Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
On completion of this chapter, you should be able to:
Identify the echocardiographic distinction between pericardial effusion and pleural effusion
Describe the echocardiographic findings in cardiac tamponade
Describe the echocardiographic findings in dilated, restrictive, infiltrative, and hypertrophic cardiomyopathy
Recognize the echocardiographic findings of tumors, thrombus, vegetations, and normal variants
In addition to valvular heart disease, the sonographer may encounter fluid within the pericardial sac secondary to pericardial disease; enlargement of the cardiac chambers with regurgitant valvular flow as a result of dilated cardiomyopathy; increased thickness of the myocardium secondary to infiltrative cardiomyopathy; abnormal thickness of the interventricular septum compared with the posterior wall as seen in hypertrophic cardiomyopathy; or abnormal lesions within the cardiac chambers that represent tumors, thrombus, or vegetations. Each of these areas will be presented briefly to provide a basic understanding and investigative pathway for the sonographer.
The normal pericardium has two layers: the outer sac is the fibrous pericardium , and the inner sac is the serous pericardium . The inner layer is further divided into the visceral layer (epicardium) that is continuous with the pericardial surface and covers the heart and proximal great vessels and the fibrous parietal layer ( Fig. 34.1 ). The visceral layer is reflected to form the parietal epicardium . The visceral and parietal layers are separated by 15 to 50 mL of serous fluid. Their normal thickness is 1 to 2 mm and is abnormal if the thickness is greater than 4 mm.
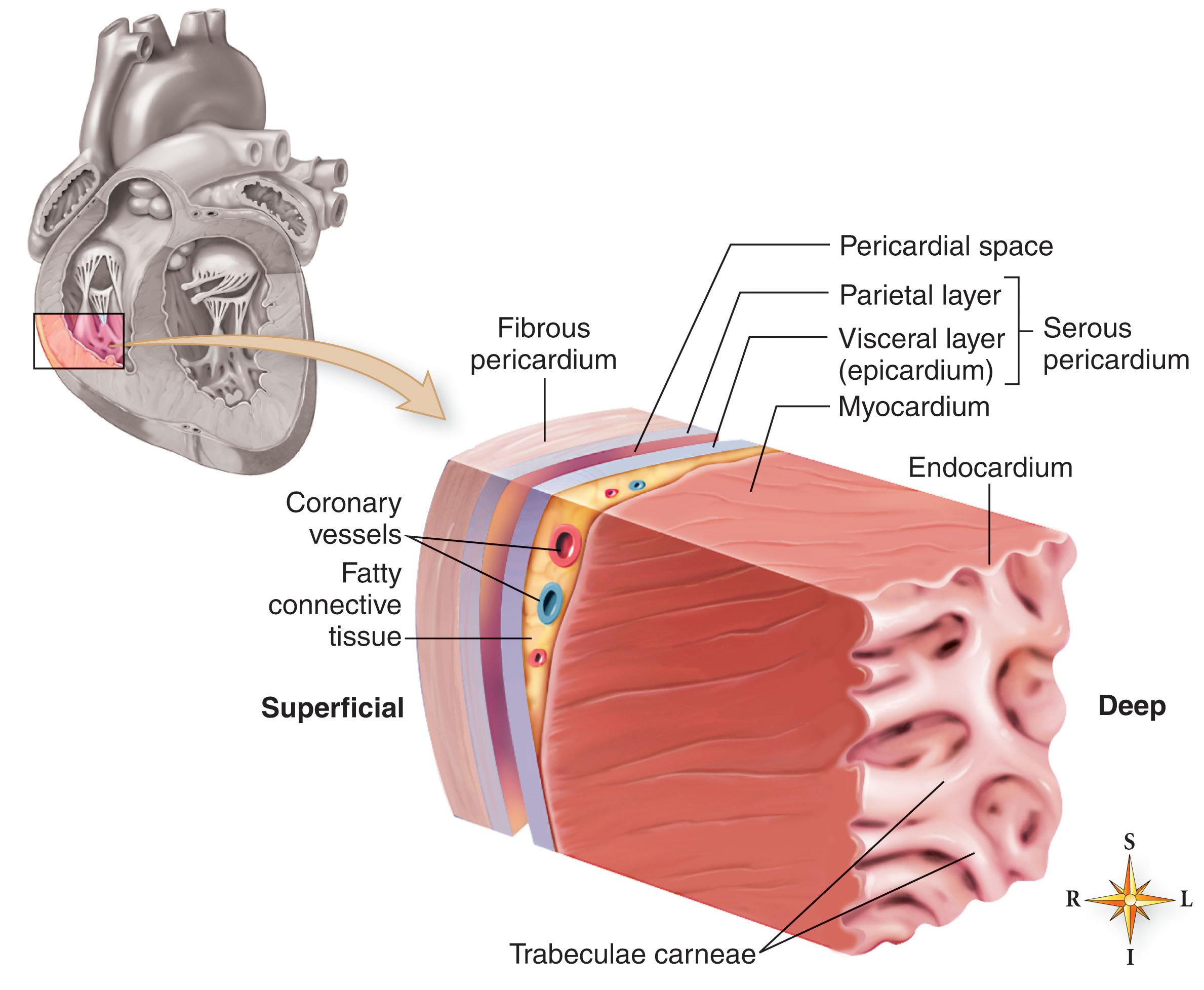
The pericardium has many functions. It provides a mechanical protection to the heart, provides a barrier to infection, reduces friction, and hemodynamically limits acute distention. The pericardial sac also contributes to the diastolic coupling and ventricular interdependence.
The pericardial effusion is recognized as an echo-free space between the visceral and parietal pericardium. This fluid can be transudative , exudative , malignant, or hemorrhagic. The effusions first accumulate posterior to the heart and, as the size increases, extend laterally and then circumferentially.
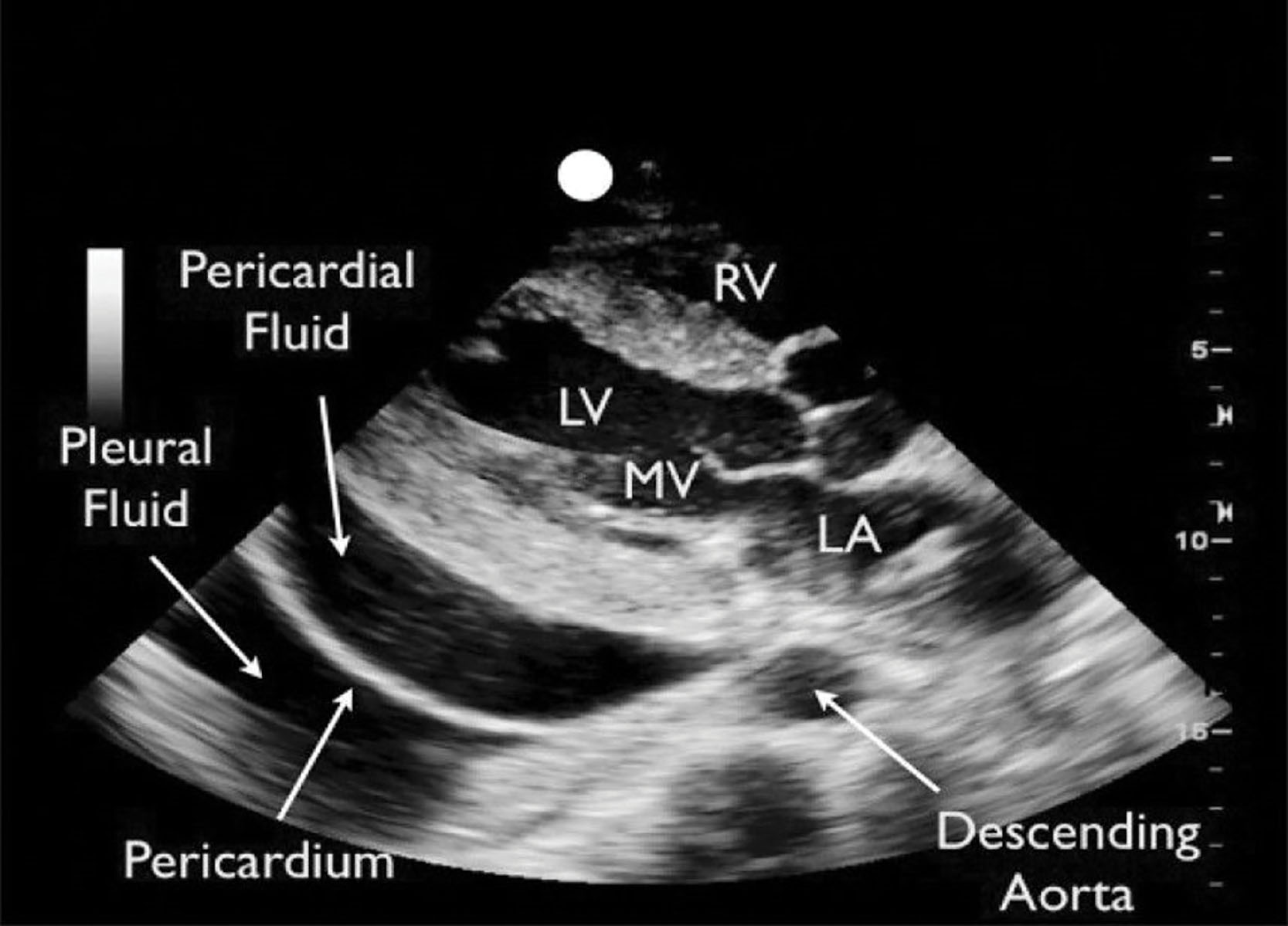
Echocardiography is the primary method for the initial detection of pericardial effusion that occurs when fluid accumulates in the pericardial sac. This fluid usually accumulates posterior to the heart because this is the most dependent surface. As the effusion increases, it extends laterally and then may surround the apex and the anterior surface of the heart. It is important to distinguish the pericardial effusion from a possible pleural effusion. This is best imaged with transthoracic echocardiography (TTE) in the parasternal long-axis view with the deep image view. The round circle of the descending aorta may be seen posterior to the left atrium and serves as a landmark to distinguish pericardial from pleural effusion ( Fig. 34.2 ). The pericardial effusion always is found anterior to the descending aorta, whereas the pleural effusion is posterior to the descending aorta. Recall that the pericardium reflects off the aorta at this junction.
The estimation of the amount of fluid around the heart may be made with echocardiographic evaluation ( Table 34.1 ). The small effusions are the most obvious during systole. If the effusion is greater than 25 mL, the echo-free space persists throughout the cardiac cycle. As the effusion increases in size, the parietal pericardium movement decreases. The small pericardial effusion would measure less than 0.5 cm in systole, a moderate effusion would measure 0.5 to 2.0 cm, and a large effusion would be greater than 2.0 cm ( Fig. 34.3 ).
| Size | Small | Medium | Large |
|---|---|---|---|
| Volume (mL) | <100 | 100–500 | >500 |
| Localization | Localized | Circumferential | Circumferential |
| Width (cm) | <1 | 1–2 | >2 |

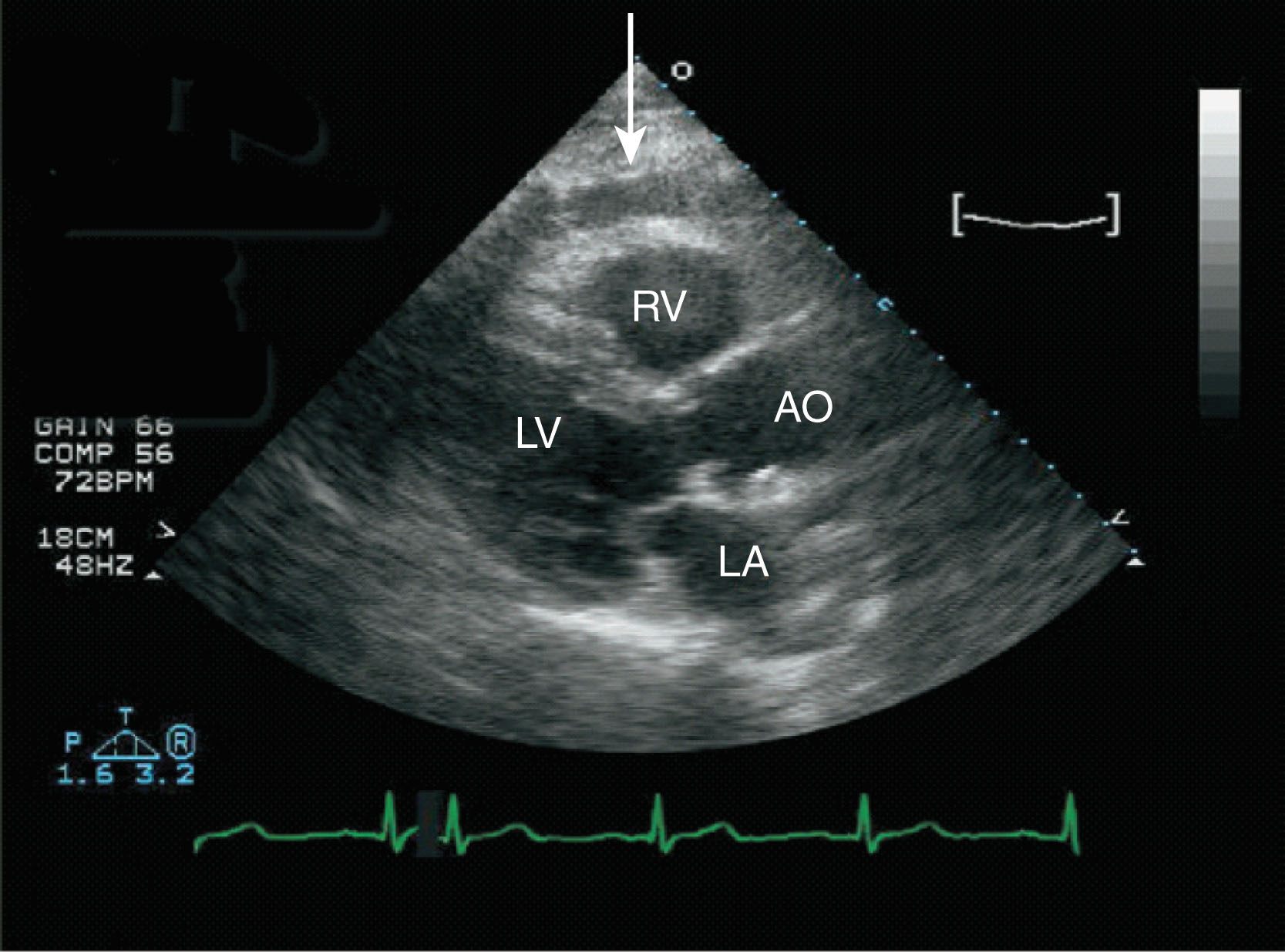
If there is only an echo-free space anterior to the right ventricle, the consideration of pericardial fat should be made ( Fig. 34.4 ). If the patient has chronic effusions, fibrinous stranding within the fluid and on the epicardial surface of the heart may be noted. Pericardial effusion with hemorrhage or purulent fluid is more heterogeneous and echogenic; such hemorrhagic effusions are more common in patients with metastatic disease. Loculated effusions may be seen in postsurgical patients ( Fig. 34.5 ).

When a large volume of pericardial fluid accumulates rapidly, it becomes an emergent clinical problem. Cardiac tamponade occurs when the pressure in the pericardium exceeds the pressure in the cardiac chambers, which results in impaired cardiac filling. The intrapericardial pressure increases to the point of compromising systemic venous return to the right atrium. The pressure and volume relationship is much stiffer when fluid accumulates rapidly.
Clinically the patient will exhibit hypotension and tachycardia as manifested by symptoms of low cardiac output. The clinical finding of pulsus paradoxus is closely related to the echo findings of reciprocal respiratory changes in both the right and left ventricular filling and emptying.
Echocardiographic findings are quite specific for tamponade: moderate to large pericardial effusion, right atrial systolic collapse, right ventricle diastolic collapse, reciprocal respiratory changes in ventricular volumes, and a dilated inferior vena cava with lack of respiratory changes ( Fig. 34.6 ). Doppler findings of the tricuspid and mitral inflow will show a respiratory variation in the right ventricular (greater than 40% variation) and left ventricular (greater than 25% variation) diastolic filling ( Boxes 34.1 and 34.2 ). There is increased right ventricular filling on the first beat after inspiration and decreased left ventricular filling on the first beat after inspiration ( Fig. 34.7 ).

Intrathoracic pressure decreases, transmitted to all cardiac chambers—intrapericardial and intracardiac pressures decrease.
SVC and IVC are mostly extrathoracic, and pressure remains relatively constant.
Pressure gradient between great veins and RA + RV increases, leading to increased right-sided heart filling. Tricuspid inflow varies by <25%.
↑ in RV volume causes small compensatory decrease in LV filling—“ventricular interdependence” (normal pulsus paradoxus <10 mm Hg).
Pulmonary veins and LA + LV are entirely intrathoracic and have an almost equal decrease in pressures; hence overall pressure gradient between PV and LV remains constant with little change in LV filling through the respiratory cycle. Mitral inflow varies by <15%.
Intrathoracic, intrapericardial, and intracardiac pressures increase, with a mild decrease in RV filling and a subsequent increase in LV filling.
On right side, decrease in intrathoracic pressure is transmitted to intrapericardial space and both pericardial and intracardiac pressures decrease relative to SVC/IVC pressures, leading to RV filling increases.
Tricuspid inflow varies >25% with respiration.
Ventricular interdependence makes septum bow toward LV to accommodate increased RV volume, leading to decreased LV filling.
On left side, there is a decrease in intrathoracic and PV (PCWP) pressures but only a small decrease in intrapericardial and intracardiac pressures. Therefore LV filling gradient decreases, leading to decreased LV filling.
Decreased LV filling allows for increased RV filling (ventricular interdependence).
Mitral inflow varies >15%.
RV filling is decreased slightly, and LV filling is restored.

In chronic pericardial disease the pericardium may become thickened and inelastic, which in turn limits the ventricular filling, leading to chronic biventricular diastolic dysfunction, right-sided heart failure, and low systemic output. This condition is secondary to long-standing pericardial inflammation with pericardial scarring, thickening, fibrosis, and calcification. Causes of constriction include prior cardiac surgery, acute or chronic/recurrent pericarditis, mediastinal radiation, tuberculous pericarditis, trauma, or idiopathic.
On echocardiography there may be thickening of the pericardial layer with increased echogenicity ( Fig. 34.8 ). There may be normal left ventricular size and systolic function. There is biatrial enlargement. The septum is quite abnormal: there may be an abrupt posterior motion of the ventricular septum in early diastole, a flat motion in middle diastole, an abrupt anterior motion following atrial contraction, or a septal “bounce” demonstrating an exaggerated interventricular dependence. The left ventricular posterior wall may be abnormal with a rapid movement during diastole, then flat during mid to late diastole, with an abrupt termination of ventricular filling. The inferior vena cava is dilated with minimal respiratory variation.

Doppler findings in constrictive pericarditis reflect abnormal hemodynamics ( Box 34.3 and Fig. 34.9 ). The mitral inflow pattern demonstrates increased early diastolic filling velocity followed by rapid deceleration, leading to a short filling time. There may be dynamic changes with respiration: early diastolic mitral inflow is reduced with the onset of inspiration, and the isovolumic relaxation time shortens and returns to normal with expiration. The pulmonary venous flow may show respiratory changes. The systolic wave and early diastolic wave velocities are increased during expiration and decreased during inspiration. The diastolic flow reversal is augmented in expiration in the hepatic vein flow. There may be a prominent y-descent on the hepatic vein or superior vena cava flow pattern.
Right atrial pressure: increased
RV/LV filling pressures: increased RV, equivalent LV
Pulmonary artery pressures: mild
RV diastolic pressure plateau: greater than one-third peak RV pressure
Diastolic filling: rapid, early, impaired late
2D echocardiography: pericardial thickening without effusion
Doppler: E > a on LV inflow
Prominent y-descent in hepatic vein
Prominent a wave in pulmonary valve flow
Respiratory variation in IVRT, E velocity
Other diagnostic testing: CT or MRI for pericardial thickening
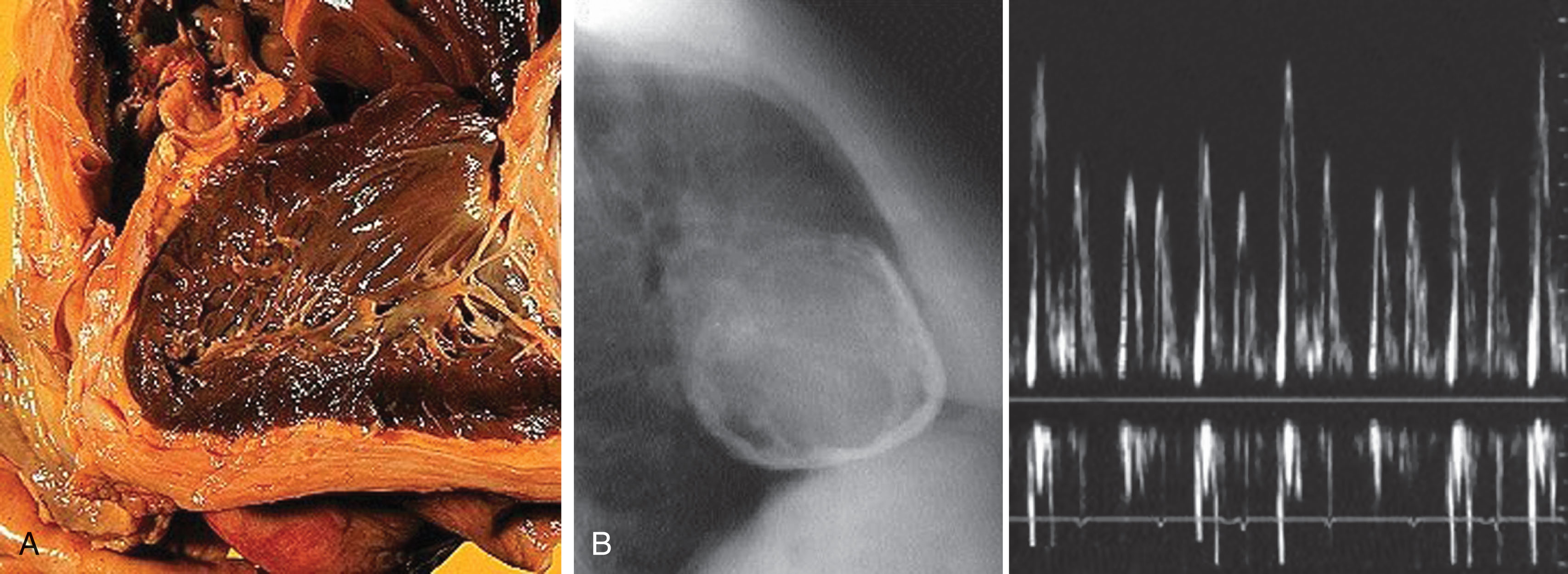
Cardiomyopathy may be defined as a primary myocardial disorder that is not related to the effects of valvular heart disease, hypertension, or coronary artery disease. Traditionally cardiomyopathies may be divided into these categories: dilated/congestive, restrictive/infiltrative, arrhythmogenic right ventricular dysplasia, hypertrophic, and unclassified (e.g., endomyocardial fibroelastosis). This section will focus on dilated, restrictive, and hypertrophic cardiomyopathies. Echocardiography plays a primary role in the assessment of the patient with congestive heart failure and suspected cardiomyopathy. The echocardiographic study not only provides prognostic information but also serves as a guide to the success of therapy.
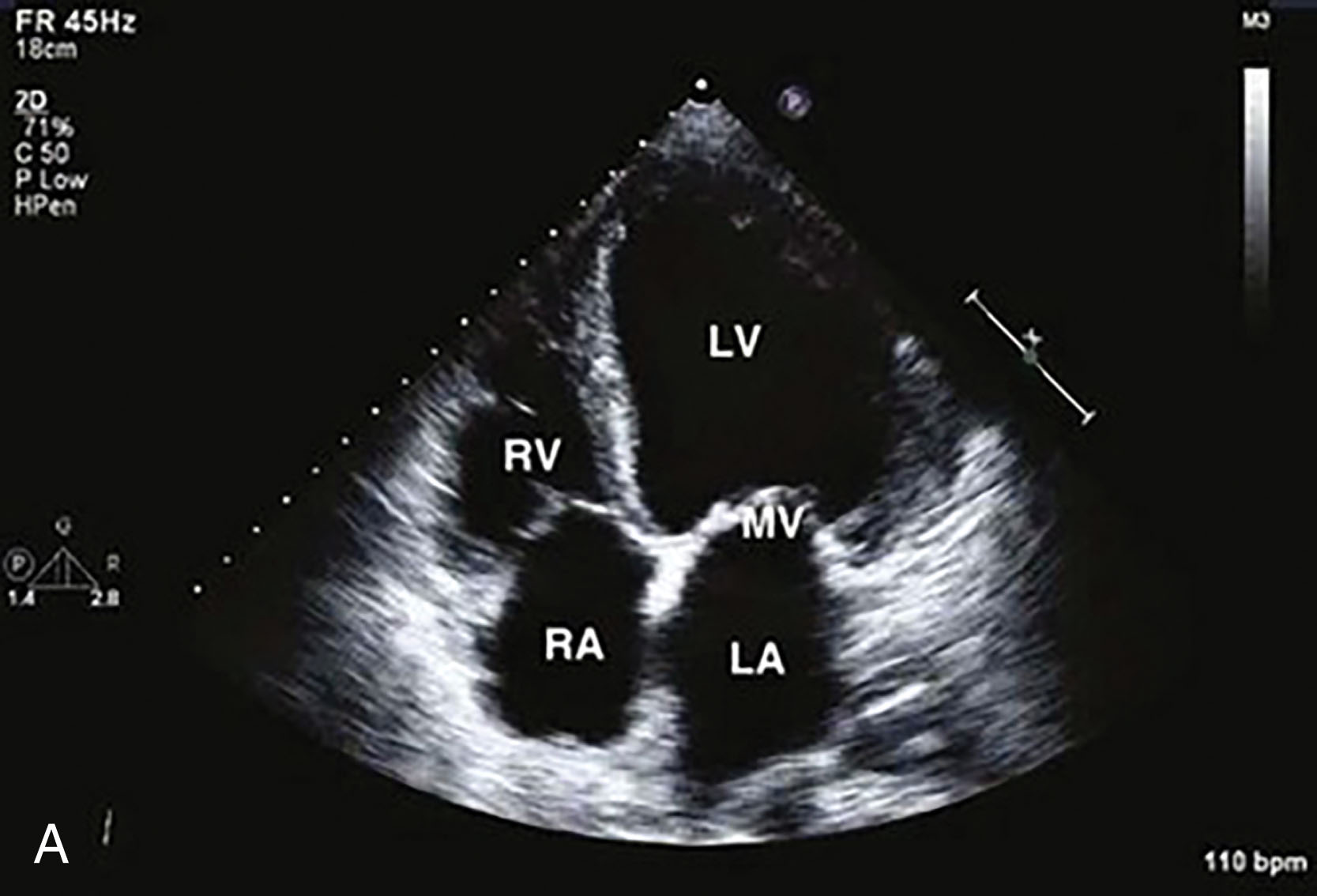
Dilated cardiomyopathy is characterized by the dilation and reduced contractility of the left ventricle or both the left and right ventricles. There are multiple causes of dilated cardiomyopathy, as noted in Box 34.4 . The abnormalities that may be seen with two-dimensional (2D) echocardiography include the following:
Left ventricular dilation ( Fig. 34.10A ).

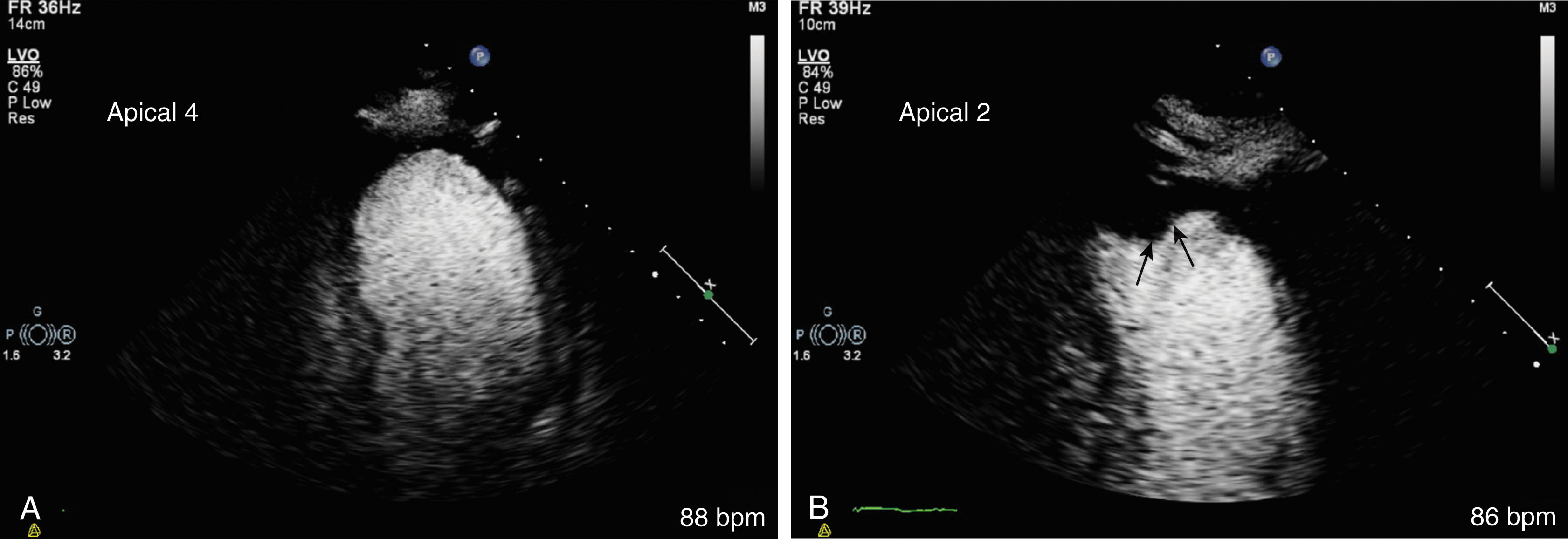
Apical and lateral displacement of the papillary muscles with resultant functional mitral regurgitation (MR) due to the reduction in the length of the mitral valve apparatus in comparison with the annulus ( Fig. 34.10B ). The mitral valve leaflets may coapt only at the tips or not at all. In addition, there may be a shortened length of coaptation at the zona coapta.
Increased distance between the interventricular septum and the E point of the anterior leaflet mitral valve ( Fig. 34.10C ).
“B” bump seen when there is a delayed closure of the mitral valve which correlates with an elevated end-diastolic pressure in dilated cardiomyopathy ( Fig. 34.10D ).
Left ventricular thrombus ( Fig. 34.11 ). Contrast should be used if there are wall motion abnormalities and the endocardial layer is not clearly identified.
Normal to reduced wall thickness ( Fig. 34.12 ).

Reduction of all of the systolic indices (see Fig. 34.12 ).
Idiopathic
Familial
Noncompacted myocardium
Postpartum cardiomyopathy
Hemochromatosis
Infectious
Postviral myocarditis
Human immunodeficiency virus
Legionella infection
Gram-negative sepsis
Toxic cardiomyopathy
Adriamycin/chemotherapy agents
Alcohol
Carbon monoxide poisoning
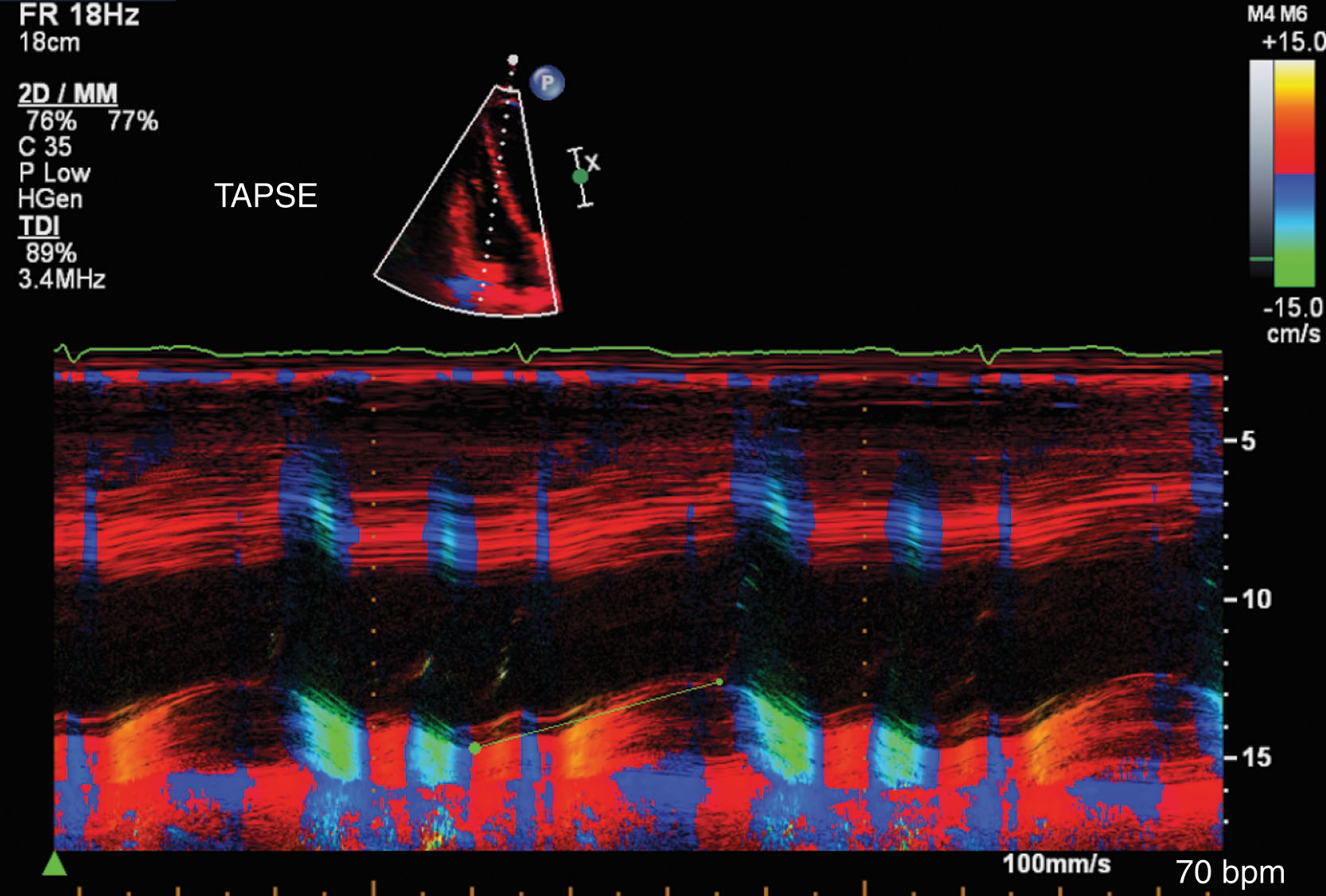
Additional echocardiographic findings may include the following changes. Left atrial dilation may be present; the degree of dilation is dependent on the duration of the cardiomyopathy. It also is a marker of more severe and long-standing ventricular dysfunction. Pulmonary hypertension may be present, as well as tricuspid regurgitation ( Fig. 34.13 ). There may be dilation of the right ventricle with pulmonary hypertension, or there may be pathologic involvement of the right ventricle. The prognosis is worse when the right ventricle is involved. Measurement of tricuspid annular plane systolic excursion (TAPSE) of less than 15 mm indicates abnormal movement of the right ventricular free wall. This measurement is made with the M-mode cursor parallel to the base of the tricuspid annulus ( Fig. 34.14 ).
It is challenging to distinguish with echo between ischemic and nonischemic cardiomyopathy. With end-stage ischemic disease, the sonographer will note regional wall motion abnormalities, calcification, and thin, bright, echogenic wall segments that may or may not have aneurysm formation. The left ventricular ejection fraction is moderately to severely depressed. The right ventricular systolic function is normal, whereas the pulmonary pressure is elevated. There is at least moderate MR.
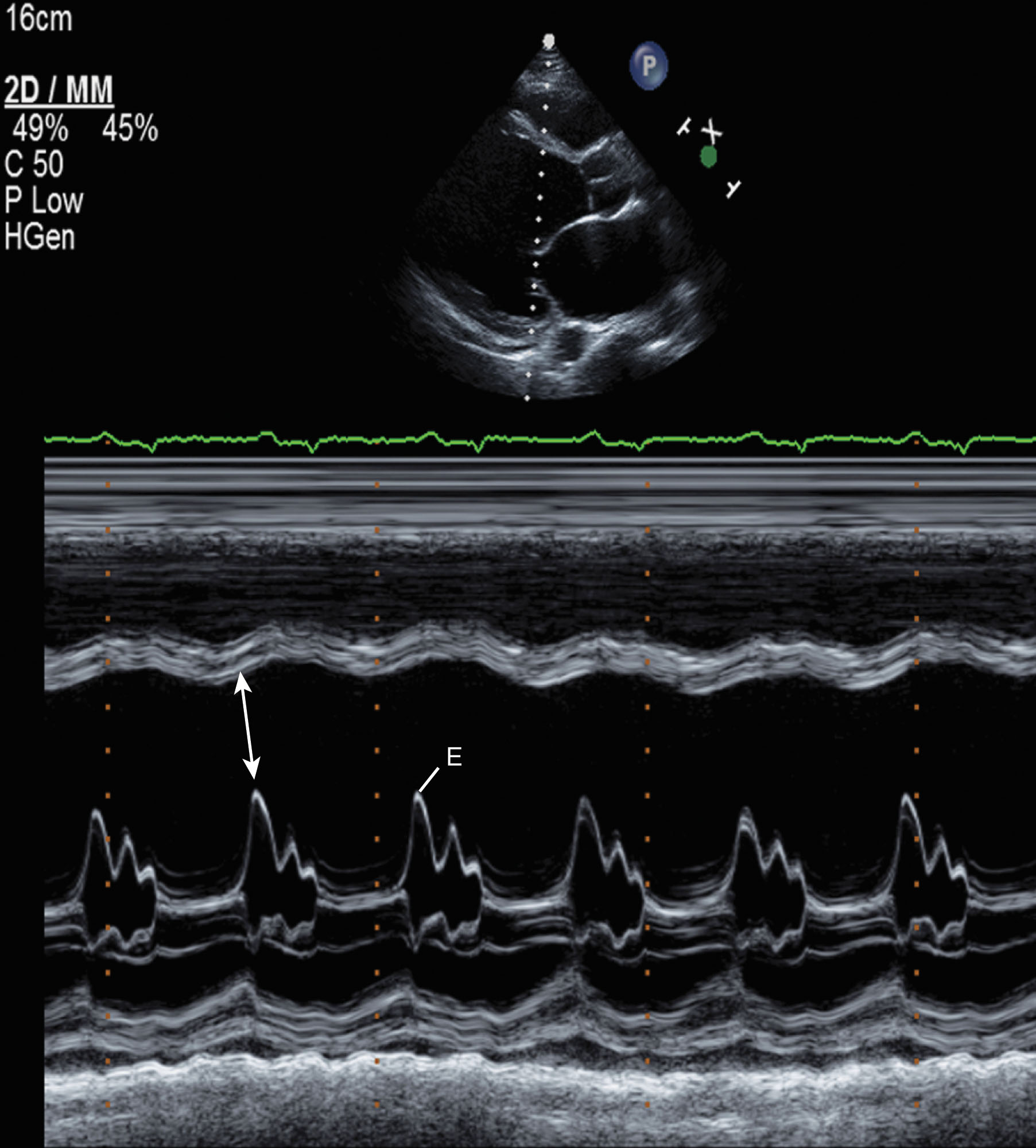
Left ventricular volumes are useful to monitor clinical progress. Quantitative measurements should be obtained from all chambers with either 2D or three-dimensional (3D) imaging ( Box 34.5 ). The end-diastolic volume index and end-systolic index should be calculated and related to body surface area. The M-mode analysis of the mitral valve tracing ( Fig. 34.15 ) may demonstrate an increased distance between the E point of the anterior leaflet of the mitral valve and the corresponding (synchronous) interventricular septal endocardium. The normal E-point separation should be less than 6 mm; with ventricular enlargement, this distance increases. Also noted on this M-mode tracing is the “B bump,” which signifies the delayed closure of the mitral leaflets between the leaflet coaptation points. This indicates increased left ventricular end-diastolic pressure (LVEDP), typically exceeding 20 mm Hg.
Quantitative measurements should be obtained from all chambers with 2D/3D imaging:
End-diastolic volume index >100 mL/m 2
Cardiac output calculations are frequently normal:
EF usually between 20% and 40%
Maintain high end-diastolic volumes and elevated heart rates
End-systolic volume index:
Exceeds 30 mL/m 2 important indicator of global LV dysfunction
ICM and global LV dysfunction with evidence of infarction, ESVI of 45 mL/m 2 identifies patients with a poor outcome
The aortic root motion may also indicate cardiac function. The systolic anterior motion (SAM) of the aortic root is proportional to the cardiac output ( Fig. 34.16 ). The diastolic backward motion of the aortic root is determined by the emptying dynamics of the left atrium during left ventricular filling. This movement reflects the left ventricular diastolic filling pattern. The aortic root motion in low-cardiac-output state will demonstrate a reduced anterior systolic motion of the root. The aortic valve opening is also reduced and its duration is abbreviated with a slow closure at end systole.

If the patient has MR, the continuous wave (CW) Doppler velocity from the MR can be used as an index of global systolic function. The rate of rise of the Doppler velocity reflects the rate of rise of the left ventricular pressure. A dp/dt (change in pressure over change in time) less than 800 mm Hg/sec indicates severe dysfunction of the left ventricle (see Fig. 34.16C ).
The Doppler evaluation of left ventricular function is outlined in Box 34.6 . Forward flow and the diastolic properties of the right and left ventricles may be assessed. The assessment of forward flow is made through time velocity interval, stroke volume, and cardiac output (see Chapters 31 and 32 ). The diastolic properties of the left ventricle may be determined by the E/A ratio, dispersion of the E-wave velocity, color Doppler M-mode velocity of propagation, deceleration time, pulmonary vein flow, and annular Doppler tissue imaging. The diastolic properties of the right ventricle are determined by the Doppler flow in the hepatic veins and the Doppler flow in the superior vena cava.
Time-velocity interval
Volume-based stroke volume
Determine cardiac output
Mitral inflow pattern:
E/A ratio
Dispersion of E-wave velocity
Color Doppler M-mode velocity of propagation
Deceleration time
Isovolumic relaxation time
Pulmonary vein flow
Annular Doppler tissue imaging
Doppler flow in hepatic veins
Superior vena cava Doppler flow
The adverse prognosis of a patient with cardiomyopathy may be determined by the left ventricular size and function ( Box 34.7 ) and the diastolic properties of the left ventricle.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here