Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Surgical risk at experienced centers can be accurately determined.
Preoperative use of functional MRI, diffusion tensor imaging, and Wada testing along with routine MRI, CT angiography, and digital subtraction angiography greatly facilitates interventional risk assessment.
Location, size, venous drainage, perforating vessels, and diffuseness are key to surgical risk assessment in association with age, medical comorbidities, and acuteness in surgical episode.
Multimodality therapy is often employed, but currently there is no combined risk assessment scale.
Considering all variables—location and vascular anatomy are the key risk factors in intervention.
Intracranial arteriovenous malformations (iAVMs) have an incidence of 1.12–1.42 per 100,000 person-years, and patients most commonly present with hemorrhage (38%–86%), followed by seizures and headaches. The annual risk of iAVM hemorrhage is 2.1%–4.14%, and these lesions account for 40% of all intracerebral hemorrhage in patients between 14 and 45 years old. Following iAVM hemorrhage, morbidity and mortality rates are as high as 50% and 10%, respectively.
Due to the high prevalence and severity of this pathology in a preferentially young population, clinicians and patients seek aggressive treatment options to lessen, and ideally eliminate, the high morbidity and mortality of iAVMs. Treatment options include endovascular embolization, radiosurgery, and microsurgery or a combination of these modalities with the overall goal of complete eradication of the iAVM, thereby eliminating the risk of hemorrhage and subsequent neurologic deficit. To aid in multidisciplinary discussion and clinical decision-making and to provide evidence-based prognostication, AVM grading systems—most notably the Spetzler-Martin system—are utilized ubiquitously. Since the introduction of the Spetzler-Martin grading system in 1986, there have been a plethora of validated grading paradigms to help the neurosurgeon develop the ideal treatment plan for each iAVM. Although many of these grading systems focus on AVM location (supratentorial or infratentorial), presence of hemorrhage, or treatment modality (microsurgery, radiosurgery, or endovascular treatment), all of them utilize a combination of similar clinical and radiographic factors, such as patient age, AVM size, location, venous drainage, arterial feeders, nidus morphology, and previous hemorrhage. Here, we aim to detail and describe the most noteworthy AVM grading systems, demonstrate their application toward predicting surgical risk, and highlight their evidence-based utility as demonstrated by both landmark publications and the most up-to-date literature.
The Spetzler-Martin grading system is an anatomic classification that assesses AVM size, location, and venous drainage pattern ( Table 19.1 ). The size of the AVM (< 3 cm, 3–6 cm, > 6 cm) is determined using a nonmagnified diagnostic cerebral angiogram and measuring maximum nidus diameter. This measurement includes the AVM nidus, but also indicates factors relating to the difficulty of AVM resection, including number of feeding arteries and degree of vascular steal. The AVM location as it relates to eloquent cerebral locations is of significant prognostic importance. Those AVMs that present in eloquent territory—as defined by Drs. Spetzler and Martin as sensorimotor, language, and visual cortex; hypothalamus; thalamus; internal capsule; brainstem; cerebellar peduncles; or deep cerebellar nuclei—earn an additional point, reflecting the increased risk of surgical morbidity and mortality. Finally, the venous drainage pattern is evaluated for deep venous drainage, especially the internal cerebral veins, basal vein of Rosenthal, or precentral cerebellar vein (vein of the cerebellomesencephalic fissure). Deep venous drainage is considered to further add to the risk associated with resection. Overall, using this system generates grades of I–V, which correlates, both retrospectively and prospectively, with morbidity and mortality following microsurgical intervention ( Table 19.2 ). Radiographic examples of each Spetzler-Martin grade are presented in Figs. 19.1–19.5 . Of note, a Spetzler-Martin grade of VI is reserved for untreatable lesions, cases in which treatment would unavoidably be associated with disabling morbidity or mortality.
| Graded Feature | Points |
|---|---|
| Size a | |
| Small (< 3 cm) | 1 |
| Medium (3–6 cm) | 2 |
| Large (> 6 cm) | 3 |
| Eloquence b | |
| Noneloquent | 0 |
| Eloquent | 1 |
| Pattern of Venous Drainage c | |
| Superficial only | 0 |
| Deep | 1 |
a Largest diameter as seen on diagnostic cerebral angiogram.
b Eloquent locations are defined as sensorimotor, language, visual cortex; hypothalamus, thalamus; internal capsule; brainstem; cerebellar peduncles; and deep cerebellar nuclei.
c Venous drainage is superficial if all drainage is via the superficial system. Drainage is deep if it involves any of the deep veins, including internal cerebral veins, basal vein, precentral cerebellar vein, etc.
| Grade a | No. of Cases | NO DEFICIT b | MINOR DEFICIT c | MAJOR DEFICIT d | Death (%) | |||
|---|---|---|---|---|---|---|---|---|
| No. | % | No. | % | No. | % | |||
| I | 23 | 23 | 100 | 0 | 0 | 0 | 0 | 0 |
| II | 21 | 20 | 95 | 1 | 5 | 0 | 0 | 0 |
| III | 25 | 21 | 84 | 3 | 12 | 1 | 4 | 0 |
| IV | 15 | 11 | 73 | 3 | 20 | 1 | 7 | 0 |
| V | 16 | 11 | 69 | 3 | 19 | 2 | 12 | 0 |
| Total | 100 | 86 | 86 | 10 | 10 | 4 | 4 | 0 |
a Grade VI is reserved for untreatable lesions that would unavoidably be associated with disabling morbidity or mortality.
b Complications lasting less than 3 days were not included in morbidity.
c Major morbidity is defined as hemiparesis, increase in aphasia, homonymous hemianopsia, severe deficit with major aphasia and hemiparesis.
d Minor morbidity is defined as mild increase in brainstem deficit, temporary increase in visual field deficit, temporary increase in aphasia and weakness, mild increase in aphasia only detectable with rapid speech, temporary mild increased weakness, increase in trigeminal nerve deficit, mild temporary increase in hemiparesis, mild residual ataxia, temporary mild dysphagia.
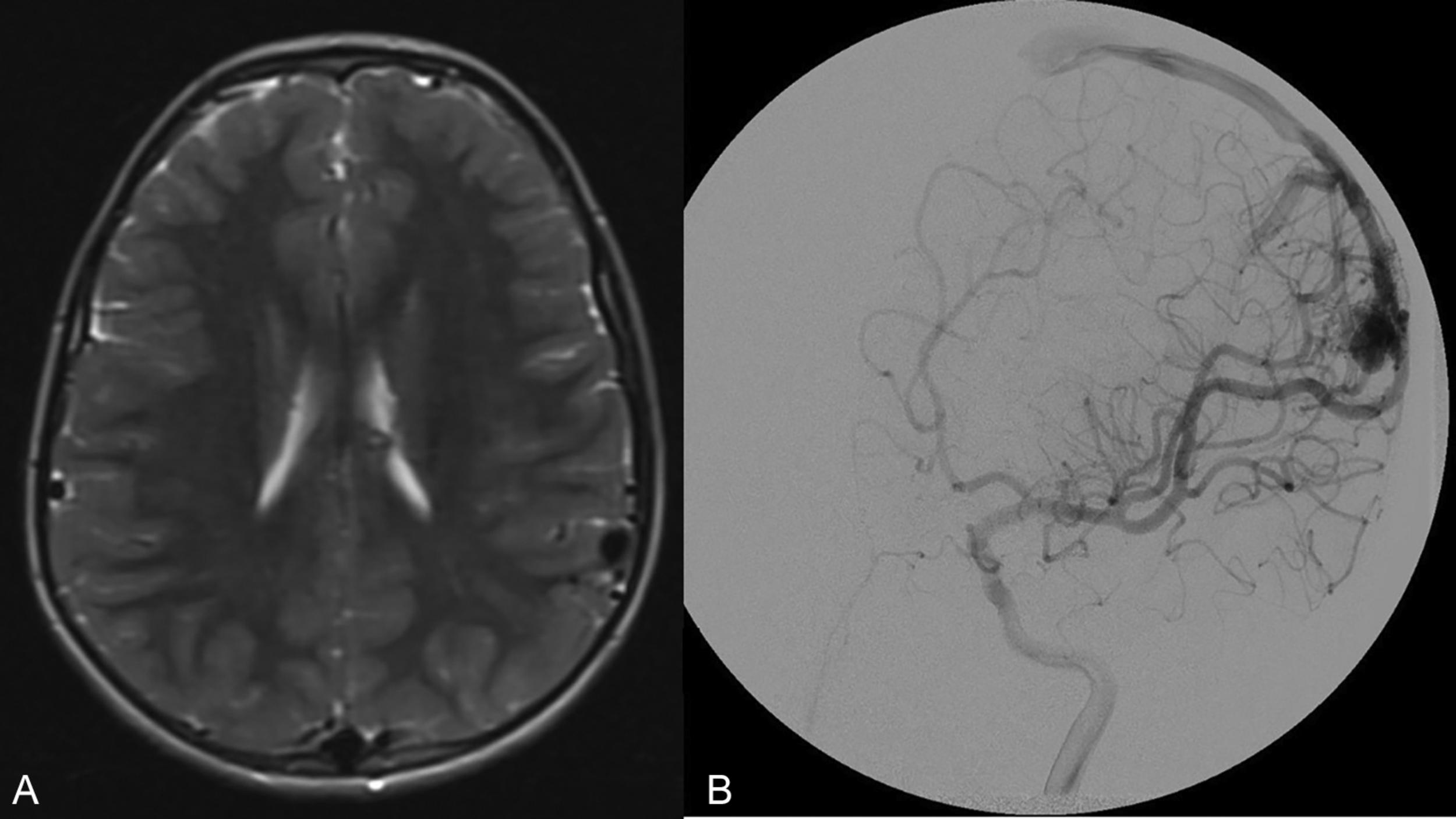
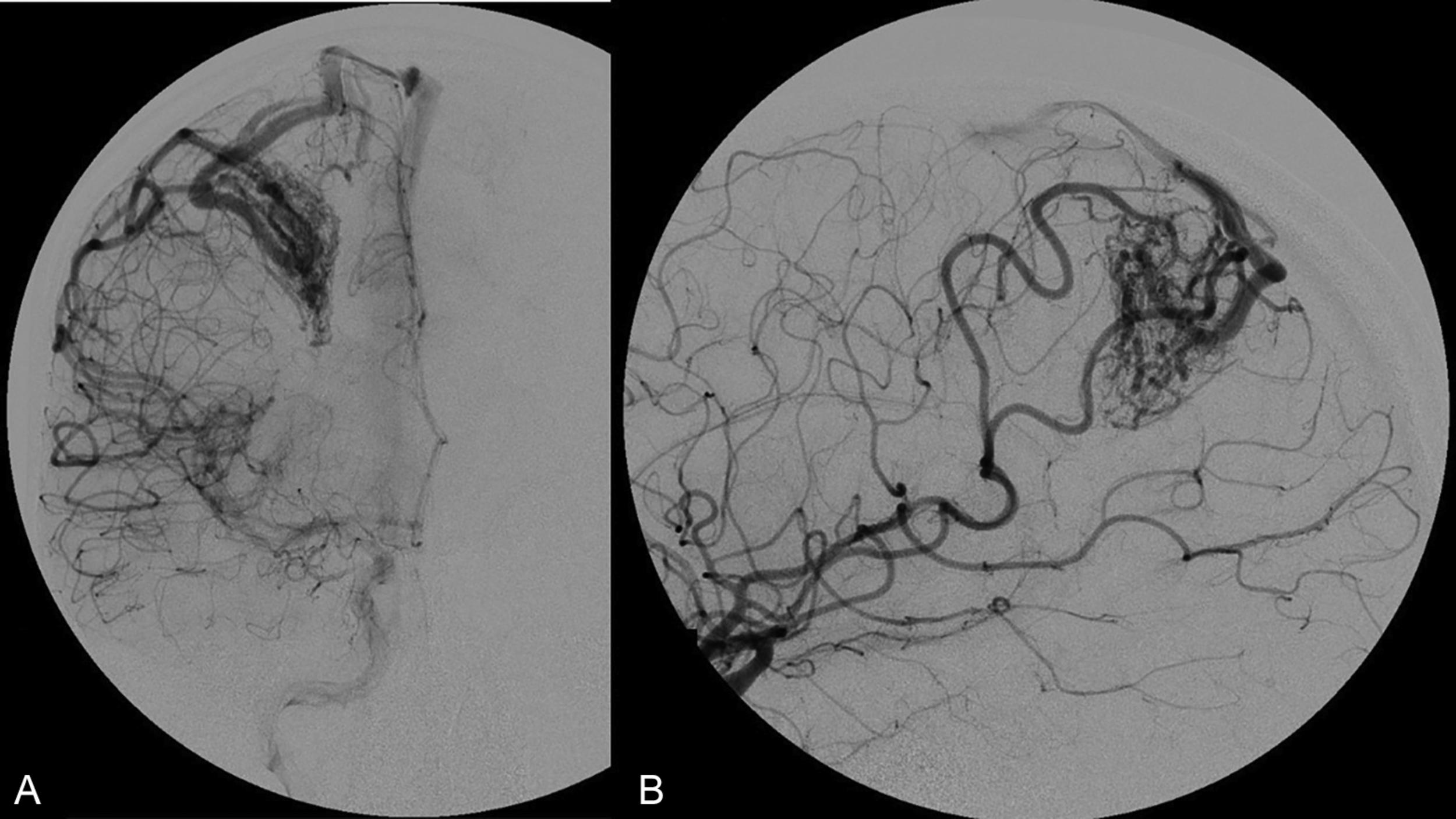
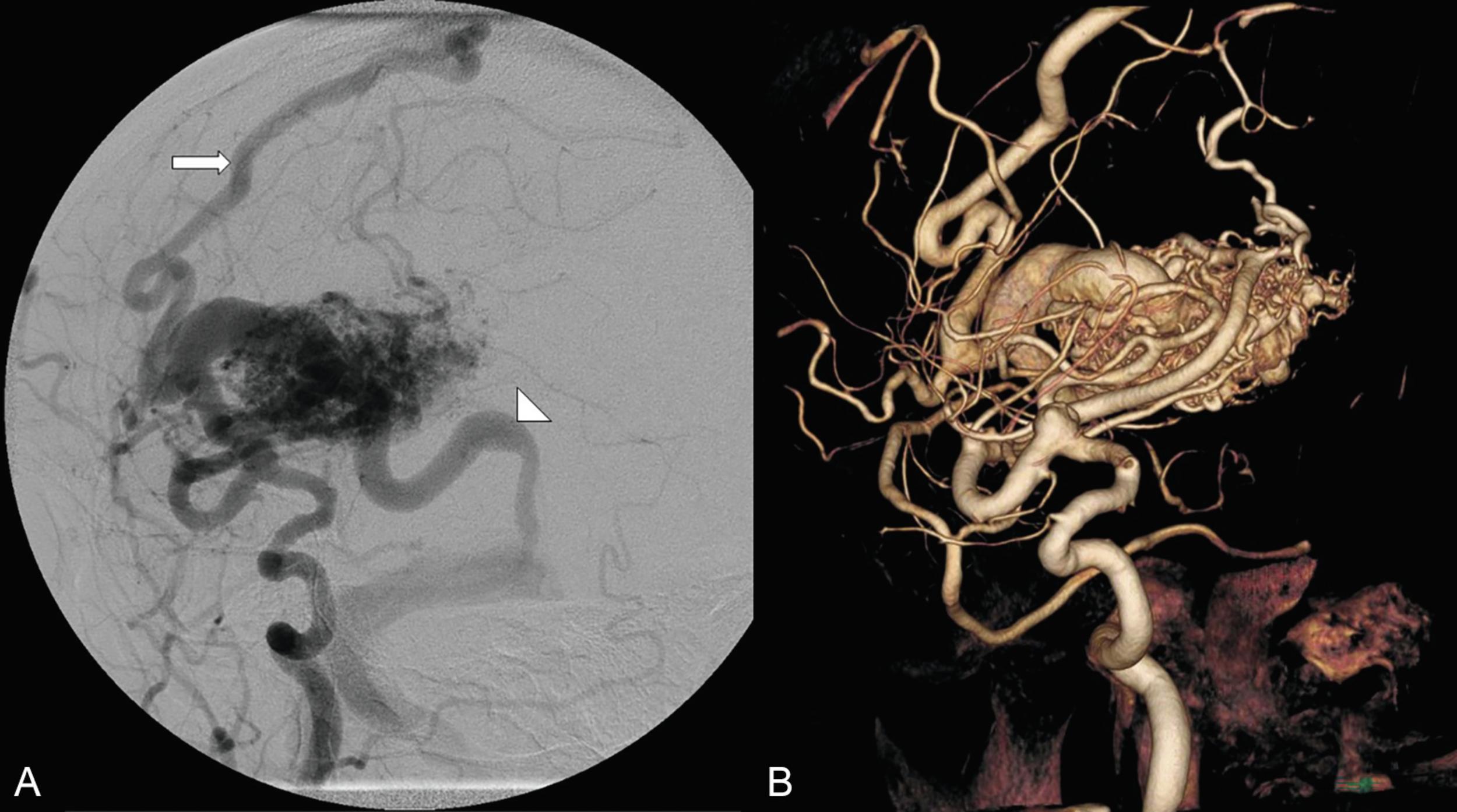
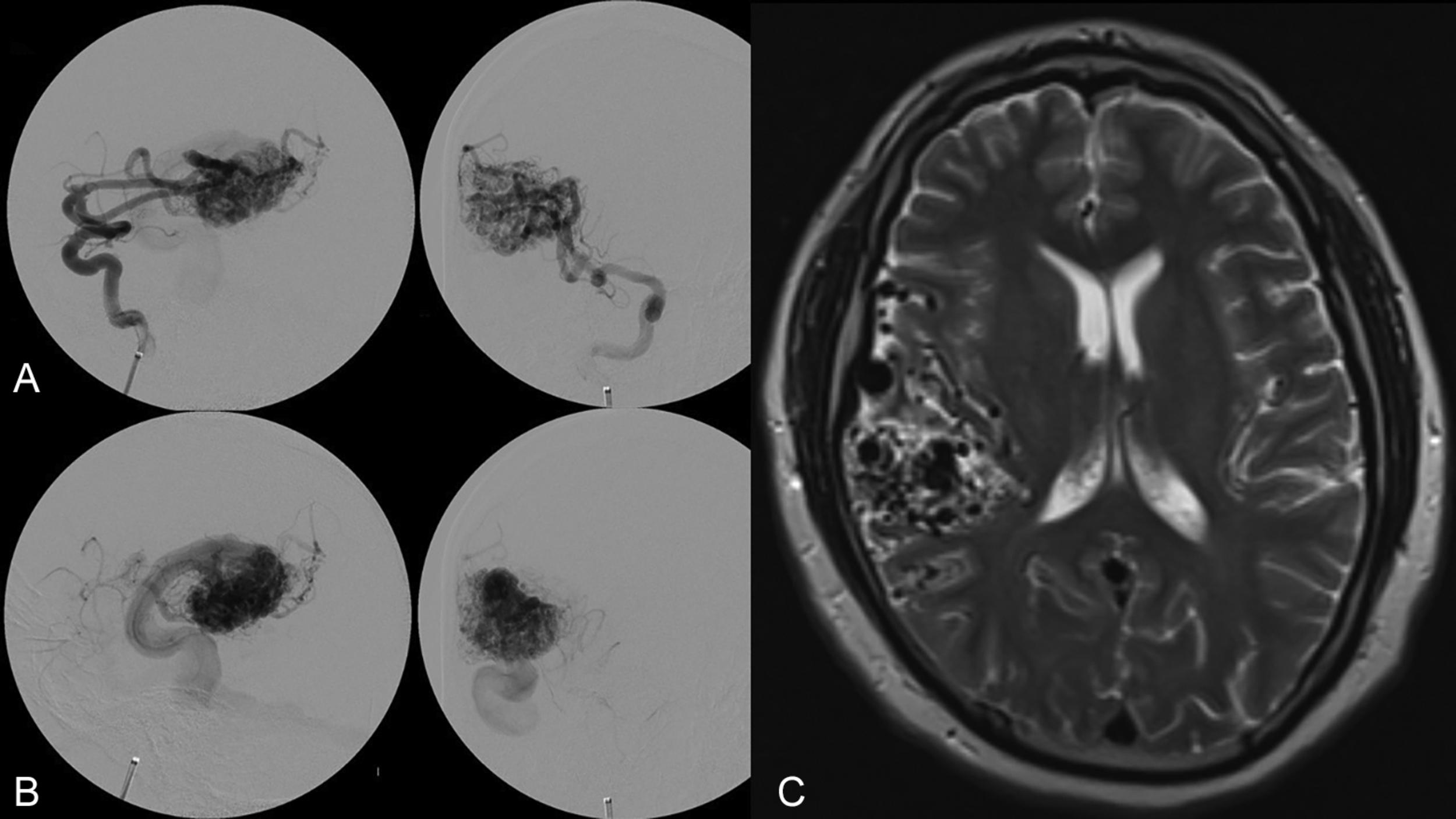
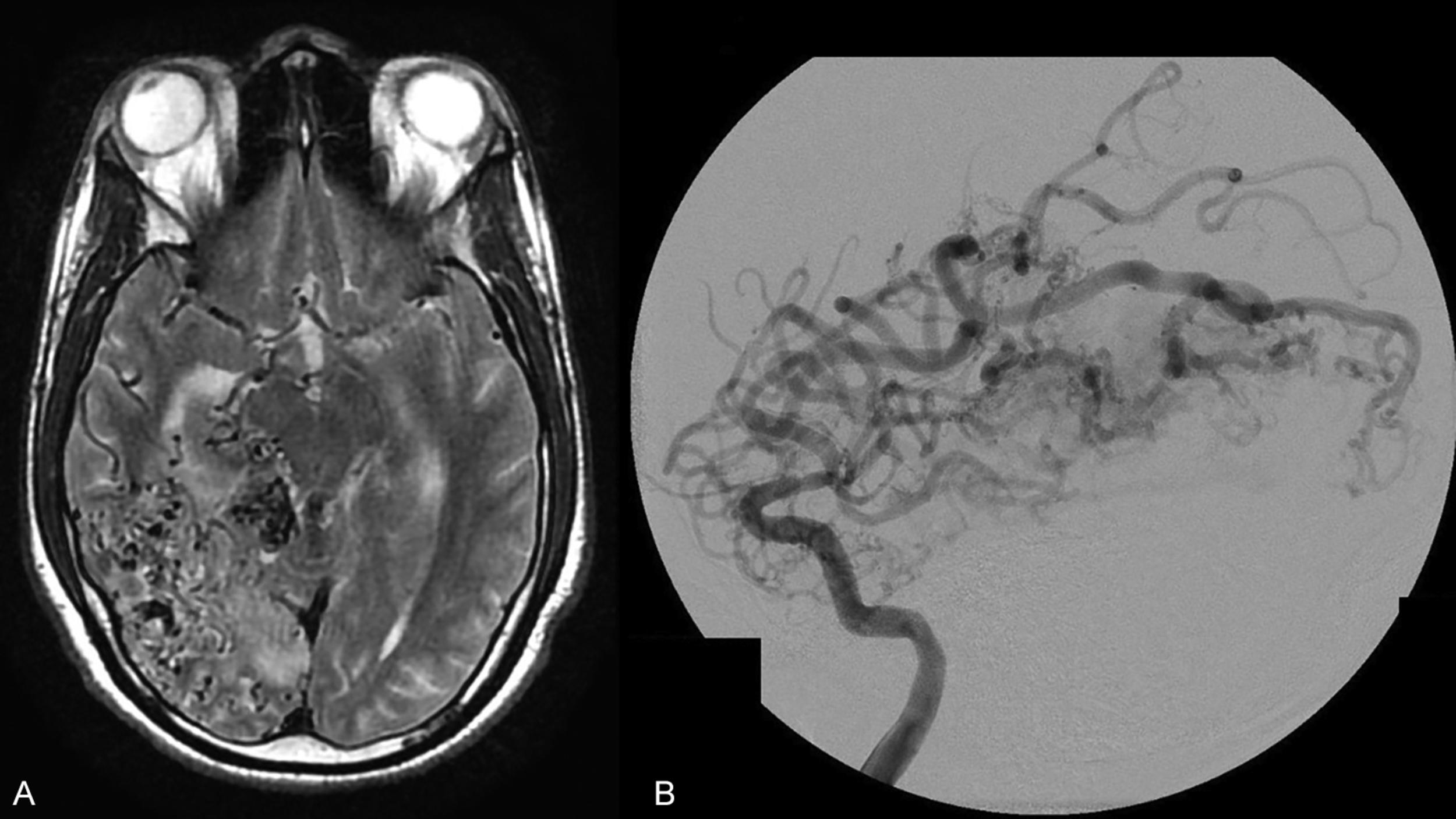
Although the Spetzler-Martin grading scale remains ubiquitous in clinical practice for neurosurgical discussion, assessing surgical risk, and predicting the outcome of microsurgical resection, clinicians often use it in conjunction with the Spetzler-Ponce classification or the supplementary grading scale of Lawton et al. described below.
In 2010, Lawton et al. proposed a supplementary grading scale that was developed with the intention of expanding the Spetzler-Martin grading system by focusing on additional factors that would help predict neurologic outcome following resection and thereby further refine patient selection for surgery. This supplementary AVM grading system awards points for clinical and AVM characteristics that are not covered by the original Spetzler-Martin grade, including patient age, ruptured AVM status, AVM diffuseness, and inclusion of a deep perforating artery, which are all predictors of worse outcome. This supplementary model was found to be more predictive of outcome than the Spetzler-Martin grade alone; however, the new scale was only intended as a supplement and not as a replacement for the already well-established Spetzler-Martin grading system.
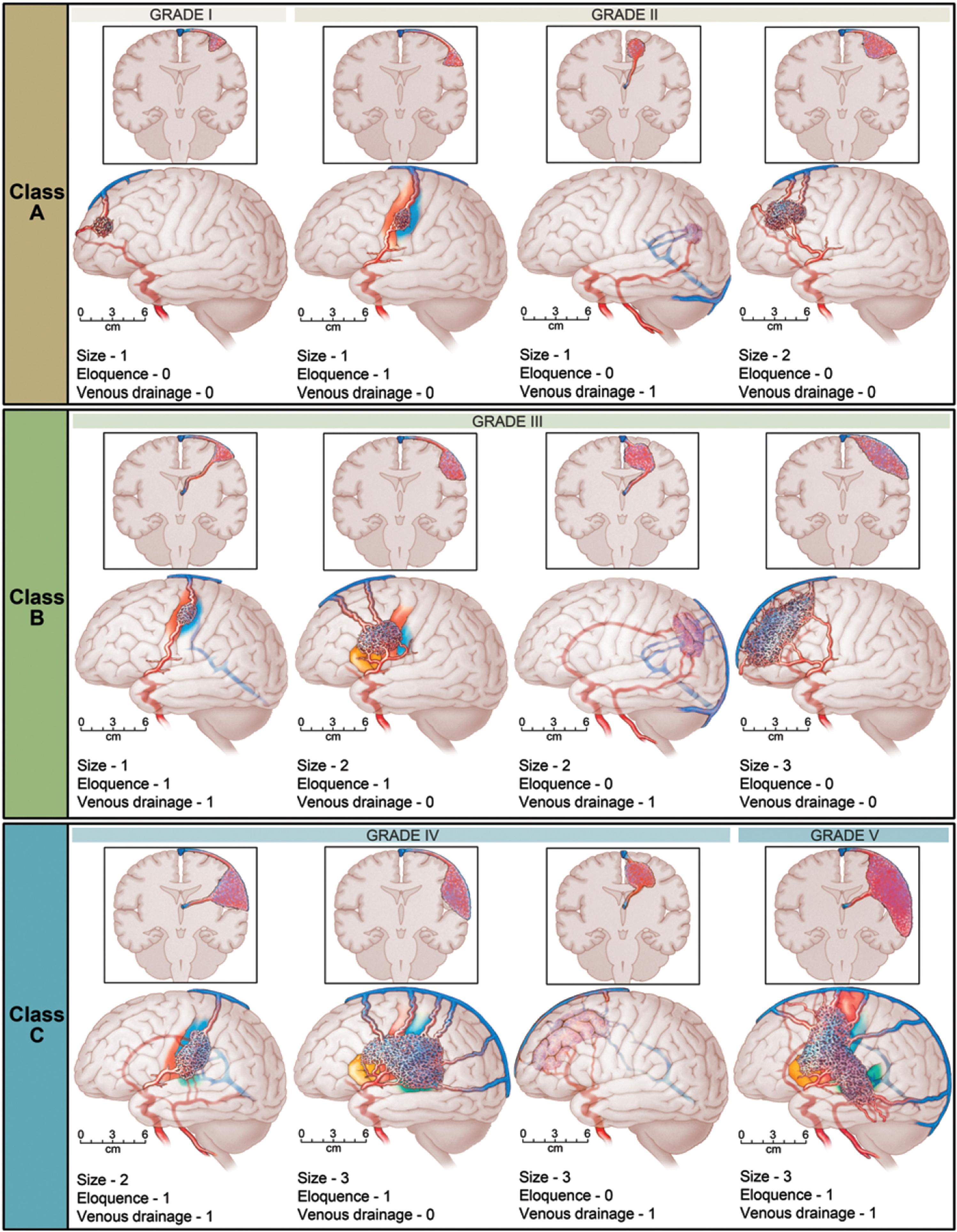
Shortly thereafter, in 2011, Spetzler and Ponce proposed a three-tier simplification of the original Spetzler-Martin grading scale. In this three-tier system, Spetzler-Martin grades I and II are combined into class A, grade III is class B, and grades IV and V are class C ( Fig. 19.6 ). This model, known as the Spetzler-Ponce classification, was intended to help guide treatment decisions as well as predict outcomes. Spetzler and Ponce retrospectively applied their three-tier classification to seven surgical case series in which results had been stratified by Spetzler-Martin grade and concluded that the predictive accuracies for surgical outcomes were equivalent in the original five-tier Spetzler-Martin grading system and the newly proposed three-tier classification. Based on their analysis, they further concluded that combining Spetzler-Martin grades I and II and combining grades IV and V were justified because of the small amount of difference in outcome between the individual grades in each of these two combined categories. Furthermore, this Spetzler-Ponce model also proposed a treatment paradigm according to the AVM class, where resection, multimodality treatment, and no treatment were proposed for class A, class B, and class C AVMs, respectively.
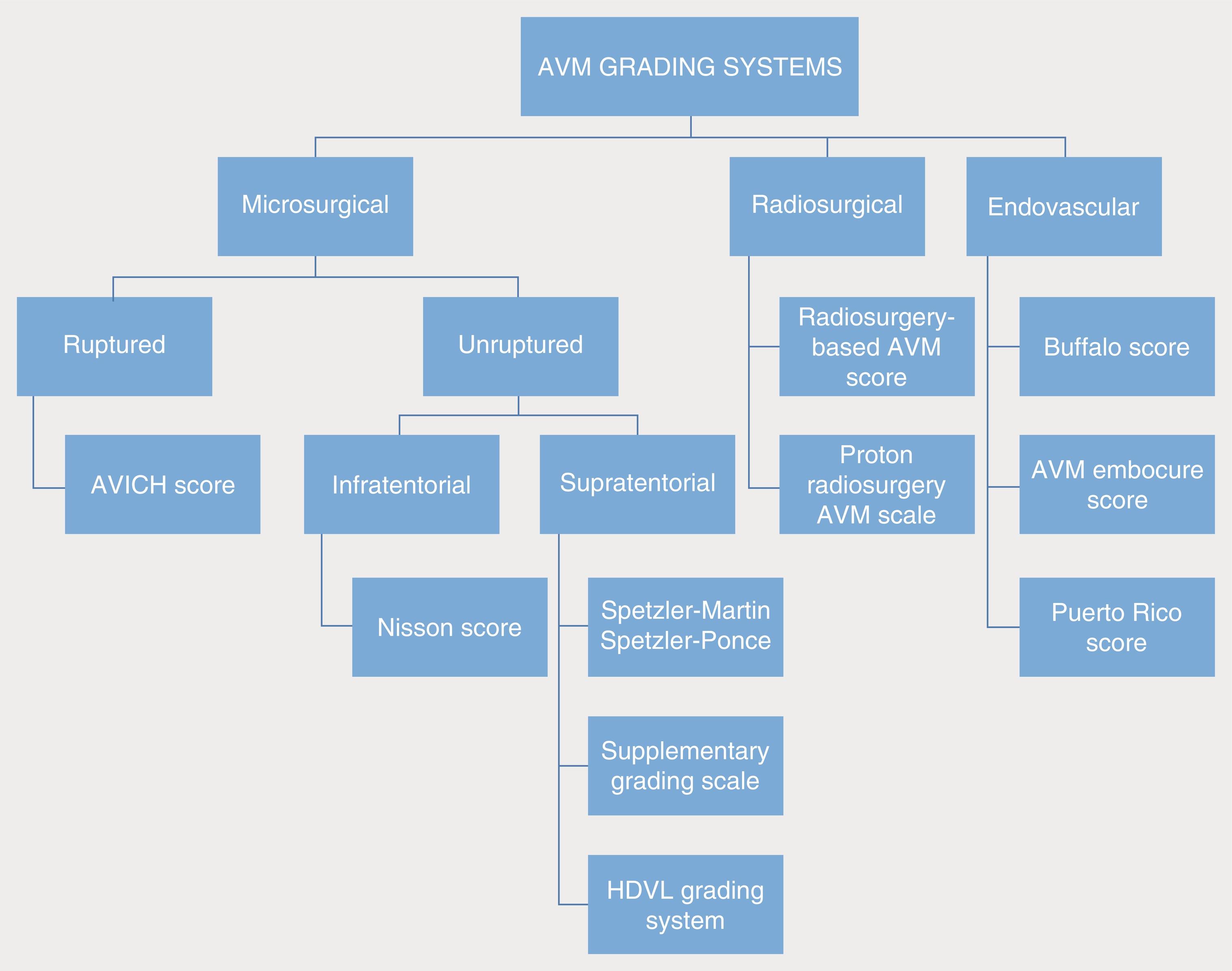
In addition to the foundational and widely employed aforementioned iAVM grading systems (the Spetzler-Martin grading system, the supplemental system proposed by Lawton et al., and the Spetzler-Ponce classification), several additional grading classifications have been proposed for iAVMs, especially for those lesions with particular clinical and radiographic criteria. Table 19.3 presents a summary of the grading systems, their significant contributing factors, and the outcome metrics used. Fig. 19.7 presents a flow diagram showing how some of the grading systems may be applied depending on the characteristics of the individual patient’s AVM and the treatment considerations. Here, we briefly explore additional AVM grading systems, specifically those focused on ruptured AVMs, posterior fossa AVMs, and AVMs in eloquent territory.
| Author | Score | Grading System Factors | Outcome | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Size | Loc/ Eloq | Venous Drainage | Age | Nidus Comp | Hem Pres | Other/ Comments | |||
| Microsurgery | |||||||||
| Spetzler and Martin | Spetzler-Martin | X | X | X | Morbidity | ||||
| Spears et al. | Toronto model | X | Internally validated | ||||||
| Lawton et al. | Supplementary | X | X | X | Externally validated | mRS score | |||
| Spetzler and Ponce | Spetzler-Ponce | X | X | X | Externally validated | Morbidity/mortality and mRS score | |||
| Nisson et al. | Posterior fossa | X | X | Preop exam, emergency surgery | mRS score | ||||
| Jiao et al. | HDVL | X (LED) | X | X | X | mRS score | |||
| Neidert et al. | AVICH | X | X | X | X | X | X | GCS, ICH volume, IVH | mRS score |
| Silva et al. | RAGS | X | X | X | Hunt and Hess score | mRS score | |||
| Radiosurgery | |||||||||
| Pollock et al. | RBAS | X a | X | X | 18.9 Gy (15–25 Gy) | AVM obliteration and clinical | |||
| Hattangadi-Gluth et al. | PRAS | X a | X | 15 Gy | AVM obliteration | ||||
| Milker-Zabel et al. | Heidelberg | X | X | 18 Gy (12–22 Gy) | AVM obliteration | ||||
| Starke et al. | VRAS | X a | X | X | 25 Gy | Clinical | |||
| Endovascular | |||||||||
| Dumont et al. | Buffalo | X | No. of vascular pedicles, pedicle diameter, externally validated | Validated to predict complication occurrence and procedural risk as component of multimodality treatment (not cure); > 85% AVM nidus reduction | |||||
| Lopes et al. | AVMES | X | X | X (no. of draining veins) | No. of vascular pedicles | Complete obliteration and complication; > 85% AVM nidus reduction | |||
| Feliciano et al. | Puerto Rico | X | Fistula present, no. of vascular pedicles | Risk assessment and outcome stratification; > 85% AVM nidus reduction | |||||
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here