Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Abnormal hemostasis leads to thrombus formation. Thrombosis in either the arterial or the venous system is a leading cause of significant morbidity and mortality. When thrombosis occurs in the arterial system, myocardial infarction and stroke may occur, whereas thrombosis in the venous system results in venous thromboembolic disease. Thrombosis and thrombotic-related events are among the most common causes of mortality in the Western world. It is estimated that 695,000 people had new thrombotic events within the coronary circulation in 2017 in the United States and that more than 325,000 people had recurrent events; an additional 165,000 silent myocardial infarctions are estimated to occur annually. Stroke also accounts for significant morbidity, with 795,000 people per year suffering from a thrombotic event within the cerebral circulation. There are 300,000 to 600,000 new cases of venous thromboembolism each year, approximately 30% of which result in death in the first 30 days after diagnosis, the majority of deaths being sudden in the setting of a pulmonary embolism. In fact, thromboembolism is the cause of one in four deaths worldwide, and, although mortality rates have declined, the cases worldwide continue to increase. This is largely due to the fact that the population is aging, because the majority of events occur in the elderly.
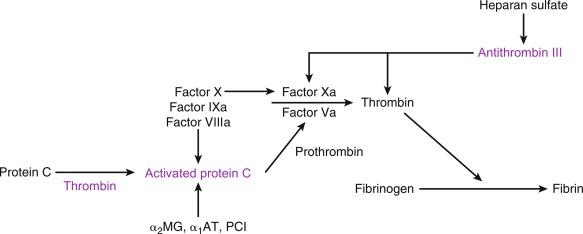
The pathogenesis of thrombosis was elucidated as early as 1856 when Virchow first described its major determinants, including abnormalities in the vessel wall, platelets, and coagulation proteins as essential for establishing a thrombus. The composition of arterial thrombi is distinct from that of thrombi that form in the venous circulation: arterial thrombi are composed mainly of platelets and occur in areas of vascular wall injury; by contrast, venous thrombi are rich in fibrin and dependent on a hypercoagulable response associated with individual coagulation factor abnormalities or mechanical issues related to blood flow limitation. Under normal circumstances, the endothelial lining is not a thrombotic surface with endothelial cells constantly interacting with other cell types, including platelets to directly inhibit thrombus formation through the release of antithrombotic factors, such as thrombomodulin, tissue factor pathway inhibitor system, plasmin, and antithrombin systems. At the same time, platelet aggregation is inhibited through prostacyclins and nitric oxide (NO), which are released from platelets directly. However, when the endothelial surface becomes damaged, release of many procoagulant proteins, especially tissue factor, and activation of platelets result in uncontrolled hemostasis at the site of vascular injury. As the thrombus begins to form, it recruits additional platelets to the area, leading to further platelet activation. Initially tethering of platelets is dependent on exposure of glycoprotein Ib-V-IX in damaged collagen, which binds to von Willebrand factor, resulting in adhesion of platelets to the area of injury. Further recruitment of platelets is mediated through activation of the glycoprotein IIb/IIIa (GPIIb/IIIa) platelet receptor, which undergoes a conformational change leading to increased affinity for fibrinogen. These events culminate with further platelet activation, which results in the release of many essential components for thrombus formation, including adenosine diphosphate (ADP), serotonin, and thromboxane A 2 (TXA 2 ). Exposure of vascular collagen also leads to activation of the normal mechanisms of hemostasis, including the coagulation cascade, through exposure of tissue factor leading to “hemostasis in the wrong place.” The coagulation regulatory system is outlined in Fig. 8.1 and is discussed in Chapter 4 . Briefly, both the tissue factor–mediated pathway (extrinsic) and the contact-mediated pathway (intrinsic) rely on activation of inactive enzyme precursors of serine proteases, which then lead to activation of another downstream protein within the cascade. The ultimate step (factor XIII activation) results in cross-linking of fibrin to stabilize a platelet plug and evolving thrombus. The tissue factor–initiated pathway is essential for thrombus formation. When tissue factor is released during cellular injury, factor VII is activated and complexes. This complex next activates factor X and factor XI. Activation of factor X is essential for conversion of prothrombin (factor II) to thrombin through the prothrombinase complex assembled on activated platelets. This cascade of coagulation proteins is essential for hemostasis but also can have deleterious effects when it occurs in an unregulated fashion, leading to unwanted thrombotic complications.
Walton and colleagues have established that erythrocytes also play a significant role in thrombus formation. The authors demonstrated that erythrocytes can affect thrombus formation by increasing platelet deposition at the site of injury. This finding was especially notable in cases where the hematocrit was elevated, demonstrating that elevations in hematocrit can have a negative impact on overall thrombus risk.
It is estimated that 100,000 to 200,000 deaths occur yearly because of a venous thromboembolic event (VTE). These events occur mainly in the vasculature at the area of the vessel sinus where stasis can lead to a hypercoagulable microenvironment. The hemostatic process is activated when tissue factor is exposed at the site of vascular injury, leading to initiation of the coagulation cascade with subsequent formation of thrombin and conversion of fibrinogen to fibrin. This process evolves at the same time that platelets are actively being recruited to the area of injury through collagen exposure, leading to platelet and fibrin thrombus formation. There are a number of physiological anticoagulants that are also present and modulate this response, including antithrombin, tissue factor pathway inhibitor, and activated protein C and its cofactor protein S. Defects in these hemostatic proteins can lead to disorders that elevate the risk of thrombus formation.
The risk factors for venous thromboembolism are associated with venous stasis or acquired and congenital hypercoagulable states and include obesity, smoking, malignancy, pregnancy, hormone therapy, and recent trauma or surgery. Immobilization resulting from prolonged hospitalization following surgical intervention and during long-distance air-travel also contributes to the risk of VTE. Genetic risk factors that are associated with increased risk of VTE include mutations in factor V (Leiden) and prothrombin 20210, as well as mutations leading to deficiencies in antithrombin, protein C, and protein S. Approximately 5% of the Caucasian population has at least one mutation for factor V Leiden, and 15% to 20% of patients who present with a VTE carry the mutation. Approximately 2% of the population carry the prothrombin gene mutation, but it may be present in approximately 5% to 15% of persons with VTE. The population frequencies of mutations in other coagulation factor genes, such as protein C, are estimated to be 1 in 500 individuals. Antithrombin III deficiency has a frequency of 1 in 300 in the general population and 3% to 5% among those with thrombotic events. Previously it was thought that genetic mutations in the genes important for methylene tetrahydrofolate reductase and hyperhomocysteinemia increased the risk of VTEs; however, recently this association has been shown to be less likely. One of the acquired risk factors known to be important in both venous and arterial thrombosis is the acquisition of antiphospholipid antibodies, which represent a family of antibodies against phospholipids, such as cardiolipins, and phospholipid binding proteins, such as beta 2 glycoprotein I. The mechanisms responsible for thrombosis are still based on speculation but may include inhibition of protein C, antithrombin, and annexin A5 expression; binding and activation of platelets; enhanced endothelial cells tissue factor expression; activation of the complement cascade; and impaired assembly of fibrinolytic proteins on the endothelial cell surface owing to antibody-mediated impairment of annexin A2 availability for tissue plasminogen activator and plasminogen binding to the endothelium. The criteria for the diagnosis of the associated disorder, antiphospholipid syndrome, include the presence of both clinical events and laboratory evidence for the presence of antiphospholipid antibodies.
Because it is clear that other genes could also be responsible for increased risk of VTE, a number of studies have been performed using genome-wide association studies (GWAS) to identify new genetic loci. Recently, Germain and colleagues published the results of a meta-analysis of 12 GWAS compiling more than 7500 cases of VTE. The results identified a number of new potential loci for genetics-associated VTE, including some genes which were previously not understood to be associated with thrombosis, thereby setting the stage for a new understanding of the pathobiology and the risk of VTE. Although it is clear that there are strong genetic links that increase the risk of VTE, the utility of uncovering these genetic predispositions often does not affect clinical therapeutic strategies, making discovery of these underlying genetic disorders less appealing. In fact, the overuse of these screening tests in patients with provoked events is greatly discouraged by the Choosing Wise Campaign of the American Society of Hematology, as well as the British Committee. Most physicians currently do not pursue a hypercoagulable work-up in those individuals who present with a provoked VTE. However, these work-ups are still used in those with spontaneous events, as well is individuals whose family members would benefit from the knowledge of an increased propensity for thrombosis.
Recently, a number of new drugs have been approved for the treatment of VTEs. In the past the main stays of treatment included mainly heparin products and the vitamin K antagonist warfarin. Owing to issues with frequent blood testing to maintain therapeutic ranges, food and drug interactions, and fluctuations in drug levels, new oral agents were developed which provided a steady state of drug delivery without concerns for drug interactions and no need for frequent monitoring. These drugs target factor Xa and thrombin in the coagulation cascade and are termed direct oral anticoagulants (DOACs). These drugs have been especially effective in providing a therapeutic option for long-term therapy for prevention of venous thrombosis recurrence in unprovoked clots. These DOACs include a direct thrombin inhibitor, dabigatran etexilate, as well as three factor Xa inhibitors, rivaroxaban, apixiban, and edoxaban. All these drugs have a rapid onset of action, hepatic metabolism, and renal clearance. Studies using these DOACs in comparison with therapeutic warfarin demonstrated noninferiority for the treatment of an acute deep vein thrombosis or pulmonary embolism. Although there was no difference in efficacy for treatment, the bleeding profile of the DOACs was better than that of warfarin, with lower risk of significant bleeding. Briefly, RECOVER I and II, which included more than 5000 patients, were randomized double-blinded control trials comparing the use of dabigatran to warfarin for VTE. The pooled analysis of these two trials demonstrated similar rates of recurrent VTE but fewer episodes of clinically relevant bleeding. Similarly the factor Xa inhibitors demonstrated noninferiority in terms of VTE, with less significant bleeding. At this time, it is unclear if there is one DOAC that offers improved VTE prevention in comparison with the others. Recently, studies have demonstrated the efficacy of this novel anticoagulant in patients with malignancy, which often leads to a hypercoagulable state, making this patient population more vulnerable to thrombosis. Edoxaban was demonstrated to be noninferior to the standard of care (low-molecular-weight heparin) with respect to risk of recurrent venous thromboembolism or major bleeding. However, there are differences that make some more well utilized than others. Dabigatran and apixaban are taken twice daily, whereas edoxaban and rivaroxaban are once-daily medications. Both dabigatran and edoxaban require bridging therapy with a parenteral anticoagulant during the first 5 to 10 days prior to primary therapy, whereas apixaban and rivaroxaban can be used as initial therapy. Perhaps the greatest difference at the time of writing this chapter is that dabigatran is the only agent with a known reversal agent (idarucizumab). Of note, the utility of all these drugs in patients with obesity, cancer, hepatic cirrhosis, antiphospholipid syndrome, and other special clinical circumstances has yet to be established, making these drugs less likely to be used in those situations.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here