulcer | |||
| Nonpolypoid with a frank ulcer | 0-III | Excavated (ulcer) | ||
| Term | Definition |
|---|---|
| Visible dysplasia | Dysplasia identified on targeted biopsies from a lesion visualized at colonoscopy |
| Polypoid | Lesion protruding from the mucosa into the lumen ≥2.5 mm |
| Pedunculated | Lesion attached to the mucosa by a stalk |
| Sessile | Lesion not attached to the mucosa by a stalk: entire base is contiguous with the mucosa |
| Nonpolypoid | Lesion with little (<2.5 mm) or no protrusion above the mucosa |
| Superficial elevated | Lesion with protrusion but <2.5 mm above the lumen (less than the height of the closed cup of a biopsy forceps) |
| Flat | Lesion without protrusion above the mucosa |
| Depressed | Lesion with at least a portion depressed below the level of the mucosa |
| General descriptors | |
| Ulcerated | Ulceration (fibrinous-appearing base with depth) within the lesion |
| Border | |
| Distinct border | Lesion’s border is discrete and can be distinguished from surrounding mucosa |
| Indistinct border | Lesion’s border is not discrete and cannot be distinguished from surrounding mucosa |
| Invisible dysplasia | Dysplasia identified on random (nontargeted) biopsies of colon mucosa without a visible lesion |
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here