Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
An independent 74-year-old man left a family wedding reception early because he did not feel well; he complained of mild nausea and general malaise. His daughter called the next day, and when he did not answer the phone, she went to his home to check on him, discovering him wandering in his backyard acutely confused. She convinced him to go to the emergency department, where he was found to be febrile with a temperature of 38.5°C (101.3°F). He soon became unresponsive to verbal stimuli, had conjugate eye deviation to the right, neck stiffness, and bilateral palmar grasps. He withdrew his extremities to noxious stimuli; plantar responses were flexor.
A noncontrast head computed tomography (CT) was unremarkable. Cerebrospinal fluid (CSF) examination demonstrated a WBC count of 45/mm 3 , predominantly lymphocytes, protein of 110 mg/dL, and a normal glucose level. Intravenous (IV) acyclovir 10 mg/kg every 8 hours was begun. Magnetic resonance image (MRI) of the brain demonstrated T2-weighted hyperintensity with edematous changes in the left insular region of the inferior temporal lobe, the parahippocampal gyrus, and the hippocampus, extending into the subthalamic nucleus suggestive of herpes simplex virus (HSV) encephalitis. Electroencephalography (EEG) demonstrated periodic lateralized epileptiform discharges (PLEDs). This diagnosis was confirmed by HSV polymerase chain reaction (PCR) 4 days after symptom onset. The patient gradually improved and was treated with a 21-day course of acyclovir. After a short stay in a rehabilitation facility, he was discharged home.
Comment: This is an example of the rapidity with which herpes simplex encephalitis (HSE) will declare itself and the urgent need to consider this diagnosis in any acutely confused patient, initiating treatment based on clinical judgment alone without waiting for definitive diagnostic proof. Unless this type of decision-making takes place, HSE would have caused irreversible temporal lobe damage, particularly affecting memory and language function.
A wide spectrum of viral agents may cause infectious encephalitis. Diagnosis and management are dependent on identifying the precise causative agent. Only a few viruses are amenable to specific antiviral therapy. Therefore, prevention strategies are particularly important, especially for arthropod-borne viruses such as West Nile virus (WNV) and Eastern Equine encephalitis (EEE).
HSE is the most common acute encephalitis in the United States, with an annual incidence of 1 in 250,000 to 1 in 500,000. It affects all ages and both sexes equally, without significant seasonal variation. Early antiviral treatment significantly reduces mortality, but morbidity remains unacceptably high. Most cases of HSE are caused by oral herpes virus (HSV-1); however, genital herpes virus (HSV-2) is more common among neonates with disseminated disease. HSE commonly develops as a recurrent infection, but occasionally may occur during primary infection. Animal data have verified the existence of retrograde viral transport into the brain via olfactory or trigeminal nerves. However, human disease pathogenic pathways are not fully clarified. There is a predilection for HSE, once established, to lead to a hemorrhagic necrosis with inflammatory infiltrates and cells containing intranuclear inclusions.
The symptoms and signs of patients with subacute or acute focal encephalitis are generally nonspecific, with fever, headache, and altered consciousness being the most common. Focal manifestations often include seizures, typically complex partial in character because of the predisposition of the temporal lobe to develop herpes infection. It is common for these patients to also develop language difficulties, personality changes, hemiparesis, ataxia, cranial nerve defects, and papilledema ( Fig. 45.1 ). The differential diagnosis includes stroke, brain tumors, other viral encephalitides, bacterial abscesses, tuberculosis, cryptococcal infections, and toxoplasmosis.
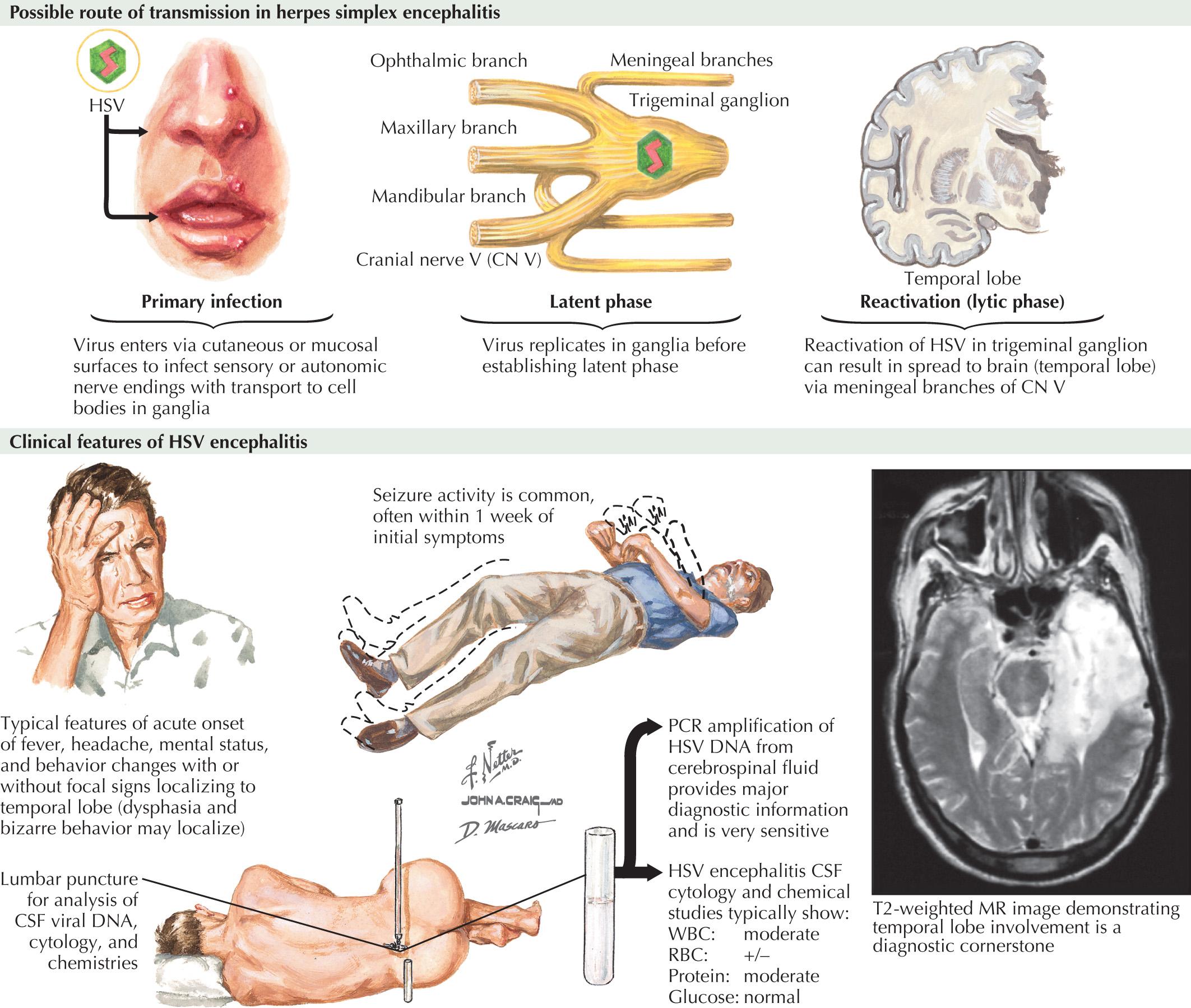
One of the major issues in diagnosis is for the examining clinician to put HSE into the diagnostic spectrum very early on in the temporal profile of the patient's illness. If this is not applied, a major therapeutic window for successful treatment is lost. One unfortunate clinical scenario is failure to diagnose a patient with HSE who presents with confusion for 3 to 5 days that is wrongly attributed to medication or minor infection of the urinary or respiratory tracts.
For patients with suspected encephalitis, the initial diagnostic studies must include a CT scan (to rule out a mass effect) followed by immediate CSF examination. CT scan results are abnormal in 50% of cases early on and usually demonstrate localized edema, low-density lesions, mass effects, contrast enhancements, or hemorrhage. MRI and EEG may be subsequently obtained for further confirmation. These usually demonstrate temporal lobe damage ( Fig. 45.2 ); however, normal studies do not exclude an HSE diagnosis. If this instance does occur, it is often wise to repeat the study within a few days, particularly if the patient continues to be confused.
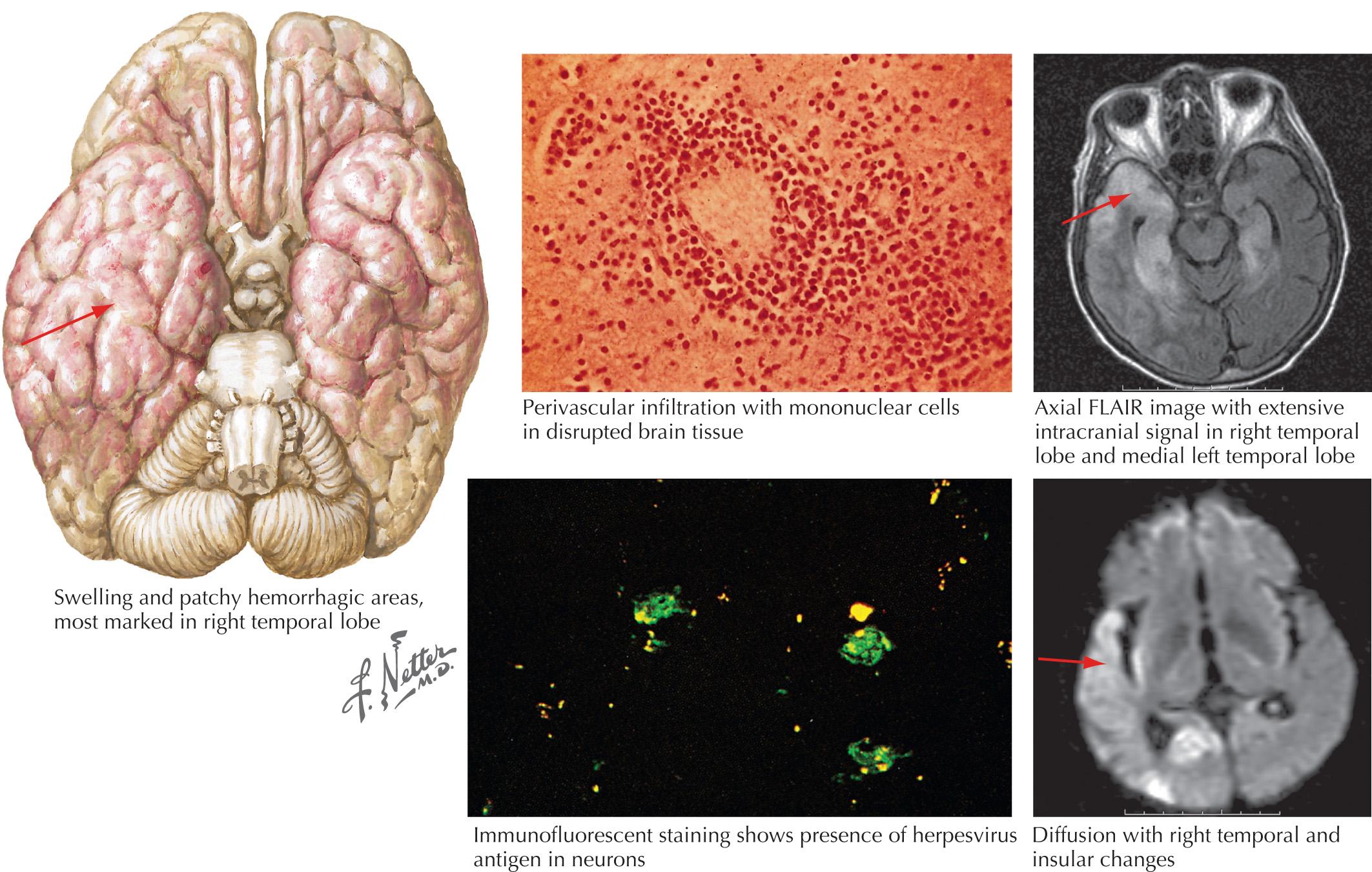
CSF findings are nonspecific, often showing lymphocytic pleocytosis with a slight protein increase. Abnormal CSF findings are found in 96% of biopsy-proven HSE cases. EEG may show repetitive spike or sharp wave discharges, and slow waves localized to the involved area often as PLEDs.
The accuracy of PCR testing for HSV-DNA to detect HSV-1 and -2 in CSF compares favorably with the previous use of brain biopsy. This methodology provides excellent sensitivity and specificity (90%–98%). The viral sequence for HSV may be detected months after the acute episode and may be negative in early disease phases. PCR should not be used to monitor therapy success. Brain biopsy was previously the gold standard for specificity, but it is rarely indicated now with the widespread availability of HSV PCR testing. If used, biopsy specimens are examined for both histopathologic changes and HSV antigens by immunofluorescence testing and appropriate culture techniques.
Immediate initiation of acyclovir is indicated after HSE is suspected. This relatively benign medication has the greatest chance of efficacy if it can be initiated very early on in the patient's clinical course. The excellent outcome of the patient in the initial vignette in this chapter emphasizes the absolute importance for primary care and emergency medicine physicians to immediately consider HSE in individuals of any age who experience relatively acute changes in mental status. Unfortunately, if therapy is not commenced immediately at presentation, the outcome is usually poor, with minimal chance of return to independent living.
Before the availability of IV acyclovir, mortality from HSE was approximately 70%. If one is able to initiate antiviral therapy within the first 24 hours of symptom onset, and if the patient is fully treated for 21 days, the prognosis for long-term outlook is much better. This approach has substantially reduced both mortality and morbidity. Overall, although morbidity remains high, with 60% to 70% of patients having significant neurologic deficits, the mortality is now at 10% to 20%.
On rare occasions, a patient seemingly doing well with initial therapy has a relapse. Inadequate early dosing of the acyclovir is the usual cause. Thus initial treatment regimens must include daily doses of 30 mg/kg intravenously, usually in three separate doses of 10 mg/kg.
A 60-year-old New Hampshire man presented in mid-August with 2 weeks of headache, followed by dizziness, unsteady gait, and nausea and vomiting. He walked his dog past a local pond in the woods every day, sustaining multiple mosquito bites. He had type 2 diabetes mellitus and a history of a prior nephrectomy for renal cell cancer. At clinical presentation, he was febrile, 38.3°C (101°F), somnolent but arousable, answering questions inconsistently; there were fine tremors in his hands, and muscle stretch reflexes were globally depressed.
Brain MRI revealed increased signal with mild mass effect in the left thalamus and basal ganglia. Spinal fluid examination demonstrated a WBC of 1860/mm 3 (81% polymorphonuclear neutrophils [PMNs], 11% lymphocytes, 8% monocytes), 26 red blood cells/mm 3 , protein 106 mg/dL, glucose 98 mg/dL (serum 209), and negative Gram staining. He had continued fevers with progressive gait ataxia, upper extremity weakness, and memory loss. Bacterial cultures and studies for Borrelia burgdorferi, Treponema pallidum, HSV, and WNV were negative. CSF IgM and plaque assay were positive for EEE. He gradually recovered motor function over the next few months with supportive care, and 1 year later, he had minimal residual cognitive deficits.
EEE virus is a member of the family Togaviridae, genus alphavirus, that is found in the eastern half of the United States. EEE is a mosquito-borne viral disease that causes disease in humans, horses, and some bird species. It generally takes 3 to 10 days to develop symptoms of EEE after being bitten by an infected mosquito. An average of five human cases occurs per year (approximately 220 confirmed cases in the United States between 1964 and 2004, most frequently in Florida, Georgia, Massachusetts, and New Jersey). EEE virus transmission is most common in and around freshwater hardwood swamps in the Atlantic and Gulf Coast states and the Great Lakes region. The main EEE virus transmission cycle is between birds and mosquitoes.
Most persons infected with EEE virus do not demonstrate a clearly discernible illness. In those individuals who do develop clinical illness, symptoms range from a mild flulike illness to fulminating encephalitis, eventually leading to coma and death. The mortality rate from EEE is approximately 33%, making it one of the most deadly mosquito-borne diseases in the United States.
Laboratory diagnosis of EEE virus infection is based on serology, especially IgM testing of serum and CSF, and neutralizing antibody testing of acute- and convalescent-phase serum. MRI is the most sensitive imaging modality for the diagnosis of EEE ( Fig. 45.3 ). The most commonly affected areas of the central nervous system (CNS) include the basal ganglia (unilateral or asymmetric, with occasional internal capsule involvement) and thalamic nuclei. Other areas include the brainstem (often the midbrain), periventricular white matter, and cortex (most often temporally). Affected areas appear as increased signal intensity on T2-weighted images.
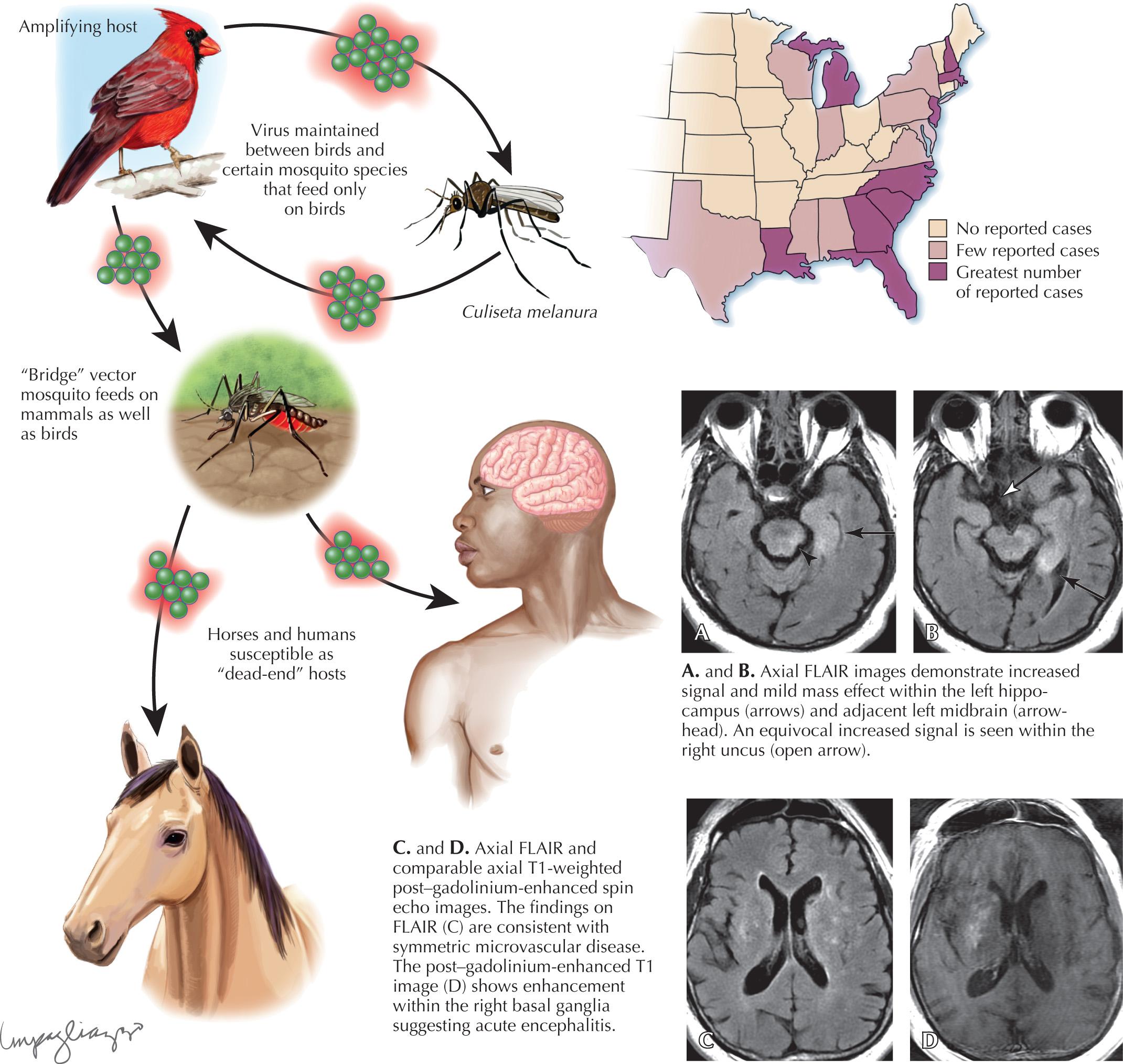
There is no specific treatment for EEE; optimal medical care includes intensive hospitalization and supportive care.
Approximately half of those persons who survive EEE will have mild to severe permanent neurologic damage, usually with cognitive impairment. Those older than age 50 and younger than age 15 years appear to be at greatest risk for developing severe EEE.
WNV is a flavivirus usually found in Africa, West Asia, and the Middle East. The Middle Eastern strains are genetically closely related to the St. Louis encephalitis virus, found in the United States. There are many potential animal, ornithologic, and insect reservoirs, including humans, horses, other mammals, birds, and mosquitoes. The WNV was not documented in the Western Hemisphere until 1999. In the temperate zones of the world, West Nile encephalitis cases occur primarily in the late summer or early autumn. In southern climates, where temperatures are milder, WNV can be transmitted year-round.
West Nile fever is typically a mild disease characterized by flulike symptoms that develop 3 to 15 days after the bite of an infected mosquito. West Nile fever usually lasts only a few days and does not seem to cause any long-term health effects. Mild fever, headache, body aches, occasional skin rash, and swollen glands are the most common symptoms.
However, there is a more severe disease spectrum that can manifest as encephalitis, meningitis, or meningoencephalitis. A poliomyelitis-like illness is also described, with an acute proximal and asymmetric flaccid paralysis, occasionally occurring during outbreaks in the United States. Neurophysiologic, radiologic, and pathologic studies suggest that WNV has a proclivity to damage anterior horn cells within the spinal cord.
Diagnosis is made by serologic assays of blood and CSF.
Treatment is entirely supportive; there is no specific drug treatment or vaccine available. In the rare instances of a polio-like illness, the long-term outcome hinges on the degree and distribution of anterior horn cell damage.
A 52-year-old man from Massachusetts presented with 2 days of fever and myalgias. He lived outside of Boston but had recently traveled to Cape Cod, New York, Pennsylvania, and Wisconsin; at the time of symptom onset, he was camping in a lake area of New Hampshire. Although his initial neurologic examination was normal, several days later he developed inattention, somnolence, and left upper extremity dysmetria. Brain MRI showed evidence of bilateral basal ganglia and thalamic T2/fluid attenuation inversion recovery (FLAIR) hyperintensity and diffusion restriction in the dorsal midbrain. The patient improved incompletely and was discharged to an acute rehabilitation facility. Two years later, he had persistent headaches as well as cerebellar dysarthria, delayed motor function, and incoordination.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here