Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The trachea is a tubular structure that extends from the cricoid cartilage superiorly to the carina inferiorly and is purely gas conducting. It is typically cylindrical in shape with slight flattening along its posterior aspect. The trachea measures approximately 11 cm in length and 2 to 2.5 cm in side-to-side diameter. Two specific segments can be identified, a shorter extrathoracic segment and a longer intrathoracic segment, based on the location of the trachea relative to the upper border of the manubrium. The trachea is composed of hyaline cartilage, fibrous tissue, muscular fibers, mucous membranes, and glands. The cartilages form incomplete C-shaped rings that occupy the anterior two thirds of the trachea. The posterior wall of the trachea is membranous and composed of fibrous tissue and nonstriated muscle fibers.
The trachea divides into two (left and right) primary or main bronchi, which branch into the lobar bronchi, which in turn divide into the segmental and intrapulmonary bronchi and bronchioles. Approximately 23 divisions exist from the trachea to the alveoli, and bronchioles are typically encountered after 6 to 20 divisions from the segmental bronchi. These airways are classified as either purely conducting or gas-exchanging regions separated by a transitional zone. The extrapulmonary bronchi are composed of hyaline cartilage, fibrous tissue, muscular fibers, mucous membranes, and glands. Compared with the cartilage of the trachea, the bronchial cartilages are shorter and narrower but maintain the same shape and arrangement. The bronchioles lack cartilage. The last generation of purely conducting airways is the terminal bronchiole. The respiratory bronchioles are the transitional branches that lead to gas-exchanging alveolar ducts, alveolar sacs, and alveoli. An acinus is lung parenchyma distal to a terminal bronchiole, and it is composed of two to five generations of respiratory bronchioles, alveolar ducts, alveolar sacs, and alveoli. Airways that are less than 2 mm in diameter are called small airways.
The secondary pulmonary lobule is the smallest portion of the lung that is surrounded by connective tissue septa, and it is described in detail in Chapter 17 .
A wide variety of imaging modalities are available to evaluate the airways in the clinical setting, including radiography, fluoroscopy, computed tomography (CT), magnetic resonance imaging (MRI), and 18-fluorodeoxyglucose ( 18 FDG) positron emission tomography (PET)/CT. The commonly accepted indications for each of these modalities are listed in Table 6.1 .
| Imaging Modality | Clinical Indication |
|---|---|
| Chest radiography (posteroanterior, lateral, oblique) | Screening examination |
| Fluoroscopy | Tracheomalacia Air trapping caused by airway obstruction |
| Multidetector computed tomography | Identification and characterization of airway neoplasms Airway diameter, wall thickness Tracheomalacia Airway compression Tracheobronchial injury |
| Magnetic resonance imaging | Airway obstruction caused by vascular rings |
| 18-Fluorodeoxyglucose ( 18 FDG) positron emission tomography/computed tomography image | Staging and restaging of airway neoplasms |
Posteroanterior and lateral chest radiography (CXR) is the most commonly performed radiologic procedure and may be the first imaging modality to evaluate patients presenting with respiratory symptoms. To reduce the visibility of the bony thorax and improve visualization of the various mediastinal interfaces, a high-kilovoltage (140 kVp) technique should be used. Bilateral, oblique radiographs improve visibility of the central airways by rotating the thoracic spine so that it is not superimposed on the trachea and main bronchi. The tracheal and proximal bronchial air columns should be identified and closely evaluated on all chest radiographs, especially when adult patients present with clinical symptoms such as stridor, wheezing, adult-onset “asthma,” recurrent pneumonia, or hemoptysis. However, abnormalities involving the airways may not be readily apparent on CXR, and further evaluation with cross-sectional imaging is typically necessary.
Computed tomography is the imaging modality of choice for evaluating the trachea and bronchi because it directly demonstrates the normal anatomy of the airways; adjacent structures such as the mediastinum, hila, and lungs; and a wide variety of abnormalities involving the tracheobronchial tree. CT is superior to CXR in demonstrating the relationship between the airways and mediastinal soft tissues, which becomes important in the setting of mediastinal masses abutting or invading the airways and primary airway neoplasms extending into the adjacent mediastinum. Additionally, CT effectively reveals the morphology of airway lesions, particularly neoplasms with fatty components, calcification, or vascular enhancement.
Modern CT scanners enable visualization and characterization of the trachea and main bronchi in a few seconds over the course of a single breath-hold. Thin section images (1–2.5 mm) can be used to generate high-resolution multiplanar reconstructions, which better delineate the craniocaudally oriented trachea and bronchi and many airway abnormalities such as neoplasms. Recent advances such as volumetric imaging, dynamic airway imaging, and other techniques such as two-dimensional (2D) minimum intensity projection (MIP) and three-dimensional (3D) volume imaging have enhanced the ability of radiologists to evaluate the airways. For instance, dynamic imaging performed during expiration and coughing is more sensitive in demonstrating specific abnormalities such as tracheomalacia than paired inspiratory and expiratory CT imaging. The two most commonly used 3D techniques include external volume rendering of the airways and internal rendering or virtual bronchoscopic images, which supplement but do not replace routine axial CT imaging and enable identification of subtle abnormalities, better demonstrate the craniocaudal extent and shape of lesions, and assist in the planning of interventions (see Chapter 1 ).
Patients may undergo CT scanning to evaluate the airways for a number of reasons, particularly individuals in whom occult airway disease is suspected based on the presence of clinical symptoms such as chronic cough, wheezing, dyspnea, or hemoptysis and patients with abnormalities on CXR that necessitate further delineation with cross-sectional imaging, such as chronic airspace opacities or consolidations or atelectasis. Other indications include discrepancy between clinical presentation and pulmonary function test (PFT) results and to evaluate potential candidates for interventional bronchoscopic procedures.
The role of MRI in the evaluation of the airways is limited. Although the large airways (trachea and main bronchi) are easily identified on standard spin-echo sequences, the segmental bronchi are difficult to visualize because of the relatively low spatial resolution of MRI compared with CT. One of the key advantages of MRI is the lack of ionizing radiation, which can be significant in children and young adults who require repeated imaging to decrease the risks associated with radiation exposure. MRI is beneficial in evaluating patients with vascular rings or tracheal compression by vascular anomalies and other mediastinal abnormalities (see Chapter 3 ).
The role of FDG PET/CT in the evaluation of the airways is primarily limited to the staging of patients with tracheobronchial malignancies (see Chapter 4 ).
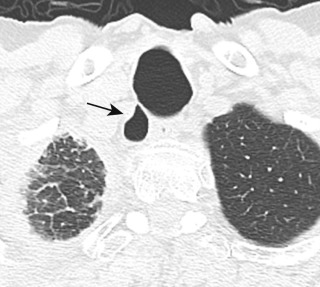
Focal outpouchings from the tracheal wall are known as tracheal diverticula, which may be congenital or acquired, and are present in approximately 1% of individuals. Acquired diverticula may be single or multiple, usually in the upper right paratracheal region, and are associated with increased intraluminal pressure and chronic cough. Most patients are asymptomatic, and these abnormalities are usually detected incidentally. However, some individuals may report signs and symptoms related to recurrent airway infections and chronic cough.
Small tracheal diverticula are often not visible on CXR but are readily identified on CT, with thin-section, multiplanar, and 3D reconstructions establishing the connection between the diverticula and the trachea ( Fig. 6.1 ). CT effectively distinguishes diverticula from other paratracheal air collections, including laryngocele, pharyngocele, Zenker diverticulum, and apical bullae or blebs.
Tracheal narrowing may be idiopathic, iatrogenic, or posttraumatic in etiology or caused by a wide variety of disease processes, including chronic obstructive pulmonary disease (COPD), tracheopathia osteochondroplastica, relapsing polychondritis, granulomatosis with polyangiitis, amyloidosis, sarcoidosis, or infection ( Table 6.2 ).
| Etiology | Airway Findings | Associated Abnormalities |
|---|---|---|
| Idiopathic | Smooth, tapered, irregular, lobulated, or eccentric narrowing 2–4 cm in length Subglottic airway typical |
± Involvement of larynx |
| Postintubation injury | Smooth narrowing (hourglass configuration) | |
| Posttraumatic | Smooth narrowing (hourglass configuration) | ± Osseous fractures (sternum, upper ribs) |
| Saber sheath | Smooth narrowing of intrathoracic trachea Coronal diameter half of sagittal diameter |
Emphysema Hyperinflated lungs Diffuse bronchial wall thickening |
| Tracheopathia osteochondroplastica | Submucosal nodularity and ossification Anterolateral walls of trachea and main bronchi affected |
|
| Relapsing polychondritis | Airway wall thickening sparing the posterior wall Diffuse narrowing of trachea and main bronchi ± Airway wall calcification ± Tracheobronchomalacia |
Nasal and auricular chondritis Arthritis |
| Granulomatosis with polyangiitis | Focal or diffuse wall thickening and narrowing ± Enlarged calcified cartilages |
Renal and pulmonary involvement |
| Amyloidosis | Diffuse wall thickening and narrowing Focal nodular masses that enhance and slowly progress ± Nodule calcification |
± Lymphadenopathy ± calcification |
| Sarcoidosis | Airway wall thickening Smooth, irregular, or nodular stenosis Lymph nodes may compress airways |
± Lymphadenopathy ± calcification Pulmonary involvement |
| Rhinosclerosis | Nodular masses or diffuse, symmetric narrowing of trachea and main bronchi Slowly progressive |
|
| Tuberculosis | Hyperplastic stage: irregular wall thickening and narrowing Fibrostenotic stage: smooth narrowing |
Mediastinal or hilar lymphadenopathy ± calcification Pulmonary involvement |
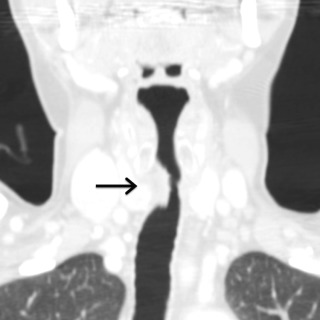
Idiopathic narrowing of the laryngeal and subglottic trachea is typically encountered in middle-aged women without a history of endotracheal intubation, traumatic injury, infection, or systemic disorder. The most common clinical symptoms include progressive shortness of breath and associated wheezing, stridor, or hoarseness.
Regions of stenosis measure 2 to 4 cm in length and result in severe compromise of the airway lumen, with affected sites measuring 5 mm or less at the point of greatest narrowing. On CT, the appearance of idiopathic laryngotracheal stenosis is highly variable and ranges from smooth and tapered regions of narrowing to irregular, lobular, and eccentric forms ( Fig. 6.2 ).
Stenoses may be treated conservatively with a wide variety of techniques such as dilation, endotracheal intubation, airway stenting, steroid injection, cryotherapy, or electrocoagulation, or they may be treated surgically.
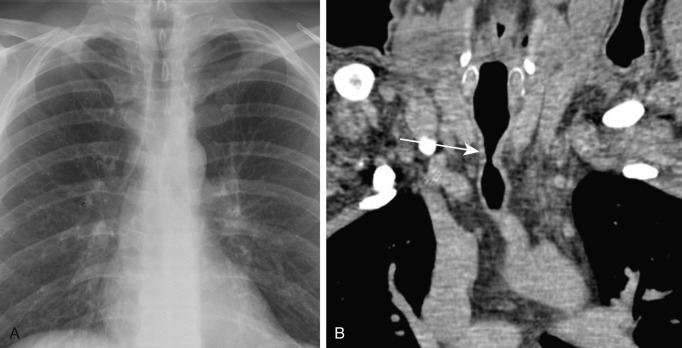
Endotracheal intubation and tracheostomy placement are the most common causes of acquired focal tracheal stenosis. Tracheal stenosis and other complications of endotracheal intubation, such as tracheoesophageal fistula and tracheoarterial fistula, are typically caused by cuff injury, specifically pressure necrosis. When the cuff pressure exceeds that of the capillaries, blood supply to the tracheal mucosa becomes compromised, and ischemic necrosis can occur, leading to softening and dissolution of the cartilage and, ultimately, tracheomalacia or scarring and stenosis. The trachea 3 to 4 cm below the cricoid cartilage is typically affected, corresponding to the level of the cuff of the endotracheal or tracheotomy tube. Less commonly, regions of tracheal stenosis may arise at the level of the tracheostomy stoma or at the site of tube tip impingement on the tracheal mucosa. Clinical symptoms of tracheal stenosis typically develop 2 to 6 weeks after extubation, although patients may present months later.
Tracheal stenosis may manifest as focal luminal narrowing on CXR. On multidetector computed tomography (MDCT), postintubation stenosis results in smooth, gradual narrowing with an hourglass configuration that is better demonstrated on coronal and sagittal reformations than axial images ( Fig. 6.3 ). Concentric or eccentric soft tissue thickening, representing edema and granulation tissue, may be present in acute stenosis. Focal expiratory airway collapse caused by destruction of airway cartilage may be identified in chronic stenosis. Stenoses are treated with surgical resection of the damaged region and end-to-end anastomosis.
The most common causes of posttraumatic tracheal stenosis include penetrating injuries, strangulation, and motor vehicle collisions. Iatrogenic airway stenosis may occur following airway anastomoses (see Chapter 12 ). Posttraumatic tracheal or bronchial stenosis results from healing of an untreated partial laceration or tear, which may be missed initially on imaging because of the lack of associated findings such as pneumomediastinum or pneumothorax in the setting of preservation of the peritracheal or peribronchial connective tissue or occlusion of the tear by a cuff or fibrin deposition. The typical hourglass configuration is usually visible on CT. In contrast, acute laceration of the trachea can occur after traumatic intubation or penetrating or decelerating injuries, discussed in more detail in Chapter 13 .
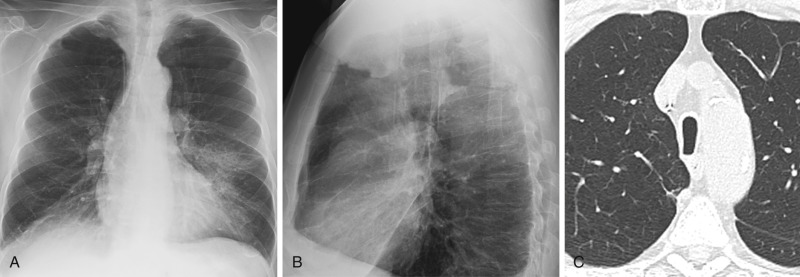
Saber-sheath trachea is an abnormal configuration of the intrathoracic trachea characterized by narrowing of the transverse diameter that typically begins at the level of the thoracic inlet. Male smokers with COPD are typically affected. In late-stage disease, the entire length of the intrathoracic trachea may be affected. The trachea is characteristically narrowed in the coronal dimension such that it is two thirds or less of the sagittal dimension ( Fig. 6.4 ). It is important to note that the main bronchi are not affected, and tracheal wall thickness is normal.
Tracheopathia osteochondroplastica is a rare, idiopathic benign disorder characterized by the presence of osteocartilaginous nodules in the submucosa of the large airways and thickening of tracheal rings. These nodules are connected to the perichondrium of the tracheal rings and protrude into the airway lumen. It is most common in men older than 50 years of age. The anterolateral tracheal walls are affected with a predilection for the upper two thirds of the trachea. Because the posterior wall of the trachea is membranous and lacks cartilage, it is characteristically spared. Most patients with tracheopathia osteochondroplastica are asymptomatic; however, clinical symptoms such as wheezing, cough, and hemoptysis have been reported at clinical presentation.
The most common abnormalities on CXR include nodular thickening and irregularity of the tracheal wall and luminal narrowing. CT effectively demonstrates these mural nodules, which can be seen to protrude into the airway lumen, frequently demonstrate calcification, and spare the membranous wall ( Fig. 6.5 ).
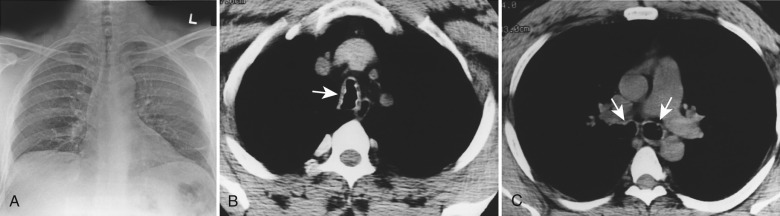
Relapsing polychondritis is a rare autoimmune disorder involving the cartilage of the ears, nose, upper respiratory tract, and joints that results in inflammation and destruction. Granulomatous tissue or fibrosis replaces the destroyed cartilage. Up to 50% of patients with the disease experience airway involvement. Associations with other autoimmune disorders such as autoimmune thyroiditis, rheumatoid arthritis, systemic lupus erythematosus, systemic vasculitis, Sjögren syndrome, and inflammatory bowel disease have been described. Airway involvement is more common in women than men, and the average age of presentation is 50 years.
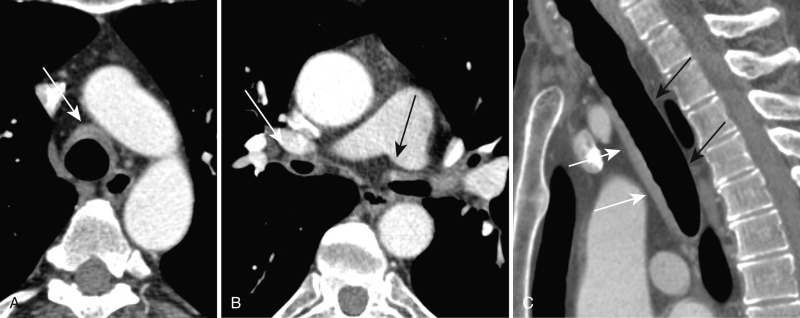
On CXR, the tracheobronchial walls are diffusely thickened, and the trachea and main bronchi show diffuse, smooth narrowing. CT demonstrates diffuse, smooth thickening of the tracheobronchial tree with characteristic sparing of the posterior or membranous segment ( Fig. 6.6 ). Stenosis of the trachea and main bronchi are usually present, and involvement of the smaller airways, including the segmental and subsegmental bronchi, can be seen. Associated findings include calcifications and distal postobstructive bronchiectasis. Dynamic imaging with CT may demonstrate excessive expiratory airway collapse caused by inflammation and destruction of cartilage, often in association with air trapping.
Early detection and treatment of the disease are necessary to delay cartilage destruction. Severe airway involvement is associated with a poor prognosis. Corticosteroids and immunosuppressive medications are the mainstay of medical therapy. Continued positive airway pressure, bronchoscopic interventions such as laser therapy, balloon dilation, and stenting, and tracheostomy in cases of severe stenosis involving the upper trachea can improve clinical symptoms.
Granulomatosis with polyangiitis (GPA), formerly known as Wegener granulomatosis, is an autoimmune, systemic vasculitis characterized by the presence of cytoplasmic antineutrophil cytoplasmic antibodies (cANCAs) in serum and granuloma formation. Small and medium-sized vessels are typically affected, and up to 50% of patients have involvement of the tracheobronchial tree. The upper and lower portions of the respiratory tract are affected in combination with the kidneys and other visceral organs. When airway involvement is present, the laryngeal and subglottic trachea is typically affected. Diffuse involvement of the tracheobronchial tree is less common and usually encountered in late-stage disease.
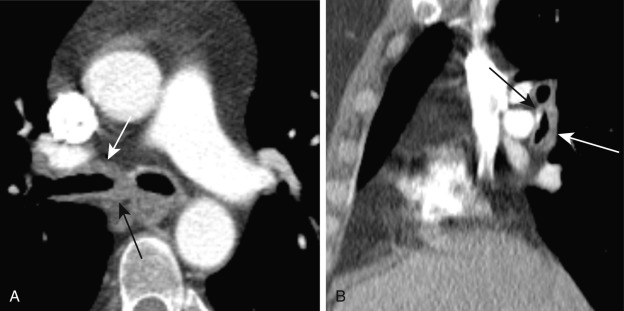
Chest radiography may demonstrate airway wall thickening, focal or diffuse wall irregularity, and luminal narrowing. On CT, involvement of the large airways results in smooth or nodular tracheobronchial wall thickening, which may progress to single or multiple regions of stenosis. Unlike in relapsing polychondritis, airway wall thickening of GPA can be circumferential, without sparing of the posterior wall ( Fig. 6.7 ). Irregular calcification of the cartilage rings, postobstructive atelectasis in the setting of airway obstruction caused by granulation tissue, and bronchiectasis may be present.
Corticosteroids and immunosuppressive medications are used for the medical treatment of GPA. In the setting of persistent airway obstruction, dilation, stent placement, laser ablation, tracheostomy, or surgical resection can improve clinical symptoms.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here