Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The first description of an epileptic seizure was written in Akkadian, the oldest written language, which dates from about 3000 years ago in Mesopotamia. The seizure was attributed to the god of the moon. Further anecdotal cases of epilepsy were found in Egyptian, Chinese, Indian, and Babylonic writings ( ). In ancient history epilepsy was believed to be caused by spirits or gods. In 400 BC Hippocrates (460–375 BC), the father of medicine, wrote his book “On the sacred disease” in which he rejected this theory. He was the first to describe epilepsy as a brain disease, and stated ( ): It is thus with regard to the disease called Sacred: it appears to me to be nowise more divine nor more sacred than other diseases, but has a natural cause from the originates like other affections.
However, his theory of the etiology of epilepsy as a superfluity of phlegm leading to abnormal brain consistency was incorrect. Hippocrates’ book was initially not translated into Latin, and after the fall of the Roman Empire epileptic seizures were again wrapped in a veil of superstition and mysticism. Translation of classical Greek and Arabic texts into Latin and the establishment of medical studies influenced the beliefs of medieval scholastic medicine. Physicians became familiar with the different types of epileptic seizures, but the causative theories for these seizures remained focused on supernatural powers. In the 18th century a growing interest in neuropathology led to the theory that “epileptic seizures arose from structural abnormalities of the brain such as hardening or infection, capable of diverting ‘animal spirits’ from their normal pathways through the brain substance or released an irritant that acted on these spirits to produce seizures.” Outside the medical circuit, beliefs of supernatural influences still persisted ( ).
Thanks to the work of Luigi Galvani (1737–1798), the founder of electrophysiology, further progress in the understanding of the pathophysiology of epilepsy was possible. In 1914 the first photos of an electroencephalogram (EEG) during an epileptic seizure were published. Pioneering work in the field of electrophysiology by Hans Berger (1873–1941) led to the standard use of EEG in diagnosis and treatment of epilepsy. Interaction during the 20th century between clinical neurology and experimental research led to the current understanding of epilepsy ( ).
Today epilepsy is defined by the International League Against Epilepsy (ILAE) as a disease of the brain characterized by an enduring predisposition to generate epileptic seizures, and by the neurobiological, cognitive, psychological, and social consequences of this condition ( ). An epileptic seizure is defined as a transient occurrence of signs and/or symptoms due to abnormal excessive or synchronous neuronal activity of the brain ( ). Because the conceptual definition of epilepsy can be difficult to apply in everyday clinical practice, the ILAE has now proposed a practical definition for epilepsy which requires that any of the following conditions are met: at least two unprovoked (or reflex) seizures occurring more than 24 h apart; one unprovoked (or reflex) seizure and a probability of further seizures similar to the general recurrence risk after two unprovoked seizures (at least 60%) occurring over the next 10 years; and/or diagnosis of an epilepsy syndrome ( ).
Worldwide, the lifetime prevalence of epilepsy is estimated to be about 65 million people ( ). In high-income countries the annual incidence of epilepsy is 46 per 100,000, whereas the prevalence approaches 7 per 1000 people a year ( ). Higher incidence rates occur in low-income countries, rural areas, and among infants and elderly people ( ). About 50% of new epilepsy cases start in those two extremes of life ( Fig. 81.1 ) ( ). In children the majority of newly diagnosed patients are younger than 1 year. In the elderly the major cause is postlesional epilepsy after a cerebrovascular disease ( ).
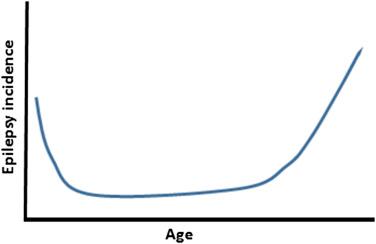
Between 5% and 10% of the population may experience one single spontaneous seizure during their lifetime. As with epilepsy, single unprovoked seizures predominate in men and in patients less than 12 months and older than 65 years ( ). Patients with a single unprovoked seizure have about a 35% risk of seizure recurrence in the subsequent 5 years. However, about 75% of patients with two or three unprovoked seizures will have further seizures within 4 years ( ). Therefore the diagnosis of epilepsy is made when a second nonprovoked seizure occurs, which is the case in 0.5%–1% of the population ( ). Treatment with antiepileptic drugs (AEDs) is then started.
The diagnosis of epilepsy is primarily based on history taking. Anamnesis of the patient is important, but preferably also a description of the seizure from a witness is of great value, since most patients have no recall of the seizure due to an alteration of consciousness. However, even with witness information, it remains a challenge to obtain a correct description of the seizures. The neurologist asks detailed questions about the specific circumstances (occupation at the time of the seizure), provoking factors (e.g., sleep deprivation, alcohol abuse, stress factors), time pattern of the seizures, hormonal influences (menstruation), personal risk factors (e.g., febrile seizures), etc. Also, a detailed family history may contribute to the diagnosis and prognosis.
During physical examination special attention is paid to (sometimes subtle) anomalies indicative of central nervous system (CNS) abnormalities (e.g., asymmetries of fine motor skills and coordination, dysmorphic features for chromosome disorders, and careful examination of the skin for neurocutaneous disorders) ( ).
Besides history taking and standard neurological examination, complementary investigations are performed, such as an EEG and an electrocardiogram (ECG) to rule out cardiac abnormalities ( ). An EEG may reveal typical epileptiformic discharges, which may help to classify the seizure type.
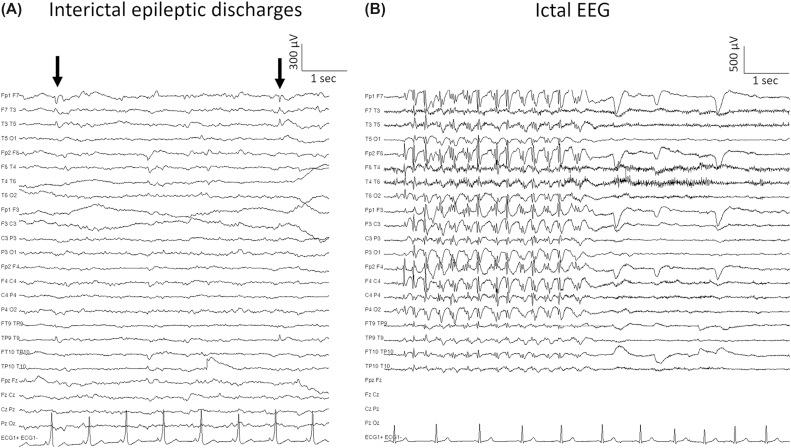
The EEG in patients with epilepsy is characterized by the occurrence of epileptiform activity, i.e., spikes, polyspikes, sharp waves, and spike–wave discharges that can be recorded during (ictal) and in between (interictal) seizures ( Fig. 81.2 ). Recording of habitual seizures during video EEG monitoring is the gold standard for confirmation of the diagnosis of epilepsy and classification of seizure type.
A brain magnetic resonance imaging (MRI) to detect structural epileptogenic lesions is required, except for patients presenting with typical syndromes such as juvenile absence epilepsy or juvenile myoclonic epilepsy ( ). If necessary, further specialized investigations such as positron emission tomography, single photon emission computed tomography, and magnetoencephalography can be performed.
Epilepsy is not a straightforward entity. The term “epilepsy” comprises many syndromes and there may be different underlying causes. Correct diagnosis and classification of the seizure type or epileptic syndrome are indispensable for appropriate treatment and prognosis. The classification is based on the clinical presentation of the seizures combined with the interictal and/or ictal abnormalities on the EEG.
Epileptic seizures are classified into two subtypes according to the networks in which they originate: focal epileptic seizures originate within networks limited to one hemisphere, while generalized epileptic seizures arise within and rapidly engage bilaterally distributed networks. Generalized seizures can be further classified into absence, myoclonic, atonic, clonic, tonic, and tonic–clonic seizures. Focal seizures used to be classified into simple or complex seizures on the basis of presumed changes in the level of consciousness. However, the ILAE recommends abandoning these imprecise terms and instead describing focal seizures according to their clinical manifestations using the “Glossary of Ictal Semiology” (e.g., aura, focal motor, autonomic events, retained/altered responsiveness or awareness) ( ).
The elements that cause epilepsy are multiple and heterogeneous, but they can be used to subcategorize it into epilepsy syndromes, as this aids in the choice of antiepileptic treatments. An epilepsy syndrome is a unique epilepsy condition defined by a typical age of onset, seizure type, and EEG features ( ). Previously the subdivisions of “symptomatic, idiopathic, and cryptogenic” epilepsy syndromes were used, but these terms were poorly delineated and led to confusion. With the current understanding it has been possible to categorize epilepsy syndromes by their specific etiologies, e.g., genetic, structural, metabolic, immune, infectious, and epilepsy syndromes that have no known cause ( ). Genetic epilepsy syndromes are a direct result of a known or presumed genetic defect(s) in which seizures are the core symptom of the disorder (e.g., Angelman syndrome). A small number of inherited epilepsies are “pure” idiopathic epilepsy syndromes ( Table 81.1 ). Almost all mutation occurs in genes that encode voltage-gated or ligand-gated ion channels. It must be clear that a single mutation can give rise to a variety of epilepsy types and a single seizure phenotype can be associated with different mutations. Seizures in structural epilepsy syndromes are attributed to distinct structural brain abnormalities that can be developmental (e.g., tuberous sclerosis) or acquired (e.g., trauma). Metabolic abnormalities are responsible for the increased brain excitability in metabolic epilepsy syndromes, such as in mitochondrial disorders. The increased risk of developing epilepsy in immune epilepsy syndromes is driven by CNS inflammation (e.g., Rasmussen syndrome). The most common epilepsy syndrome worldwide, however, is the infectious syndrome, where infections of the CNS (e.g., bacterial meningitis) can cause both acute symptomatic seizures and epilepsy.
| Syndrome | Gene | Gene Product |
|---|---|---|
| Benign familial neonatal convulsions | KCNQ 2 KCNQ 3 |
Voltage-gated potassium channel subunits |
| Benign familial neonatal infantile seizures | SCN 2 A | Voltage-gated sodium channel, α subunit, type 2 |
| Autosomal-dominant nocturnal frontal lobe epilepsy | CHRNA 4 CHRNB 2 CHRNA 2 |
Nicotinic acetylcholine receptor subunits |
| Autosomal-dominant lateral temporal lobe epilepsy | LGI 1 | Leucine-rich glioma-inactivated protein |
| Generalized epilepsy with febrile seizures plus (GEFS+) | SCN 1 A, SCN 1 B, SCN 2 A GABRG 2 |
Voltage-gated sodium channel subunits GABA A receptor, γ subunit, type 2 |
| Severe myoclonic epilepsy of infancy | SCN 1 A | Voltage-gated sodium channel, α subunit, type 1 |
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here