Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Of all organ systems, the skin has the greatest number of pathological lesions, perhaps in part because the skin is subject to a wide variety of environmental exposures, no special procedures are necessary to visualize its surface (therefore pathologic lesions are readily apparent), and skin is the largest organ!
In addition to age and gender, relevant clinical history is often necessary or helpful for interpretation ( Table 15.1 ).
| HISTORY RELEVANT TO ALL SPECIMENS | HISTORY RELEVANT FOR DERMATOPATHOLOGY SPECIMENS |
|---|---|
| Organ/tissue resected or biopsied | Anatomic site, duration, and appearance of the lesion (especially for incisional biopsies) |
| Purpose of the procedure | |
| Gross appearance of the lesion sampled | Clinical differential diagnosis—photographs of the lesion are very helpful |
| Any unusual features of the clinical presentation | Systemic diseases and relevant comorbidities |
| Any unusual features of the gross appearance | Family history of skin disorders |
| Prior surgery/biopsies—results | Previous similar lesions |
| Prior malignancy | Duration of the lesion and history of change over time |
| Prior treatment (radiation therapy, chemotherapy, drug use can change the appearance of tissues) | Sampling considerations (biopsy of edge or center) |
| Travel history and environmental exposures | |
| Compromised immune system | Symptoms that may produce secondary changes (e.g., pruritis leading to scratching) |
| Endocrine changes, pregnancy |
The ability to clearly visualize the entire epidermis in a perpendicular section is critical for diagnosis and at times for prognosis (e.g., malignant melanoma). Therefore vertical orientation must be maintained at all times in sections.
Any specimen that is labeled “excision,” regardless of the type of specimen, must have the margins evaluated by inking and submission of appropriate sections. While the term “excision” may seem to relate to the nature of the specimen (e.g., punch biopsy vs. ellipse), it is ultimately the intent of the clinician that must be considered. Thus, designations for specimens on the requisition form as “excisional biopsies,” “shave removals,” etc. that imply an intent to remove the entire lesion require an attempt at margin assessment.
Red or black inks should be avoided as they can be difficult to see in some skin specimens. Blue and green inks are preferred. Diagrams are helpful for any difficult or complicated specimens.
Small vesicular lesions should not be cut through when present in any type of specimen. The overlying tissue layer is important for diagnosis and is fragile. If sectioned, it is easily detached and lost. Small specimens can be submitted in a cassette without sectioning. The histotechnologist can be instructed to bisect the specimen after tissue processing. After this treatment, the tissue is stiffened and a delicate vesicle is less likely to be lost or damaged during sectioning. If the specimen is of a size that sectioning is required, the sections can be made on either side of the lesion so as to leave the vesicle intact.
Very small, centrally located lesions (e.g., a small 0.1 cm pigmented macule or a small papule possibly representing a hair follicle) also must be processed with great care. Bisecting such a centrally located small lesion runs the risk of resulting in the central most diagnostic areas of the lesion being discarded as waste sections when the paraffin block is faced by the histotechnologist in order to create a flat surface. Small biopsies may be submitted whole in a cassette with instructions to obtain levels into the center of the specimen. If the specimen requires sectioning due to size, the specimen should be cut such that the lesion remains intact in a central section.
Immunofluorescence (IF), electron microscopy (EM), or frozen tissue: Fresh (unfixed) or frozen specimens (usually punch biopsies) may be submitted for special studies for the evaluation of specific diseases:
Immunofluorescence: lupus erythematosus, dermatitis herpetiformis, bullous pemphigoid, pemphigus, IgA-mediated vasculitis, others.
Immunophenotyping on frozen tissue: leukemias and lymphomas. However, the majority of studies can be performed using formalin-fixed, paraffin-embedded tissue.
Electron microscopy: epidermolysis bullosa, other blistering diseases, some melanomas (e.g., S100 negative tumors), unusual tumors, amyloid, Cerebral Autosomal Dominant Arteriopathy with Subcortical Infarcts and Leukoencephalopathy disease (CADASIL; this disease can be diagnosed due to changes in vessels in skin biopsies), certain lysosomal storage disorders.
Operating room consultations with frozen sections: Frozen section examination may be requested in cases of suspected toxic epidermal necrolysis (TEN) or staphylococcal scalded skin syndrome (SSSS). Skin stripping or a sloughed epidermal specimen (sectioned and wound to produce a “jelly roll”) may be submitted for frozen section to determine the level of cleavage and degree of necrosis in the epidermis. Frozen section examination of skin may also be helpful in the early diagnosis of necrotizing fasciitis. However, frozen sections should rarely, if ever, be used for the primary diagnosis of small skin lesions, especially pigmented lesions.
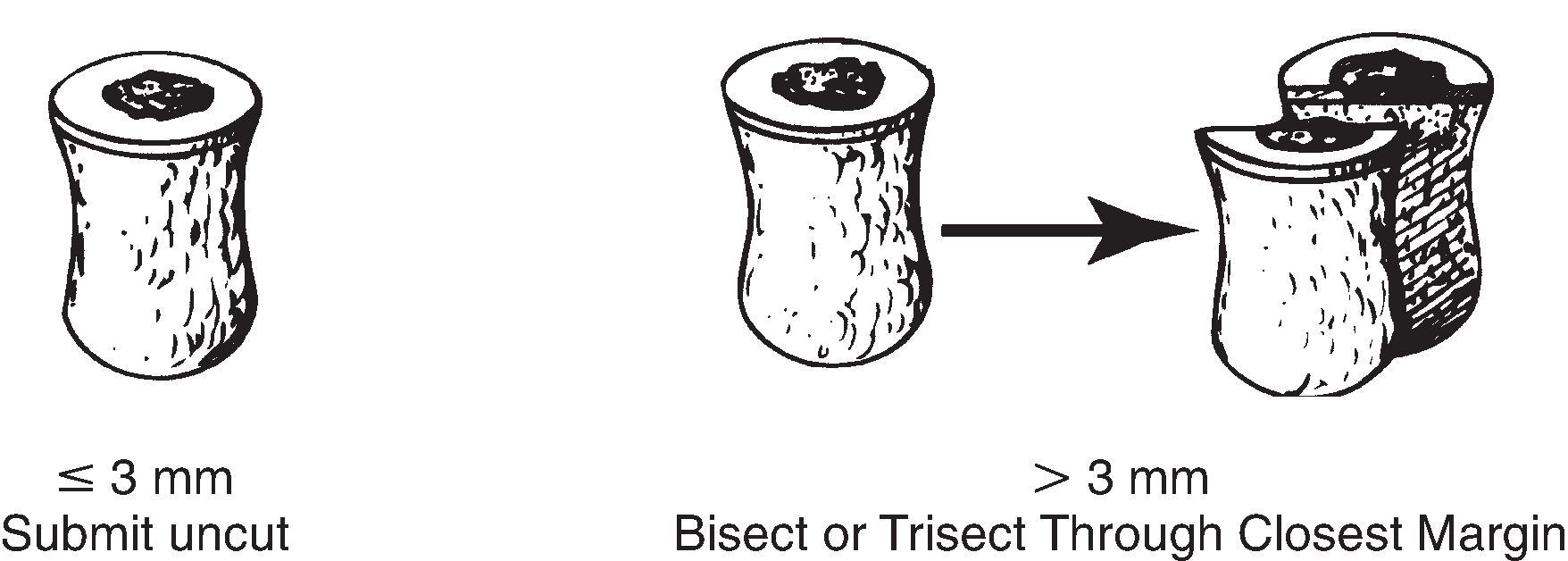
Punch biopsies are performed to completely excise small lesions, to sample large lesions, or to evaluate an inflammatory process or a systemic disease (e.g., pustular psoriasis). Punches can be 2, 3, 4, 5, 6, 8, 10, or 12 mm in diameter ( Fig. 15.1 ). Smaller punch biopsies are processed as described below (i.e., not as skin ellipses with tips). However, some larger punch biopsies (that in some cases are labeled “punch excision”) may remove an entire lesion, and can be processed similar to a skin ellipse excision (i.e., inked and sectioned into at least three perpendicular sections including one or more central slices and two “tips”).
Describe the type of specimen (“punch biopsy”) including diameter, depth, and skin color.
Describe lesions including size, type (macular, papular, vesicular, plaque), borders (well circumscribed, irregular), color (brown, black, variegated), shape (verrucous, lobulated), and distance from the closest margin. Ink all biopsies.
An important goal of processing any specimen, even a punch biopsy, is to determine if the lesion has been completely removed. Therefore, sections must be obtained at the closest point of the lesion to the margin. If the margin is grossly present at the margin, document this finding in the gross description.
Punch biopsies 3.5 mm or smaller are best submitted entire without sectioning. These specimens can be bisected by the histology laboratory after tissue processing. Alternatively, the specimen can be embedded without sectioning and step sections (levels) may be cut at intervals to insure adequate sampling throughout the entire specimen. Careful attention to specimen processing is particularly important to ensure adequate representation of small, centrally located, lesions.
Punch biopsies 4 mm or larger are bisected unless en face tips can be obtained without creating a “false positive” margin due to including a portion of the lesion. If there is a discrete lesion, the specimen should be cut in a plane to demonstrate the closest margin. If the lesion is very small (i.e., leveling the block might remove the lesional tissue), the punch biopsy should be cut on either side of the lesion, leaving the lesion intact in a central section.
Larger punch biopsies used to remove an entire lesion may be processed as small ellipses, obtaining tips, and internal serial sections.
Vesicles or blisters should not be sectioned. Small specimens should be submitted in a cassette without sectioning. Larger specimens can be sectioned on either side of the lesion, leaving the lesion intact in a central section.
Biopsies to evaluate alopecia: A number of different methods of sectioning these specimens have been developed in order to adequately visualize hair follicles (see “Special Studies” below).
Request two H&E levels per block.
Received in formalin labeled with the patient’s name and medical record number and “5 mm punch, left leg” is a 5 mm in diameter by 5 mm (depth) punch biopsy with tan/white skin. There is a flat homogeneously brown lesion with slightly irregular margins, 0.3 × 0.3 cm, on the skin surface. The lesion is less than 0.1 cm from the nearest margin, but does not grossly involve the margin. The specimen is not oriented. The margins are inked blue. The entire specimen is submitted for microscopic evaluation.
Micro A1: bisected, two frags.
Suspected infection: If a biopsy is from a potentially immunocompromised patient (e.g., post bone marrow transplant or with leukemia), and/or if the requisition states to rule out infection, stains for bacteria, fungi, and mycobacteria should be ordered immediately (typically Gram stain, periodic acid-Schiff-diastase [PAS-D] stain, a silver stain such as methenamine silver, and stains for acid-fast bacilli such as Ziehl–Neelsen and Fite–Faraco). These are often specimens requiring expedited (rush) interpretation due to the clinical setting. Diagnosing an infection even a few hours earlier can be lifesaving.
Alopecia: Both vertical and horizontal sections can be helpful for evaluating diseases of hair follicles that result in alopecia. Multiple methods of specimen processing have been suggested. The best method may vary with the disease process. In standard specimen processing, only vertical sections are evaluated. Bisecting at a 30° angle to the epidermis can improve the visualization of hair follicles. If more than one punch biopsy is taken for diagnosis, one may be processed for vertical sections and one for horizontal sections.
Headington method (horizontal sections) —The punch biopsy is bisected horizontally at 0.1 cm above the junction of the dermis and subcutis. The two cut surfaces are inked. These two surfaces are embedded face up for sectioning.
Tyler technique (horizontal and vertical sections) —The punch biopsy is bisected into two vertical sections. One half of the biopsy is then bisected horizontally and embedded as in the Headington method.
HoVert (horizontal and vertical) technique (horizontal and vertical sections) —The punch biopsy is transected horizontally 0.1 cm below the epidermal surface to create an epidermal disk. The disk section is then sectioned vertically, perpendicular to the epidermal surface. The cut surface of the lower portion is inked. This portion is then transected horizontally at the junction of the dermis and the subcutaneous tissue. The upper surface of the new lower portion is inked. The two inked surfaces are embedded face down.
Re-embedding technique (horizontal and vertical sections) —The punch biopsy is sectioned vertically according to standard processing. After initial H&E sections are taken, the tissue is re-embedded with the subcutaneous tissue facing downward and the epidermal surface upward. Additional horizontal sections are obtained.
Shave biopsies are usually performed to sample or remove nonmalignant lesions (e.g., seborrheic keratoses, actinic keratoses, or fibroepithelial polyps) or for diagnosis of basal and squamous cell carcinomas. Shave biopsies of pigmented lesions should be strongly discouraged and interpreted with caution. The diagnosis of melanoma may be difficult in such a specimen due to limited sampling and the depth of invasion may be impossible to assess. Specimens are inked if designated “shave excisions” or “shave removals” (often seen with deep “scoop” or “saucerized” shave biopsies) as this indicates there is the intent to completely remove the lesion.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here