Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Parasitic infections remain an important cause of pulmonary disease throughout the world, although less frequently encountered in resource-rich countries. The most common protozoal parasite–associated lung diseases throughout the world are caused by Plasmodium falciparum (falciparum malaria) and Toxoplasma gondii (toxoplasmosis). Helminths cause a variety of pulmonary diseases. The most common diseases caused by nematodes include the transient disease associated with transpulmonary migration, the aberrant migration of the larvae of zoonotic pathogens (ie, visceral larva migrans [VLM]), hypersensitivity reactions to parasite antigens, and infection by Strongyloides stercoralis, which includes Strongyloides hyperinfection syndrome. Echinococcosis is the most important pulmonary disease caused by a cestode, but the recognition of pulmonary cysticercosis is also critical. Paragonimus is the most commonly recognized trematode that causes pulmonary disease, but schistosomes, because of their wide range of endemicity and the vast number of people infected, also are an important cause of pulmonary disease. The recognition of these infections is important, because specific antiparasitic chemotherapy and/or surgery are required to produce a cure.
In excised tissues, parasites may be dead, degenerated, incompletely sampled, or tangentially sectioned, posing a diagnostic dilemma for the pathologist. Under such conditions, a specific diagnosis based on parasite morphology can only be achieved when unique morphologic features of the organisms are present. The definitive identification of parasites in human tissue may not be possible in some instances. A thorough understanding of the parasites most likely to be encountered in the lungs and their morphologic features, however, helps to suggest the correct possibility. The definitive diagnosis is often achieved by using a combination of studies, which in addition to the morphologic findings includes serology, a thorough history to evaluate the possibility of exposure (eg, travel to an endemic area) and molecular diagnostics.
More than 40 types of parasites can be found in the lungs of humans ( Table 14.1 ). Two of these parasites, Paragonimus and Echinococcus species, preferentially infect the lungs. The remainder are either lost in the wrong tissue or host in transit to another anatomic site, or pulmonary involvement is part of disseminated disease. This review is not comprehensive and describes only the parasites that are most likely to be encountered by the practicing pathologist and clinician in North America. It focuses primarily on pulmonary manifestations of these organisms. The reader who desires additional information is referred to the references at the end of this chapter.
| Protozoa | Metazoa | |
|---|---|---|
| More Common ∗ | Plasmodium falciparum and Toxoplasma gondii | Dirofilaria immitis, Strongyloides stercoralis, Toxocara species, Paragonimus species, Schistosoma species, Taenia solium, Echinococcus species |
| Less Common ∗ | Entamoeba histolytica, Cryptosporidium species, Leishmania donovani, Trypanosoma cruzi, Acanthamoeba species, and Trichomonas tenax | Wuchereria bancrofti, Brugia malayi, Onchocerca volvulus, Capillaria aerophila, Mammomonogamus laryngeus, Ascaris lumbricoides, Ascaris suum, Mansonella perstans, Angiostrongylus cantonensis, Halicephalobus deletrix, Metastrongylus elongates, Enterobius vermicularis, Lagochilascaris minor, Baylisascaris procyonis, anisakids, Gnathostoma spinigerum, Alaria species, Fasciola hepatica, Clinostomum complanatum, Spirometra species, mites, fly larvae, Armillifer species, Linguatula serrata, and Limnatis nilotica |
∗ More common and less common do not refer to the overall prevalence of infection, but rather to the frequency of the parasite reported in histologic sections from the lung. For example, although Ascaris is one of the most common nematode infections in the world, it causes minimal pulmonary disease during pulmonary transmigration and therefore is infrequently encountered in histologic sections from the lung.
Protozoal parasites that infect the lungs are not limited to this anatomic site but are usually part of a disseminated, multiorgan infection. The following discussion is focused on infections caused by Plasmodium species ( P. falciparum [falciparum malaria] and Toxoplasma gondii [toxoplasmosis]), because of their wide distribution and the severity of the diseases they may cause.
Five Plasmodium species, P. falciparum, P. vivax, P. ovale, P. malariae, and P. knowlesi, cause human malaria
P. falciparum causes the most serious form of malaria and can cause pulmonary disease
Malaria, transmitted by the Anopheles mosquito, kills approximately 2 million people each year
Disease is present in all hemispheres but is more prevalent in tropical and subtropical areas
Malarial lung presents approximately 2 to 3 days after the onset of fever and chills
Pulmonary presentation is variable, but progression is usually rapid from cough/dyspnea to severe hypoxia and respiratory arrest
Iatrogenic fluid overload and secondary bacterial pneumonia are also risks for these patients
Radiographic changes manifest from 6 to 24 hours after the onset of dyspnea
Chest radiographs show signs of pulmonary edema
The mortality rate for those with severe falciparum malaria is 15% to 30% even with intensive care treatment; pregnant women are at a particularly high risk
Pulmonary edema in falciparum malaria is a poor prognostic sign, with 80% mortality without ventilatory support
Death usually occurs within 3 to 8 days after the onset of malaria and usually within 24 hours of the recognition of pulmonary involvement
Typical treatments for cardiogenic pulmonary edema are not helpful
Successful treatment will include antimalarials, maintaining optimal ventilation, managing metabolic acidosis and hypovolemia while avoiding overhydration, and prompt treatment for secondary bacterial infections
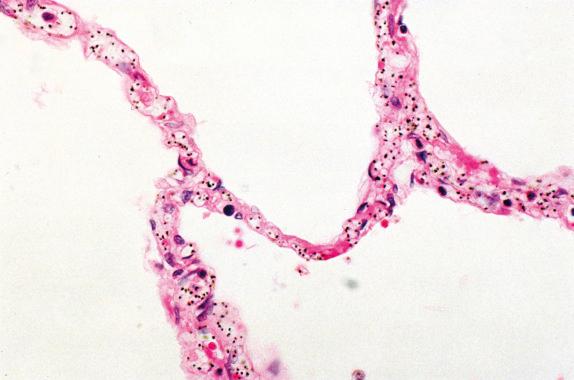
Severe pulmonary edema, with capillary congestion and thickened alveolar septa
Macrophages with hemozoin pigment and infected erythrocytes within the capillaries
Diffuse alveolar damage occurs in 50% of patients
Although malaria is far less common in North America than in previous centuries, it remains one of the most serious public health problems in the world. This disease kills approximately 2 million people each year, mostly children. Five species of Plasmodium are responsible for human disease: P. falciparum, P. vivax, P. ovale, P. malariae, and the very infrequently encountered P. knowlesi. The most serious form is falciparum malaria; this is the type that most commonly causes pulmonary manifestations (ie, malarial lung). Although pulmonary involvement is most common with P. falciparum, P. vivax has been shown to rarely cause acute respiratory distress syndrome.
The clinical descriptions of soldiers with pulmonary malaria has varied, with symptoms ranging from dry cough to acute respiratory distress similar to that encountered in patients with asthma. Pulmonary disease is usually associated with evidence of multisystem progression, such as central nervous system involvement (ie, cerebral malaria) and kidney involvement (acute renal failure). Characteristically, the onset of malarial lung is abrupt and progresses rapidly from cough/dyspnea to severe hypoxia and respiratory arrest. The terminal event is usually preceded by breathing abnormalities, such as Cheyne-Stokes breathing, suggestive of brainstem dysfunction.
Acute pulmonary insufficiency, without cardiac decompensation or fluid overload, occurs in approximately 7% of nonimmune patients with falciparum malaria and presents usually 2 to 3 days after the onset of fever and chills. Although initially the respiratory rate, blood pressure, and clinical examination may be normal, the clinical features of these patients change rapidly. The first clinical signs of pulmonary involvement may be circumoral cyanosis, dyspnea, and increased respiratory rate. The cyanosis spreads to the face and extremities. A spasmodic cough may develop, and there is a decrease in the hematocrit. Scattered rhonchi and rales may be heard in the basal aspects of the lungs, and foamy, blood-tinged sputum may be produced. These are poor prognostic signs, and death usually occurs 24 hours after the recognition of malarial involvement of the lung. In addition to the malaria-induced lung injury, iatrogenic fluid overload and secondary bacterial pneumonias are perils for these patients.
The chest radiograph is normal in the early phases of pulmonary involvement, with changes becoming discernible 6 to 24 hours after the onset of dyspnea. Signs of pulmonary edema appear, such as a generalized increase in the interstitial markings, and progress to fluffy infiltrations in both lungs. Evidence of a secondary bacterial pneumonia may also be detected.
The lungs of patients with pulmonary involvement by P. falciparum are congested, edematous, and heavy. Focal hemorrhages may be seen throughout, and pink foamy fluid fills the airways. A serosanguineous pleural effusion may be present. Areas of consolidation will be evident if secondary bacterial pneumonia is present.
Microscopically, there is severe pulmonary edema with capillary congestion and thickened alveolar septa. Hyaline membranes are present in approximately 50% of patients, but thrombosis and infarcts are absent. The edema that fills the alveoli contains macrophages that, in turn, contain hemozoin pigment ( Fig. 14.1 ). The astute observer may note the presence of infected erythrocytes within the capillaries of the thickened alveolar septa. Evidence of disseminated intravascular coagulation is usually absent, but rarely has been described. Secondary bacterial pneumonia, usually bronchopneumonia, is often present.
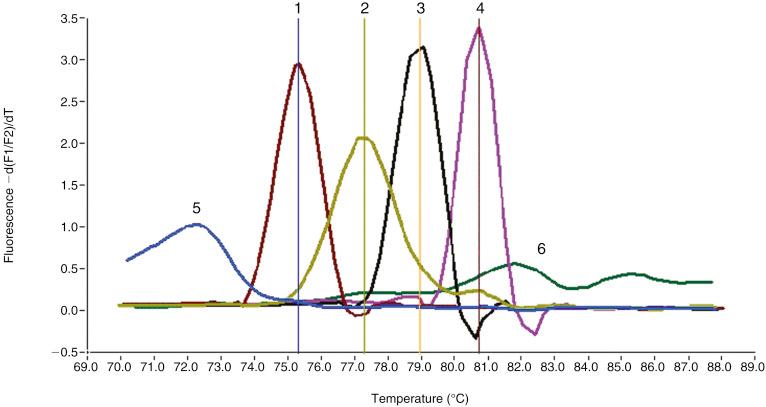
Rapid antigen detection assays are useful in the field and are comparable to good microscopy. At least three thick and thin blood films performed 8 to 12 hours apart should be performed before malaria is excluded from the differential diagnosis. A variety of nucleic acid amplification methods have been described for the detection and differentiation of Plasmodium species, including real-time polymerase chain reaction (PCR) assays that detect all four Plasmodium species and differentiate them based on postamplification melt curve analysis ( Fig. 14.2 ).
The differential diagnosis of malaria is extensive and includes diseases such as typhoid fever, typhus, dengue, and the cyclic fevers caused by filarial worms, among others. Other differential diagnostic considerations for the pulmonary manifestations in patients with falciparum malaria include secondary bacterial pneumonia and metabolic acidosis.
The severity of falciparum malaria depends on a variety of factors, such as a previous infection (ie, some degree of immunity), the host inflammatory response, and pregnancy, among others. The mortality for those who develop severe falciparum malaria is high, with a rate of 15% to 30% even with intensive care treatment. In contrast, the mortality of uncomplicated acute falciparum malaria is about 0.4%. There is a high mortality associated with development of malarial lung.
Pulmonary edema associated with severe falciparum malaria may present at any time during the course of illness, even when the patient is improving clinically and there is a resolution or reduction in the parasitemia. Pregnant women are at a particularly high risk. The mortality rate for patients with pulmonary edema who do not have ventilatory support is approximately 80%. However, even with ventilatory support, the mortality rate of affected patients is higher than those without pulmonary involvement. Patients so affected often die within 3 to 8 days after onset of malaria, usually within 24 hours of the recognition of pulmonary involvement.
The prompt diagnosis and treatment of falciparum malaria may curtail the development of malarial lung. Successful treatment of malaria includes the administration of antimalarials and maintenance of optimal ventilation. The use of exchange transfusion is controversial, but may benefit some patients. Patients with complicating bacterial pneumonia should be treated with appropriate antibacterial agents.
Pulmonary toxoplasmosis, which is part of disseminated disease caused by Toxoplasma gondii, occurs with congenital toxoplasmosis and with infections predominantly of immunocompromised patients
T. gondii infection is highly prevalent throughout the world
Oocysts are passed in the feces of the definitive hosts, members of the family Felidae (ie, cats), and are consumed by humans and other intermediate hosts, wherein they asexually replicate and encyst
Infection of people with an intact immune system is subclinical or consists of a mononucleosis-like syndrome, with or without localized lymphadenopathy
Disseminated infections occur in congenital disease and in patients with AIDS and other types of immuncompromising conditions
Pulmonary involvement occurs in more than 70% of immunocompromised patients with disseminated toxoplasmosis, and the likelihood of death is high in this situation
A nonproductive cough and dyspnea are the most common symptoms, and fever is the most common sign
Radiographic findings include bilateral diffuse pneumonia, miliary nodules, and interstitial and lobar infiltrates
CT scans may show ground-glass opacities and superimposed septal thickening and intralobular linear opacities
Pleural effusions may be present
The prognosis is poor for patients with disseminated toxoplasmosis; the death rate is up to 92% in bone marrow transplant recipients who develop pulmonary toxoplasmosis
The prophylaxis and treatment of choice is pyrimethamine/sulfadoxine, but this therapy kills only the proliferating tachyzoites, not the quiescent bradyzoite cysts
Prevention of primary toxoplasmosis includes avoiding contact with cat feces, good hand-washing, washing of fruits and vegetables, and thorough cooking of meats before consumption
The proliferative form of Toxoplasma, which is morphologically diagnostic, is the crescentic, subtly pyriform tachyzoite
Pseudocysts and true cysts are also present
Coagulation necrosis, an alveolar fibrinous exudate, interstitial chronic inflammation, and edema are present
Toxoplasma gondii infection is highly prevalent throughout the world, and it is estimated that 50% of the world population is infected. Herein, we concentrate on the far less common pulmonary manifestations of toxoplasmosis.
People with an intact immune system who become infected respond to and contain the parasite. Such patients may be asymptomatic, develop a mononucleosis-like syndrome or develop localized lymphadenopathy. Disseminated T. gondii infections, which may have pulmonary involvement, occur in three clinical situations: (1) congenital toxoplasmosis, (2) in patients with AIDS, and (3) in patients with a non-HIV-associated immunosuppressive condition. In congenital toxoplasmosis, the infection is acquired during pregnancy and transmitted from the mother to the fetus. Congenital toxoplasmosis may be acute, subacute, or chronic. Acute severe disease results in death in utero. Parasitic infection with T. gondii is an important cause of morbidity and mortality for patients with advanced HIV infection or AIDS. Cerebral toxoplasmosis is most common in these patients, secondary to the reactivation of latent bradyzoite cysts. Patients with AIDS will less commonly (approximately 2%) have extracerebral toxoplasmosis, with a 0.5% prevalence of pulmonary toxoplasmosis. The non-AIDS immunocompromising conditions associated with toxoplasmosis are hematopoietic malignancy, transplantation (particularly stem cell/bone marrow transplantation), and conditions wherein high-dose or prolonged corticosteroid use is employed. Disseminated toxoplasmosis in these patients may have a prominent pulmonary component. Far less commonly, pulmonary toxoplasmosis may occur in immunocompetent individuals who become infected or in patients with other conditions (eg, lung cancer).
Pulmonary involvement occurs in more than 70% of patients with disseminated toxoplasmosis. A nonproductive cough and dyspnea are the most common symptoms, and fever is the most common sign. In some patients, the presenting or most significant features may be empyema or pleural effusion. Infection of lung tissue indicates dissemination of the parasite. There is a high likelihood of death in these patients from bronchopneumonia or meningoencephalitis.
Chest radiographs lack both sensitivity and specificity; chest computed tomography (CT) scanning is superior for more sensitive detection of findings. Radiographic patterns described in patients with pulmonary toxoplasmosis include bilateral diffuse pneumonia, miliary nodules, and interstitial and lobar infiltrates. CT scans may show ground-glass opacities and possibly superimposed septal thickening and intralobular linear opacities. Hilar and mediastinal lymphadenopathy is usually absent, but may be present depending on the degree of immunocompromise. Pleural effusion has also been reported.
At autopsy, the lungs are heavily congested, with petechial hemorrhages and areas of consolidation and necrosis.
The proliferative form of Toxoplasma is the crescentic, subtly pyriform tachyzoite. It may be detected in cytologic preparations from sputa or bronchoalveolar lavage (BAL) fluid. It is 4 to 8 microns in smear preparations, but in histologic sections it is approximately half this size. Tachyzoites, which are plentiful in fulminant disease, stain well with the routine hematoxylin-eosin stain but can also be highlighted with the Giemsa or eosin-methylene blue stains. Oil immersion microscopy may be necessary because of the small size of these organisms. Tachyzoites can likely replicate in any nucleated cell but are most frequently seen in the brain, heart, liver, intestine, lungs, and lymph nodes.
Coagulation necrosis and an alveolar fibrinous exudate are seen in the lungs, with chronic inflammation and edema. Many alveoli may be collapsed and contain cells packed with tachyzoites. Pseudocysts are prevalent in areas of necrosis. True cysts may also be observed ( Fig. 14.3 ). True cysts, which are present in chronic disease, may be differentiated from the pseudocysts of acute toxoplasmosis with histochemical stains. The periodic acid–Schiff (PAS) and Gomori methenamine silver stains highlight true cysts; the cyst wall is usually argyrophilic, and bradyzoites are usually PAS-positive. In contrast, pseudocyst walls and tachyzoites stain weakly with those reagents but are readily identified in hematoxylin and eosin–stained sections in tissues from patients with fulminant disease.
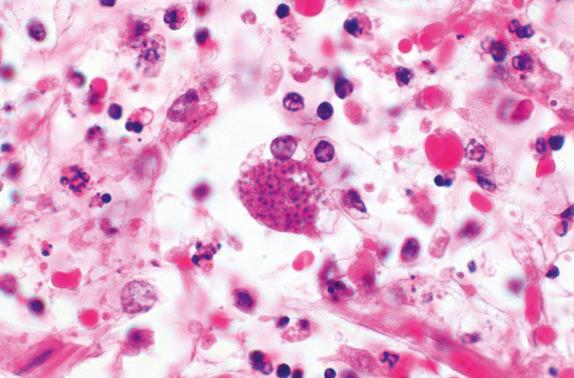
Serologic studies are useful for the diagnosis of toxoplasmosis. The presence of IgM antibody or a fourfold rise in the IgG antibody is indicative of acute infection. The presence of stable IgG antibody is indicative of prior infection, which means the patient likely has bradyzoite cysts dormant in his or her tissues and is at risk for reactivation toxoplasmosis in the event of profound immunosuppression.
Immunohistochemical staining has been described as likely superior to histochemical staining for the detection of rare organisms. However, because a large number of organisms are usually present in dissemination disease, it is questionable whether immunohistochemistry is truly necessary to achieve the diagnosis. Nucleic acid amplification assays, such as the PCR, hold the greatest promise as a useful ancillary method of detection. PCR assays have been used to detect toxoplasmosis in amniotic fluid, cerebrospinal fluid, aqueous humor, blood, and BAL fluid. Currently, there are no Food and Drug Administration–approved molecular diagnostic assays for the detection of T. gondii.
The differential diagnosis of congenital toxoplasmosis includes other infectious diseases known to cause intrauterine fetal demise, such as cytomegalovirus (CMV) and herpes simplex virus. There are numerous causes of pneumonia in immunocompromised patients, including opportunistic pathogens such as Pneumocystis jirovecii, Aspergillus, zygomycetes, Nocardia, and multiple viruses, as well as agents causing pneumonia in healthy hosts.
The prognosis is poor for patients with disseminated toxoplasmosis. A 92% death rate has been reported for bone marrow transplant recipients who develop pulmonary toxoplasmosis, with approximately half of the deaths occurring within 3 days after the onset of symptoms. The prophylaxis and treatment of choice is with pyrimethamine/sulfadoxine. This therapy kills the proliferating tachyzoites but is not active against quiescent bradyzoite cysts. Therefore the immunocompromised patient remains at risk for reactivation toxoplasmosis at any time.
Preventive measures to avoid contracting primary toxoplasmosis include avoidance of contact with cat feces, good hand-washing, thorough washing of fruits and vegetables, and complete cooking of meats before consumption. These measures are helpful in preventing congenital toxoplasmosis associated with a primary infection but are not helpful in reducing reactivation toxoplasmosis associated with immunosuppression.
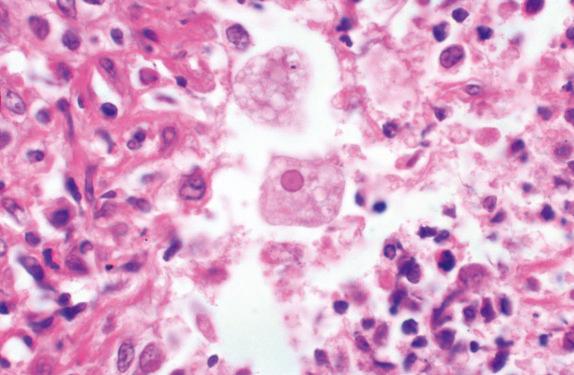
A variety of other protozoa have been identified as causes of human pulmonary infections, including Cryptosporidium species, Entamoeba histolytica ( Fig. 14.4 ), Trypanosoma cruzi, and Leishmania donovani.
Ascaris lumbricoides and other roundworms, such as hookworms and filariae, infect millions of people throughout the world but are a relatively rare cause of clinically apparent pulmonary disease. Part of the success of these parasites is that the migration, maturation, and subsequent dwelling in the intestine usually cause minimal tissue damage (ie, the most biologically successful parasites do not significantly harm the host). The significant conditions caused by nematodes, for the most part, may be separated into several categories: (1) hyperinfection of the immunocompromised host by Strongyloides stercoralis; (2) the aberrant migrations of animal nematodes (ie, VLM) and, more rarely, nematodes that normally infect humans; and (3) hypersensitivity reactions to parasite antigens.
Strongyloidiasis, an infection by Strongyloides stercoralis, causes a spectrum of acute disease, uncomplicated chronic disease, or complicated chronic disease, which includes the hyperinfection syndrome of the immunocompromised
S. stercoralis is a geohelminth that exists in either a parasitic life cycle or a free-living life cycle and is endemic throughout the world, predominantly in tropical and subtropical regions
Prevalence figures range from 7% to 48%; rural villages with poor sanitation are common sites for the transmission of Strongyloides
Acute disease begins with a transient, mild dermatitis that is pruritic and erythematous, followed about a week later by cough and possibly sore throat, and then by nonspecific abdominal complaints
Uncomplicated chronic strongyloidiasis may result in nutritional deficits, but infection is often subclinical and remains undetected and persistent for years
Complicated chronic strongyloidiasis manifests in the elderly and those with an immunocompromising condition or taking immunosuppressive medications; the severity of pulmonary symptoms correlates with the number of migrating larvae; hyperinfection syndrome may cause death
The radiologic findings after initial infection range from normal to the transient pulmonary infiltrates of Löeffler syndrome, whereas patients with uncomplicated chronic strongyloidiasis usually have normal chest radiographs
The radiologic findings associated with the Strongyloides hyperinfection range from a normal chest radiograph (less common) to nonsegmental patchy infiltrates, nodular infiltrates, or multiple infiltrates.
No appreciable mortality is associated with acute and uncomplicated chronic disease, whereas the mortality of Strongyloides hyperinfection syndrome is 50% to 86%
Thiabendazole or ivermectin are excellent therapeutic options
Hyperinfection requires hospitalization, prolonged courses of an antiparasitic agent, and possibly long-term antiparasitic suppressive therapy and reduction of immunosuppression when feasible
Serum immunoglobulin levels may be used to follow therapy
Morphologic detection and identification of larvae in sputum, BAL fluid, other body fluids, or feces confirms the diagnosis; stool examination also reveals rhabditiform larvae
Invasive filariform larvae of S. stercoralis can be seen in the lung
Pulmonary host response includes neutrophils, macrophages, and eosinophils; abscesses and cavities can occur
Strongyloidiasis refers to an infection with Strongyloides stercoralis, a geohelminth that has both a parasitic and a free-living life cycle. The areas of endemicity are widespread in tropical and subtropical regions, including the southeastern United States. The infective filariform larvae of this helminth, like the cercariae of schistosomes and the larvae of hookworms, have the ability to penetrate intact human skin. After skin penetration, the larvae migrate to the lungs, where they penetrate the alveoli and enter the airways. The larvae are then swallowed and mature into adult females in the distal stomach and small intestine. The parasitic female S. stercoralis is parthenogenetic and thus does not require a male for reproduction. Unlike other intestinal helminths that produce eggs that are passed intact in the stool and embryonate in the soil, the eggs of Strongyloides release rhabditiform larvae within the intestinal tract that are then passed in the feces. Rhabditiform larvae may differentiate into either infective filariform larvae or mature into male and female adults. Male and female adults copulate, and the gravid female produces eggs; this is the free-living, nonparasitic cycle. Alternatively, the rhabditiform larvae may differentiate into the infective filariform larvae in the intestinal tract. This feature affords the parasite the opportunity to autoinfect the host, which may occur internally through the bowel wall or externally through the skin in the perianal region. This difference between Strongyloides and other intestinal geohelminths is important, because it is pivotal for the establishment of chronic strongyloidiasis and, in immunocompromised hosts, the commonly fatal hyperinfection.
The clinical manifestations of strongyloidiasis may be separated into acute disease, uncomplicated chronic disease, and complicated chronic disease, which includes the hyperinfection syndrome.
In acute disease, there is a transient, mild dermatitis that is pruritic and erythematous, which occurs after filariform larvae penetrate the skin and begin migration. About a week later, cough and sore throat may develop, followed by nonspecific abdominal complaints, such as diarrhea and a feeling of fullness.
Patients who have chronic strongyloidiasis may have nutritional deficits, but most often the infection is subclinical, remains undetected, and persists for years secondary to low-grade reinfection. Between 15% and 30% of patients with uncomplicated chronic strongyloidiasis are asymptomatic. The remainder have nonspecific findings similar to those described earlier. Gastrointestinal symptoms are most common at this stage, followed by pulmonary complaints that may include wheezing and dyspnea.
Complicated chronic strongyloidiasis manifests in the elderly and those with an immunocompromising condition or those who are taking immunosuppressive medications. Although the reproductive activity of the worms in patients with chronic uncomplicated strongyloidiasis is regulated in some manner so that the host is not in danger from an enormous worm burden, the host–parasite regulatory mechanism is lost in patients with compromised immunity, and increasing autoinfection leads to life-threatening hyperinfection. In the gut of hyperinfected patients, numerous adult worms produce abundant rhabditiform larvae that molt into filariform larvae that migrate through most organs, particularly the lungs. The severity of pulmonary symptoms correlates with the number of migrating larvae. Pulmonary symptoms consist of dyspnea, cough, hemoptysis, cyanosis, and respiratory distress. Eosinophilia may be present, but is often suppressed due to the effects of corticosteroids.
The initial infection and the transient migration of Strongyloides larvae, and for that matter, the transient transpulmonary migration of any human intestinal helminths (eg, A. lumbricoides ), result in changes that range from normal to the transient pulmonary infiltrates of Löeffler syndrome. Patients with uncomplicated chronic strongyloidiasis usually have normal chest radiographs. The radiologic findings associated with the Strongyloides hyperinfection include normal chest radiographs, nonsegmental patchy infiltrates, nodular infiltrates, or multiple infiltrates. Lung abscesses, nodules that mimic tuberculosis, diffuse infiltrates, and pleural effusions may also be seen.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here