Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Lung transplantation, whether single, bilateral, or, less often, heart–lung, has been an accepted mode of therapy for a variety of end-stage lung diseases for about 25 years. The most common indications for transplantation in adults are emphysema, cystic fibrosis (CF), idiopathic pulmonary fibrosis, sarcoidosis, and primary pulmonary hypertension (PPH), whereas for children most common indications are CF and PPH. Advances in donor management, surgical techniques, and immunosuppressive drugs increased the early survival to 90% in the first 3 months and 85% in the first year. Infections and acute rejection continue to be the most common causes of death in the first year posttransplantation. However, in contrast to other solid organ transplantation, over half of the lung transplant recipients continue to suffer and die of chronic lung allograft dysfunction (CLAD), generally occurring 3 to 10 years posttransplantation. Methods evaluating allograft dysfunction are variable and depend on the clinical differential diagnosis, posttransplant time, and complications suspected ( Table 24.1 ).
| Posttransplant Time of Onset | Complication | Method of Evaluation |
|---|---|---|
| Immediate (0–7 days) | Primary graft dysfunction | Clinical, TBB |
| Vascular anastomotic: bleeding, obstruction, thrombosis | Reexploration | |
| Acute antibody-mediated rejection (hyperacute rejection) | Clinical, DSA, TBB | |
| Early (8–30 days) | Infection of lung | BAL, culture, PCR TBB |
| Airway anastomotic infection, dehiscence | Culture, EBB | |
| Acute rejection | TBB | |
| Late (>30 days) | Acute rejection | TBB |
| Infection of lung | BAL, culture, PCR, TBB | |
| Airway stenosis | Bronchoscopy, culture | |
| Chronic rejection | PFT, TBB, wedge biopsy | |
| PTLD (mass lesion) | FNAB, TBB, wedge biopsy |
Postsurgical complications of the arterial or venous anastomoses, although rare, are associated with high morbidity and mortality rates. Several types of complications have been described, from kinking of anastomosis, thrombosis, stricture, and extramural mass pressure (with use of omental pedicle) to problems with vessel orientation (inversion of the donor vessel). Venous anastomoses are more prone to complications due to their extensive length. Management of a vascular complication depends on the underlying cause and includes medical, catheter-based, and surgical interventions.
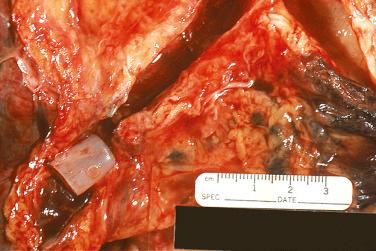
Multiple factors are involved in the development of airway anastomotic complications. These include (1) donor and recipient procedural problems involving ischemia and length of donor bronchus, size mismatch, type of immunosuppressive therapy, and postoperative tracheostomy; (2) type of surgical procedure performed, including end-to-end or telescoping technique; and (3) the presence of infection in preoperative and postoperative periods. Bronchial stenosis usually develops 2 to 9 months after transplantation and is the most common complication, occurring in 1.6% to 32% of transplanted patients. Necrosis at the anastomotic site is very common 1 to 5 weeks posttransplantation and is part of the normal healing process. In rare instances, extensive necrosis and ischemia lead to dehiscence of the anastomosis. Patients with dehiscence may clinically present with evidence of pneumothorax or pneumomediastinum, or may develop tracheobronchomalacia. Ischemia is also an important factor in the development of fistula (bronchopleural, bronchomediastinal, and bronchovascular). In 7% to 24% of patients, excessive granulation tissue is formed at the site of anastomosis causing airway obstruction. Management depends on the type of airway complication and on its severity ( Fig. 24.1 ).
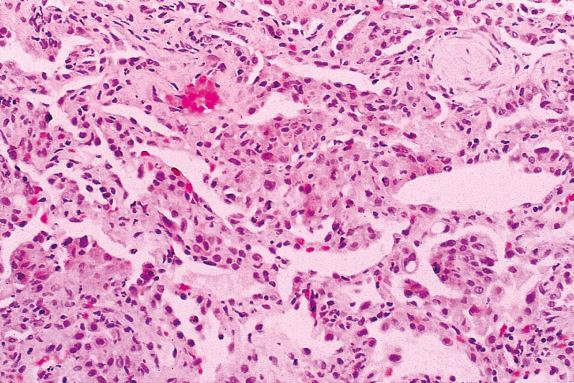
Primary graft dysfunction (PGD), which occurs in 10% to 25% of posttransplanted patients, is the result of a combination of ischemia, reperfusion, donor brain death, and cold organ preservation. PGD is the leading cause of morbidity and mortality in the early posttransplantation period and is a risk factor for development of bronchiolitis obliterans syndrome (BOS). Mechanisms leading to acute lung injury with diffuse alveolar damage (DAD) ( Fig. 24.2 ) include ischemia-induced endothelial and epithelial injury, abnormalities in coagulation and fibrinolysis, alterations in cell adhesion, and chemotaxis. The absence of perivascular and airway mononuclear cell infiltrates helps distinguish PGD from acute rejection.
Transbronchial biopsies (TBBs) from pulmonary allograft are evaluated for the presence or absence of vascular and airway rejection and are graded according to a grading system developed by the International Society for Heart and Lung Transplant (ISHLT) in 1990 and revised in 1996 and in 2007 ( Table 24.2 ). The biopsies are obtained either as a part of surveillance protocol or from symptomatic patients. Because rejection is a patchy process, it is recommended to obtain at least five fragments of alveolar lung tissue. Three different levels are stained with hematoxylin and eosin. Connective tissue stains and silver stain can be performed to assess submucosal fibrosis and fungal infection, respectively.
|
Grade 0—None Grade 1—Minimal Grade 2—Mild Grade 3—Moderate Grade 4—Severe |
|
Grade 0—None Grade 1R—Low-grade Grade 2R—High-grade Grade X—Ungradeable |
|
Grade 0—None Grade 1—Present |
|
Acute antibody-mediated rejection (AMR) presents a diagnostic challenge due to the lack of widely accepted diagnostic criteria. AMR is caused by binding of preformed or de novo donor-specific human leukocyte antigen (HLA) antibodies (DSA) to the antigens on the capillary endothelial cells. Interaction between antibody and antigen leads to allograft injury through activation of complement and complement cascade and leukocyte recruitment. Currently, the diagnosis of AMR is based on clinical evidence of allograft dysfunction, presence of circulating DSA, and pathologic findings of acute lung injury. The incidence of AMR is not known.
A small number of patients with preformed DSAs may develop hyperacute rejection during or immediately after the transplant leading to respiratory failure. For those patients that develop de novo DSA posttransplantation, the majority will develop clinical symptoms in the first year, presenting with breathlessness, hypoxia, and radiographic finding of diffuse pulmonary infiltrates.
The histologic findings in AMR are not well documented, but recent studies have shown that pathologic features of acute lung injury that include capillary injury (neutrophilic capillaritis/neutrophilic septal migration), epithelial injury with type 2 pneumocyte hyperplasia, endothelial injury with at least some intraalveolar fibrin, and neutrophils may be present in the septal capillaries. Biopsies from patients with AMR have an increased incidence of acute cellular rejection (ACR) (either high-grade acute cellular rejection—A3 or more—or persistent/recurrent acute cellular rejection of any grade), lymphocytic bronchiolitis (either high-grade lymphocytic bronchiolitis—grade B2R—or persistent low-grade lymphocytic bronchiolitis—B1R), obliterative bronchiolitis, and arteritis not associated with infection or cellular rejection. In those patients with preformed DSAs who develop hyperacute rejection, the pathologic features are most commonly DAD with evidence of significant vascular injury, including pulmonary infarction associated with vascular thrombosis, vascular fibrinoid necrosis, vasculitis, and neutrophilic margination.
Immunostain for C4d deposition can be demonstrated in various pulmonary structures; however, only staining of the vascular endothelium of the interstitial alveolar capillaries is considered significant in diagnosing AMR. C4d is considered positive when >50% of interstitial capillaries stain with it. Performing immunohistochemistry (IHC) for C4d should be considered in cases with the previously described histologic patterns, in cases with clinical diagnosis of graft dysfunction that cannot be explained by histologic findings, and in cases with detected DSA regardless of the histologic findings.
The diagnosis of AMR is a diagnosis of exclusion because histologic patterns of injury are nonspecific, and requires a multidisciplinary approach. Clinical evidence of allograft dysfunction, presence of circulating DSA, and pathologic changes are required to suggest a diagnosis of AMR. Positive C4d immunostain should always be interpreted together with clinical and pathologic findings. It is important to keep in mind that capillary C4d immunoreactivity can occasionally be seen in the absence of the circulating DSAs and in other conditions that may activate complement, such as infection. Unlike other organs, the presence of C4d in the donor lung tissue has been shown to be an insensitive marker for the presence of AMR in the lung and is most commonly negative.
Hyperacute rejection is usually fatal; however, most patients with AMR initially improve but are at a higher risk of developing CLAD. Treatment includes removal of the circulating antibodies by plasmapheresis, intravenous immunoglobulins, and B-cell–mediated therapy.
Acute rejection is a cell-mediated process with progressive infiltration of the graft by host mononuclear cells. Immune cell activation causes release of inflammatory chemokines and up-regulation of adhesion molecules. Major cellular targets include endothelial and epithelial cells.
Although acute rejection can develop as early as 3 days to many years posttransplant, patients experience rejection commonly within the first 3 months, with most episodes occurring between 2 and 9 months. Thirty-four percent of posttransplant patients develop at least one episode of rejection during the first year. Noncompliance with immunosuppressive medications is a significant cause of late episodes of acute rejection. Patients with acute cellular rejection (ACR) often present with a low-grade fever, cough, and dyspnea, and some are hypoxemic with greater than 10% decrease in pulmonary function tests. Aspiration and infection may precipitate episodes of acute rejection.
Chest imaging studies in the setting of ACR can show perihilar or lower zone alveolar or interstitial infiltrates, septal lines, subpleural edema, peribronchial cuffing, and pleural effusion. Computed tomography (CT) scans show ground-glass opacities, septal thickening, and pleural effusions. These findings are suggestive of acute rejection; however, sensitivity is only 35%.
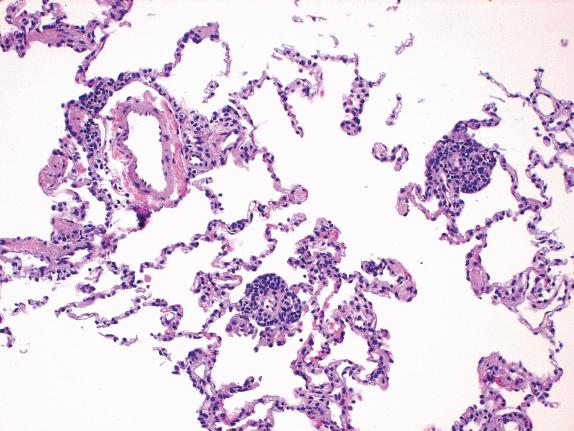
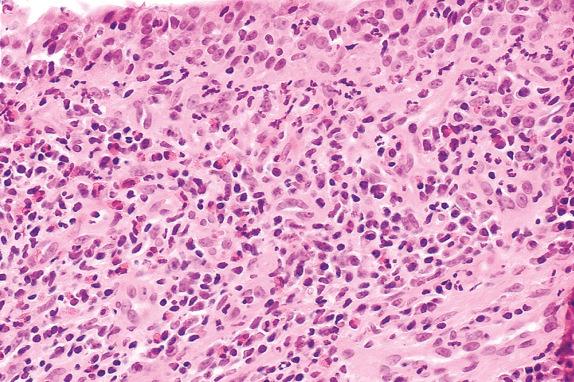
Acute rejection is characterized by mononuclear cell infiltrates. The intensity and distribution of the infiltrate form the base of the ISHLT grading system. In minimal acute rejection (grade A1) perivascular infiltrates are infrequent. Blood vessels, particularly venules, are cuffed by small, round, plasmacytoid and transformed lymphocytes forming a layer two to three cells thick ( Fig. 24.3 ). In mild acute rejection (grade A2) perivascular infiltrates are more frequent and consist of more than three layers of lymphocytes, with eosinophils around venules and arterioles ( Figs. 24.4 and 24.5 ). Grade A2 or higher-grade acute rejection is often accompanied by lymphocytic airway inflammation ( Fig. 24.6 ). Moderate acute rejection (A3) is characterized by an extension of the inflammation into alveolar septa ( Fig. 24.7 ) in contrast to grades A1 and A2, in which infiltrate is limited to the perivascular adventitia. In severe acute rejection (grade A4), diffuse perivascular, interstitial, and airspace infiltrates associated with pneumocyte damage, macrophages, hyaline membranes, hemorrhage, and neutrophils or epithelial ulceration with fibrinopurulent exudates are seen.


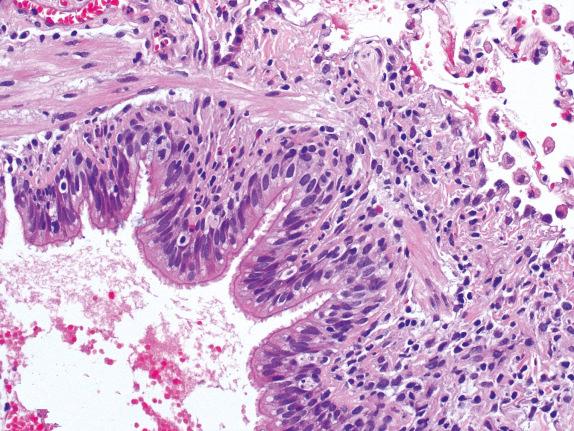
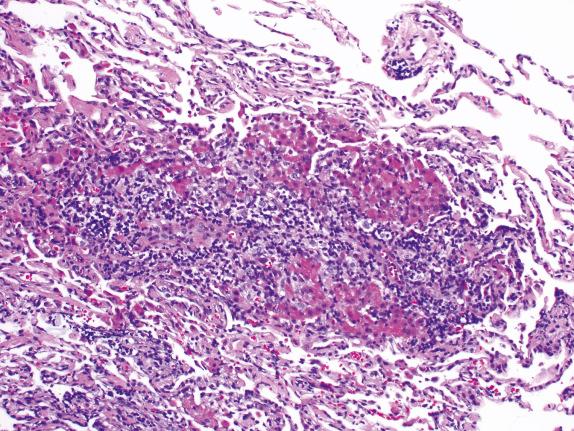
In the 2007 revision, airway rejection (lymphocytic bronchiolitis) is graded from low to high grade (B1R–B2R). In low-grade lymphocytic bronchiolitis, mononuclear cells, with occasional eosinophils, are present in submucosa of the bronchioli either as scattered cells or as a circumferential band, without infiltration or necrosis of the epithelium. High-grade lymphocytic bronchiolitis ( Fig. 24.8 ) is characterized by dense bronchiolar inflammatory cell infiltrates with epithelial cell dropout, lymphocytic infiltration of the epithelium, and sometimes ulceration. The inflammatory infiltrate is the same as seen in perivascular rejection with the lymphocyte as the predominant cell and smaller numbers of eosinophils and neutrophils.
Although the inflammatory cells, as well as mediators of inflammation, during acute rejection have been extensively studied in bronchoalveolar lavage (BAL) fluid, neither has been shown to accurately distinguish rejection from infection.
The main differential diagnosis for acute rejection is infection caused by bacteria, viruses, or fungi. The clinical and radiologic features are very similar to those of rejection. Microbiologic cultures and TBB are most useful. Special stains, including IHC for cytomegalovirus (CMV), Gomori methenamine silver (GMS) stain for fungus and Pneumocystis jirovecii, and Ziehl-Neelsen stain for acid-fast bacteria (AFB), should be performed as indicated.
Treatment of grade A1 minimal rejection is variable among institutions, with most opting not to treat if no symptoms are present. Mild (grade A2) and higher grades of acute rejection are treated irrespective of symptoms. Most acute rejection episodes are effectively treated by increasing the immunosuppression (usually three boluses of IV methylprednisone followed by an oral prednisone taper). Persistent or repeated episodes need more aggressive therapy, which carries additional risk of infection; however, this treatment is needed because these patients are at high risk of developing chronic rejection. Although the vascular rejection (grade A2 and higher) are routinely treated, there is no consensus in treatment of airway rejections. Most institutions do not treat airway rejection without concurrent vascular rejection; however, this is variable among lung transplant centers. Recent literature suggests that any airway inflammation/rejection increases the risk of developing CLAD.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here