Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This chapter is not intended to provide a comprehensive review of all the bedside procedures that can be performed in the ICU. Rather, it will illustrate the indications, management, and complications of common surgical procedures that can be performed outside of the OR and propose an algorithm with basic principles to follow for bedside procedures ( Fig. 1 ).
The basic concept of bringing the surgeon to the site of the injured patient is not novel. During the Korean War, mobile army surgical hospital (MASH) units allowed injured soldiers to receive essential surgical care close to the battlefield before they could be transported to hospitals for definite care. This concept is widely used today by the military around the world and more recently has been adopted by humanitarian organizations to provide medical care to injured civilians. It has also been recently modified and used in response to new challenges created by critically ill patients in the ICU.
Critically injured patients are more likely to survive their injuries today due to a multitude of improvements in trauma systems and critical care. As a result, “diseases of survivorship” have become more prevalent and are posing new and complex challenges to the trauma surgeon. Clearly, surgeons are most comfortable operating in the OR where conditions are optimal. However, the safe performance of bedside elective surgical procedures has already been demonstrated with tracheostomy and percutaneous feeding access. Currently, there are now circumstances where it is not safe to transport the patient to the OR, and the surgeon is forced to operate under less optimal conditions, in the patient’s best interest, in the surgical ICU SICU. This situation arises if the patient is too critical to travel to the OR but needs urgent or emergent surgery, or if the patient needs an emergent surgery but the OR is not immediately available due to other emergencies. This chapter will provide indications and management of both elective and emergent bedside procedures.
Patients with persistent respiratory failure following major trauma frequently require tracheostomy since the complications related to the presence of an endotracheal tube for more than 7 days increases and can be life-threatening. Patients who have a high likelihood of requiring prolonged mechanical ventilation undergo tracheostomy at the earliest possible time when conditions are stable and optimal. Early tracheostomy is advocated with patients with head injury (Level II) and may decrease total days off mechanical ventilation and ICU length of stay in patients without head injury (Level III) (The Eastern Association for the Surgery of Trauma Practice Management Guidelines Work Group). Some of the theoretic and proven benefits of tracheostomy include reduction of dead space and airway resistance, facilitation of weaning, improved pulmonary toilet and oral care, better toleration by the patient, and establishing a secure long-term airway.
The indications for tracheostomy include (1) need for prolonged mechanical ventilation (>7 days), (2) an inability to protect airway such as in severe traumatic brain injury, severe maxillofacial trauma, extensive neck, or vocal cord edema/trauma/injury, (3) complex tracheal repair; (4) cervical spinal cord injuries, and (5) ventilator dependency due to frequent trips to the OR.
Bedside tracheostomy can be performed via an open or percutaneous technique based on the surgeon’s preference. Tracheostomy is not advised at times when respiratory complications related to the procedure will be poorly tolerated by the patient. These include conditions such as severe hypoxemia, severe hypercarbia, or respiratory acidosis. Redo tracheostomies in patients with difficult anatomy (short neck, goiter or cervical spine injury) should preferably be performed in the OR under more favorable conditions with optimal lighting and instruments. No clear advantages have been found in percutaneous technique compared to open techniques. Relative contraindications to the percutaneous tracheostomy include a redo tracheostomy, moderate to severe coagulopathy, and unstable cervical spine injuries or an inability to extend the neck.
Adequate preparation for this procedure is critical, as errors can quickly lead to major complications. A complete surgical tracheostomy tray and percutaneous tracheostomy kit are present at the bedside ( Table 1 ). Lighting in the SICU should be optimized, or surgeons may prefer using a portable headlight. Assisting personnel include a surgical team with a surgeon and one assistant surgeon, one respiratory therapist, one anesthesiologist, and an ICU nurse. All personnel in the room have protective headwear, masks, and gloves, and the surgeons also wear sterile gowns and gloves.
| Bedside Tracheostomy | |
| Tracheal set |
|
| Airways |
|
| Sutures |
|
| Anesthesia |
|
| Field |
|
| Percutaneous Tracheostomy | |
| Percutaneous tracheostomy kit | |
| Bronchoscope | |
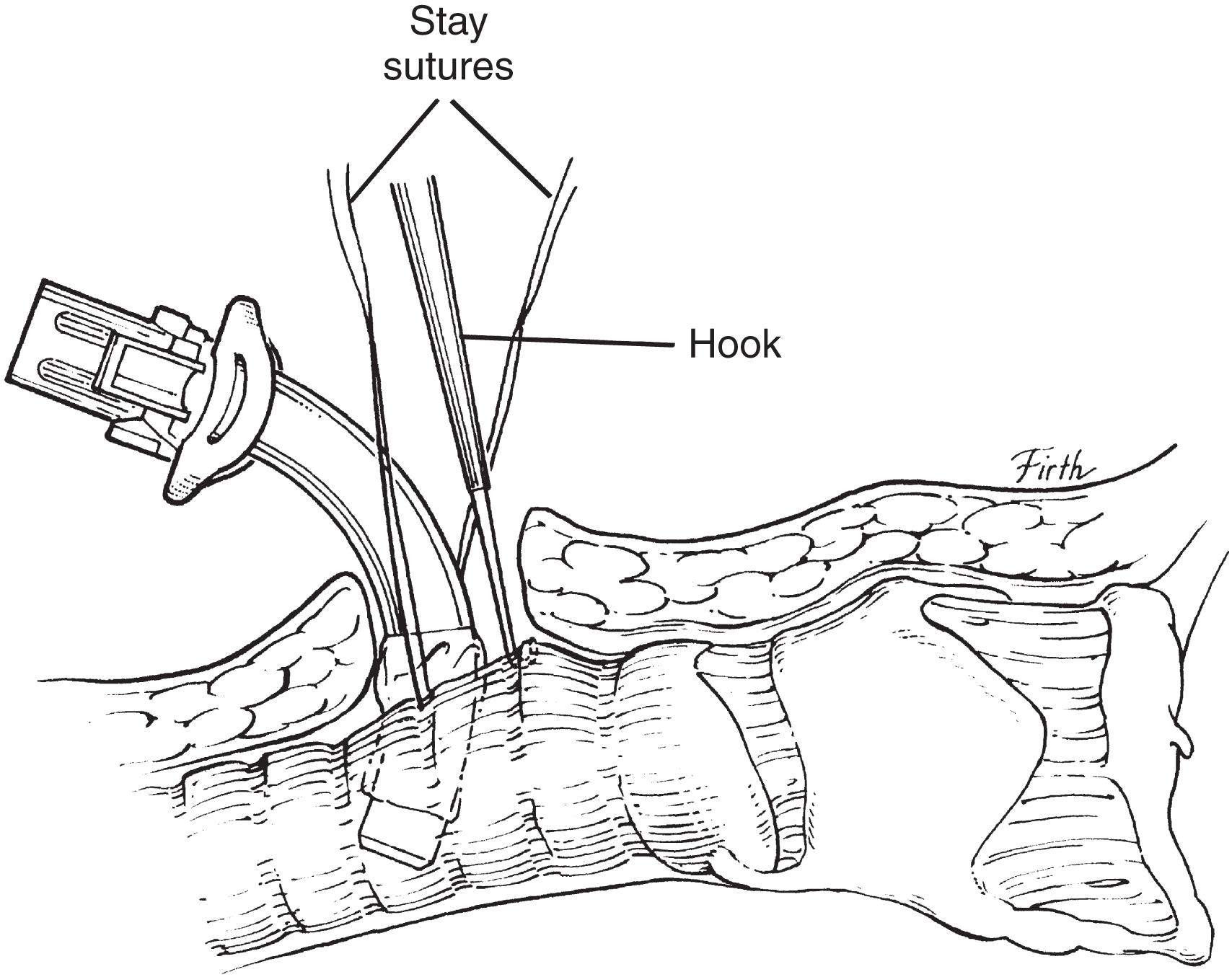
Once the patient is paralyzed and sedated, the neck is prepared and draped. Local anesthetic is injected at the surgical site, and then a 2-cm vertical midline incision is made below the cricoid. The platysma is divided, and the strap muscles are retracted laterally. The second to fourth tracheal rings are exposed by retracting the isthmus of thyroid superiorly (using a vein retractor) or by dividing the isthmus of the thyroid. Stay sutures can be placed at the lateral aspect of trachea; note that the balloon of endotracheal tube should be deflated while placing stay sutures. Before the procedure begins, the surgeon should test the balloon of the tracheostomy and ensure that the anesthesiologist suctions the endotracheal tube and mouth and that all equipment works and is within reach. A tracheotomy is performed using an 11-blade scalpel, the opening is dilated, and under direct vision the endotracheal tube is pulled back to just above the tracheotomy site. The tracheostomy is inserted, the inner cannula is placed, and the balloon is inflated. Then capnography is performed, adequate return of tidal volume is assessed, and chest wall movement is visualized. When placement is confirmed, the tracheostomy is sutured to the skin and secured with tracheal ties, and then the endotracheal tube is removed ( Fig. 2 ).
There are several kits available today with various modifications for the percutaneous tracheostomy using the Ciaglia technique. Use of bronchoscopy has been advocated by some, but this procedure has low morbidity and mortality without this adjunct. The authors recommend the following percutaneous technique, which includes making a 2-cm vertical midline incision below the cricoid, and gentle dissection and retraction of the strap muscles with the ability to identify the thyroid and palpate the tracheal rings. With or without the use of a bronchoscope in the endotracheal tube, the endotracheal tube is pulled back superiorly so that a 10-mL saline-filled syringe is inserted into the trachea just below the endotracheal tube at about the second or third tracheal ring. Once air bubbles enter the syringe, the syringe is removed, and a guidewire is advanced into the tracheal lumen. The needle is removed, and the tract is dilated with the short dilator. With the use of a guiding catheter, either serial dilation (12- to 36-French) or a single tapered dilation is performed. The 28-Fr dilator within a #8 tracheostomy tube is placed over the guiding catheter and the entire unit is inserted into the trachea ( Fig. 2 ). Once in place, the guidewire, the guiding catheter, and the 28-Fr dilator are removed; the inner cannula is placed; and the balloon is inflated. Placement is confirmed and securing of the tracheostomy is performed as previously explained.
Due to the ongoing coronavirus 19 (COVID-19) pandemic, there are increased numbers of infected patients in the ICU. The performance of bedside tracheostomy in critically ill intubated COVID-19-positive patients exposes the ICU and operative team to a risk of infection. It is recommended that these procedures are performed in a negative-pressure room since this is an aerosol-generating procedure. A number of modifications and precautions are needed to reduce the risk of infection. First attempt to delay the procedure if possible until the patient is COVID-19-negative because a delay of tracheostomy to ensure a reduced viral load and likelihood of survivorship is recommended. Tracheostomy followed by patient death or institution of comfort care soon after should be avoided.
A bedside tracheostomy limits the risk of transportation of COVID-19 patient to the OR but it does require the following modifications:
Use of a negative pressure room with limited but experienced staff to minimize accidental spread.
Use of full personal protective equipment (PPE) by everyone in the room, including gown, glovers, protective eye wear, cap and N-95 mask. The use of a hooded device such as powered air-purifying respirator (PAPR) may provide additional protection but limit comfort during the procedure.
Team discussion about the steps and good communication prior to the tracheostomy is recommended since communication with full PPE is less effective and clinical decompensation can occur due to limited team size.
Keep the patient on the closed ventilator circuit for as long as possible. Disconnecting the circuit should ideally be preceded by 10 to 20 seconds of apnea, followed by clamping of the circuit to reduce virus aerosolization. The used ventilator circuit and endotracheal tube should be directly discarded in a closed biohazard bag to avoid virus spread.
Bedside tracheostomy is considered a safe procedure when performed meticulously and using the suggestions stated here. It has a low mortality rate (0.1%–1%) and minimal morbidity (up to 3%). Complications related to tracheostomy occur intraoperatively, early postoperatively, and late postoperatively. Intraoperative complications include bleeding, perforation of posterior wall of the trachea or anterior wall of esophagus, hypoxia, and loss of the airway. Early postoperative complications include bleeding, hematoma, pneumothorax, and tracheoesophageal fistula. Late postoperative complications include subglottic stenosis, laryngeal nerve injury, tracheal granulation, and tracheoinnominate fistula.
A meta-analysis comparing open surgical and percutaneous tracheostomies found that the rate of serious complication was similar in the two groups. The authors also noted that perioperative complications occurred more often with the percutaneous technique, but that postoperative complications were more frequent with the open surgical tracheotomy. However, most of the differences in complication rates were attributed to minor complications. Another meta-analysis by Freeman et al found no difference in overall operative complication rates but lower postoperative complications and bleeding in the percutaneous technique. Subglottic stenosis is a rare late complication but can occur after percutaneous and open tracheostomy.
Seriously injured patients are at risk for malnutrition and all of its complications. In addition, failure to use the gastrointestinal tract for a prolonged period leads to atrophy of the intestinal mucosa and bacterial translocation. Short-term enteral nutrition is often provided via a naso- or orogastric tube. Due to the high incidence of patient discomfort, accidental dislodgment, and sinusitis from prolonged use of nasoenteric tubes, percutaneous cannulation of the gastrointestinal tract is preferred for long-term feeding access. Percutaneous endoscopic gastrostomy (PEG) can be performed by the surgeon at the bedside in the SICU. PEG was introduced in 1980 as an alternative to laparotomy for placement of gastrostomy. The surgeon intensivist operator has the advantage of being familiar with the patient’s condition and possible recent abdominal surgery.
Many patients undergoing major surgery for trauma will have a jejunostomy tube or gastrostomy tube inserted at the time of surgery. In the SICU setting, patients needing access to the gastrointestinal tract are those who remain ill and are unable to eat. Any organ failure may cause inability to tolerate oral feedings, especially failure resulting from prolonged ventilatory dependency, traumatic brain injury, or prolonged sepsis. If the duration of injury is expected to be 1 to 2 weeks, feeding can be given by naso- or orogastric tube. Longer periods of naso- or orogastric intubation are associated with sinusitis, increased reflux, aspiration, and rarely esophageal stricture. Other indications for PEG include facial trauma and dysphagia.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here