Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Hereditary hemochromatosis is an inherited disorder that leads to iron overload due to excessive absorption of dietary iron because of a deficiency of hepcidin, which is an iron regulatory peptide hormone. A homozygous C282Y mutation in HFE is responsible for the vast majority of hereditary cases. However, iron overload due to hyperabsorption of dietary iron may result from other genetic mutations, chronic anemias associated with ineffective erythropoiesis (e.g., thalassemia; Chapter 148 ), chronic liver disease, and iatrogenic causes (see Table 196-1 ).
| HEREDITARY HEMOCHROMATOSIS |
| Type 1- HFE Related Autosomal Recessive |
| C282Y/C282Y C282Y/H63D Other mutations |
| Type 2-4-Non– HFE Related |
| Hemojuvelin ( HJV ) mutations (autosomal recessive) Hepcidin ( HAMP ) mutations (autosomal recessive) Transferrin receptor 2 ( TFR2 ) mutations (autosomal recessive) Ferroportin ( SLC40A1 ) mutations (autosomal dominant) Aceruloplasminemia Divalent metal transporter 1 ( SLC11A2 ) mutations (rare) Atransferrinemia |
| Miscellaneous |
| African iron overload Neonatal iron overload (rare) |
| SECONDARY IRON OVERLOAD |
| Anemia Caused by Ineffective Erythropoiesis |
| Thalassemia major Sideroblastic anemias Congenital dyserythropoietic anemias |
| Liver Disease |
| Alcoholic liver disease Chronic viral hepatitis B and C Porphyria cutanea tarda Nonalcoholic steatohepatitis After portacaval shunt |
| Miscellaneous |
| Transfusional iron overload Excessive parenteral iron administration |
Three common HFE mutations (C282Y, H63D, and S65C) have been described in the general population, but only the homozygous C282Y and the C282Y/H63D compound heterozygous mutations have been associated with the hemochromatosis phenotype in the absence of another contributing factor for iron overload. A North American study that screened almost 100,000 patients found an estimated prevalence of C282Y homozygosity in 0.44% of non-Hispanic Whites, 0.11% of Native Americans, 0.027% of Hispanics, 0.014% of Blacks, 0.12% of Pacific Islanders, and 0.000039% of Asians. The H63D mutation is associated with phenotypic hemochromatosis only when present along with the C282Y mutation, whereas the S65C mutation is considered a polymorphism and has not been shown to contribute to iron overload. The true prevalence of these other, nonclassic forms 2 to 4 are unknown.
Dietary iron absorption is regulated by body iron needs. Under circumstances of iron deficiency, iron absorption is increased until body iron stores are replete, at which time iron absorption decreases to basal levels. Iron from the diet is absorbed in the proximal duodenum either as inorganic iron or as heme iron ( Fig.196-1 ). Iron balance in humans is regulated primarily at the level of intestinal absorption because the body does not have a mechanism to excrete iron other than by the physiologic loss of desquamated cells or by menses. Hepcidin, which is a 25–amino acid peptide hormone synthesized mainly in the liver and secreted into the blood, is considered the master regulator of iron homeostasis. Hepcidin acts by blocking iron absorption from the intestine and blocking iron release from reticuloendothelial cell storage sites, which are mainly within the liver (see Fig. 196-1 ). Genetic iron overload disorders are caused when mutations in iron regulatory genes result in reduced production and inappropriately low levels of hepcidin in response to stored and circulating levels of iron, the net effect of which is lifelong hyperabsorption of dietary iron. Progressive iron loading of various organs can eventually cause the characteristic tissue injuries of the hemochromatosis phenotype.
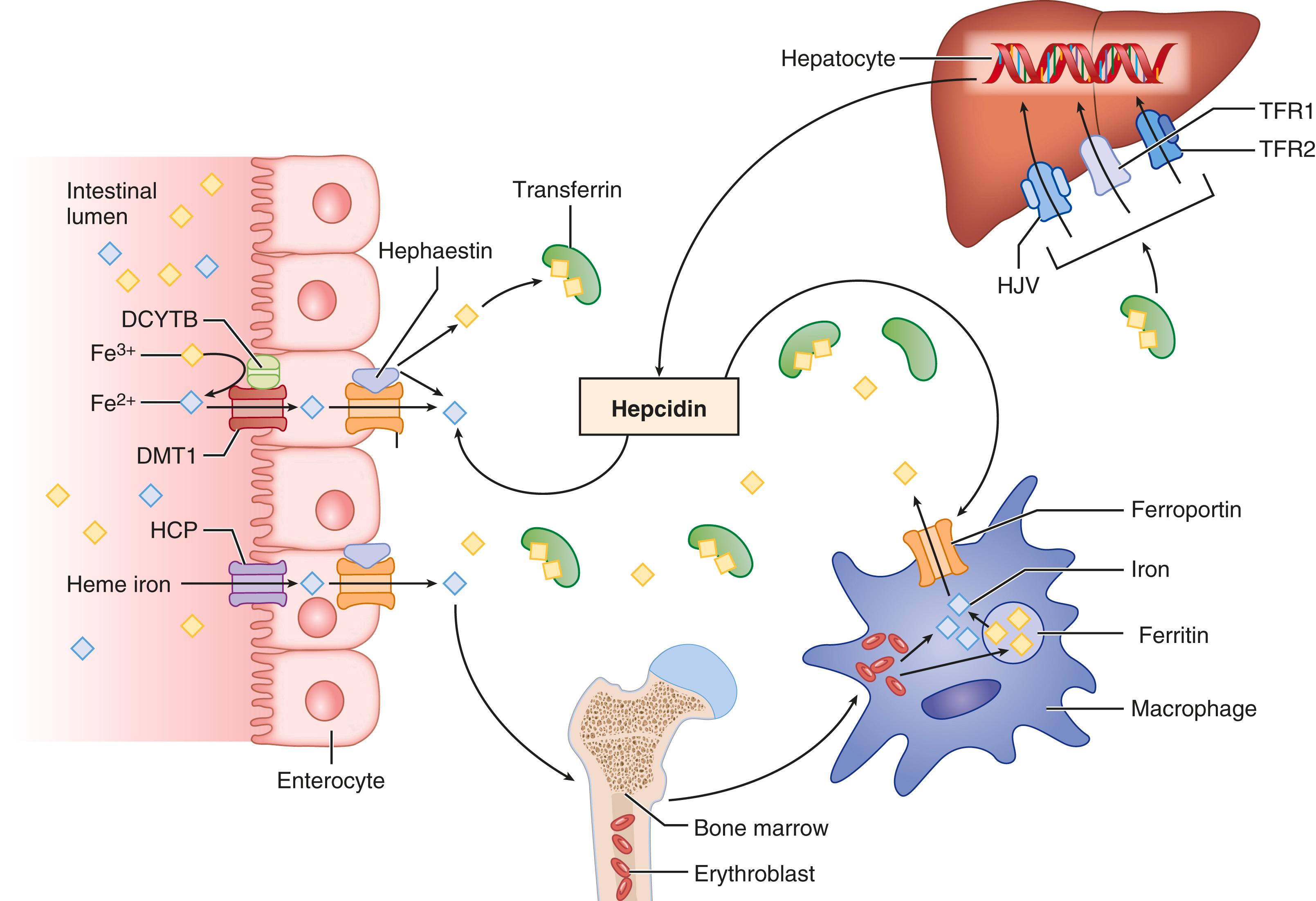
Absorption of inorganic iron follows a coordinated process beginning with conversion of iron from the ferric form to the ferrous form by the duodenal cytochrome b–related ferric reductase (dcytb) that is present on the luminal surface of duodenal enterocytes. Ferrous iron then traverses the apical membrane of enterocytes via the divalent metal transporter 1. The absorbed iron may be utilized for intracellular processes, stored as ferritin in enterocytes, or converted back to ferric iron by hephaestin, thereby allowing it to be transferred across the basolateral membrane of enterocytes into circulating plasma via the iron export protein, ferroportin. Iron is bound to transferrin at the transferrin receptor (TfR1) in the circulation. Hepatocytes take up transferrin-bound iron via the receptors TfR1 and possibly TfR2, as well as by non–receptor-mediated mechanisms. Hepatocytes may also absorb free iron from the circulation when transferrin becomes highly saturated. In contrast, reticuloendothelial system cells (Kupffer cells in the liver) take up iron primarily by phagocytosis of senescent erythrocytes or possibly apoptotic hepatocytes. Kupffer cells can also sequester transferrin-bound iron via TfR1.
Mutations in the HFE , TfR2 , and HJV genes result in markedly depressed hepcidin levels, thereby permitting persistently increased absorption of iron from a normal diet. Mutations in the HAMP (hepcidin) gene may likewise lead to inappropriately low levels of hepcidin, thereby resulting in iron overload. Parenchymal iron overload may also be caused by mutations in the ferroportin gene.
Classical ( HFE ) hemochromatosis is defined as type 1 hemochromatosis, whereas other rare types of hereditary hemochromatosis are now classified as types 2 to 4. , Juvenile hemochromatosis, which is a severe form of hemochromatosis that is associated with severe iron overload and that presents in the second to third decade of life, consists of type 2A and type 2B forms. Type 2A is caused by a mutation in the hemojuvelin ( HJV ) gene, which is located on chromosome 1 and which encodes the hemojuvelin protein. Type 2B is caused by a mutation in the hepatic antimicrobial protein ( HAMP ) gene, which encodes hepcidin. Type 3 is due to a mutation in the transferrin receptor 2 ( TFR2 ) gene that is located on the long arm of chromosome 7; TFR2 is similar to TFR1 but expressed predominantly in the liver. Type 4 is caused by dominant gain-in-function mutations in the ferroportin ( SLC40A1 ) gene, which is located on chromosome 2, thereby leading to hepcidin resistance. A different mutation resulting in loss of function in ferroportin has been termed “ferroportin disease” because its clinical, biochemical, and histologic features are very different from other forms of hereditary hemochromatosis.
Population-based screening studies have shown that the penetrance and clinical expressivity of the homozygous C282Y mutation is low, especially in women. A number of factors may be associated with phenotypic expression, such as physiologic blood loss due to menses or factors leading to increased iron absorption (e.g., excess alcohol intake or other liver diseases such as chronic hepatitis C or steatohepatitis) and increased intake of ascorbic or citric acid.
Iron overload is associated with reduced function in immune cells. It is also associated with altered regulation of CD8 T lymphocytes from both HFE patients and Hfe -null mice. Siderophilic microbes grow rapidly in niches where iron is more available, and these organisms may cause severe infections in patients with iron overload. The putative mechanisms for diabetes include direct damage to pancreatic beta cells by iron, with some component of insulin resistance in the liver due to the organ’s iron loading. Hypothyroidism is thought to be due to direct iron toxicity in the thyroid gland, whereas rare hypogonadotropic hypogonadism and adrenal insufficiency in hereditary hemochromatosis may be due to iron deposition in the anterior pituitary. Arthropathy is believed to be caused by the deposition of calcium pyrophosphate crystals in joint spaces, thereby leading to inflammation, joint space narrowing, chondrocalcinosis, formation of subchondral cysts, and osteopenia.
The clinical features vary among the different forms of genetic hemochromatosis ( Table 196-2 ). Many patients who carry HFE mutations, whether heterozygous or homozygous, do not express the clinical phenotype of iron overload disease ( Tables 196-3 and 196-4 ) or even biochemical evidence of iron overload (increased serum transferrin-iron saturation, ferritin), and the majority of patients with the homozygous C282Y mutation may never develop end-organ damage from iron overload. For example, among C282Y homozygotes with normal serum ferritin values at diagnosis, less than 15% develop ferritin values >1000 ng/mL over 12 years of follow-up. An estimated 50% of female and 25% of male adults who are homozygous for the C282Y mutation have normal serum ferritin levels and will never require phlebotomy therapy. However, about 1 in 10 male C282Y homozygotes will eventually develop liver disease if hemochromatosis is not diagnosed early, usually by screening, and treated.
| TYPE | AGE | MUTATION/GENE | INHERITANCE | COMMON CLINICAL FEATURES |
|---|---|---|---|---|
| 1A | Middle-age | C282Y+/+ HFE |
AR | Arthropathy, diabetes, hypothyroidism, hypogonadism, adrenal insufficiency, liver disease, endocrine dysfunction, cardiomyopathy, skin discoloration |
| 1B | Middle-age | C282Y/H63D HFE |
AR | |
| 2A | <30 years | G3290V /HJV | AR | Hypogonadism, cardiomyopathy |
| 2B | <30 years | Several /HAMP | AR | |
| 3 | Intermediate | Several/ TfR2 | AR | May have juvenile hereditary hemochromatosis or type 2 features |
| 4 | Variable | Several/ SLC40A1 | AD | Hepcidin resistance, fatigue, joint pain |
| ASYMPTOMATIC |
| Abnormalities of serum iron studies on routine screening chemistry panel Abnormal liver test results Identified by family screening Identified by population screening |
| NONSPECIFIC SYSTEMIC SYMPTOMS |
| Weakness Fatigue Lethargy Apathy Weight loss |
| SPECIFIC ORGAN-RELATED SYMPTOMS |
| Abdominal pain (hepatomegaly) Arthralgias (arthritis) Symptoms of diabetes mellitus (pancreas) Amenorrhea (cirrhosis) Loss of libido, impotence (pituitary, cirrhosis) Heart failure symptoms (heart) Arrhythmias (heart) |
| ASYMPTOMATIC |
| No physical findings Hepatomegaly |
| SYMPTOMATIC |
| Liver |
| Hepatomegaly Cutaneous stigmata of chronic liver disease Splenomegaly Signs of liver failure: ascites, encephalopathy |
| Joints |
| Arthritis Joint swelling |
| Heart |
| Dilated cardiomyopathy Congestive heart failure |
| Skin |
| Increased pigmentation |
| Endocrine |
| Testicular atrophy Hypogonadism Hypothyroidism |
Furthermore, the H63D mutation does not result in iron overload unless present in compound heterozygous form with C282Y. In addition, the S65C mutation is now recognized to be a polymorphism that is not associated with iron overload.
Based on genetics, phenotypic expression, and long-term natural history, hereditary hemochromatosis may be classified into three clinical stages, as shown:
Stage 1: genetic predisposition for hereditary hemochromatosis without evidence for increase in iron stores (normal serum iron studies)
Stage 2: genetic predisposition for hereditary hemochromatosis and some phenotypic characteristics of iron overload in the absence of organ damage
Stage 3: genetic predisposition for hereditary hemochromatosis accompanied by evidence for iron overload with tissue injury or organ damage
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here