Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Perineal and urogenital reconstruction has particular challenges due to the anatomic and functional complexity and differences between the male and female genitalia. Furthermore, genital defects and abnormalities are associated with psychological and social implications, which must be considered by the reconstructive surgeon. The complexity of genital anomalies requires a multidisciplinary approach with close cooperation between the reconstructive surgeon and the urologist or gynecologist.
Pertinent functions to consider from a reconstructive standpoint:
General
Micturition
Urinary and fecal continence
Intercourse
Erogenous sensation
Protective sensation
Female
Bidirectional vaginal/cervical patency (insemination, menstruation, birth)
Lubrication
Male
Erection
Emission
Ejaculation
Extracorporal testicular location
Upright micturition
The perineum consists of a complex fibromuscular system, which aids in actively supporting the abdominal and pelvic viscera, and plays active roles in sexual function, micturition, and defecation. We focus on the recipient neurovascular structures located in the perineum.
The perineum can be divided into an anterior, urogenital, and a posterior anal triangle by a transverse line connecting the ischial tuberosities ( Fig. 19.1 ). In the urogenital triangle we find the urogenital diaphragm, which spans between both pubic rami and is perforated by the urethra and vagina. It also serves as the insertion point for the musculature of the external genitalia (i.e., ischiocavernal and bulbospongiosal muscles). In the posterior lateral corner of the urogenital triangle, the neurovascular bundle enters the perineal region from the pudendal canal (Alcock canal). It consists of terminal branches of the pudendal nerve, the internal pudendal artery and vein. The pudendal nerve is split into the deep and superficial perineal nerve as well as the dorsal nerve of the penis/clitoris. The artery too splits into three branches: dorsal penile/clitoral artery, deep dorsal penile/clitoral artery, and bulbous artery. The venous drainage happens via the deep dorsal vein, the internal pudendal system, as well as the earlier mentioned superficial dorsal vein. The latter runs in the retropubic space and drains into the vesical plexus. Branches of the internal pudendal, obturator, and external pudendal vessels ensure the blood supply to the vulva and scrotum.
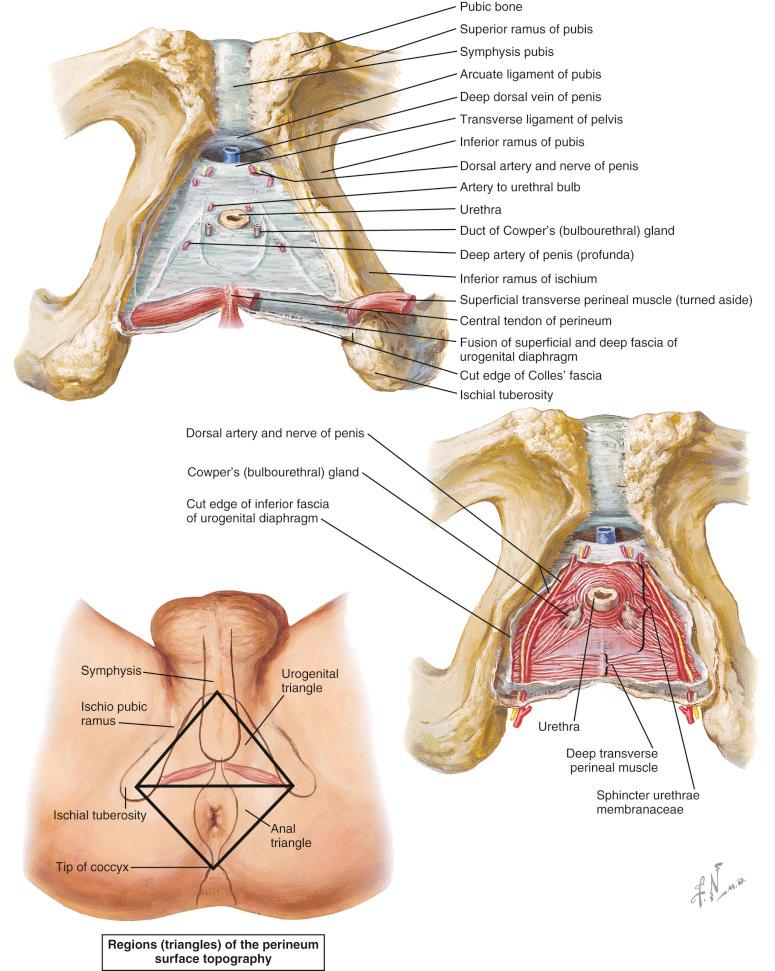
The posterior anal triangle includes the ischioanal fossa, the pudendal canal, and the anal sphincter system.
Pertinent neurovascular structures for the reconstructive surgeon (see Chapter 11 for detailed discussion; also Figs 11.6 and 11.10 ):
External pudendal artery and vein – superficial and deep branch
Medial circumflex femoral artery and vein
Superficial circumflex iliac artery and vein
Femoral artery and vein
Saphenous vein
Obturator artery and vein
Obturator nerve
Perineal branch of posterior femoral cutaneous nerve
Iliohypogastric nerve
Ilioinguinal nerve
Genital branch of the genitofemoral nerve
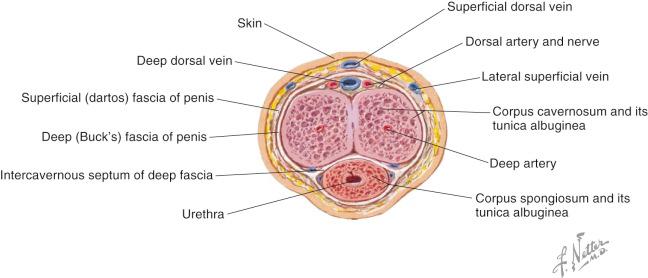
Skin and fascial layers of the penis ( Fig. 19.2 ):
Skin
Dartos fascia (continuation of Scarpa's and Colles' fascia)
Areolar tissue and superficial dorsal venous system
Buck's fascia (continuum of suspensory ligament of penis and perineal membrane)
Unpaired deep dorsal vein, two dorsal arteries, two dorsal penile nerves, spongy urethra
Tunica albuginea
Corpora cavernosa including deep penile arteries
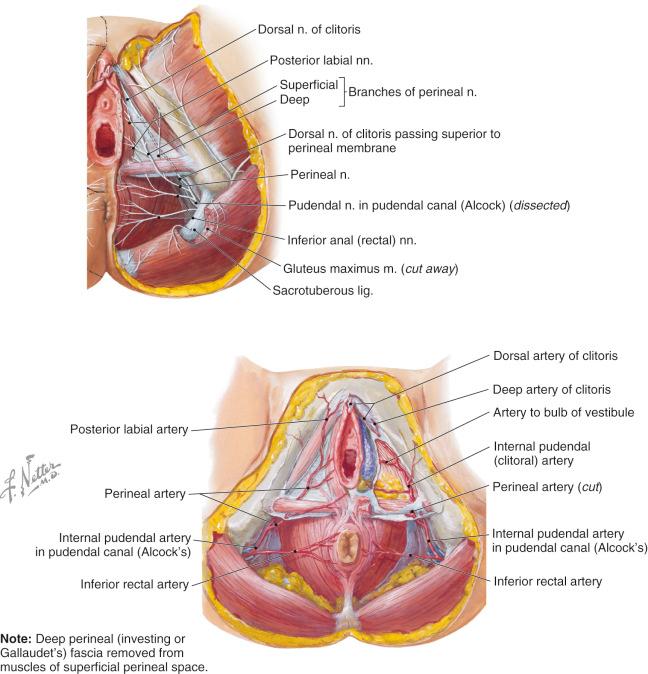
The vagina is an elastic muscular tube that measures 5–10 cm in length and 2 cm in width ( Fig. 19.3 ). It enters the perineum at an angle of inclination of around 30°. It is bordered anteriorly by the bladder and urethra and posteriorly by the rectum and it is attached laterally to the pelvic musculature. An abundance of autonomic innervation is responsible for secretions and involuntary contractions. Pain fibers are derived from the sacral plexus, and the tactile and erogenous sensation is provided via the deep perineal branch of the pudendal nerve. There is a rich vascular supply consisting of multiple arcades that are derived from the vaginal artery via the internal iliac system.
Perineal and urogenital defects can have a varying degree of functional, psychological, and aesthetic impairment. For example, a short penile stump may be sufficient for upright micturition but insufficient for sexual penetration. In the pediatric population, emotional, cognitive, and body image development may be affected by both the genital deformity and the reconstructive surgery. In addition, donor site morbidity and stigma can play a significant role in the choice of reconstructive options.
The preoperative evaluation for the reconstructive surgeon includes an assessment of the anatomic defect, evaluation of the functional and aesthetic impairment and availability of local or distant tissue used for the reconstruction. A thorough sexual history should be obtained and realistic expectations defined. A multidisciplinary approach, including the reconstructive surgeon, urologist, and gynecologist, can play an important role. It is also important to consider the psychological implications of a genital deformity prior to undertaking any complex reconstruction.
A preoperative urologic, gynecologic, and anorectal exam should be performed to determine the following:
Evaluate the defect and local anatomy
Determine baseline fecal continence
Assess the ejaculatory duct and amount of erectile tissue (particularly important in congenital cases)
Assess urinary tract and bladder function (i.e., presence of perineal urethrostomy)
Assess sensation and vascular supply to the region
For the purpose of this chapter, perineal and genital reconstruction will be divided into acquired and congenital deformities for both males and females:
Penis/scrotum
Acquired
Phallic inadequacy
Absent phallus
Hidden penis
Congenital
Micropenis
Aphallia
Ambiguous genitalia
Revision hypospadias repair
Epispadia
Bladder or cloacal exstrophy
Vagina/vulva
Acquired (trauma, oncologic resection)
Vaginal defect
Vulvar defect
Congenital
Vaginal agenesis
Mayer–Rokitansky–Küster–Hauser
Androgen insensitivity syndrome
Ambiguous genitalia
Bladder and cloacal exstrophy
Pelvic floor
Bladder and cloacal exstrophy
Acquired from trauma or tumor extirpation
Gender dysphoria
Please see flap chapters for exact anatomy and harvest of each respective flap. We focus on pertinent technical variations here.
Phallic inadequacy that is not amenable to standard, local penile lengthening procedures (i.e., penile stump advancement, suspensory ligament release, V-Y advancement, penoscrotal web excision) can be treated with reconstructive phalloplasty using local or distant flaps. The goals for penile reconstruction include:
The procedure should be reproducible.
The penile reconstruction should result in a neourethra that is capable of passing urine while standing in those patients who are continent.
The neophallus should have both tactile and erogenous sensation.
The reconstruction should provide enough bulk to accept a prosthetic for erection and sexual penetration.
The neophallus should be aesthetically acceptable to the patient.
Groin flap
Suprapubic flap
Anteromedial thigh flap
ALT
DIEP
Radial forearm free flap phalloplasty (RFFFP)
Free fibula
Latissimus dorsi (± innervation)
ALT
Gracilis myocutaneous flap
Chimeric ALT/sartorius flap
SCIA
Scapular
LAF/ELAF
Free tissue transfer with neurovascular coaptation to create a sensate flap
Glansplasty
Delayed or immediate urethroplasty
Glansplasty: no mention of timing.
Tattooing of phalloplasty and laser hair removal (if needed) before return of protective sensation (usually performed between 3 and 6 months)
Implantation of IPP after return of flap protective sensation – usually at 1 year
Scrotal plasty using local tissue or pedicled medial thigh flaps. The tissue can be augmented with scrotal silicone implants.
These key steps can be performed in a single reconstruction or staged depending on the surgeon's preference and the patient's needs. However, the safest and most predictable approaches that lead to the lowest rates of urethral stricture or fistula formation seem to be the following:
Initial operation: Phalloplasty including vascularized neourethra (either tube within a tube or a pedicled SCIP), including glans
At 12 months: IPP, scrotal plasty
or
Initial operation: phalloplasty including glans
At 3 months: creation of neourethral plate (FTGS or buccal graft)
At 6 months: 2nd stage of urethroplasty – tubularization
At 12 months: IPP; scrotal plasty
The options for recipient neurovascular structures ( Fig. 19.4 , see also Fig. 11.3 , 11.6 , and 11.8 ) for microvascular phalloplasty include:
End-to-side: femoral artery (with or without reversed saphenous interposition graft)
End-to-end: femoral side branches, inferior epigastric
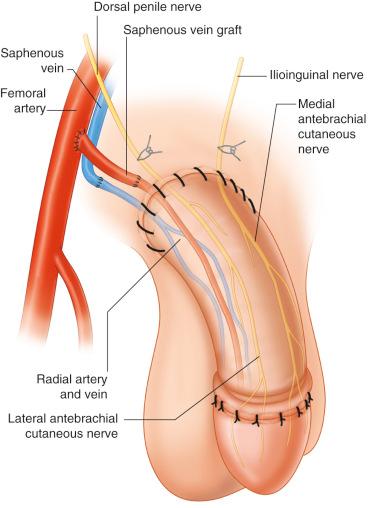
Recipient vein:
End-to-end: saphenous vein or inferior epigastric
Recipient nerves:
Erogenous (S4-Co): dorsal penile nerves (branch of pudendal nerve)
Dorsal nerve of the clitoris (in Gender affirming surgery)
Protective sensation: ilioinguinal, iliohypogastric, genito-femoral, (L1–L4) saphenous, femoral nerve
There is a variable amount of native donor tissue that can be used as part of the reconstruction. This is particularly true for phalloplasty in gender affirming surgery and congenital conditions, where the remnant glans or clitoris can be incorporated into the base of the neophallus. Similarly, preserving the clitoral hood or parts of the labia is useful for scrotal reconstruction. In the case of inadequate phallic length in both congenital and acquired conditions, it is prudent to preserve native corpora cavernosa that can be used to proximally secure the IPP or fibula.
The overall size and design of the phalloplasty is similar, regardless of the flap choice ( Figs 19.5 , 19.6 ). The surgeon may decide to incorporate a urethral section in the flap if immediate urethral reconstruction is desired.
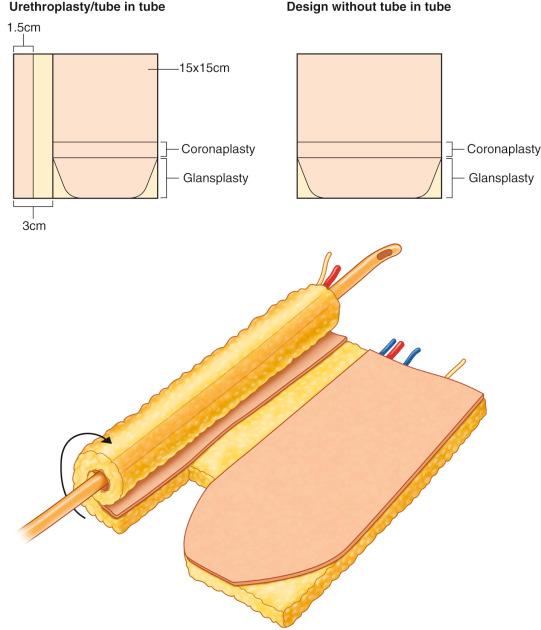
There are four accepted methods of urethroplasty that can be offered and discussed with the patient:
( Fig. 19.7 )
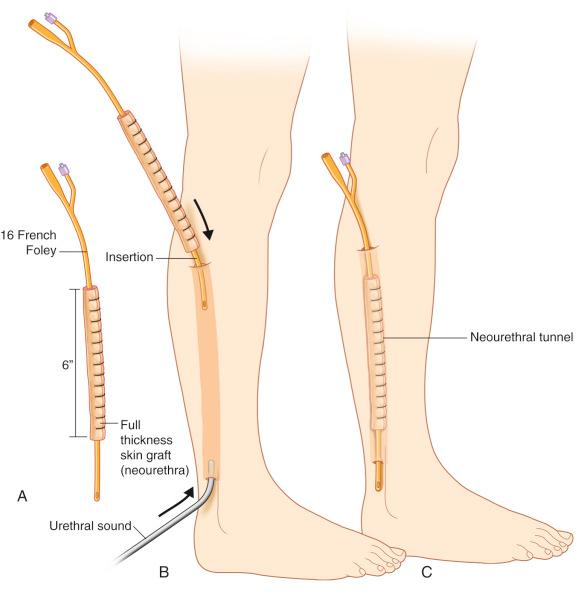
A full-thickness skin graft is wrapped around a Foley catheter and placed under the proposed flap 3 months prior to the phalloplasty. This method has been shown to provide a viable continuous urethra of sufficient length, as well as a trend to a lower incidence of urethral fistulas and better-controlled girth.
This is the most commonly described method, where a 1.5 cm wide longitudinal strip is de-epithelialized 3 cm to the lateral border of the flap. The lateral skin bridge is then rolled over a Foley catheter and sutured to itself. The main fasciocutaneous flap is rolled in the counter direction around the neourethra ( Fig. 19.5 ). For a radial forearm free flap phalloplasty, the ulnar side of the flap is generally chosen for the urethra, as it tends to contain less hair.
A thin SCIP flap or a free ulnar forearm flap can be used to create a vascularized neourethra in cases where a tube within a tube is not feasible. Most often this is combined with a pedicled ALT.
A full-thickness skin graft (groin, expandable donor site, labia minores) is wrapped around a Foley and inserted into the phalloplasty. In order to prevent downstream fistula formation, the tissue edges should overlap.
At 3 months following phalloplasty, the ventral surface is opened and a full-thickness skin graft or buccal mucosal graft is placed to re-create the urethral plate. At 3–6 months later, the healed graft is tubularized over a Foley catheter and the skin edges of the phallus are closed over the tubed graft. A sheet of acellular dermal matrix can be inserted as a layer of tissue that separates the two suture lines. This repair is similar to certain methods used for hypospadia repair.
Prelamination and delayed formation of a neourethral plate and subsequent tubularization seem to offer the best long-term result with the downside being multiple-staged surgeries.
When not performing a prelaminated or immediate urethroplasty in congenital cases of micropenis or penile insufficiency, the native glans/urethra can be preserved and placed coming out of the proximal, ventral surface of the free flap in case of flap failure. At a subsequent stage, the native urethra can be internalized for the urethroplasty and the glans can be de-epithelialized and inserted into the phallus. When reconstructing the urethra, a 12–14 French catheter is used in pediatric and exstrophy patients and a 16–18F in the adult population.
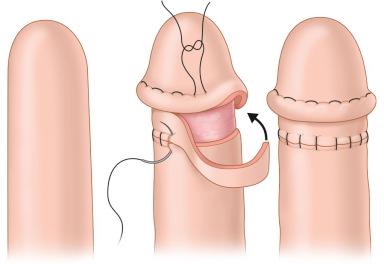
For a glansplasty ( Fig. 19.8 ), we prefer to create the neocoronal ridge before elevating the flap. To create a more natural appearance of the glans, the neocoronal ridge can be marked out in a boomerang shape but a plain circumferential incision is also acceptable. The incision is made about 3.5–4.0 cm from the distal tip of the neophallus. A proximally based 1 cm skin flap is elevated in the deep dermal plane circumferentially around the phallus. The thin skin flap is folded onto itself and secured with interrupted 5-0 polyglyconate suture to create the corona. The palmaris longus tendon or a thin strip of acellular dermal matrix can be inserted within the neocoronal ridge in an attempt to reduce subsequent flattening. The exposed deep dermal/subcutaneous donor site is then covered with a full-thickness skin graft. The skin graft usually heals, appearing like a scar, similar to that seen following circumcision. Over time, the neocoronal ridge can flatten. Further reconstruction of the ridge can be accomplished by inserting acellular dermal matrix or injectable fillers. The slightly darker color of the glans can be achieved with tattoo techniques. Reconstruction of foreskin is technically not feasible or has at least, not been described.
To reduce the risk of implant erosion or extrusion, insertion of the implantable penile prosthesis (IPP) is usually delayed 12 months for the return of sensation. For the same reason, it is prudent to account for sufficient distal soft tissue coverage at the time of phalloplasty. Various techniques are described to reduce the risk of erosion and extrusion, such as reinforcing the most distal area of the IPP prosthesis with fascia latae, acellular dermal matrix, or a polyethylene terephthalate sleeve. In congenital cases, the IPP should only be implanted in the late adolescence to early adulthood, as a high degree of maturity is needed to avoid complications.
The main complications of IPP are:
Mechanical dysfunction
Leakage of reservoir
Erosion and extrusion
Infection
Penile fibrosis/disfigurement
The normal male genitalia experiences exponential growth during puberty secondary to increased testosterone receptor density. A neophallus lacks these receptors and will only grow longitudinally with the child. Therefore, reconstruction is usually delayed until after puberty to ensure adequate size of the phalloplasty. This, however, has to be balanced against the potential downside of delayed psychosexual development.
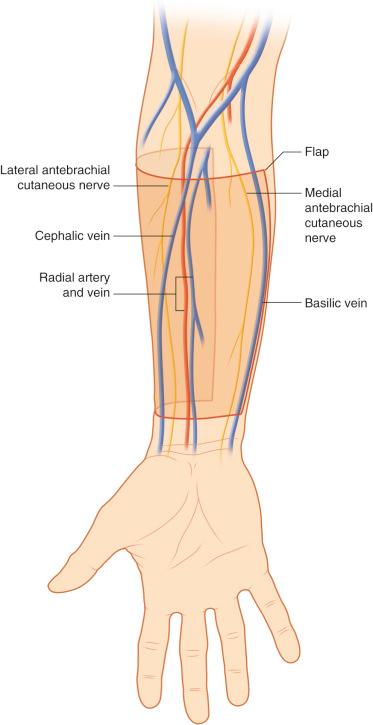
Since its first description by Chang et al. in 1984, the radial forearm free flap phalloplasty (RFFFP) has become the gold standard due to the tissue pliability, limited donor site morbidity, dual sensory innervation, and aesthetic outcome. Provided there is mixed radial–ulnar vascular supply, the flap should be harvested from the nondominant hand (see also Chapter 47 ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here