Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Acute aortic dissection is an uncommon but life-threatening emergency that requires prompt diagnosis, rapid triage, and immediate treatment. A unified effort across several international centers over the past 20 years has resulted in the establishment of a detailed, prospective registry that describes the major aspects of the presentation, management, and outcomes of patients with acute aortic dissection. This longitudinal experience has led to important clinical insights into an old disease and generated additional efforts to explore its genetics and pathobiology.
Despite advancements in diagnostic and treatment modalities for acute aortic dissection, in-hospital mortality rates remain significantly elevated at 20%, with little change over the past decade. Thus high-quality, prospective data to guide clinical management for acute aortic dissection are needed. In addition, enhanced awareness among practitioners regarding risk factors, presentation, diagnostic pathways, and treatment options for aortic dissection is a critical first step towards improving patient outcomes.
Published figures on the incidence of aortic dissection likely underestimate the actual occurrence rate because misdiagnosis is common and the percentage of acute aortic dissection patients who expire before hospital presentation is not known. Nonetheless, acute aortic dissection may, by conservative estimates, constitute 7% of all out-of-hospital cardiac arrests. However, overall, acute aortic dissection is a rare event, which, in turn, may be responsible partly for its underrecognition in clinical practice. For example, analysis of the Swedish National Cause of Death Register between 1987 and 2002 estimated the incidence of thoracic aortic aneurysm or dissection to be 16.3 per 100,000 men and 9.1 per 100,000 women. A population-based study in the United Kingdom reported an incidence of acute aortic dissection of 6 per 100,000 people per year among the general population. More recently, a prospective cohort of patients with an average age of 58 years was longitudinally followed for a median of 16 years in Sweden. The incidence of acute dissection was 15 per 100,000 patient years; others have reported acute aortic dissection affecting 30 per million individuals per year. Acute myocardial infarction is 140-fold more common than aortic dissection, even by conservative estimates. Data from the International Registry of Aortic Dissection (IRAD) support these epidemiological trends and show that the mean age of patients at presentation with aortic dissection is 62 years, with men accounting for 67% of cases.
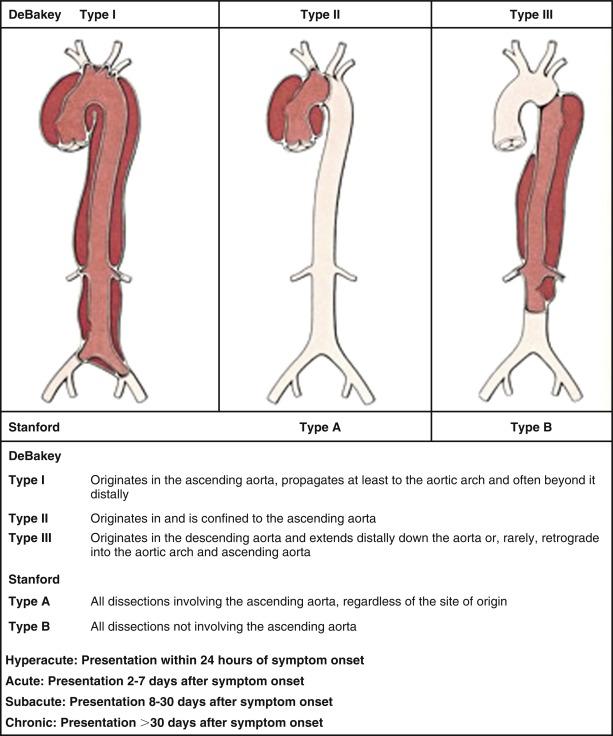
Classifying aortic dissection according to anatomic location and time from onset of symptoms helps to stratify risk and guide selection of initial treatment strategy ( Fig. 32.1 ). The Stanford classification system designates dissections that involve the aorta proximal to the brachiocephalic artery (i.e., root and ascending aorta) as type A and those that do not as type B. This distinction is clinically important because dissection involving the ascending aorta is a key determinate of early death and major morbidity. The location of the intimal tear does not influence Stanford dissection type. In the older DeBakey classification, a type I dissection originates within the ascending aorta and extends for a variable distance beyond the take-off of the innominate artery. A DeBakey type II dissection is confined to the ascending aorta, whereas a type III dissection originates in the descending thoracic aorta beyond the origin of the left subclavian artery and either terminates above (type IIIA) or extends below (type IIIB) the level of the diaphragm. Although there is no single universally accepted classification system, the Stanford classification scheme is most often used currently in practice and will be used throughout this chapter. The terms “communicating” and “noncommunicating” refer to the presence or absence, respectively, of blood flow between the true and false lumens of the aorta.
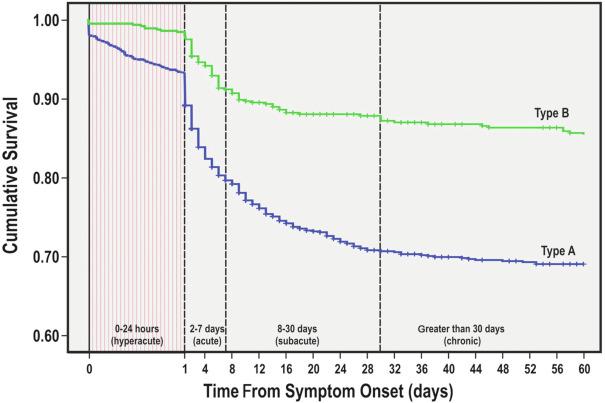
Traditionally, aortic dissection was considered acute or chronic if presentation occurs within or after 14 days, respectively, of symptom onset. However, adverse remodeling of the aorta is evident 2 to 12 weeks following presentation, which is also a time frame associated with increased risk of complications and mortality ( Fig. 32.2 ). For this reason, a European interdisciplinary consensus group has proposed a subacute phase, defined as 2 to 6 weeks after presentation. Based on data from IRAD, postaortic dissection chronology was refined further as follows: hyperacute (< 24 hours), acute (2 to 7 days), subacute (8 to 30 days), and chronic (> 30 days).
In clinical practice, aortic dissection is diagnosed by imaging data that demonstrate an intimal flap with separation of true and false lumens. In type A dissection, the true lumen is usually displaced along the inner curvature of the aortic arch and continues caudally along the medial aspect of the descending thoracic aorta. Aortic branch vessel blood flow may derive from either the true or false lumen. Alternatively, flow may be sluggish or absent within the false lumen, or branch vessels may be completely occluded at or near their origins.
Forces that weaken the medial layer of the aorta increase the probability of dilation, aneurysm formation, and dissection ( Box 32.1 ). Acquired and genetic diseases that mediate this process are discussed next. In classic aortic dissection, the initiating event is an intimal tear through which blood rapidly surges into the media under systolic pressure, splitting the layers of the aortic wall and creating an intimal flap that separates the true from the false lumen. The dissecting hematoma most commonly propagates distally (anterograde), although proximal (retrograde) or bidirectional migration is also observed.
Marfan syndrome
Ehlers-Danlos syndrome
Familial thoracic aortic aneurysm disease
Bicuspid aortic valve disease
Aberrant right subclavian artery
Aortic coarctation
Noonan syndrome
Turner syndrome
Polycystic kidney disease
Loeys-Dietz syndrome
Hypertension
Iatrogenic
Pregnancy
Inflammatory aortitis
Cocaine, chronic amphetamine use
Contemporary imaging modalities or autopsy findings identify the primary entry tear in approximately 90% of cases. The entry tear is most frequently located a few centimeters above the level of the aortic valve along the greater curvature of the aorta in cases of type A dissection and accounts for nearly 60% of all cases. Compared with other locations in the ascending aorta, the proximal few centimeters of the greater curvature are exposed to relatively greater hemodynamic, shear, and torsional forces. A pivot region located in the descending thoracic aorta just beyond the insertion of the ligamentum arteriosum, where the relatively mobile arch meets the fixed descending thoracic aorta, is the second most common entry site for intimal tears, which will then propagate as a type B dissection (30% of cases). Arch entry occurs in 7% of cases. The abdominal aorta is the least common site for entry (3% of cases) despite the high prevalence of intimal-medial ulcers in patients with atherosclerotic disease in this segment.
The dissecting hematoma usually propagates in an anterograde direction, although retrograde extension can occur. By this mechanism, as many as 20% of dissections that originate in the distal arch or descending thoracic aorta may involve the ascending aorta through retrograde extension. Such dissections appear to have a comparable 5-year survival rate to those type B dissection without retrograde extension. In rare cases, a second tear may occur, resulting in a three-channel dissection.
Blood within the false lumen may reenter the true lumen anywhere along the length of the dissection. Reentry may be protective because of spontaneous decompression of the false lumen, which, in turn, may reduce the risks of rupture and/or the development of malperfusion syndromes.
Aortic rupture, defined as a tearing in the vessel wall that results in extravascular hemorrhage, most commonly occurs with trauma and aortic transection but may occur secondary to dissection. Rupture into the pericardial space resulting in cardiac tamponade may occur in type A dissection, whereas rupture into the left pleural space can be encountered with type B dissection. Dissection-mediated end-organ ischemia or infarction occurs from (i) mechanical compression of aortic branch vessels by false lumen hematoma, (ii) extension of the dissection plane across the ostium of the branch vessel, or (iii) dynamic vessel inlet obstruction caused by an oscillating intimal flap. Compromise of the coronary, brachiocephalic, mesenteric, renal, spinal, and iliac circulations can occur and result in a myriad of clinical presentations. Occlusion of the left ventricular (LV) outflow tract by an intimal flap has also been reported.
Thrombosis of blood within the false lumen may seal the entry tear, thus eliminating communication with the true lumen and preventing false lumen expansion. However, partial thrombosis of the false lumen has been identified as a risk factor for long-term death in patients with type B dissection. Elevation of pressure within the false lumen with partial thrombosis may lead to further extrinsic compression of the true lumen and impairment of blood flow to critical organs. Alternatively, it has been proposed that partial thrombosis of the false lumen is associated with worse clinical outcomes via the promotion of vascular inflammation, tissue hypoxia, and/or neovascularization with weakening of adjacent vascular structures and an increased risk for aortic rupture.
Persistent patency of the false lumen is also associated with a higher risk of long-term complications, such as late rupture or false aneurysm formation requiring operative intervention. Although native aortic aneurysm disease is often a risk factor for dissection, a dissection need not result in aneurysm formation. The term “dissecting aneurysm” is an inaccurate anachronism because these diseases are not synonymous, a distinction that is particularly important when considering their natural histories and treatments.
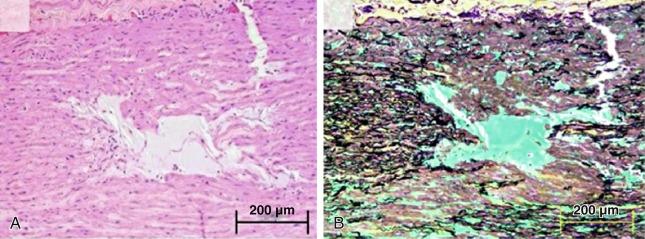
As is true for aneurysms, any process that leads to the destruction or degeneration of the major supporting elements of the aortic media (elastin, collagen, smooth muscle cells) can predispose to the development of dissection. The histopathologic term medial degeneration refers to the noninflammatory destruction or fragmentation of elastic lamellar units, dropout of vascular smooth muscle cells (VSMCs), and accumulation of mucopolysaccharide ground substances (not always in distinct cystic spaces), which characterize the final common pathway for a variety of processes that affect the integrity of the aortic media ( Fig. 32.3 ). Although such changes predispose to aneurysm development, dissection may occur prior to significant aneurysm enlargement. In the GenTac registry of patients with a genetically associated thoracic aortic aneurysm, only 4 of the 31 patients (13%) with acute aortic dissection met criteria for intervention on predissection imaging.
Marfan syndrome (MFS) is the most common inherited connective tissue disorder, with an estimated prevalence of 1 case per 3000 to 5000 individuals irrespective of racial, ethnic, or geographic considerations. If untreated, aortic disease in MFS is progressive and incompatible with normal longevity. Of patients with aortic dissection younger than 40 years of age, half have a history of MFS. However, medial degeneration is not pathognomonic of MFS and may be present in numerous other conditions.
The clinical spectrum of MFS is heterogenous, although the presence of an aortic root aneurysm (aortic diameter z score ≥ 2) and ectopia lentis is sufficient to make the diagnosis even in the absence of a family history. Conversely, in the absence of either of these two clinical features, the current Ghent nosology for MFS provides a scoring system based on the presence of an FBN1 mutation and/or other key systemic features of MFS that can be used to make the diagnosis ( Boxes 32.2 and 32.3 ). Systemic features that are suggestive of MFS include: positive wrist sign (i.e., entire distal phalanx of the adducted thumb extends beyond the ulnar border of the palm); positive thumb sign (i.e., the tip of the thumb covers the entire fingernail of the fifth finger when wrapped around the contralateral wrist); pectus excavatum; pneumothorax; dural ectasia; hindfoot deformity; and protrusio acetabuli. Other clinical features less strongly associated with MFS include mitral valve prolapse, various abnormal facial features, and thoracolumbar kyphosis.
Ao ( z ≥ 2) and EL = MFS a
Ao ( z ≥ 2) and FBN1 = MFS
Ao ( z ≥ 2) and Syst (≥ 7pts) = MFS a
EL and FBN1 with known Ao = MFS
EL with or without Syst and with an FBN1 not known with Ao or no FBN1 = ELS
Ao ( z < 2) and Syst (≥ 5 with at least one skeletal feature) without EL = MASS
MVP and Ao ( z < 2) and Syst (< 5) without EL = MVPS
EL and FH of MFS (as defined above) = MFS
Syst (≥ 7 pts) and FH of MFS (as defined above) = MFS a
Ao ( z ≥ 2 above 20 years old, ≥ 3 below 20 years old) + FH of MFS (as defined above) = MFS a
a Caveat: without discriminating features of Shprintzen-Goldberg syndrome, Loeys-Dietz syndrome, or vascular Ehlers-Danlos syndrome and after TGFBA1/2, collagen biochemistry, COL3A1 testing if indicated. Other conditions/genes will emerge with time.
In general, MFS is diagnosed in the presence of aortic root dilation/dissection and ectopia lentis; aortic root dilation/dissection plus FBN1 mutation; aortic root dilation plus sufficient systemic findings ( Box 32.3 , ≥ 7 points); ectopia lentis plus a FBN1 mutation previously associated with aortic disease; or, in an individual with a positive family history of MFS, the diagnosis is made in the presence of ectopia lentis, or a systemic score ≥ 7 points or aortic root dilation.
Ao , Aortic diameter at the sinuses of Valsalva above indicated z score or aortic root dissection; EL , ectopia lentis; ELS , ectopia lentis syndrome; FBN1 , fibrillin-1 mutation; FBN1 not known with Ao , FBN1 mutation that has not previously been associated with aortic root aneurysm/dissection; FBN1 with known Ao , FBN1 mutation that has been identified in an individual with aortic aneurysm; MASS , myopia, mitral valve prolapse, borderline ( z < 2) aortic root dilation, striae, skeletal findings phenotype; MFS , Marfan syndrome; MVPS , mitral valve prolapse syndrome; Syst , systemic score (see Box 32.3 ); z , z score.
Wrist and thumb sign: 3 (wrist or thumb sign: 1)
Pectus carinatum deformity: 2 (pectus excavatum or chest asymmetry: 1)
Hindfoot deformity: 2 (plain pes planus: 1)
Pneumothorax: 2
Dural ectasia: 2
Protrusio acetabuli: 2
Reduced US/LS and increased arm/height and no severe scoliosis: 1
Scoliosis or thoracolumbar kyphosis: 1
Reduced elbow extension: 1
Facial features (3/5): 1 (dolichocephaly, enophthalmos, downslanting palpebral fissures, malar hypoplasia, retrognathia)
Skin striae: 1
Myopia > 3 diopters: 1
Mitral valve prolapse (all types): 1
Maximum total: 20 points; ≥ 7 indicates systemic involvement.
US/LS , Upper segment/lower segment ratio.
From Loeys BL, Dietz HC, Braverman AC, et al. The revised Ghent nosology for the Marfan syndrome. J Med Genet. 2010;47:476–485.
Limited forms (i.e., forme frustes) of MFS that feature cardiovascular manifestations include mitral valve prolapse syndrome (MVPS) (mitral valve prolapse, pectus excavatum, scoliosis, and mild arachnodactyly) and the MASS phenotype (myopia, mitral valve prolapse, borderline and nonprogressive aortic root dilation, skeletal findings and striae). There is increasing awareness of familial thoracic aortic aneurysm and dissection (FTAAD), although candidate genes affecting both matrix and smooth muscle cell components have been identified in only 20% of such patients. Vascular-type Ehlers-Danlos syndrome (EDS) is associated with arterial rupture and dissection, including of the aorta.
MFS is a monogenic disorder caused by a variety of mutations in the FBN1 gene, which encodes fibrillin-1, an extracellular matrix protein that is associated with elastic fibers. It was initially believed that loss of function of the FBN1 gene directly affected the structural integrity of the extracellular matrix. Additional investigations have demonstrated that abnormal cell-signaling plays an important role. Using an in vivo murine model of MFS, Dietz and colleagues demonstrated that fibrillin-1 is a key target for binding of latent transforming growth factor (TGF)-β by cytokines that acts as a “master-switch” regulator of inflammatory, profibrotic, and metalloproteinase signaling pathways in vascular endothelial cells and VSMCs. Thus deficiency in fibrillin-1 leads to reduced TGF-β binding and the potential for enhanced activation of numerous cell-signaling pathways with alteration of the extracellular matrix.
Angiotensin I (AT-1)-mediated TGF-β activation has been studied as a pharmacologic target to inhibit or reverse aortic dilation in MFS. Habashi and colleagues studied the effects of losartan, an AT-1 receptor antagonist, on aortic aneurysm formation in transgenic mice encoding a cysteine to glutamine substitution at position 1039 in the FBN-1 gene (Fbn1 C1039G/- ). Mice expressing this particular fibrillin-1 mutation, the most common mutation with MFS, demonstrate significant and progressive aortic root dilation compared with control mice. Treatment with losartan attenuated TGF-β signaling in the aortic wall and resulted in full normalization of aortic wall thickness, marked by improvement in aortic wall architecture (i.e., a decrease in elastin fiber disruption), compared with treatment with placebo or with the β-adrenergic receptor antagonist, propranolol.
In one small clinical trial of young MFS patients, losartan decreased the rate of aortic root growth significantly (mean change 0.46 ± 0.62 mm/year vs. 3.5 ± 2.8 mm/year prior to treatment, P < .001). These findings were supported by the results of a randomized trial of losartan versus placebo in 116 adults with MFS, which demonstrated a decreased rate of aortic root dilatation in the losartan group, including those with prior aortic surgery. Notably, more than 70% of patients in both groups were also using β-adrenergic receptor antagonist therapy, raising the possibility that these two complimentary therapies may be synergistically effective. Indeed, one small study of 28 patients with MFS found losartan added to a β-adrenergic receptor antagonist was associated with a slower rate of dilatation compared with the use of a β-adrenergic receptor antagonist alone. The comparative efficacy of losartan versus atenolol was assessed in a larger randomized trial of 608 children and young adults with MFS. Contrary to expectations, neither the 3-year primary end points (rate of change in aortic root z scores) nor secondary end points of the trial differed between treatment groups. Both strategies resulted in a decrease in the rate of aortic root growth over time. In clinical practice, and in the absence of more effective therapies, MFS patients are frequently placed on maximally tolerated doses of both an angiotensin II type 1 receptor antagonist and a β-adrenergic receptor antagonist.
MFS is associated with a sixfold increased risk of dissection compared with the other monogenic causes of thoracic aneurysms. In a Dutch review of 600 patients with MFS, risk factors for type B dissection included prior prophylactic aortic aneurysm surgery and proximal descending aortic diameter ≥ 27 mm. In this nonrandomized study, use of angiotensin II receptor blockade therapy was associated with a decrease in type B dissection of (HR 0.3; 95% CI: 0.1 to 0.9; P = .03) compared with patients not receiving angiotensin II receptor blockade.
The Loeys-Dietz syndrome (LDS) is a related disorder caused by mutations in TGFBR1 and TGFBR2 , which regulate signal transduction of the TGF-β ligand. Phenotypical features of LDS include chest wall deformity, high-arched palate, bifid uvula, and hypertelorism. LDS is a diffuse arteriopathy that can cause tortuosity, dissection, and aneurysm disease of the peripheral arteries and aorta, and is a particularly aggressive form of arteriopathy with a mean age of death of 26 years. It is recommended that aortic repair be considered at an internal diameter of ≥ 4.2 cm measured by echocardiography or ≥ 4.4 cm measured by computed tomography or magnetic resonance imaging (MRI) (external diameter), because dissection in these patients frequently occurs at a diameter < 5.0 cm.
The EDS (1:5000 births) comprises a heterogeneous group of disorders characterized clinically by hypermobile joints, hyperextensile skin, and tissue fragility. Vascular-type EDS (type IV) is associated with spontaneous vascular rupture and dissection. Aortic involvement occurs in EDS type IV, an autosomal dominant disorder attributed to structural defects in the pro-α1 (III) chain of type III collagen, encoded by the COL3A1 gene on chromosome 2q31. Associated clinical features of EDS type IV include thin skin, easy bruising, and visceral and uterine rupture. Median survival is 48 years, and the most common cause of death is arterial dissection or rupture.
The FTAAD phenotype is specific for aortic pathology and does not involve extra-aortic disease. FTAAD has been mapped to several genetic loci, including 16p13.11 ( MYH11 gene), 5q13-14, and 11q23.2-q24, which are not associated with abnormalities of fibrillin or collagen. More than five mutations in the FBN-1 gene have been identified in patients with familial or spontaneous thoracic aortic aneurysm and dissection, with histopathologic changes characteristic of medial degeneration, yet with no demonstrable abnormalities of collagen or fibrillin in fibroblast culture.
Polymorphisms encoding vitamin K epoxide reductase complex subunit 1 (VKORC1) result in undercarboxylation of specific matrix proteins and are associated with calcification of the arterial wall. In patients carrying the C allele (CT or CC), a relative twofold increase in the probability of developing aortic dissection has been observed. A missense mutation in the ACTA2 gene that encodes for actin filaments in VSMCs is linked to 14% of patients with FTAAD. As a consequence of this mutation, intracellular actin filament assembly is disrupted, promoting focal areas of VSMC disarray, decreased VSMC contraction, and medial degeneration of the aorta. This phenotype is believed to be associated with weakening of the aortic wall and increased predisposition to dissection.
More recently, defects in the MYLK and PRKG1 genes have been implicated in the development of thoracic aortic aneurysm via disruption of normal actin-myosin interaction. MYLK encodes myosin light chain kinase, and PRKG1 disinhibits activity of the type 1 cGMP-dependent protein kinase, which is an inhibitor of myosin light chain phosphatase and thus a negative regulator of actin-myosin physical associations.
Bicuspid aortic valve (BAV) disease is the most common congenital cardiac anomaly in adults (4:1000 live births) and is often accompanied by an aortopathy that is histologically similar to but less severe than that observed in patients with MFS. Dilatation of the root and more commonly of the ascending aorta is present in up to 40% of patients with BAV disease and is a risk factor for dissection or rupture. It appears to be inherited in an autosomal dominant pattern with incomplete penetrance. Pathogenesis of aortic dissection in BAV patients is possibly due to interplay between hemodynamic and histologic processes that weaken the aortic wall. In particular, some reports indicate that BAV patients have a thinner intima and decreased smooth muscle actin expression.
Although BAV is associated with aortic aneurysm and dissection, recent guidelines have increased the recommended size threshold for prophylactic surgical intervention from 5.0 to 5.5 cm in the absence of a family history of acute aortic syndrome, an annual increase in size of ≥ 0.5 cm, or the need for surgical aortic valve replacement for severe valve disease. This is the same size criterion applied to patients with ascending aortic aneurysm disease without BAV. Single-center observational data of patients with acute dissection have demonstrated that BAV patients have larger aortic diameters at presentation than patients with trileaflet aortic valves.
Similar histopathological changes have been described in patients with aortic coarctation (many of whom have BAV disease), Noonan syndrome, Turner syndrome, and polycystic kidney disease.
There are numerous acquired disorders that contribute to aortic dissection. The incidence of aortic dissection increases with age (27 per 100,000 for 65 to 75 years old and 35 per 100,000 for ≥ 75 years old). Dissection is also more common in men; women tend to be older with more atypical symptoms, delayed diagnosis, and an overall higher mortality (30% vs. 21%). Systemic hypertension is the most common modifiable risk factor for aortic dissection and is present in approximately 75% of patients. Hypertension accelerates the normal aging process and leads to intimal thickening, VSMC apoptosis, vascular fibrosis, loss of elasticity, and compromise of nutritive blood supply (see Chapter 3 ). Decreased aortic compliance and vulnerability to pulsatile forces predispose to injury and create a substrate for dissection. In one report, ambulatory blood pressure was higher in those who did not survive to hospital admission with aortic dissection. However, aortic diameter is the strongest predictor of dissection in patients without an underlying genetic predisposing disorder.
In the IRAD registry, iatrogenic aortic dissection after cardiac surgery or catheterization accounted for 5% of the total reported, although the association of dissection with prior cardiac surgery may be even more frequent, because 16% of those in the registry had a history of cardiac surgery. Older age, hypertension, and severe peripheral vascular disease are risk factors associated with procedure-related dissection. Pain may be absent in iatrogenic dissection. In one review of three European hospitals, aortic dissection complicated 0.06% of the 108,083 cardiac catheterizations. Short- and long-term outcomes following iatrogenic dissection were good, with stent implantation when a coronary artery was involved and conservative therapy in the majority of the remainder. Need for cardiac surgery was rare, with an overall low mortality and long-term complication rate. Retrograde dissections created at the time of catheterization usually seal spontaneously on withdrawal of the catheter. Aortic atherosclerotic plaques may prevent the longitudinal propagation of a dissection. Dissections arising from sites where the aorta has been incised or cross-clamped may occur intraoperatively or at any time following surgery.
Deceleration injury from high-speed accidents results in aortic transection with false aneurysm formation and rupture, most commonly in the region of the aortic isthmus just beyond the origin of the left subclavian artery. Transection results in a transmural tear that is different both pathologically and etiologically from aortic dissection.
Risk of aortic dissection is fourfold higher during pregnancy but is a rare complication with an estimated incidence of 5.5 per 1 million. However, by some estimates, 50% of all dissections in women younger than the age of 40 occur during labor, delivery, or early following birth. Histopathologic changes affecting the aortic media of pregnant women have been described, including alterations in elastic fibers and VSMCs. Both estrogen and relaxin are associated with alterations in matrix metalloproteinase (MMP) homeostasis and contribute to vascular remodeling and a susceptibility to injury, independent of the hemodynamic stress of labor and delivery. In many cases, pregnancy “unmasks” primary conditions that predispose to aortic dissection (e.g., MFS). In those patients with preexisting MFS or BAV disease, aortic root size > 4.0 cm is a contraindication to pregnancy due to the increased risk for spontaneous rupture or dissection.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here