Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Infection is the major threat to recovery among most trauma patients that survive the successful resuscitation and intervention of the initial 24 hours following severe injury. The infectious risk may occur from environmental contamination that attended the injury process or from endogenous microflora of disrupted organ structures within the patient. Infection may complicate the surgical site employed for injury repair. Similarly, the trauma patient receives a large measure of care in the critical care unit with multiple invasive tubes and monitoring devices where each serve as a portal for microbial entry and potential life-threatening infection. From injury until recovery, the trauma patient is exposed to pathogens at the site of injury and everywhere “the hands of man” have been in the process of treatment.
Not only are there abundant opportunities for infection, but the trauma patient sustains a remarkable compensatory suppression of immune responses. Virtually every measurable component of the innate and adaptive immune response is suppressed. The exact benefit for the suppressed host is often hard to rationalize from an evolutionary perspective, but modulation of the wholesale systemic activation of the innate inflammatory response that is activated by major tissue injury must provide some protection for the injured host. However, the millennia of evolution did not anticipate aggressive resuscitation, surgical interventions, and critical care units. The contemporary end result is survival through incredible multisystem injury, only to have a recovering host that is a human Petri dish. The critical care unit becomes the common ground of immunosuppression and infection risk that is shared by all trauma/critically ill patients. It is a wonder that all do not die of infection after major injury.
This overview will briefly discuss a model of common clinical issues that cause infection in the care of trauma patients. This will not be a detailed discussion of each anatomic site of infection or a detailed discussion of pathogens and antibiotic choices, since most are discussed in detail in other chapters. It will highlight the common features that are associated with the important infections of the trauma patient and will use examples of specific infections and specific pathogens to emphasize these features. This overview will hopefully give a perspective to the many areas that deserve continued attention and investigation.
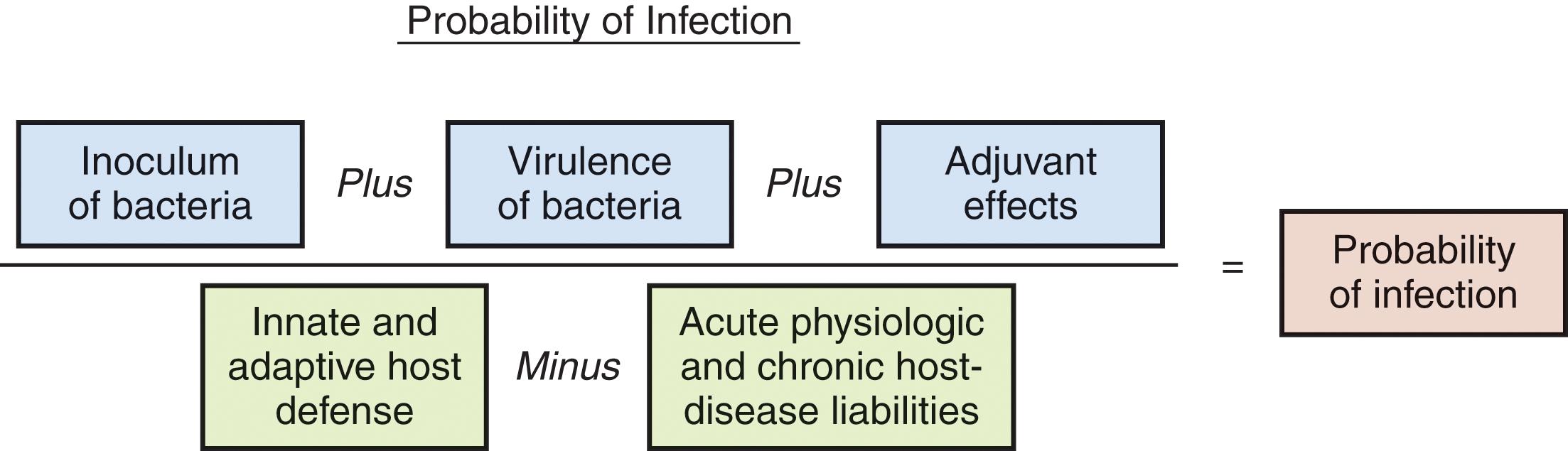
All injuries and all patients are not at the same risk for the development of infection. Infection occurs because of the complex interactions of numerous variables of the pathogen and the host ( Fig. 1 ).
One of the most commonly studied areas of infection is the number of microbes that contaminate an anatomic site and the critical threshold that is required for infection to result. All injured sites, all surgical incisions, and all invasive medical devices will have recoverable bacteria when cultured. Our environment is not sterile, and man himself has more microbial colonists than eukaryotic cells. The presence of bacteria does not uniformly determine infection, but the numbers of bacteria per gram of tissue or unit of surface area of tissue are predictive.
Classic studies of experimental and clinical wounds have created the magical 10 5 organisms per gram of tissue as the necessary microbial threshold for infection as an outcome. The 10 5 threshold is held as the standard in the definition of infection in the urinary tract. A threshold of 10 4 bacteria is considered the defining limit of bacteria in bronchoalveolar lavage samples of lung secretions in the critical care unit patient. There must be a definable limit of Clostridioides difficile spores that are ingested to create the enterocolitis syndrome. While there is some biological variability around the 10 5 threshold, the supporting research has clearly defined that there is a limit to the magnitude of contamination that can be handled without resultant infection by the host at each anatomic site ( Fig. 2 ).
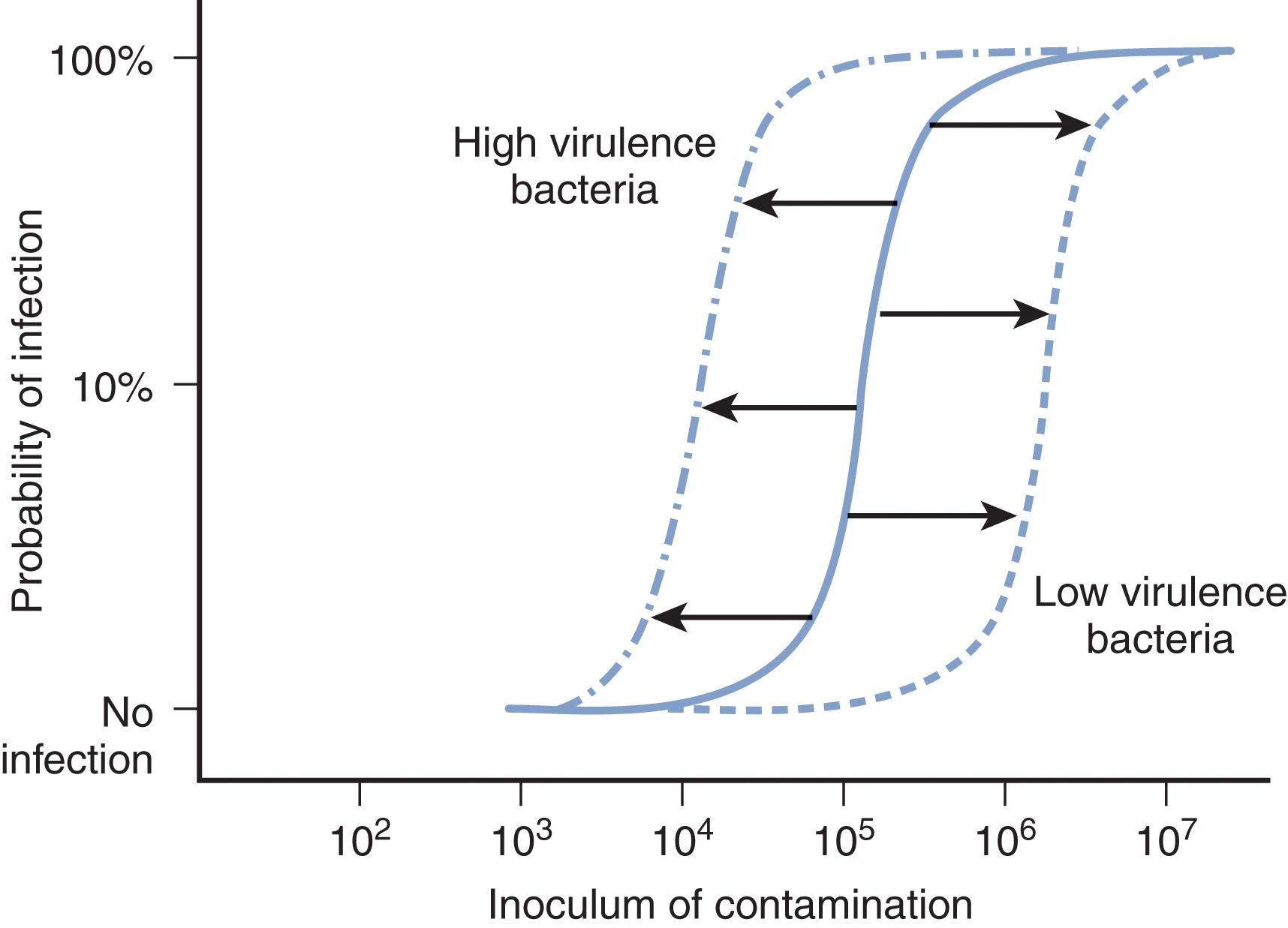
Bacteria have individual profiles of virulence characteristics. Virulence factors include structural components of the bacterial cell (e.g., endotoxin), secreted bacterial protein products that damage tissues or specifically retard phagocytic cell function (e.g., toxins), and antimicrobial resistance factors that impair the effectiveness of treatment strategies. The presence of lipid A in gram-negative endotoxin, potent exotoxins produced by Clostridium perfringens , superantigens from Streptococcus pyogenes, and coagulase from Staphylococcus aureus means that selected bacteria pose a greater infectious risk. When bacterial contaminants possess these unique virulence factors, it means that fewer microbes are required to cause clinical infection than the traditional view of 10 5 bacteria per gram.
Conversely, many bacterial species lack potent virulence factors but still pose infection risks. Pseudomonas aeruginosa, Staphylococcus epidermidis, Enterococcus spp., and Candida albicans are microbes that demonstrate poor virulence characteristics in experimental models but have proven to be very real pathogens in the clinical setting. They share the capacity to resist the antimicrobial activity of many drugs and emerge as major pathogens in the immunosuppressed critical care patient when prior treatment has eliminated the more susceptible common bacterial pathogens. Intrinsically low virulence organisms in terms of structural or secreted protein products can assume major importance in critically ill patients because of resistance. In the critical care unit, virtually any microbe can assume a role as a pathogen when competing microflora have been eliminated and the host response is suppressed.
At the end of his spectacular scientific career, Pasteur is alleged to have said, “The microbe is nothing, the terrain is everything.” While bordering on hyperbole, the comment by Pasteur emphasizes the importance of the local environment surrounding the bacterial contaminant in the trauma patient as being paramount in determining infection as an outcome. The presence of hemoglobin and hematoma in the traumatic wound or surgical site provides a rich source of nutrients and critical amounts of iron to promote microbial growth. Necrotic tissue provides a haven for bacterial proliferation that is poorly accessible by host phagocytic cells.
Foreign bodies assume importance as adjuvant variables in the development of infections in all surgical patients, but especially following injury. Traumatic wounds may contain foreign debris from the injury process, or fecal material from colonic disruption that is laden with microbes that promote infection. The surgical site will have suture material that is well documented to lower the required bacterial inoculums to cause infection. Endotracheal tubes, urinary catheters, drains, and indwelling intravascular devices that are necessary components of patient management may provide foreign body surfaces for bacteria to colonize and become the nidus for infection. The foreign body surface retards the phagocytic function of the host but also provides a surface for biofilm production as a unique virulence expression of bacteria to promote infection.
Not only do indwelling devices compromise host functions, but they may directly damage the nonspecific primary epithelial and endothelial barriers of the patient. Endotracheal tubes defeat the normal barrier of the glottis and provide a direct conduit from the external environment into the lower airway passages. Foley catheters not only create an avenue for microbial access but erode the protective transitional cell lining of the bladder. Similarly, the indwelling intravascular device creates access for treatment, but clot and endothelial damage at the site of entrance must have a role in infection.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here