Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Inflammation is an almost universal response to tissue damage by a wide range of harmful stimuli, including mechanical trauma, tissue necrosis and infection. The purpose of inflammation is to destroy (or contain) the damaging agent, initiate repair processes and return the damaged tissue to useful function. Inflammation is somewhat arbitrarily divided into acute and chronic inflammation , but, in reality, the two often form a continuum. Many causes of tissue damage provoke an acute inflammatory response but some types of insult may bring about a typical chronic inflammatory reaction from the outset (e.g. viral infections, foreign body reactions and fungal infections). Acute inflammation may resolve or heal by scarring but may also progress to chronic inflammation and it is common for a mixed acute and chronic response to co-exist. This chapter describes acute inflammation and its sequelae, while chronic inflammation is discussed in Ch. 4 . Many examples of acute and chronic inflammation are illustrated throughout this book.
There are three major and interrelated components of the acute inflammatory response
Vascular dilatation
Relaxation of vascular smooth muscle leads to engorgement of tissue with blood (hyperaemia)
Endothelial activation
Increased endothelial permeability allows plasma proteins to pass into tissues
Expression of adhesion molecules on the endothelial surface mediates neutrophil adherence
Production of factors that cause vascular dilatation
Neutrophil activation and migration
These are outlined in Fig. 3.1 .
A alveolus C alveolar capillary F interlobar fissure Fi fibrin M alveolar macrophage N neutrophils
While the basic process of acute inflammation is the same in all tissues, there are frequently qualitative differences in the inflammatory response seen under different circumstances. Terms describing these variations are widely used in clinical practice and are summarised below:
Suppurative inflammation (purulent inflammation) refers to acute inflammation in which the acute inflammatory exudate is rich in neutrophils. Suppurative inflammation is most commonly seen due to infection by bacteria where the mixture of neutrophils (viable and dead), necrotic tissue, and tissue fluid in the acute inflammatory exudate form a semi-liquid material referred to as pus , hence the term purulent inflammation (see Fig. 3.4 ). Within tissues, a circumscribed collection of semi-liquid pus is termed an abscess (see Fig. 3.12 ). The destruction of tissue may be due as much to release of neutrophil lysosomal enzymes as to tissue destruction by bacteria. Bacteria that produce purulent inflammation are described as pyogenic bacteria . They initiate massive neutrophilic infiltration with subsequent destruction of infected tissues. Pyogenic bacteria include staphylococci, some streptococci (Streptococcus pyogenes , S. pneumoniae), Escherichia coli and the neisseriae ( Neisseria meningitidis, N. gonorrhoeae ).
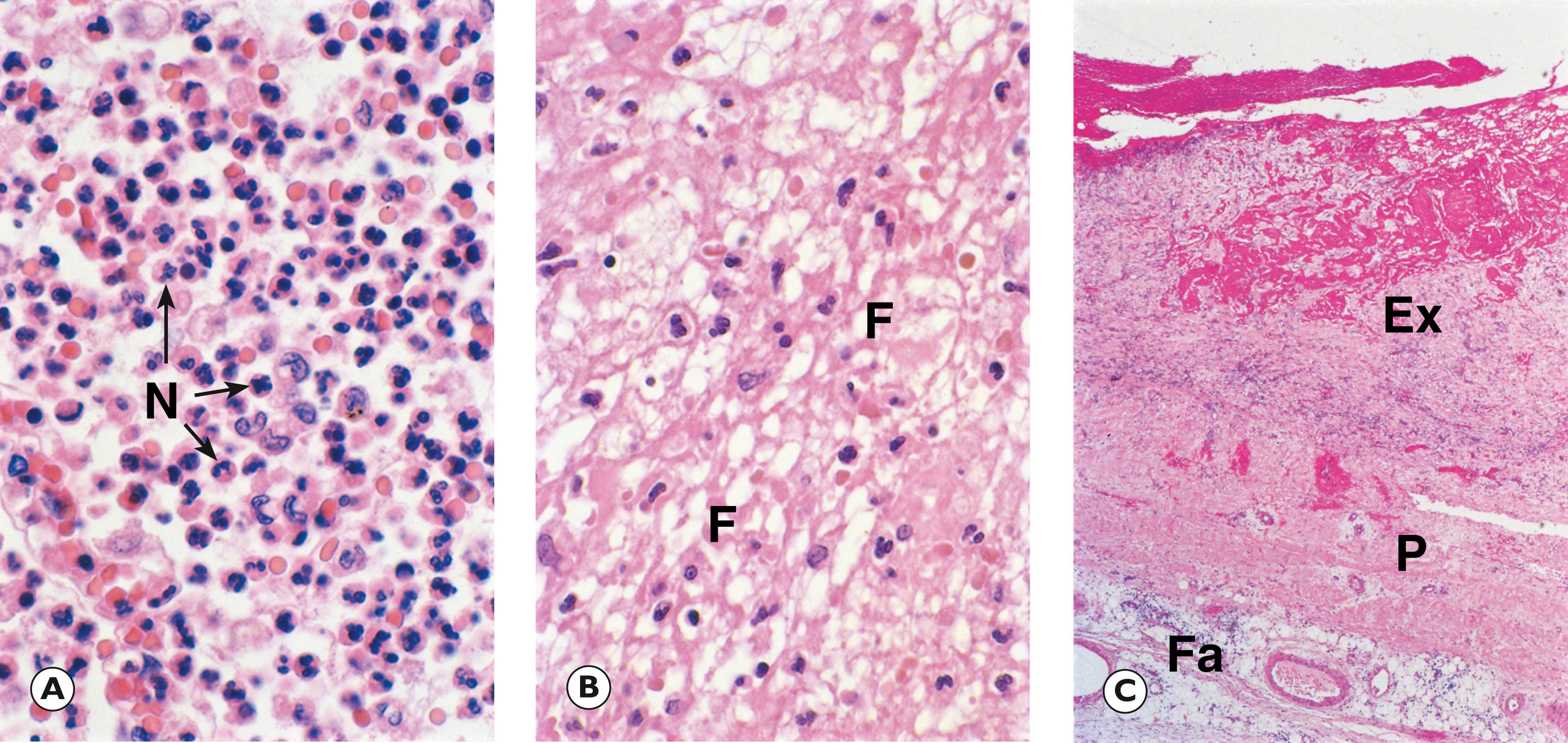
Fibrinous inflammation refers to a pattern of acute inflammation where the acute inflammatory exudate has a high plasma protein content (see Fig. 3.4 ). Fibrinogen, derived from plasma, is converted to fibrin, which is deposited in tissues. This pattern is particularly associated with membrane-lined cavities such as the pleura, pericardium and peritoneum, where the fibrin strands form a mat-like sheet causing adhesion between adjacent surfaces.
Serous inflammation describes a pattern of acute inflammation where the main tissue response is an accumulation of fluid with a low plasma protein and cell content. This is often called a transudate , which by definition has a specific gravity of <1.012 or protein content of <25 g/L in contrast to an exudate, with a specific gravity of >1.020 and protein content of >25 g/L. This pattern of response is most commonly seen in the skin in response to a burn.
The vascular and exudative phenomena of acute inflammation are responsible for the clinical features and were described by Celsus in the first century AD. The cardinal signs of Celsus are:
redness (rubor) caused by hyperaemia
swelling (tumor) caused by fluid exudation and hyperaemia
heat (calor) caused by hyperaemia
pain (dolor) resulting from release of bradykinin and PGE 2 .
Virchow later added:
loss of function (functio laesa) caused by the combined effects of the above.
Clinically, patients who have significant acute inflammation feel unwell and have a fever. This is mediated by cytokines released into the blood (interleukins 1 and 6 (IL-1 and IL-6 respectively), tumour necrosis factor (TNF) and prostaglandins), acting on the hypothalamus. Laboratory investigations commonly reveal a raised neutrophil count in the blood (neutrophil leukocytosis).
The nomenclature used to describe inflammation in different tissues employs the tissue name (or its Greek or Latin equivalent) and the suffix ‘-itis’. For example, inflammation of the appendix is referred to as appendicitis , inflammation of the Fallopian tube is termed salpingitis and inflammation of the pericardium is termed pericarditis . Notable exceptions to this rule include pleurisy , for inflammation of the pleura and acute cellulitis for inflammation of subcutaneous tissues. Many examples of acute inflammatory diseases are presented in the systematic pathology chapters, which form the second half of this book. Common examples are outlined in Table 3.1 .
Ex exudate F fibrin Fa pericardial fat N neutrophils P pericardium
| Tissue | Acute inflammation | Typical causes |
|---|---|---|
| Meninges | Meningitis | Bacterial and viral infections |
| Brain | Encephalitis | Viral infections |
| Lung | Pneumonia | Bacterial infections |
| Pleura | Pleurisy | Bacterial and viral infections |
| Pericardium | Pericarditis | Bacterial and viral infections, myocardial infarction |
| Oesophagus | Oesophagitis | Gastric acid reflux, fungal infection ( Candida albicans ) |
| Stomach | Gastritis | Helicobacter pylori infection, reflux/chemical gastritis |
| Colon | Colitis | Bacterial infections, inflammatory bowel disease |
| Rectum | Proctitis | Infections, ulcerative colitis |
| Appendix | Appendicitis | Faecal obstruction |
| Liver | Hepatitis | Alcohol abuse, viral infections |
| Gallbladder | Cholecystitis | Bacterial infections, chemical irritation |
| Pancreas | Pancreatitis | Obstructed pancreatic duct, alcoholism, shock |
| Urinary bladder | Cystitis | Bacterial infections |
| Bone | Osteomyelitis | Bacterial infections |
| Subcutaneous tissues | Cellulitis | Bacterial infections |
| Joints | Arthritis | Infections, autoimmune diseases |
| Arteries | Arteritis | Immune complex deposition |
| Kidney | Pyelonephritis | Bacterial infections |
| Peritoneum | Peritonitis | Spread from intra-abdominal inflammation, e.g. appendicitis, salpingitis Ruptured viscus, e.g. perforated peptic ulcer |
The process of acute inflammation is designed to neutralise injurious agents and to restore the tissue to useful function. There are four main outcomes of acute inflammation (if the patient survives): resolution , healing by fibrosis , abscess formation and progression to chronic inflammation . Three factors determine which of these outcomes occurs:
the severity of tissue damage
the capacity of stem cells within the damaged tissue to divide and replace the specialised cells required, a process termed regeneration
the type of agent that has caused the tissue damage.
Resolution involves complete restitution of normal tissue architecture and function. This can only occur if the connective framework of the tissue is intact and the tissue involved has the capacity to replace any specialised cells that have been lost (regeneration). Neutrophils and damaged/dead tissue are removed by phagocytosis by macrophages ( E-Fig. 3.5 ), which leave the tissue via the lymphatics. Regeneration of tissues plays an important part in resolution, for example re-growth of alveolar lining cells following pneumonia: this regrowth is dependent on the intrinsic ability of resident stem cells to divide and differentiate into mature tissue cells. Examples of resolution are recovery from sunburn (acute inflammatory response in the skin as a result of ultraviolet radiation exposure) and the restitution of normal lung structure and function following lobar pneumonia (see Figs 3.3 and 3.7 ).
Healing by fibrosis (scar formation) occurs when there is substantial damage to the connective tissue framework and/or the tissue lacks the ability to regenerate specialised cells. In these instances, dead tissues and acute inflammatory exudate are first removed from the damaged area by macrophages (see Fig. 3.6 ), and the defect becomes filled by ingrowth of a specialised vascular connective tissue called granulation tissue (see Fig. 3.8 ). This is called organisation . The granulation tissue gradually produces collagen to form a fibrous (collagenous) scar , constituting the process of repair (see Figs 3.8 and 3.9 ). Despite the loss of some specialised cells and some architectural distortion caused by the fibrous scar, structural integrity is re-established. Any impairment of function is dependent on the extent of loss of specialised cells. Modified forms of fibrous repair occur in bone after a fracture when new bone is created (see Fig. 3.11 ), and in brain with the formation of an astrocytic scar (see Fig. 23.2).
Abscess formation takes place when the acute inflammatory reaction fails to destroy/remove the cause of tissue damage and continues, usually with a component of chronic inflammation. This is most common in the case of infection by pyogenic bacteria. As the acute inflammation progresses, there is liquefaction of the tissue to form pus. At the periphery of this acute abscess, a chronic inflammatory component surrounds the area and fibrous tissue is laid down, walling off the suppuration (see Fig. 3.12 ).
| Concept | Definition/main features | Figure |
|---|---|---|
| Three major components of the acute inflammatory response |
|
3.1, 3.2, 3.3, 3.4 |
| Five cardinal signs of acute inflammation |
|
|
| Mediators of acute inflammation |
|
3.1, 3.2 |
| Outcomes of acute inflammation |
|
3.5, 3.6, 3.7, 3.8, 3.12 and clinical box (Inflammation out of control). |
| Wound healing |
|
3.9, 3.10, 3.11 |
Chronic inflammation may result following acute inflammation when an injurious agent persists over a prolonged period, causing concomitant tissue destruction, inflammation, organisation and repair. Some injurious agents elicit a chronic inflammatory type of response from the outset. Chronic inflammation is discussed fully in Ch. 4 .
Inflammation can do harm as well as good. In most people, inflammation is activated appropriately in response to some kind of adverse stimulus and terminated promptly when the infective organisms have been eliminated and tissue damage repaired. In some individuals, control of the inflammatory process is lost; this is called autoimmune disease .
One of the major stimuli of acute inflammation is a type of adaptive immune response where specific antibodies recruit components of the acute inflammatory response as effector mechanisms, thereby directing the non-specific components of the acute inflammatory system at specific targets ( E-Fig. 3.6 ).
Normally, the immune system is programmed to ignore antigenic stimuli within the individual’s own body, a process known as self-tolerance . When this self-tolerance is lost for one or more antigens, autoimmune disease results. These may be organ-specific antigens as in the organ-specific autoimmune disease Hashimoto’s thyroiditis (see Fig. 20.3 ) or, at the other end of the spectrum, the antigens triggering the response may be widespread in many tissues, as in systemic lupus erythematosus (SLE) , where the body reacts to double-stranded DNA as well as many other cellular antigens, causing systemic disease.
In many autoimmune disorders, inflammation and pain may cause major morbidity. Also, the misdirection of the immune response causes local tissue damage such as pain and swelling in the joints in rheumatoid arthritis (see Fig. 22.11 ) or renal failure due to renal damage in SLE. Many anti-inflammatory drugs are available but side effects are common and can be potentially serious. Recently, immunomodulating therapies have been used to treat some inflammatory conditions, e.g. anti-TNF antibodies in rheumatoid arthritis and inflammatory bowel disease. These therapies can have side effects, including an increased propensity to some infective organisms such as Mycobacterium tuberculosis, highlighting the complex immunomodulating role of biological therapies.
Many factors can delay or stop healing. A common factor is continuing inflammation where the initial stimulus to inflammation persists: a good example of this would be an infected wound or persistent peptic ulceration due to Helicobacter pylori infection (see Ch. 13 ). The presence of dead tissue or a foreign body in a wound can delay healing, as well as host factors such as poor nutrition, poor circulation, pre-existing diabetes mellitus or medications such as steroids.
C capillary F fibroblasts M macrophage My myocardial remnant P proteinaceous debris
Keloid is the name given to excessive formation of scar tissue. Some individuals have a tendency to form excessive amounts of collagen. When this occurs but does not extend beyond the bounds of the pre-existing wound, it is called a hypertrophic scar . However, when is extends beyond the pre-existing scar, it is called keloid and can give rise to a very unsightly appearance. Unfortunately, attempts to remove the excessive scar tissue surgically can give rise to even more keloid formation and a poor cosmetic outcome.
C capillaries D dermis F fibroblasts S scar
B bone C provisional callus Ca capillaries F fibrin G granulation tissue N neutrophils O osteoblasts Os osteoid
While most fractures heal completely and well with modern management, in some individuals, there is delay in complete healing of a fracture; in others there may be cessation of the healing process without restitution of functional bone, known as non-union . A wide range of factors may contribute to delayed or failed healing, the most important of which are poor immobilisation, poor blood supply, poor nutrition (lack of protein, vitamin C, etc.), the presence of infection and foreign bodies or fragments of necrotic bone in the fracture. Old age, drugs such as steroids and non-steroidal anti-inflammatory drugs (NSAIDs), diabetes mellitus, burns and irradiation may also delay or stop healing.
Fractures are described as complete or incomplete depending on whether the entire thickness of the bone is fractured. In closed fractures the overlying skin is intact, while compound fractures include disruption of the overlying skin. A comminuted fracture has multiple separate bone fragments. Pathological fractures occur in bone with pre-existing disease, such as osteoporosis or metastatic tumour. Greenstick fracture is an incomplete fracture where there is initial bending with partial fracture of the side under tension: it typically occurs in children.
Activation of T cells is dependent on antigen presenting cells APC . The antigen is taken up by an APC (e.g. macrophage, B lymphocyte, dendritic cell, Langerhans cell of skin) and broken down to short peptides. Processed antigen PA is then bound to a major histocompatibility complex molecule MHC , and the MHC-peptide complex is incorporated into the cell membrane so that the bound antigenic peptide is exposed to the extracellular fluid. Contact with a mature T cell bearing a T cell receptor with appropriate specificity activates the T cell. The type of response depends on whether the peptide is presented bound to MHC class I or II. Antigenic peptides bound to class II MHC molecules induce a T helper cell T H response needed to activate B cells B and cytotoxic T cells T C . B cell receptors (sIg) or T C receptor must also bind to the antigen for activation to occur. T H cells secrete a variety of interleukins IL that mediate activation, clonal expansion and maturation of the B or cytotoxic T cell response.
Antigen synthesised within a body cell (e.g. tumour cell, virus-infected cell) is presented on the APC plasma membrane bound to a class I MHC protein where it is recognised by cytotoxic T cells T C . Cytotoxic T cells are able to kill the abnormal cells directly. T H activation is also required for a T C response to be mounted.
B lymphocytes interact with unprocessed antigens. They recognise antigen by means of the BCR (surface immunoglobulin, sIg). In most cases, the unprocessed antigen is presented to the B cell on the surface of an APC such as a follicular dendritic cell in a lymphoid follicle. The majority of antigens can only activate a B cell if there is ‘help’ from an activated T helper cell T H . Activation without T cell help will occur if sIg binds to a protein or polysaccharide antigen with a repeating chemical structure (e.g. the polysaccharide coat of the bacterium Pneumococcus ). Such antigens are often known as T cell–independent antigens . Few naturally occurring antigens are of this type (not illustrated).
Production of antibodies by plasma cells. Mechanisms of antibody-mediated antigen elimination are as follows:
Antibody blocks the entry of organisms (such as viruses) into cells by binding to viral surface antigens.
Antigen-antibody complexes ( immune complexes ) activate complement to produce (among other factors) the membrane attack complex MAC , which punctures the outer membrane of the attacking organism.
Bound antibody with or without complement opsonises organisms and facilitates phagocytosis by neutrophils and macrophages .
Antibody is essential for antibody-dependent cell cytotoxicity ( ADCC ) (see below).
Antibody bound to toxins inactivates them and facilitates their removal by phagocytic cells.
Cell-mediated cytotoxicity is the destruction by apoptosis of abnormal cells by cytotoxic T cells, natural killer (NK) cells or antibody dependent cytotoxic cells. Certain types of organism, such as Mycobacterium tuberculosis , the cause of tuberculosis, activate T helper cells (T H 1) to secrete cytokines that in turn activate macrophages. Activated macrophages are more effective at killing phagocytosed organisms. This is the mechanism of type IV hypersensitivity ( chronic granulomatous inflammation ) (not illustrated).
Which ONE of the following combination of processes is an essential component of the acute inflammatory response?
Vascular dilatation, endothelial activation, macrophage activation and migration.
Vascular dilatation, vascular smooth muscle activation, macrophage activation and migration.
Vascular dilatation, endothelial activation, neutrophil activation and migration.
Vascular dilatation, vascular smooth muscle activation, neutrophil activation and migration.
Vascular constriction, endothelial activation, neutrophil activation and migration.
Which ONE of the following causes vasodilatation?
C5a
Leukotriene C4
Nitric dioxide
Histamine
Serotonin (5-HT)
Which ONE of the following is not one of the cardinal clinical features of acute inflammation?
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here