Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Parasitic infections of the nervous system range from acute syndromes such as diffuse cerebritis in cerebral malaria to more chronic mass lesions causing seizure disorders such as neurocysticercosis. This chapter will focus on the most common parasites causing central nervous system (CNS) infections.
A 45-year-old previously healthy Indian male working as an engineer in the United States returned from India in August after a 6-week stay visiting with his parents. One week after his return, he presented to the emergency ward with 4 days of fever to 102°F, headache, and diarrhea. His face was flushed; he had mild confusion, severe lethargy, a moderately stiff neck, and a temperature of 39.4°C (103°F). A lumbar puncture demonstrated a normal cerebrospinal fluid (CSF). His peripheral white blood cell (WBC) was 12,000/mm 3 , with a hemoglobin of 10 g and a platelet count of 40,000/mm 3 . His blood glucose level was 56 mg/dL. A peripheral blood smear demonstrated multiple intraerythrocytic ring forms consistent with the trophozoites of Plasmodium falciparum with a parasite count of 3%.
The patient was treated with intravenous artesunate, obtained from the Centers for Disease Control and Prevention (CDC), and doxycycline. He became afebrile, alert, and oriented after 3 days of intravenous therapy. His oral treatment regimen was completed after 7 days of doxycycline.
Comment: Malaria remains a major cause of morbidity and mortality in the developing world and the most important treatable cause of acute parasitic infection in travelers returning to their Westernized homelands. In the United States, 1564 imported cases were reported during 2006; 39% were attributable to P. falciparum . Immigrants who have recently visited with friends and relatives in their countries of origin often do not take antimalarial prophylaxis and are at higher risk of acquiring malaria.
Malaria continues to have a global presence, primarily affecting individuals living in South and Central America, Africa, and Asia ( Fig. 46.1 ). Close to a half billion individuals are affected annually with up to a million deaths each year. Previously endemic in the United States, public health measures have greatly decreased its incidence here. However, at least a thousand cases are reported annually here and are primarily related to P. falciparum affecting travelers to endemic geographic areas.
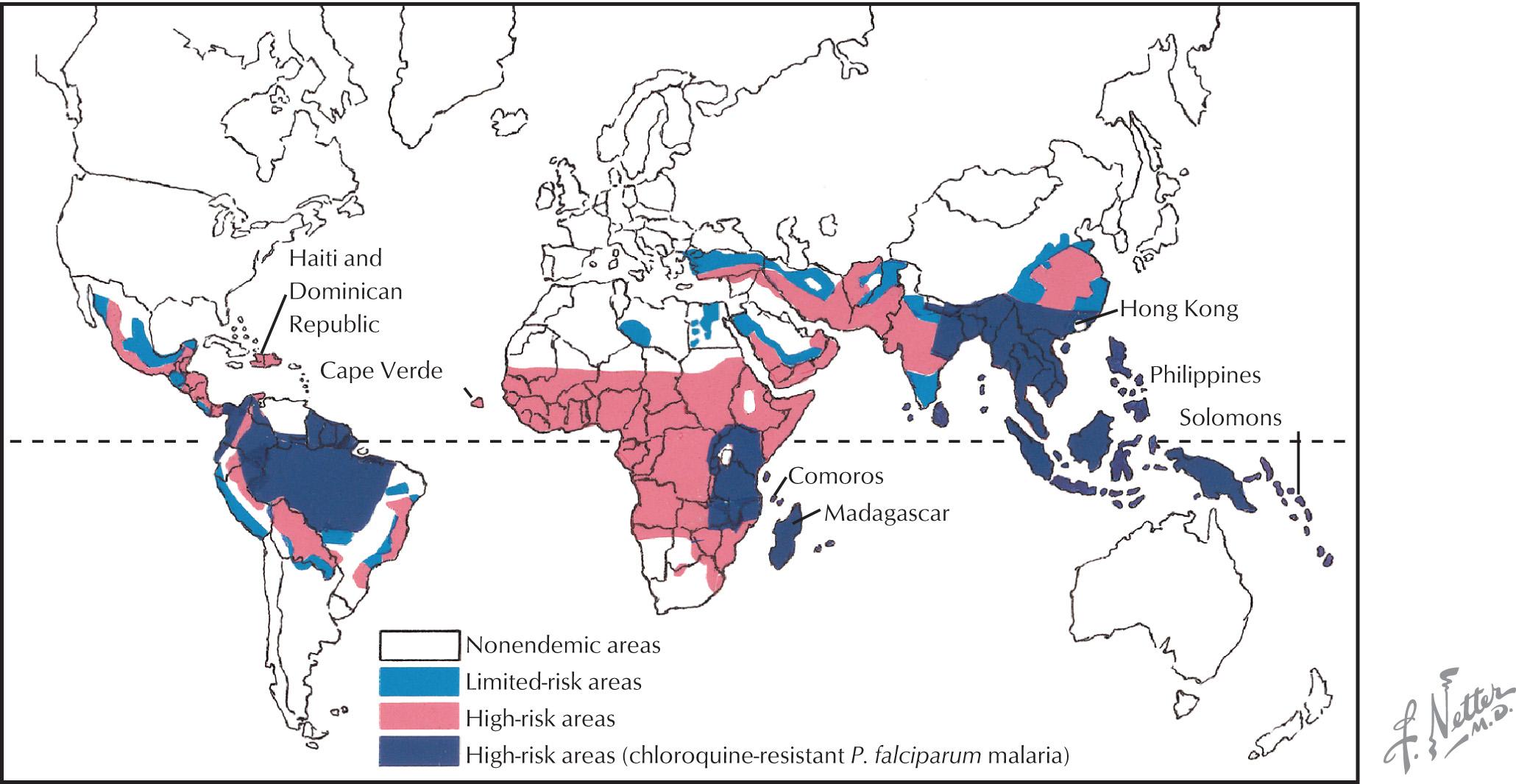
Malaria is caused by four common species of parasites: P. falciparum, P. vivax, P. ovale, and P. malariae. Each form is transmitted to the human from the bites of infected Anopheles mosquitoes. Erythrocytes infected with mature parasites of P. falciparum adhere to endothelial cells in the microvasculature of many organs, including the brain, and undergo a complex interplay with host factors, leading to the manifestations of cerebral malaria.
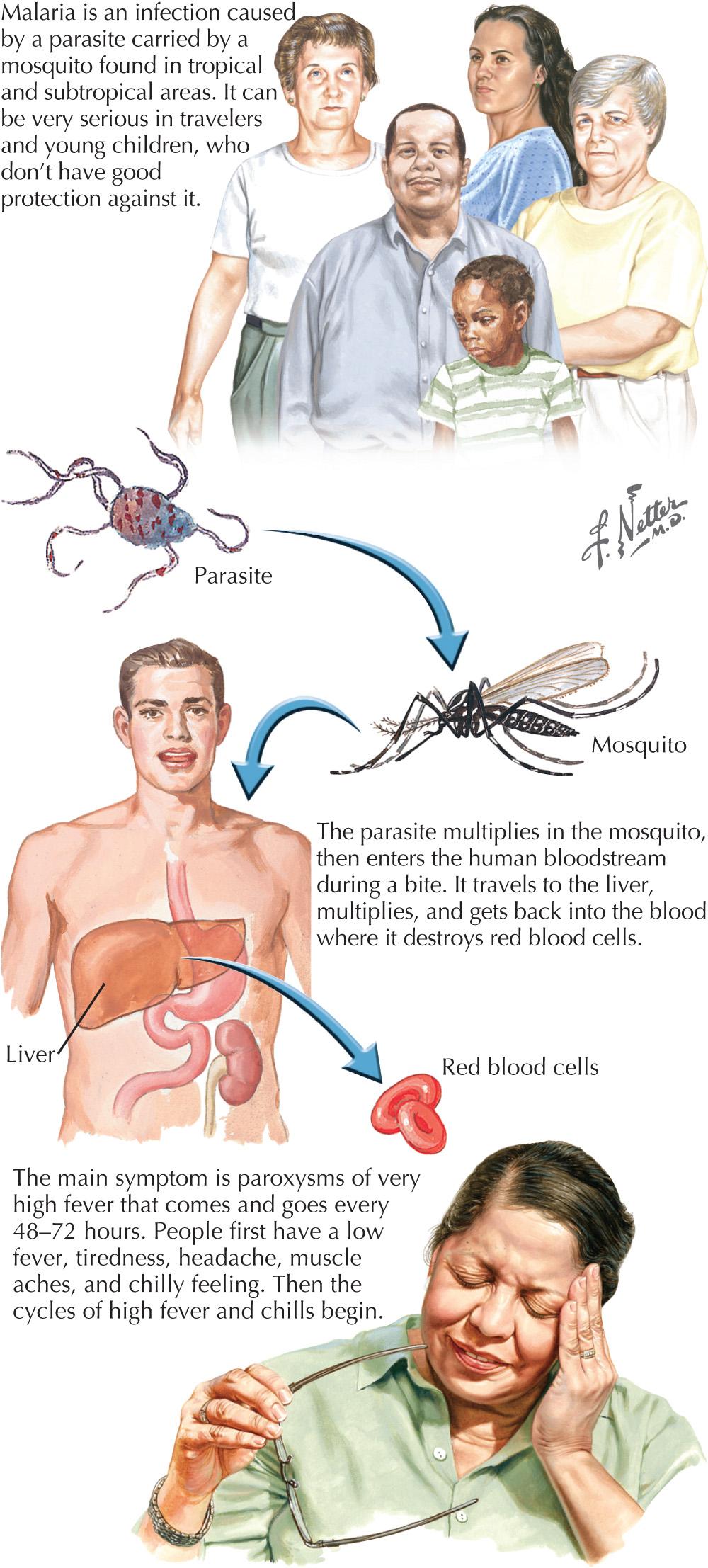
Cerebral malaria is the most life-threatening form, having an adult mortality rate of 20%–50%; this is even worse in children. It is caused by P. falciparum, with the rare exceptional instance of P. vivax. Its primary neurologic features range from irritability and confusion to seizures and coma. Early on there are usually several days of fever and other nonspecific symptoms indistinguishable from those of uncomplicated malaria. Patients may gradually develop coma or in contrast deteriorate suddenly and persistently after a generalized seizure. Grand mal convulsions occur in about half of adult patients. The seizures are often exacerbated by hypoglycemia and lactic acidosis metabolic features that often accompany severe malaria. Hypoglycemia is a common and important abnormality in patients with cerebral malaria and may not be suspected clinically because the symptoms (anxiety, restlessness, and tachycardia) are attributed to the infection itself.
If malaria is suspected, peripheral blood smears need to be examined every 8–24 hours by an experienced microscopist. In a patient with fever and abnormal mental status who was potentially exposed to malaria, antimalarial chemotherapy must be started immediately even if blood smears are repeatedly negative. Microscopic examination and culture of CSF is also essential in patients with cerebral malaria to exclude other treatable CNS infections.
Increased drug resistance has led to combination therapy for malaria. The treatment of cerebral malaria consists of either intravenous quinidine or artesunate accompanied by doxycycline ( Fig. 46.2 ). Intravenous quinidine has to be administered in an intensive care unit (ICU) setting with electrocardiographic monitoring, as it may lead to severe arrhythmias. Exchange transfusion should be strongly considered for persons with a parasite density of more than 5%–10% or even with a lower level of parasitemia if the cerebral malaria is severe or other complications of the malaria occur, including non–volume overload pulmonary edema, or renal complications.
A 38-year-old West African woman, who migrated from her native country 4 months ago, was evaluated in an emergency department for a few weeks of bizarre behavior. In the preceding months, she noted modest weight loss and progressive failure to thrive. She was referred to an inpatient psychiatric service, where she became more lethargic. Her physical examination revealed a low-grade fever with a suspicion of hepatomegaly but was otherwise normal.
Laboratory tests demonstrated a white cell count of 6400/mm, hemoglobin 10 g, and normal platelets. Her liver function tests revealed a mild transaminitis. A careful exam of her peripheral blood smear showed a trypomastigote. HIV antibody was negative. The patient's basic CSF parameters were normal. However, both her indirect fluorescent antibody (IFA) and enzyme-linked immunosorbent assay (ELISA) to Trypanosoma gambiense in her CSF were positive. Treatment was started with melarsoprol but was stopped because of progressive encephalopathy. The patient's family signed her out of the hospital against medical advice and she was lost to follow-up.
Comment: With the world becoming “smaller,” previously “exotic” infectious diseases may now be seen anywhere, including economically highly developed countries. Cultural issues also arise, as illustrated here, where the family made a decision not to allow attempts at a second line of therapy such as intravenous eflornithine when the first medication trial was not successful.
After being brought under control for many years, ever since the 1970s, African trypanosomiasis has reemerged as a new epidemic of enormous proportions. This disease is divided into two different forms, each characterized by meningoencephalitis when it reaches more advanced stages. Both are transmitted by the bites of infected tsetse flies.
West African sleeping sickness accounts for more than 90% of reported cases of sleeping sickness and causes a chronic infection primarily occurring in West and Central Africa. It is caused by Trypanosoma brucei gambiense. Individuals can be infected for months or even years without experiencing any major symptoms or signs. Initially, the systemic disease presents with fever, fatigue, weight loss, cervical lymphadenopathy, and hepatosplenomegaly.
Once the neurologic manifestations emerge, the patient often has developed an advanced stage of CNS infection. Personality changes are common; patients are frequently mistaken as having psychiatric disorders, as in the vignette. In the early phases of CNS disease, a disruption of the normal circadian sleep rhythm occurs ( Fig. 46.3 ).
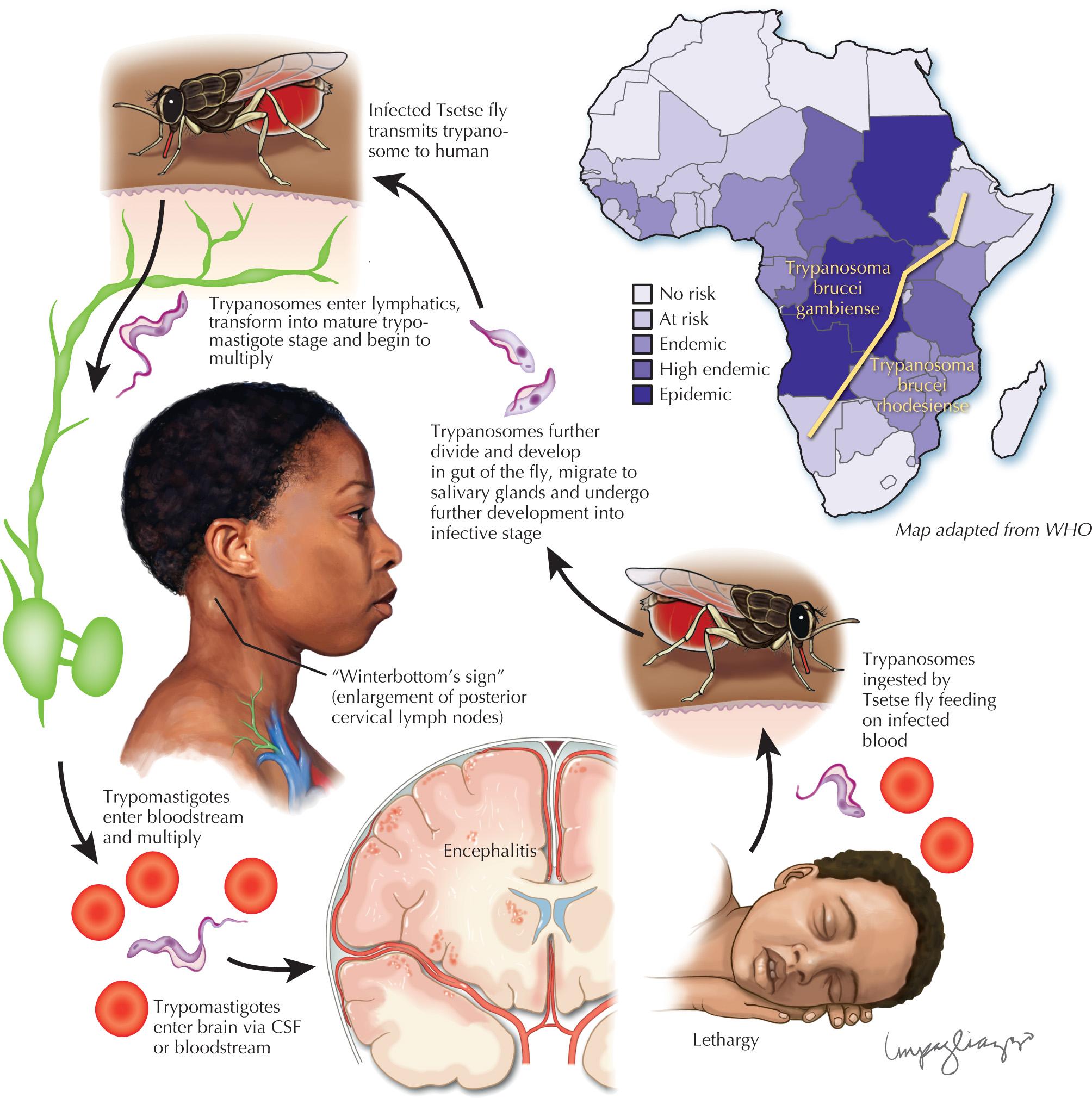
East African trypanosomiasis, caused by Trypanosome brucei rhodesiense, causes a more acute infection in contrast to the more chronic West African form. Neurologic symptoms develop rapidly after just a few months or weeks.
An African sleeping sickness diagnosis is not an easy one to establish in the early phases of the disease when there may not be any CSF changes. Patients are classified as early- or late-stage disease based on CSF findings: those with a CSF WBC greater than 5/mm, often with mild lymphocytic pleocytosis (rarely above 400/mL) and an increased protein content accompanied by a high level of IGM or with trypanosomes demonstrated in CSF seen, are in late-stage disease. Electroencephalography (EEG) can aid in the diagnosis by demonstrating the characteristic impairment in vigilance.
Although visualization of the trypanosome on peripheral smear is the gold standard for diagnosis, immunologic tests such as the ELISA and IFA for antibody levels in the CSF are relatively sensitive and specific for the diagnosis of West African trypanosomiasis. However, no reliable serologic tests for East African trypanosomiasis are currently available for practical diagnostic use.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here