Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The spotted fevers comprise a large group of tick-, mite-, and flea-borne zoonotic infections that are caused by closely related rickettsiae. These include Rocky Mountain spotted fever (RMSF), boutonneuse fever, African tick bite fever, North Asian tick typhus, lymphangitis-associated rickettsiosis, Queensland tick typhus, Flinders Island spotted fever, Japanese spotted fever, tick-borne lymphadenopathy, Far Eastern spotted fever, flea-borne spotted fever, and rickettsialpox. Rickettsiae are emerging or reemerging pathogens in many parts of the world. Associated diseases have a broad spectrum of severity; the most virulent, RMSF (historically in Montana), exhibited a case-fatality rate of 66%. Even young and previously healthy people may die of RMSF. In recent years, the wide distribution and potential severity of the other spotted fevers have been recognized, especially in Europe, Africa, Australia, Asia, and Japan. Early diagnosis remains deceptively difficult.
RMSF was first described in Idaho in the late 19th century. Ricketts established the infectious nature of the illness and demonstrated the role of ticks as the vector in western Montana in 1906. Wolbach, in 1919, clearly identified the causative rickettsiae in endothelial cells. The causative agent was designated Rickettsia rickettsii. Geographic origin, clinicoepidemiologic observations, and serotyping were the historical basis for species designation of subsequent rickettsial isolates. Phylogenetic analyses have more accurately revealed the evolutionary relationships among the rapidly increasing number of clinical and environmental isolates. It is likely that virulence was a relatively late mechanism of evolutionary survival for Rickettsia , which arose as vertically transmitted symbiotes of insects, arachnids, leeches, and amebas, with gain of virulence associated with genome reduction. Contemporary phylogenomics based on whole-genome sequences has defined the ancestral, typhus, transitional, and spotted fever groups. Rickettsia australis, Rickettsia felis , and Rickettsia akari are members of the transitional group. Criteria for species designation of Rickettsia based on historically named species, an approach considered inappropriate by some respected taxonomists, have been proposed to be determined by the divergence of 16S ribosomal RNA (rRNA), citrate synthase, outer membrane proteins A and B, and Sca4 of the most closely related, previously named species. These criteria have resulted in proposals for species designation of rickettsial strains that are much more closely related than other bacterial species. For example, a proposed criterion of 0.2% divergence of the 16S rRNA gene has created many more species names than the usual 1% to 1.3% divergence of other bacterial taxa. An attractive approach, concatenated phylogeny based on multilocus sequence typing using eight loci of Rickettsia genes has revealed that species designation of Rickettsia sibirica, Rickettsia africae , and Rickettsia parkeri should be subject to further discussion. The spotted fever group (SFG) rickettsial strains that have been strongly or weakly associated with human infections ( R. rickettsii, R. conorii, R. africae, R. sibirica [including the mongolitimonae strain], R. honei, R. japonica, R. slovaca, R. parkeri, R. massiliae, R. monacensis, R. aeschlimannii, R. heilongjiangensis, R. amblyommatis , and R. helvetica ) definitely merit identification that is useful for clinical and epidemiologic purposes. It is controversial whether they and a rapidly growing number of candidates merit separate designations as different at the species level. An excellent guide for diagnosis and management of spotted fever rickettsioses has been published by the Centers for Disease Control and Prevention (CDC).
SFG rickettsiae are obligately intracellular bacteria that reside in the cytosol and less often in the nuclei of host cells. These rickettsiae are small, measuring approximately 0.3 by 1.0 µm. They have one of the smallest bacterial genomes, ranging between 1.1 and 1.6 Mb. The cell wall, which has the ultrastructural appearance of a gram-negative bacterium, contains peptidoglycan and lipopolysaccharide (LPS). Rickettsiae are difficult to stain with ordinary bacterial stains but are conveniently stained with the Gimenez method or with acridine orange. They have not been cultivated in cell-free medium. Growth requires living host cells, such as the yolk sac of embryonated eggs, experimental animals, or cell culture (e.g., Vero, HEL, and L-929 cells). Rickettsiae have undergone remarkable genome reduction with exploitation of their cytosolic environment by being highly adapted for intracellular survival with effective transport systems for adenosine triphosphate, amino acids, and phosphorylated sugars, in addition to their own independent metabolic enzymes. Rickettsiae exhibit a large family of surface proteins, (autotransporters) that are a major source of antigenic differences. Among these, OmpA (190 kDa) and OmpB (135 kDa) contain conformational epitopes that are targets of humoral immunity and were the antigenic basis for serotyping; other antigens are also shared among the SFG. The LPS of SFG rickettsiae contains highly immunogenic antigens that are strongly cross-reactive among all members of the group. However, antibodies to LPS do not provide protection against infection.
The role of a tick bite in the transmission of RMSF was demonstrated by McCalla and Brereton and reported in 1908 ; a tick obtained from a patient with RMSF transmitted the disease to two volunteers. The seasonal distribution of RMSF parallels tick activity. The tick is both the vector and the main reservoir. Dermacentor variabilis, the American dog tick, is the prevalent vector in the eastern two-thirds of the United States and the Far West; Dermacentor andersoni, the Rocky Mountain wood tick, in the western states; Rhipicephalus sanguineus, in Mexico and Arizona ; and Amblyomma cajennense, Amblyomma sculptum, Amblyomma mixtum, Amblyomma patinoi, Amblyomma tonelliae, and Amblyomma aureolatum in Central and South America. Amblyomma tenellum is a potential vector for R. rickettsii in Mexico and is likely involved in its maintenance in nature. Causes for the variation in infection rates among populations of ticks are not clear, although in Dermacentor only a small portion of ticks (generally 4%) carry any rickettsiae, and fewer than 1 in 1000 ticks carry virulent R. rickettsii. One limiting factor is the deleterious effect that R. rickettsii has on ticks; another is the inhibition of establishment of transovarial transmission of R. rickettsii by the presence of another Rickettsia species in the tick. Humidity, climatic variations, human activities altering the vegetation and fauna, and the use of insecticides have been suspected to play a role in the fluctuation of tick populations and the prevalence of human rickettsiosis.
R. rickettsii is transmitted transstadially (stage to stage) and transovarially in ticks, thus maintaining the agent in nature. Horizontal transmission through vertebrate hosts would also appear to occur to a small degree and to be a necessary factor for the maintenance of R. rickettsii in nature. In most mammals, rickettsemia is of very short duration and low titer, allowing infection of only a small proportion of feeding ticks. Of the three tick stages—larva, nymph, and adult—only adult Dermacentor ticks feed on humans. The prevalence of pathogenic rickettsiae in various populations of ticks, their efficiency at transstadial and transovarial transmission, and their ability to infect uninfected ticks while cofeeding on the same host are variable. Many rickettsiae of unknown pathogenicity have been isolated and characterized in the United States, including Rickettsia bellii, Rickettsia montanensis, Rickettsia rhipicephali , and Rickettsia peacockii.
The tick transmits the disease to humans during a prolonged period of feeding that may last for 1 to 2 weeks. The blood meal and host temperature associated with tick feeding modulate the expression of genes that may upregulate virulence factors of rickettsiae. Furthermore, salivary products of the tick may enhance establishment of rickettsial infection. The bite is painless and frequently goes unnoticed. After the attached tick has fed for 6 to 10 hours, rickettsiae begin to be injected from the salivary glands. An even longer period may be required for reactivation of rickettsial virulence in unfed ticks. Humans can also be infected by exposure to infective tick hemolymph during the removal of ticks from persons or domestic animals, especially when the tick is crushed between the fingers.
The mean infectious dose of R. rickettsii is 23 organisms, but as few as one bacillus can cause infection. Laboratory-acquired infection transmitted by infectious aerosols or parenteral inoculation of R. rickettsii may be prevented with careful technique and the use of biohazard containment hoods, masks, and gloves.
From the 1870s until 1931, RMSF was recognized as existing only in the western United States. At present, the prevalence of the disease is higher in the South Atlantic states and in the South Central regions than in the Rocky Mountain states ( Fig. 186.1 ). Local prevalence in highly endemic areas, such as North Carolina, has been as great as 14.59 per 100,000. The incidence reported among American Indians is as high as 16.8 per 100,000 according to passive national surveillance data and 94.6 per 100,000 with use of International Classification of Diseases, Ninth Revision (ICD-9) codes from Indian Health Service records. Tribal lands in Arizona have been found to be highly endemic areas for RMSF. These populations are affected economically through medical costs, loss of productivity, and loss of life related to the disease. Indeed, the 10% case-fatality rate in Arizona is currently higher than in other areas of the United States. The report of a focus in the South Bronx emphasizes that the ecologic conditions that permit the establishment of RMSF are widely distributed. Most cases are diagnosed during late spring and summer. However, especially in the southern states, a few cases occur during the winter.
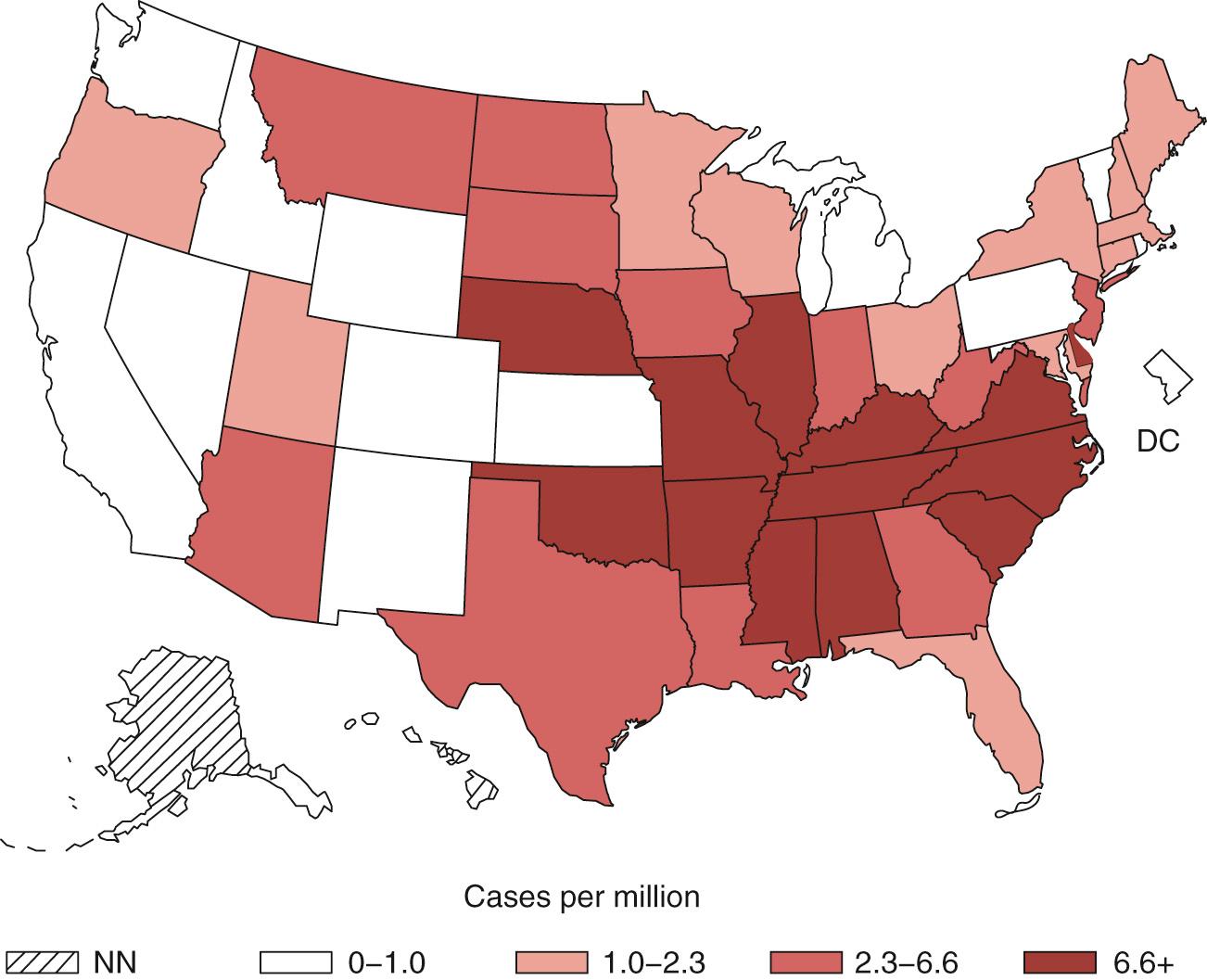
In the southern states, incidence is highest in children, adults 60 to 69 years old, and patients who are known to be exposed more often to ticks than are matched controls. In the western mountainous states, because of transmission by the wood tick D. andersoni, a higher proportion of men contract the disease because of occupational exposure. On tribal lands of Arizona, incidence is highest in children younger than 10 years, and cases are associated with exposure to ticks and contact with dogs. The case-fatality rates reported from 1999 to 2012 are highest for children younger than 10 years old and those older than 70.
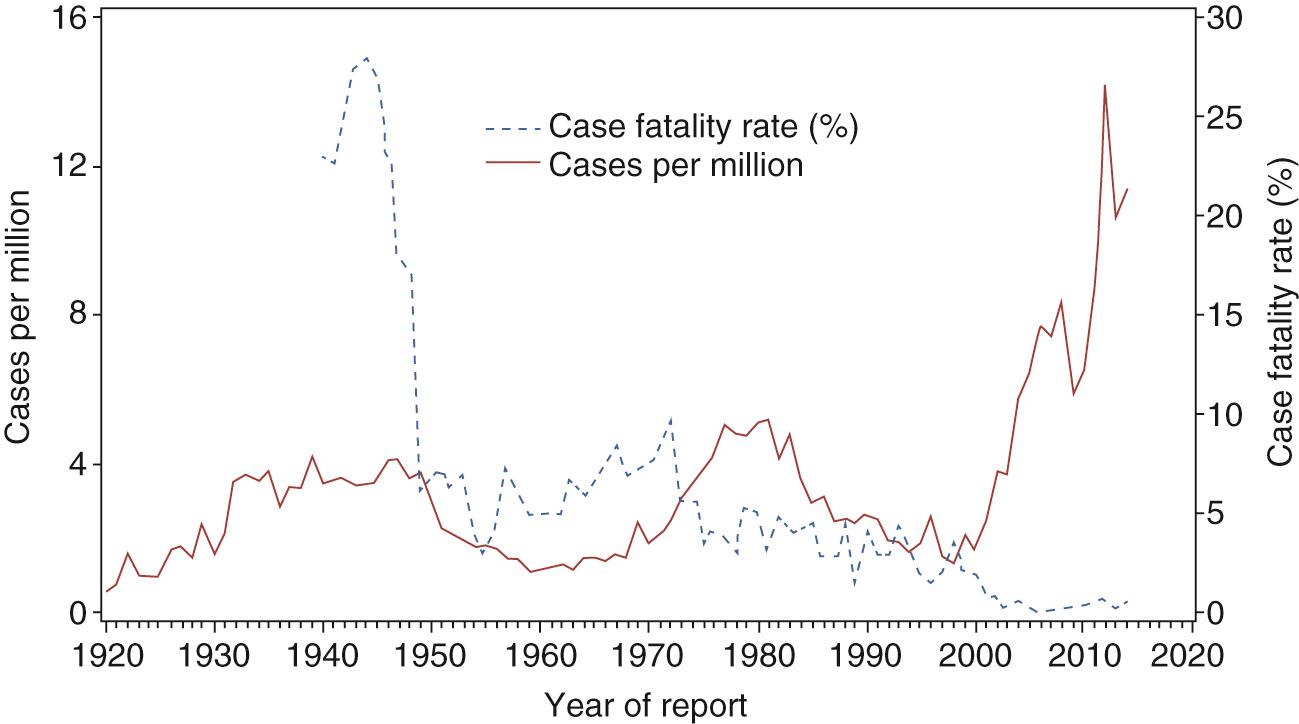
Consistent with unexplained 30- to 40-year cycles of waxing and waning incidence of RMSF, the number of cases reported to the CDC has skyrocketed (13,561 cases from 2008 to 2012); Fig. 186.2 ). Problematic issues are the low proportion of laboratory-confirmed cases (15%, with only 5% with specific evidence for R. rickettsii ); a significant population of healthy persons with serum antibodies reactive with R. rickettsii, which shares antigens with other SFG rickettsiae; discovery that R. parkeri transmitted by Amblyomma maculatum causes human infections; evidence that highly prevalent R. amblyommatis carried by Amblyomma americanum ticks causes mild or subclinical infections; and a reported RMSF case-fatality rate of 0.4% compared with 23% in the preantibiotic era and 5% in recent years. It is likely that many of these patients had human monocytotropic ehrlichiosis and were misdiagnosed based on the presence of antibodies stimulated at a previous time by R. amblyommatis. Some of them likely were infected with R. parkeri.
Phylogenetic analysis of isolates of R. rickettsii has demonstrated that the organism exists in several clades, likely originated in North America, and subsequently drifted into Central and South America, where it is currently emerging in Argentina and reemerging in Brazil, Colombia, Panama, Costa Rica, and Mexico, largely unrecognized, and misdiagnosed as dengue or other febrile exanthems. R. rickettsii infections appear to be more severe in these regions, with case-fatality rates of up to 40% in Brazil and Mexico. The alarming situation in northern Mexico has prompted the Mexican Ministry of Health to declare an epidemiologic emergency. Cases originating from northern Mexico have spilled over into the United States with fatal outcomes due to delay in early treatment. Cases have also occurred with community and familial clustering likely associated with large populations of R. sanguineus .
Rickettsiae introduced into the skin apparently spread via lymphatics and small blood vessels to the systemic and pulmonary circulation where, by means of OmpA, OmpB, Sca1, Sca2, and fibroblast growth factor receptor-1, they attach to and induce phagocytosis by their target cells—the vascular endothelium—to establish numerous disseminated foci of infection. Comparison of the genomes of highly passaged isolates of R. rickettsii originating from the western and eastern United States, three virulent and one attenuated, has revealed numerous deletions and amino-acid substitutions between western and eastern isolates. A notable mutation in the avirulent stain was disruption of the ompA gene leading to absence of its expression. Targeted knockout of ompA in the virulent Sheila Smith strain failed to attenuate disease in an animal model, indicating alternative mechanisms contributing to pathogenesis. Other mutations in the eastern isolates include four amino-acid substitutions in OmpB, which in the case of the attenuated Iowa strain does not undergo normal posttranslational processing. After entry, the rickettsiae escape rapidly from the phagosome into the cytosol in association with expression of membranolytic phospholipase D and hemolysin C, and less frequently invade the nucleus. Rickettsiae proliferate intracellularly by means of binary fission. The movement of spotted fever rickettsiae within the cytoplasm, into projections invaginating into the nucleus and into cell projections from which they are released extracellularly or spread into the adjacent cell, is caused in part by propulsion by the host cell's actin filaments. SFG rickettsial protein Sca2 is associated with activation of Arp2/3 and polymerization of actin at one pole of the rickettsiae, and RickA also plays a role during the early stage of infection in actin-based mobility. Evolutionary and experimental disruption of actin-based motility seems to attenuate SFG rickettsial virulence.
The consequence of cell-to-cell spread in the body is a focal network of hundreds of contiguous infected endothelial cells corresponding to the lesions (e.g., maculopapular rash). The presence of greater quantities of rickettsiae in damaged cells supports the concept of direct cell injury. No convincing data support endotoxin or exotoxin as a pathogenic mechanism. Rickettsial injury to the host cell is caused, at least in part, by free radical–induced damage to host cell membranes and rickettsial phospholipase and protease activities. More pathogenic rickettsiae are associated with increased endothelial injury and cell death. The major pathophysiologic effect of endothelial cell injury is increased vascular permeability, which in turn results in edema, hypovolemia, hypotension, and hypoalbuminemia. Endothelial cell induction of heme oxidase and cyclooxygenase, production of prostaglandins, and phosphorylation-induced destabilization of vascular endothelial cadherin may contribute to vasodilatation and increased vascular permeability. Hyponatremia is the result of secretion of antidiuretic hormone as an appropriate response to hypovolemia. High quantities of rickettsiae infecting the pulmonary microcirculation are associated with increased vascular permeability and cause noncardiogenic pulmonary edema. Vascular injury and the subsequent host lymphohistiocytic response correspond to the distribution of rickettsiae and include interstitial pneumonia, interstitial myocarditis, encephalitis, and similar vascular lesions in the rash, gastrointestinal tract, pancreas, liver, skeletal muscles, and kidneys. However, even severe vascular injury rarely leads to clinically significant hemorrhage. Platelets are consumed locally in numerous foci of infection; consequently, thrombocytopenia is observed in 32% to 52% of patients. A procoagulant state ensues, including endothelial injury, release of procoagulant components, activation of the coagulation cascade with thrombin generation, platelet activation, increased antifibrinolytic factors, consumption of natural anticoagulants, activation of the kallikrein-kinin system, and secretion of coagulation-promoting cytokines. These observations are supported by numerous studies of endothelial cells in culture, such as the demonstration that tissue factor is secreted by Rickettsia -infected endothelial cells, but true disseminated intravascular coagulation occurs only rarely, and occlusive vascular thrombosis is not the basic pathophysiologic event.
The host immune and inflammatory responses are critical for clearance of infection, but cytokines may contribute also to increased vascular permeability, and T-regulatory cells may contribute to suppression of immunity in overwhelming fatal illness. T lymphocytes (particularly CD8 cells) are important effectors of immune clearance of rickettsiae, and interferon-γ (IFN-γ) and tumor necrosis factor-α activate infected endothelial cells to kill intracellular rickettsiae. Dendritic cells stimulated via Toll-like receptor 4 activate natural killer cells in the early innate immune response and secrete IFN-γ, which dampens the rickettsial burden. The adaptor molecule MyD88 is a key component of Toll-like receptor–mediated recognition of Rickettsia. In macrophages, activation of the inflammasome contributes to host control of the bacterium. The ability to invade and proliferate in macrophage-like cells has been noted in pathogenic but not in nonpathogenic rickettsiae. Cytotoxic T lymphocytes are crucial to the clearance of rickettsial infection. OmpA and OmpB cell wall proteins are important immunogens.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here