Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The most common indication for histologic assessment of small bowel mucosa is to evaluate for a malabsorption disorder. Although the spectrum of small intestinal diseases that can result in symptoms of malabsorption can seem daunting at first, most diseases produce histologic findings that fall into a limited number of injury patterns. A pattern-based approach is useful because, in practice, it is often difficult, if not impossible, for the pathologist to diagnose a specific cause. Furthermore, lack of available clinical history can add to this challenge. The small bowel disorders presented here mostly fall into one of two major patterns of injury based on assessment of the architecture at low magnification: the flattened villi pattern and the intact villi pattern ( Table 5.1 ). Small bowel mucosa with intact villi pattern can be further subdivided based on the nature of the inflammatory infiltrate into neutrophilic inflammation, eosinophilic inflammation, lymphoplasmacytic inflammation, and disorders with minimal or no inflammation. Lastly, some disorders can alter the lamina propria and result in villous blunting without significant inflammation. These include infectious enteritis caused by Whipple disease, Mycobacterium avium-intracellulare infection, and histoplasmosis and conditions that alter the lacteals, including primary and secondary lymphangiectasia.
| Flattened Villi Pattern (with or without Intraepithelial Lymphocytosis) |
|
| Intact Villi with Neutrophilic Inflammation |
|
| Intact Villi with Eosinophilic Inflammation |
|
| Intact Villi with Lymphoplasmacytic Inflammation |
|
| Intact Villi with Paucicellular Inflammation |
|
The flattened villi pattern of injury has also been called the malabsorption pattern and is characterized by villous blunting, crypt hyperplasia with or without significant intraepithelial lymphocytosis, and expansion of the lamina propria by lymphocytes and plasma cells. The prototypical malabsorption disorder is celiac disease (CD), but a number of other disorders can produce similar histologic findings, including peptic injury, tropical sprue, collagenous sprue, nongluten protein sensitivity, small intestinal bacterial overgrowth (SIBO), autoimmune enteropathy, common variable immunodeficiency (CVID), and drug-related sprue-like enteropathy, among others.
Several conditions result in small bowel mucosal injury without altering the villous architecture. These may or may not be accompanied by prominent inflammation and can be further categorized based on the inflammatory component into conditions with neutrophilic inflammation, eosinophilic inflammation, lymphoplasmacytic inflammation, and disorders with minimal or no inflammation.
The characteristic feature of this pattern is the presence of neutrophilic inflammation within the epithelium. In most cases, the lamina propria is also expanded by a mixed inflammatory cell infiltrate composed of neutrophils, eosinophils, lymphocytes, and plasma cells. The epithelium shows regenerative epithelial changes in the form of reactive mucin loss and slight nuclear hyperchromasia, with preservation of the nucleus-to-cytoplasmic ratio. The potential causes are drug injury (especially nonsteroidal antiinflammatory drugs [NSAIDs]), infections (including Helicobacter pylori ), peptic injury, inflammatory bowel disease (IBD), Zollinger-Ellison syndrome (ZES), and early ischemia.
This pattern is characterized by excessive number of eosinophils in the mucosa along with eosinophil-mediated epithelial injury in the form of cryptitis or crypt abscesses. Similar to the rest of the gastrointestinal (GI) tract, increased eosinophils can be associated with idiopathic eosinophilic enteritis, parasitic infection, allergies, drug- or medication-induced injury, connective tissue disease, vasculitis, systemic mastocytosis, and Langerhans cell histiocytosis (LCH).
This pattern of injury is characterized by expansion of the lamina propria by lymphocytes and plasma cells. The possible causes of this pattern of injury include CD, IBD, and drug- or medication-related injury (especially NSAIDs).
Some conditions are associated with intact villous architecture and minimal or no inflammation. These disorders include certain infections, such as giardiasis, cytomegalovirus (CMV), radiation injury, amyloidosis, chemotherapeutic agents, drugs or medications (especially mycophenolate mofetil [MMF]), graft-versus-host disease (GVHD), and transplant rejection.
Identifying the two major patterns of small bowel injury (flat villi and intact villi) is helpful in generating a differential diagnosis and determining the cause of mucosal injury. However, the patterns are not etiologically specific. For instance, many conditions can manifest with flattened villi pattern of injury. Furthermore, one specific etiology may result in a variety of patterns of injury. For instance, NSAID-related injury can manifest with all major patterns of injury. It is therefore important to identify the pattern of injury and provide a comment enlisting all the potential etiologies that may contribute to injury. The sections in this chapter have been organized based on causes of small bowel injury.
The term malabsorption is broadly used to describe any type or degree of dysfunction in the uptake of substances that are normally retained or absorbed by the small intestine. Malabsorption can result from a multitude of causes, which can be broadly categorized into disorders of inadequate digestion (pancreatic disorders, disorders of decreased intestinal bile salt concentration), endocrine or metabolic disorders (diabetes mellitus, thyroid dysfunction, ZES), biochemical or genetic diseases (CD, disaccharidase deficiency, abetalipoproteinemia, hypogammaglobulinemia), mucosal disorders (infections, immunologic conditions, infiltrative disorders), and postmucosal disorders (lymphatic obstruction, cardiac failure, vascular obstruction). Conditions that affect the small bowel mucosa may drastically reduce the amount of villous surface area, preventing adequate uptake of nutrients. Microscopically, malabsorption disorders often show villous blunting, crypt hyperplasia, and intraepithelial lymphocytosis— the flattened villi pattern of injury . Having said that, some malabsorption disorders have no specific pathologic findings (e.g., disaccharidase deficiency, transporter defects), and lack of inadequate nutrient uptake is not caused by global surface area loss but by defects in specific protein function.
Once considered to be a rare childhood disorder, CD (also known as gluten-sensitive enteropathy or celiac sprue) is a chronic immune-mediated disorder induced by dietary exposure to gluten in genetically predisposed individuals. Confirmation of a diagnosis also requires prompt improvement of clinical symptoms and mucosal pathology after withdrawal of dietary gluten.
Celiac disease occurs both in adults and children with a female predominance (female-to-male ratio, 2–3 to 1). It is estimated to affect between 0.5% and 1% of individuals in both Europe and the United States. The peak age of incidence is the third to fourth decades of life. However, up to 20% of individuals may be diagnosed after 60 years of age.
The classification and diagnosis of CD are based on GI manifestations; however, patients with extraintestinal complications are increasingly being recognized ( Table 5.2 ). The clinical presentation of CD ranges from asymptomatic to severe malnutrition. CD may be divided into four clinical subtypes, two symptomatic types and two asymptomatic types. In the classic symptomatic subtype, patients present with chronic diarrhea, abdominal distension, and pain. In the atypical symptomatic subtype, GI symptoms may be less pronounced to absent. Instead, extraintestinal features such as anemia, short stature, neurologic symptoms, dermatitis herpetiformis, delayed puberty, osteoporosis, infertility, vitamin and protein deficiencies, and elevated liver enzyme levels are predominant. Some patients have asymptomatic or silent CD; they lack classic or atypical symptoms but have unequivocal villous atrophy found incidentally for other reasons or after serologic screening. Finally, latent CD is defined by a positive serology but lack of villous atrophy on initial biopsies. Individuals are asymptomatic but may develop symptoms, histologic changes, or both.
| Endocrine disorders |
| Type 1 diabetes mellitus |
| Autoimmune thyroid disorders |
| Addison’s disease |
| Reproductive disorders (infertility, miscarriages) |
| Osteoporosis |
| Alopecia areata |
| Neurologic disorders |
| Cerebellar ataxia |
| Neuropathy |
| Epilepsy |
| Migraines |
| Cardiac disorders |
| Idiopathic dilated cardiomyopathy |
| Autoimmune myocarditis |
| Hepatic disorders |
| Primary biliary cirrhosis |
| Autoimmune hepatitis |
| Autoimmune cholangitis |
| Other |
| Dermatitis herpetiformis |
| Anemia |
| Selective immunoglobulin A deficiency |
| Sjögren syndrome |
| Juvenile chronic arthritis |
| Turner syndrome |
| Down syndrome |
| Dental enamel defects |
High-risk groups to be screened for CD are those with other autoimmune disorders such as type 1 diabetes mellitus and autoimmune thyroiditis, a family history of CD, selective immunoglobulin (Ig) A deficiency, or dermatitis herpetiformis. CD is also more common in certain pediatric populations, including those with Down syndrome, in whom the frequency of CD is as high as 10%. In this group, CD is usually not detected on the basis of GI symptoms.
Celiac disease is a clinicopathologic diagnosis that is most often based on positive serology and specific histologic findings. Antitissue transglutaminase (anti-tTG) antibodies are both highly sensitive (90%–98%) and specific (95%–97%) for CD. Antiendomysial (EMA) antibodies are specific (97%–100%) but have slightly inferior sensitivity (85%–98%). Antigliadin antibody testing should no longer be used because of its low positive predictive value. A diagnosis of CD may be made if both tissue biopsy and serologic testing are consistent with CD. CD may be excluded if results of both are negative.
Sometimes there is discordance between the histologic and serologic findings, which may occur for a number of reasons. First, IgA deficiency is more common in individuals with CD than in the general population. Because tTG and EMA serologic tests are based on IgA, they may be falsely negative in patients with IgA deficiency. Total IgA can be measured to identify these patients, and IgG-based assays, particularly deamidated gliadin peptide or IgG-tTG, may be used in these situations. Second, the flattened villi pattern of injury is not specific for CD. Consideration of an alternative cause (e.g., SIBO, tropical sprue, drug-associated enteropathy) should be explored in patients with negative celiac serologies. Sometimes HLA genotyping is useful from excluding the possibility of CD. HLA-DQ2 or HLA-DQ8 is present in almost all cases of CD. Negative testing for these alleles virtually eliminates the possibility of CD. Human leukocyte antigen (HLA) typing is particularly useful in patients who may have initiated a gluten-free diet (GFD) before proper diagnostic work-up. This complicates diagnosis because both serologic and histologic abnormalities may normalize with GFD. HLA genotyping can be used to rule out CD in a subset of these patients.
In patients who test positive for HLA-DQ2 or HLA-DQ8, a gluten challenge can be performed, in which biopsy and serologic analysis takes place after gluten has been reintroduced for a period time. In patients with CD, gluten challenge should elicit typical serologic and histologic features. However, some patients may experience a return of their symptoms (e.g., abdominal pain, bloating) but without serologic or histologic evidence of CD. An alternate diagnosis should be considered in these patients, such as IgE-mediated wheat allergy or nonceliac gluten sensitivity.
The pathogenesis of CD is related to a combination of environmental, genetic, and immunologic features and can be viewed as both luminal events and cellular events leading to the activation of immune cells and ensuing tissue damage. Enteric exposures to certain glutamine-rich proteins in the dietary grains wheat, rye, and barley are essential for the development of CD. The actual proteins that trigger the disease are the gliadins in wheat, the hordeins in barley, and the secalins in rye. In CD, many of these peptides are poorly digested by the intestinal tract proteases and traverse through the epithelium into the lamina propria as intact molecules. tTG is a multifunctional enzyme that is essential for the pathogenesis of CD. tTG has two essential functions, deamidation of glutamine to glutamic acid and cross-linking glutamine and lysine residues. After absorption, intact peptides are deamidated by tTG, converting the abundant glutamine residues to glutamic acid, thus rendering them negatively charged. The negatively charged peptides are more efficiently bound to HLA-DQ2 or HLA-DQ8 receptors on the surface of the antigen presenting cells. Intestinal DQ2- or DQ8-restricted CD4+ T cells then recognize the deaminated gliadin peptides and elicit a cytokine-mediated inflammatory response. Importantly, tTG also forms complexes with gliadin, mediated by its glutamine-lysine cross-linking function. In an environment already primed by gliadin peptide presentation on DQ2/8 T cells, tTG–gliadin complexes serve as neoepitopes that stimulate the formation of anti-tTG autoantibodies.
Finally, it was recently discovered that infection with certain viruses can trigger immunopathologic responses to dietary gluten. Specifically, infection with reovirus, which is nonvirulent and otherwise nonpathogenic, can trigger certain events critical for CD pathogenesis. In mouse models, reovirus infection represses regulatory T-cell (T reg ) conversion and promotes T helper-1 (T H 1) immunity to dietary antigen. Reovirus infection also breaks oral tolerance to gluten and promotes activation of transglutaminase 2. In humans, patients with CD were much more likely to have circulating antibodies against reovirus than normal control participants, suggesting a link between CD and prior reovirus infection. It remains to be seen if other infectious agents can elicit similar immunologic derangements.
The major genetic risk factors for CD are the HLA class II genes HLA-DQ2 and HLA-DQ8 . Approximately 95% of all patients with CD have a DQ2 heterodimer, and almost all remaining patients with CD have a DQ8 heterodimer. Gene dosage correlates with the risk for developing CD, with patients homozygous for DQ2 carrying the greatest risk. CD is concordant in 70% to 80% of monozygotic twins, 30% to 40% of HLA-identical siblings, and fewer than 20% of dizygotic twins, a rate similar to that for all first-degree relatives. However, recent linkage studies suggest that non-HLA disease-associated genes may also play a role.
A multisystem autoimmune disorder caused by an immune response to dietary gluten and related proteins
1% of the US population
1 in 133 patients with no risk factors
1 in 29 patients with at least one first-degree relative with celiac disease (CD)
20-fold higher risk for enteropathy-associated T-cell lymphoma
30-fold higher risk for small intestinal adenocarcinoma
Two- to fourfold higher risk for oropharyngeal and esophageal carcinoma
Wide age range: children and adults
Common in Northern Europeans; rare in Southeast Asians and Middle East
Variable: ranges from asymptomatic to severe malnutrition
Most common presentation is weight loss, abdominal pain, and diarrhea
Extraintestinal manifestations include infertility, osteoporosis, dermatitis herpetiformis, and various neuropathies
Complete resolution of mucosal injury with a strict gluten-free diet (GFD)
Refractory CD refers to patients on GFD for a period of 1 year or longer without resolution of symptoms
Type II refractory CD is commonly associated with a clonal T-cell population and is at high risk for developing enteropathy-associated T-cell lymphoma
If the mucosal biopsy specimen is examined under a dissecting microscope (which is seldom done), delicate villi are found to be attenuated or even absent. Endoscopically, a reduction in folds, scalloping, a mosaic pattern, or nodular mucosa may be seen ( Fig. 5.1 ).

Histologic abnormalities in the duodenum may be patchy. Current guidelines from the American College of Gastroenterology (ACG) recommend multiple biopsies of the duodenum, with one or two biopsies from the bulb and at least four biopsies from the distal duodenum. CD produces the flattened villi pattern of injury—intraepithelial lymphocytosis, villous blunting, and crypt hyperplasia ( Figs. 5.2 and 5.3 ). The earliest change in CD is the presence of increased intraepithelial lymphocytes (IELs), most conspicuously within the villous tips, with intact villous architecture ( Fig. 5.4 ). With persistent disease, epithelial damage ensues with reactive mucin loss. The lamina propria is expanded by lymphocytes and plasma cells, thus contributing to partial or complete villous blunting. Villous blunting is accompanied by crypt hyperplasia. Active inflammation is occasionally seen but is generally not prominent. Marked neutrophilic epithelial injury should suggest an additional or alternative cause, such as peptic duodenitis, NSAID injury, infection, or Crohn’s disease.
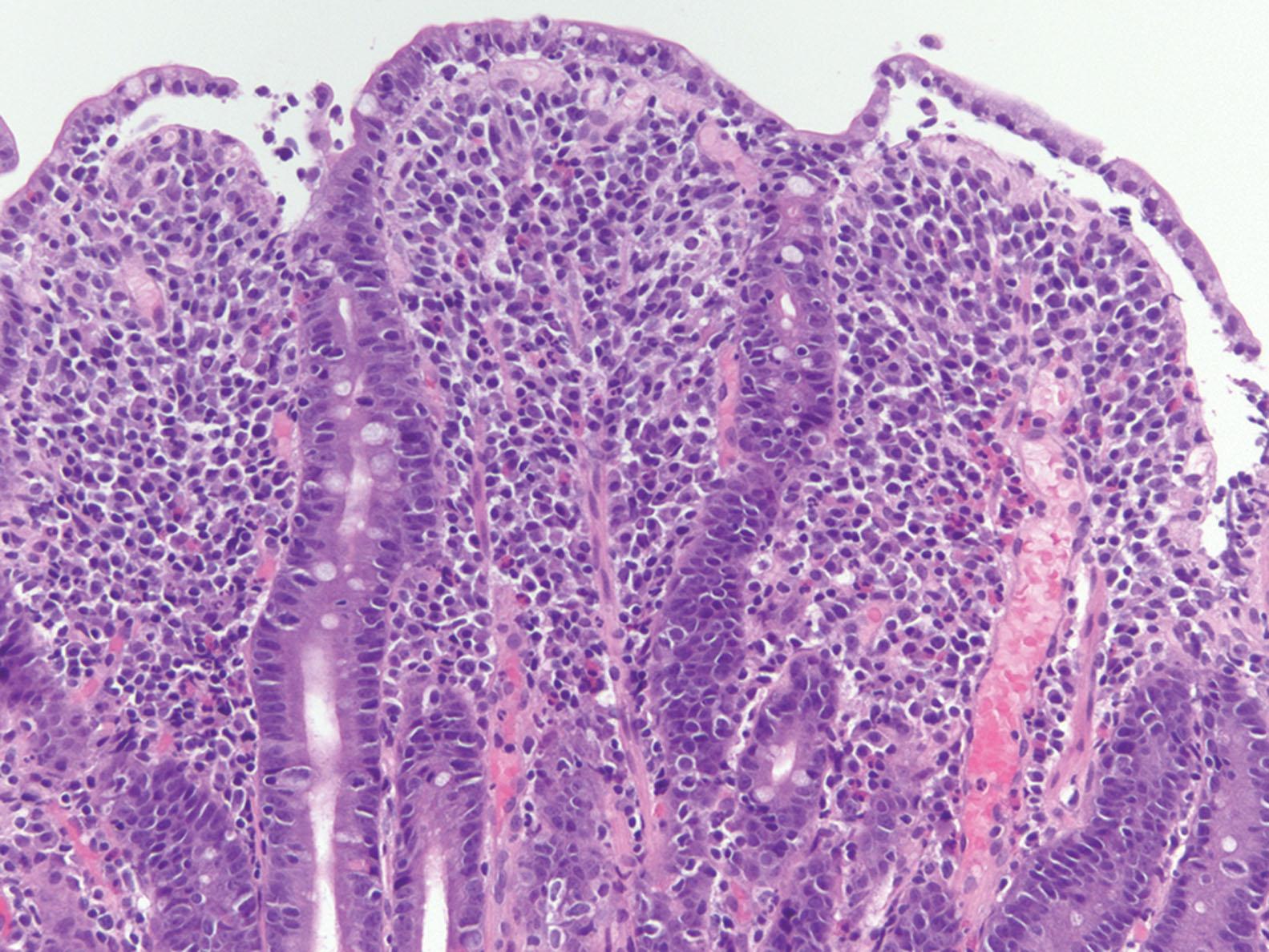


Duodenal biopsies exhibiting all the features of CD—increased IELs, villous blunting, crypt hyperplasia—are easy to recognize. However, changes of early CD can be quite subtle and include intact villous and crypt architecture with increased IELs. The increase in IELs is sometimes obvious, but in some cases, an objective measure is needed to resolve borderline cases. The normal range of IELs has been reported as 11 to 23 per 100 enterocytes. According to ACG guidelines, more than 25 IELs per 100 enterocytes is abnormal. One rapid method to determine this number is to evaluate 5 villous tips, counting the number of lymphocytes present within the distal-most 20 enterocytes. The final values can be expressed as IEL per 100 enterocytes. When possible, evaluate only well-oriented villi that are anchored by intact muscularis mucosae because tangentially sectioned lateral aspects of the villi may result in overestimation and inaccurate estimation of the lymphocyte count. A CD3 immunostain can also be used to highlight IELs. However, the utility of this approach has not been established or validated for clinical practice. Some studies have shown that CD3 immunohistochemistry (IHC) does not improve detection of CD in cases in which the hematoxylin and eosin–stained sections are normal.
Upon initiation of a GFD, patients often report a marked improvement in clinical symptoms. Microscopically, intestinal biopsies show diminished surface epithelial injury, a reduced number of IELs, and partial to complete resolution of villous architectural abnormalities. However, in some cases, despite resolution of clinical symptoms and normal serology, the inflammatory component may persist for up to a period of 1 to 2 years after GFD. If patients consume gluten-containing products, there is a rapid return of all lesions, and malabsorption ensues.
Attenuated to absent villi (as seen under a dissecting microscope)
Scalloping or mosaic appearance of the mucosa
Variable degrees of villous blunting
Intraepithelial lymphocytosis, most prominent on villous tips (>25 IELs per 100 enterocytes)
Crypt hyperplasia
Bacterial overgrowth
Infection
Tropical sprue
Nongluten protein sensitivity
Crohn’s disease
Protein intolerance
Autoimmune enteropathy
Medications (nonsteroidal antiinflammatory drugs, olmesartan)
The flattened villi pattern of injury is not specific for CD, and therefore, a diagnosis of CD can only be confirmed with the knowledge of positive celiac serologies. Without this information, the pathologist must be descriptive in the report by stating the pattern of injury (e.g., duodenal mucosa with villous blunting and increased IELs) and providing a comment with the differential diagnosis. In biopsies showing well-developed features of the flattened villi pattern of injury, the differential diagnosis includes infection, protein intolerance, tropical sprue, nongluten protein sensitivity, bacterial overgrowth, Crohn’s disease, CVID, autoimmune enteropathy (AIE), and medication injury (particularly from NSAIDs and olmesartan). In biopsies with increased IELs and normal villous and crypt architecture, the differential diagnosis is slightly different and includes viral infections, H. pylori infection, NSAID injury, peptic injury, tropical sprue, nongluten protein sensitivity, bacterial overgrowth, Crohn’s disease, and AIE.
A lifelong adherence to a GFD is the mainstay of safe and effective treatment of patients with CD. Commonly substituted grains in the GFD include rice, corn, quinoa, and buckwheat. Serologic testing may be used to check the effectiveness of a GFD. Patients whose disease does not respond to dietary therapy should undergo a systematic evaluation, including review of the patient’s diet by an expert dietician.
Patients with CD have more than a 30-fold increased risk for small bowel adenocarcinoma compared with the general population, and 13% of patients with a small bowel adenocarcinoma have underlying CD. Other malignancies potentially associated with CD include enteropathy-associated T-cell lymphoma (EATL), papillary thyroid carcinoma, and malignant melanoma.
Nonresponsive CD is defined as persistent symptoms, signs, or laboratory abnormalities despite 6 to 12 months of gluten avoidance. It is common, occurring in 7% to 30% of patients. The most common cause is inadvertent gluten ingestion. Other causes include SIBO, other food intolerances, irritable bowel disease, pancreatic insufficiency, and microscopic colitis. In patients with nonresponsive CD, the diagnosis of CD should be reconfirmed, and the patient’s diet should be closely examined to rule out inadvertent gluten ingestion. Additional studies, including upper and lower endoscopy, may also be indicated to exclude other diseases in the differential diagnosis.
If there are persistent signs and symptoms after 12 months on a strict GFD and if alternative disorders have been ruled out, the diagnosis of refractory celiac disease (RCD) is appropriate. RCD is rare (1%–2% of patients with CD) and can be subdivided into two types based on the immunophenotype of the lymphocyte population. In type I RCD, the infiltrating lymphocytes are morphologically and immunophenotypically similar to those seen in untreated CD. Patients with type I RCD usually respond to immunosuppressive regimens and are at low risk for progression to lymphoma. In type II RCD, the lymphocytes display an abnormal immunoprofile, lacking one or more normal T-cell markers (CD3, CD4, and CD8). Furthermore, monoclonal or oligoclonal T-cell receptor rearrangements can be detected. Having said that, a recent study has shown clonal T-cell populations even in typical patients with CD. Thus, the utility of TCR gene-rearrangement studies in this setting is somewhat limited. Given these findings, some consider RCD II to be a form of in situ T-cell lymphoma, and there is a much higher risk of progression to malignancy compared with RCD I. Patients with RCD II are often refractory to steroid therapy. Optimal treatment is unclear, but a variety of immunomodulatory and chemotherapeutic agents have been used.
Enteropathy-associated T-cell lymphoma is a high-grade T-cell non-Hodgkin’s lymphoma of the upper small intestine. This rare T-cell disorder is 20 times more common in patients with CD. Two types of EATL are recognized and have slightly different clinical and pathologic features, though both are very aggressive. Type I EATL is closely associated with CD. Type II EATL is less common and is not associated with CD. Pathologically, type I EATL most commonly involves the jejunum or ileum. Plaques, nodules, or strictures can be seen grossly. Microscopically, there is a dense, diffuse proliferation of atypical medium to large lymphoid cells, usually in a background of mixed inflammatory cells ( Fig. 5.5 ). Immunohistochemically, the neoplastic cells usually express CD45 and cytoplasmic CD3 while lacking other T-cell markers, including CD4, CD5, and CD8. CD30 expression is present in some cases. The neoplastic cells have a cytotoxic T-cell phenotype with expression of TIA-1, granzyme B, and perforin. Type II EATL usually involves the jejunum, but in contrast to type I EATL, it may also involve the stomach or colon. Microscopically, EATL type II is composed of a more monotonous population of smaller cells, and there are fewer admixed non-neoplastic inflammatory cells. Immunohistochemically, type II EATL is similar to type I but more often expresses CD8 but less often expresses CD30 (see Chapter 19 for additional details). The prognosis is similarly poor for both types; the 5-year survival rate is approximately 10%. About half of patients require laparotomy for complications of hemorrhage, perforation, or obstruction.
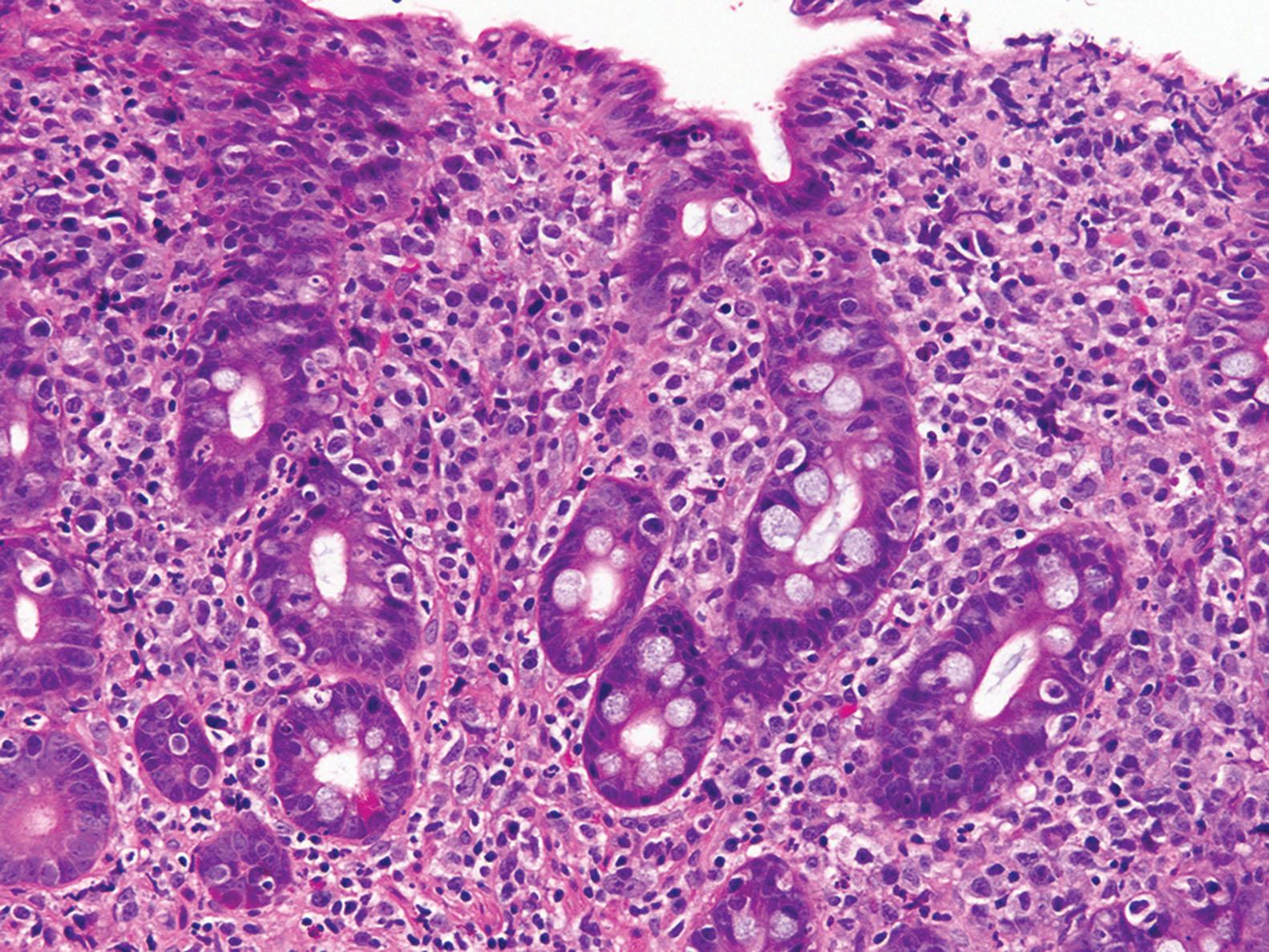
Collagenous sprue, also known as collagenous enteritis, is a rare but severe form of malabsorption disorder. Given the limited number of small case series reported in the literature, the nature of collagenous sprue and its relationship to other malabsorption disorder are unclear. Initially, collagenous sprue was thought to represent an uncommon variant of CD, enriched in patients who have refractory disease. However, it is now clear that a proportion of cases occur in patients with no other evidence of CD (based on serology, genetics, or response to a GFD). Therefore, collagenous sprue seems to represent an unusual, nonspecific injury pattern associated with a variety of pathogenic mechanisms. Beyond its association with CD, collagenous sprue has also been reported in the context of collagenous gastritis, microscopic colitis, and olmesartan-induced injury. It is most common in middle-age to older women, and, unlike collagenous gastritis, it is rare in children.
Histologically, the key feature is a thick subepithelial collagen layer ( Fig. 5.6 ). The required thickness of the collagen layer is not well defined, but some reports use greater than 12 μm as a cut-off. Similar to collagenous colitis, there are entrapment of capillaries and irregular extension of the collagen layer into the lamina propria. The background mucosa shows villous atrophy. However, in contrast to CD, the crypts are typically atrophic rather than hyperplastic. The degree of intraepithelial lymphocytosis can be variable.

Early descriptions of this entity indicated that patients usually have a poor prognosis. However, more recent studies have revealed that the clinical outcome can be quite heterogeneous. GFD is sufficient treatment for some patients, though about half are refractory to GFD, in which case immunosuppressive or chemotherapeutic agents are needed. Some patients die of intractable disease.
Tropical sprue (also known as postinfective tropical malabsorption) is a rare disorder that mimics several aspects of CD. It is an intestinal malabsorption of unknown etiology that occurs among residents in or visitors to the tropics. No single causative agent has been identified to account for tropical sprue. However, the following evidence favors an infectious cause: infection often initiates and sustains tropical sprue, tropical sprue occurs in specific geographic areas (West Indies and the Indian subcontinent) where enteric infections are common, tropical sprue is endemic in some areas, and recovery from tropical sprue with antibiotics is usually rapid and dramatic. Protozoan infections such as with Cyclospora have been suggested to play a role.
Gross findings are not well described but may include an abnormal villous pattern. Microscopically, tropical sprue produces a marked increase in IELs. The villous architecture of the mucosa is typically preserved in tropical sprue and a completely flat mucosa—like that seen in patients with CD—is never seen. Intraepithelial lymphocytosis is very prominent in tropical sprue and is seen in both the villi and crypts. In addition, there may be a significant increase in the number of lamina propria eosinophils. Finally, unlike CD, in which mucosal changes are greatest in the proximal small bowel, the lesions are similar throughout the entire small bowel in tropical sprue. Mechanistically, this fits with the resulting secondary vitamin B 12 and folate deficiency states that are commonly a consequence of tropical sprue. In fact, vitamin B 12 and folate deficiency may produce megaloblastic changes in the epithelium with enlarged nuclei and reduced mitotic activity.
After the diagnosis is made, tropical sprue is eminently treatable with antibiotics; a 3-month course of doxycycline is generally sufficient. Folic acid vitamin B 12 supplementations are also useful to correct nutritional deficiencies. Unfortunately, given the rarity of the disorder, this diagnosis is often not considered either clinically or pathologically.
Peptic duodenitis and peptic ulcer disease (PUD) represent a continuum of the same disease process, namely damage to the duodenal mucosa caused by exposure to excessive gastric acid. It is estimated to affect up to 10% of the Western population. Peptic disease is more common among men who are older than 40 years. Patients often present with burning epigastric pain relieved by eating. In severe cases, the pain may be constant and accompanied by nausea and vomiting.
Chronic infection with H. pylori is highly correlated with peptic disease of the duodenum and is associated with more than 80% of peptic ulcers. Mechanistically, this occurs with antrum-predominant H. pylori infection, which stimulates gastrin secretion, and in turn, increases acid production. Furthermore, peptic injury often results in gastric foveolar metaplasia, which can become secondarily colonized with H. pylori , further contributing to mucosal injury. Gastric foveolar metaplasia may represent an adaptation to chronic exposure to hyperacidity. Other factors, such as smoking, NSAIDs, renal insufficiency, or duodenal dysmotility, compromise mucosal defense mechanism and allow prolonged contact with gastric acid, resulting in peptic injury.
Patients with multiple duodenal ulcers represent the most severe form of the disease; multiple duodenal or jejunal ulcers should raise concern for ZES. In the absence of ZES, refractory ulcers may be seen in patients with chronic NSAID use, gastric outlet obstruction or duodenal stenosis, or those with a history of gastric bypass procedure.
Peptic duodenitis is most commonly seen in the duodenal bulb. The endoscopic appearance varies from simple erythema to friability, erosions, and nodularity of the mucosa ( Fig. 5.7 ). Nodularity and polypoid lesions most often correspond to Brunner’s gland hyperplasia or prominent foveolar hyperplasia. In PUD, the ulcers are usually circular and rarely exceed 3 cm in diameter. They are typically located along the posterior wall of the duodenal bulb and are more likely to present with perforation and massive hemorrhage because of their proximity to pancreaticoduodenal and gastroduodenal arteries.

In general, peptic duodenal disease shows the intact villi with a neutrophilic inflammation pattern of injury. There are expansion of the lamina propria by neutrophils and neutrophil-mediated epithelial injury ( Fig. 5.8 ). There are an accompanying increase in lamina propria plasma cells, reactive epithelial changes (mucin loss, nuclear hyperchromasia), gastric foveolar metaplasia ( Fig. 5.9 ) and Brunner’s gland hyperplasia. In some instances, the Brunner’s glands may extend from the submucosa into the basal aspect of the mucosa. In severe cases, villous blunting can be present as well—a feature more in keeping with the flattened villi pattern of injury. Severe peptic injury leads to ulceration (PUD).


Ancillary stains are not generally used for diagnosis of peptic disease but may be used to highlight a few interesting features. Gastric foveolar metaplasia can be highlighted by a combination periodic acid–Schiff (PAS) and Alcian blue (AB) stain that highlights the presence of neutral mucin characteristic of the gastric foveolar metaplasia ( Fig. 5.10 ). H. pylori may colonize areas of metaplastic gastric epithelium because these cells express the same surface receptors as the native gastric epithelium. The organisms may be highlighted by special stains such as IHC, Giemsa, or Diff-Quik ( Fig. 5.11 ). However, evaluation of the gastric mucosa, when provided, is a preferred method to detect H. pylori infection.


Peptic duodenal disease typically shows a neutrophilic pattern of inflammation, and other causes that produce this pattern of injury include drug or medication injury (particularly from NSAIDs), infections (other than from H. pylori ), and IBD. Occasionally, there are specific clues to distinguish between these possibilities. For example, crypt distortion and granuloma formation in conjunction with involvement of the rest of the GI tract would be highly suspicious for Crohn’s disease. Some infectious causes, including CMV and adenovirus, can be detected microscopically.
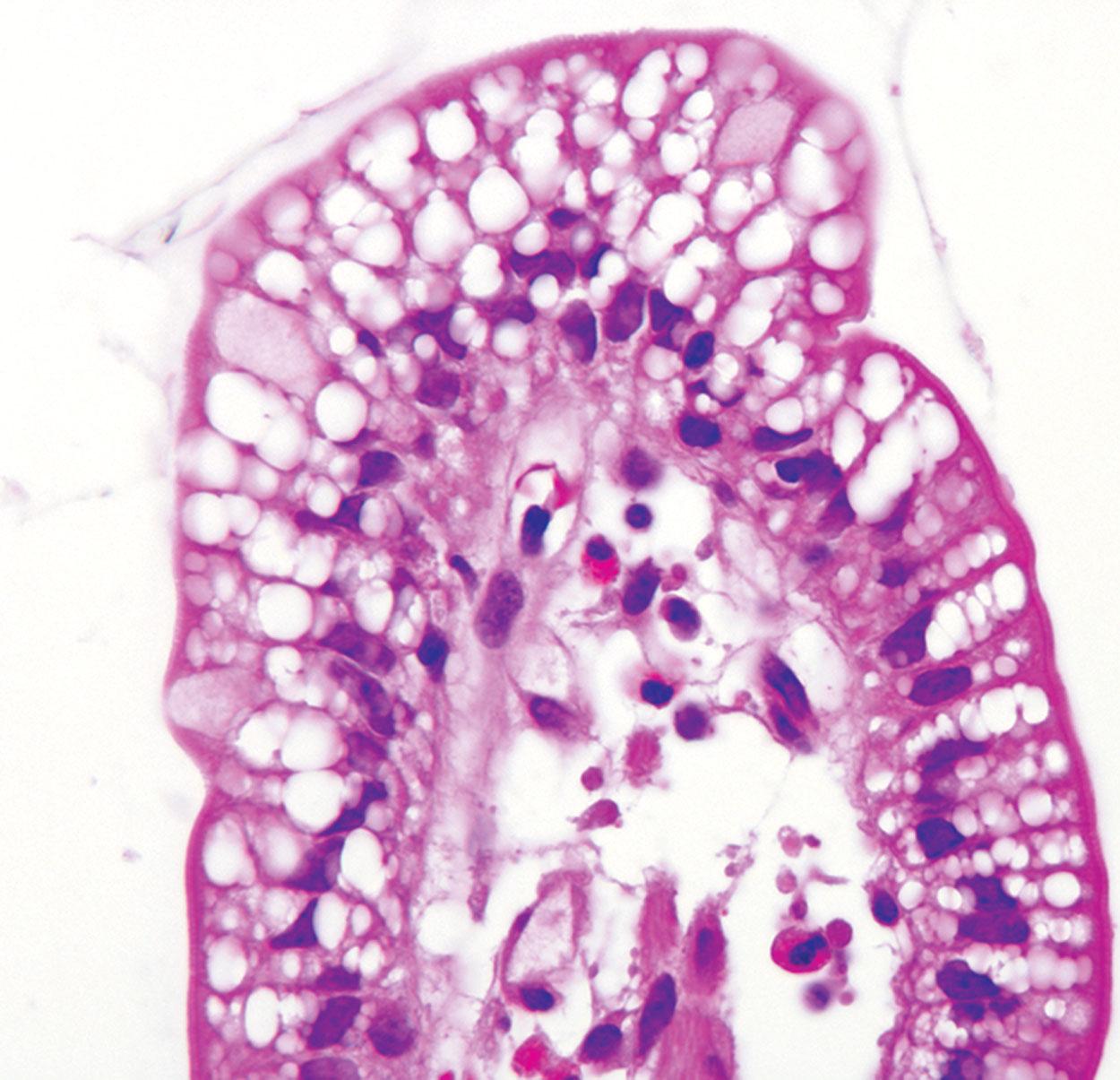
Although gastric foveolar metaplasia is a common feature of peptic duodenal disease, “lipid hang-up” is a common mimic. It results from physiologically normal vacuolization of the enterocyte cytoplasm resulting from a recent fatty meal ( Fig. 5.12 ). Compared with gastric foveolar metaplasia, which is PAS positive, these vacuoles are PAS–AB negative and are only localized to the tips of the villi. A similar finding is seen in abetalipoproteinemia; however, the vacuolization affects the tips as well as the lateral aspect of the villi. Gastric heterotopia may also be included in the differential diagnosis with peptic duodenitis resulting from the prominent gastric foveolar epithelium. However, unlike peptic duodenitis, gastric heterotopia can be recognized by normal-appearing oxyntic glands in association with the surface gastric mucinous epithelium. Finally, chronic peptic injury can also cause villous blunting with a variable degree of intraepithelial lymphocytosis. Therefore, these findings should prompt consideration of other disorders that produce the flattened villi pattern of injury (e.g., CD).
Finally, although marked histologic abnormalities are highly correlated with H. pylori infection, many duodenal biopsies show mild changes, with increased lymphoplasmacytic inflammation in the lamina propria, gastric foveolar cell metaplasia, and reactive epithelial changes (hyperchromasia and apical mucin attenuation). Neutrophils may or may not be present. If a concurrent stomach biopsy shows no suggestion of H. pylori infection, it is unclear what diagnosis is most appropriate. Various terms are commonly used, including peptic duodenitis and peptic duodenopathy . However, in the absence of H. pylori , there is little evidence that these changes are caused by stomach acid or any other “peptic” etiology. Therefore, when these cases are encountered, it is probably best to be descriptive and simply list the specific histologic abnormalities that are present.
The eradication of H. pylori and the use of acid-suppressive therapies improve symptoms in most patients with peptic duodenitis. Medical therapy is mainly intended to reduce gastric acid by small meals, antacids, and histamine-2 (H 2 ) blockers. Patients should avoid substances that promote gastric acid secretion, such as alcohol. Aspirin and other NSAIDs should also be avoided because they inhibit mucosal defenses.
Bleeding ulcers are estimated to be responsible for up to 50% of all cases of acute hemorrhage from the upper GI tract. Up to 5% of patients may have bleeding that is brisk enough to cause hematochezia. Even if initially controlled, rebleeding can affect up to one-third of patients, most commonly patients in the seventh decade of life or beyond or those with a visible vessel in the ulcer base. Refractory ulcers heal slowly or follow a relapsing and remitting course. Scarring of duodenal ulcers may lead to duodenal stricturing and obstruction.
Free perforation of a duodenal ulcer into the peritoneal cavity can be a catastrophic, life-threatening event. Most patients with this complication are older adults, and the perforations are associated with NSAID use in up to half of cases. However, in patients younger than 75 years, smoking may be a stronger risk factor for perforation than NSAIDs.
Damage to the duodenal mucosa resulting from exposure to excess gastric acid
Common; up to 10% of population in Western countries
Severe disease can cause perforation or life-threatening bleeding
More common among men who are older than 40 years
70% of patients are asymptomatic
Dyspepsia is most common symptom, classically 2 to 5 hours after a meal
In severe cases, bleeding and perforation may occur
Antisecretory therapy to reduce acid production
Helicobacter pylori eradication if H. pylori is present
Prognosis usually favorable, though life-threatening ulcer perforation is possible
Surgical therapy occasionally indicated for refractory ulcers
Circular ulcers in the duodenum, most commonly involving the duodenal bulb
Nodularity or polypoid areas may represent Brunner’s gland hyperplasia
Neutrophilic inflammation (both in the lamina propria and epithelium)
Increase plasmacytic inflammation
Gastric foveolar metaplasia
Brunner’s gland hyperplasia
Reactive epithelial changes (apical mucin loss, hyperchromasia)
Villous blunting is variable
Nonsteroidal antiinflammatory drug injury
Infection
Inflammatory bowel disease
Malabsorptive disorders (if villous blunting is prominent)
Bacterial overgrowth is another cause of small intestinal malabsorption. Although the causes of bacterial overgrowth are numerous, most result in intestinal stasis; thus, these disorders are also known as stasis syndrome. Causes of stasis in the small bowel include motor or neural disorders that affect intestinal motility such as diabetic neuropathy and scleroderma, as well as structural lesions, such as diverticula and surgical anastomoses. Certain drugs may also slow intestinal motility (e.g., narcotics and benzodiazepines). Stasis allows accumulation of anaerobic bacteria that deconjugate bile salts, deplete vitamin B 12 , and damage surface epithelium. Patients may experience bloating, abdominal pain, diarrhea, dyspepsia, or weight loss, though some patients are asymptomatic. The pathologic features can be quite variable. There is often mild to moderate villous blunting, but complete villous blunting is uncommon. IELs are also increased but typically not to the degree that is seen in CD ( Fig. 5.13 ). The gold-standard test for SIBO involves aspiration of small intestinal fluid and demonstration of greater than 10 5 colony forming units per milliliter of fluid. A carbohydrate-based breath test can also be used, which is simple and noninvasive. Breath tests are based on the principle that a test dose of carbohydrate will be metabolized by bacterial flora, and certain metabolites can be detected in the breath.

The two main goals of SIBO treatment are to correct the underlying disease process, if possible, and to decrease bacterial load. Antibiotics reduce the bacterial burden, which leads to symptomatic improvement. Dietary changes may also be helpful to provide nutrients that are quickly absorbed and less metabolized by bacteria, such as a high-fat, low-carbohydrate diet. Finally, prokinetic agents (e.g., metoclopramide) may be useful, though their benefit is not well-established.
Nonsteroidal antiinflammatory drugs are a widely prescribed class of medication and can cause injury throughout the GI tract in those consuming the medication. NSAID injury occurs via both local and systemic mechanisms. Locally, NSAIDs may pass directly into epithelial cells and become ionized and trapped within. Inside the cell, these compounds can disrupt oxidative phosphorylation and deplete adenosine triphosphate stores. Systemically, NSAIDs nonselectively inhibit cyclooxygenase isoenzymes and suppresses prostaglandin synthesis. Certain prostaglandins downregulated by NSAIDs are important for maintaining several aspects of mucosal protection, including supporting mucin production, mucosal blood flow, and bicarbonate secretion. Thus, although ulceration is relatively common, chronic use can cause fibrosis, stricture, perforation, and formation of mucosal diaphragms—so-called diaphragm disease .
Ulcers, strictures, and mucosal diaphragms associated with nonsteroidal antiinflammatory drug (NSAID) use
Overall unknown
Ulcers are most common, strictures less so, and “diaphragm disease” is rare
Data from 1997 showed 107,000 hospitalizations and 16,500 deaths resulting from NSAID consumption in the United States alone (data are not specific to the gastrointestinal [GI] tract)
The incidence of GI perforations, symptomatic gastroduodenal ulcers, and upper GI bleeding in persons known to be taking NSAIDs is about 3% over 2 years
A minority of these patients die of these complications
Most commonly, NSAIDs injury consists of localized erosions and ulcers. More rarely, NSAID may cause chronic damage that includes varying degrees of mucosal and submucosal fibrosis, ultimately broadening and forming flat strictures. There may also be mucosal membranes that divide the lumen of the small intestine into compartments, and this appearance is termed diaphragm disease .
Typically, NSAID damage shows the intact villi with neutrophilic inflammation pattern of injury, with expansion of the lamina propria by neutrophils. Epithelial injury is usually present and may be associated with erosion or ulcer surrounded by reactive epithelial changes. NSAID injury may also take the form of prominent mucosal eosinophilia, presumably via a hypersensitivity mechanism ( Fig. 5.14 ). In resection specimens, these changes are patchy in distribution with multifocal superficial ulcers.

In chronic NSAID injury, there are villous blunting and crypt hyperplasia—features of the flattened villi pattern of injury. In other cases, crypt distortion and pyloric gland metaplasia are often present. With ongoing injury, there are thickening of the muscularis mucosae and a disorganized proliferation of smooth muscle fibers, nerves, and vessels within the submucosa. These findings indicate reparative changes that are most prominent in areas with stricture formation. It has been suggested that ischemia, inflammation, and repeated mucosal injury contribute to these pathologic changes.
Rarely, chronic injury can lead to diaphragm disease . The pathogenesis of this phenomenon is not well understood but likely involves repeated cycles of injury and repair. There may be superficial annular or longitudinal ulcers. The submucosa is widened, creating annular constriction of the lumen, and it is replaced by disorganized fascicles of smooth muscles (i.e., muscularization of the submucosa), bundles of unmyelinated nerve fibers with scattered ganglion cells, and numerous blood vessels. These changes may extend to the serosa.
Nonsteroidal antiinflammatory drugs can produce any major patterns of small intestinal injury that includes neutrophilic inflammation, eosinophilic inflammation, the flattened villi pattern, and the architectural distortion pattern. Again, NSAID injury can also produce lesions with a paucity of chronic inflammation, which is a relatively distinct feature. Overall, the diagnosis of NSAID injury always requires correlation with clinical findings and exclusion of other causes.
Perforations and ulcers associated with nonsteroidal antiinflammatory drugs (NSAIDs) do not have a specific appearance, nor do the strictures of the small bowel
Diaphragm disease has a relatively unique pattern, consisting of mucosal membranes separating segments of the small bowel
Intact villi with numerous neutrophils within the lamina propria along with neutrophilic epithelial injury; occasionally mucosal eosinophilia
Erosions and superficial ulcers may be present
Chronic NSAID injury shows crypt architectural distortion, pyloric gland metaplasia, thickening of muscularis mucosae, submucosal fibrosis, and proliferation of nerves, vessels, and smooth muscle
Chronic NSAID injury may present as villous blunting with or without intraepithelial lymphocytosis
In patients with diaphragm disease , the submucosa is widened, creating annular constriction of the lumen, and is replaced by disorganized fascicles of smooth muscles (i.e., muscularization of the submucosa), bundles of unmyelinated nerve fibers with scattered ganglion cells, and numerous blood vessels
Peptic duodenal disease
Infection
Ischemia
Inflammatory bowel disease
Behçet’s disease
Malabsorption disorders (if villous blunting is prominent or IELs are increased)
Because NSAIDs injury can produce each of the major patterns of small intestinal injury, the differential diagnosis depends on which pattern is present. The neutrophilic inflammation pattern is most common, the differential diagnosis for which includes H. pylori infection, Crohn’s disease, and reactive duodenopathy. In patients with chronic NSAID use, chronic ischemia, Crohn’s disease, and Behçet’s disease are in the differential diagnosis. Clinical findings are key in distinguishing NSAID injury from Behçet’s disease. Behçet’s syndrome is characterized by lymphocytic vasculitis that affects veins and less commonly the arteries. Crohn’s disease is distinguished from NSAID injury by the presence of deep fissuring ulcers, transmural lymphoid aggregates, and granulomas. Chronic ischemia is a close mimic and requires assessment of mesenteric vessels. When villous blunting is a prominent feature, it should prompt consideration of other malabsorptive disorders. Ultimately, as with any other medication, confirmation of causal association requires correlation with history of drug intake and resolution of symptoms after discontinuation of the drug.
The prognosis is excellent for patients with NSAID ulcers if the medication is discontinued. For patients requiring resections owing to strictures or diaphragm disease , the prognosis is good, and the risks are typically those associated with the surgical procedure itself.
Olmesartan is an antihypertensive drug and a member of the angiotensin receptor blocker (ARB) family of compounds. Recently, this drug has shown to cause a sprue-like enteropathy in some patients who have negative celiac serologies. Patients typically present with diarrhea and weight loss. The length of time between olmesartan use and onset of symptoms is highly variable and ranges from a few weeks to 10 years. Histologically, olmesartan enteropathy may mimic all of the features of CD, with most cases showing villous blunting and increased IELs. Nearly one-third of cases also show features of collagenous sprue ( Fig. 5.15 ). Finally, these patients often have concomitant collagenous or lymphocytic gastritis or colitis. Symptoms quickly resolve with cessation of drug. Other members of the ARB family result in a similar sprue-like enteropathy. In general, it is prudent to consider ARB-induced mucosal injury in a patient with celiac-seronegative enteropathy.

Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here