Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Venous disease, both acute (deep vein thrombosis [DVT], pulmonary embolism [PE]) and chronic (postthrombotic syndrome, ulcers, varicose veins), is widely prevalent, affecting up to a quarter of the population.
Superficial vein disease can be highly symptomatic but often easily treated with minimally invasive ablative techniques.
The superior vena cava (SVC) syndrome manifests as severe congestion and edema of the face, arms, and upper thorax, and may progress to dyspnea, cognitive dysfunction, and headache. It is commonly associated with malignancy, indwelling catheters, or pacemaker leads.
Patients with Paget-Schroetter syndrome (PSS) develop spontaneous DVT of the upper extremity, usually in the dominant arm, after strenuous physical activity. Heavy repetitive exertion causes microtrauma to the vessel intima and leads to activation of the coagulation cascade.
Upper extremity DVT not associated with indwelling catheters or pacemaker leads should prompt further workup for a vascular compression syndrome.
Duplex ultrasonography is generally the initial test of choice for the diagnosis of DVT (in upper or lower extremities) due to its widespread availability, portability, reproducibility, and cost. However, it has a limited role for central venous stenosis.
Clinical PE has a high prevalence in untreated proximal DVT and is associated with a high mortality rates. Under treatment, the incidence of PE decrease to 5%, and the mortality to less than 1%.
Complete inferior vena cava occlusion is becoming increasingly recognized as a major source of venous morbidity. Iliocaval reconstruction using modern endovenous techniques has excellent long-term patency.
Retrievable inferior vena cava filters are important adjuncts in the care of patients with contraindications to anticoagulation or recurrent PE. They can also be beneficial in the treatment of patients with “free-floating” iliac vein thrombus, or in patients with compromised cardiopulmonary reserve.
Percutaneous mechanical thrombectomy is an important adjunct to catheter-directed thrombolysis and may result in a shorter time to vein patency, shorter length of hospitalization, reduction in hemorrhagic risk, and overall cost savings.
As interventional cardiologists have expanded their skill set from coronary interventions to the endovascular management of peripheral arterial disease, venous intervention has also gradually become a common part of the “global cardiovascular interventionalist’s” repertoire. In fact, as more and more interventional cardiologists recognize the high prevalence, morbidity, and mortality associated with venous disease in their patient populations, more have become interested in the management of venous disorders. These include both the medical and catheter-based treatment of central venous stenosis, upper and lower extremity deep venous thrombosis (LEDVT), and the insertion and retrieval of inferior vena cava (IVC) filters, to name a few.
Veins are larger in caliber and more numerous then arteries. Arteries of the extremities run in the deep compartments, protected from superficial injury. Veins, on the other hand, course in both the deep and superficial spaces. In addition, the venous system has a much greater volume capacity than the arterial system, with thinner and less elastic walls. Most anatomists distinguish three layers in the walls of veins: tunica intima, tunica media, and tunica adventitia. While the distinctions between the layers are subtle, in general the venous internal elastic membrane is poorly defined, and the tunica media is much less developed compared to that of arteries.
Similar to the arterial system, veins are commonly categorized into three major groups: large size veins, medium size veins, and venules and small size veins. Only medium and large size veins will be discussed in this chapter; for a complete review of venous embryology and anatomy, please refer to an anatomic text book.
Medium size veins range between 2 and 9 mm in diameter. These include veins from extremities distal to the axillary or inguinal crease, and cutaneous veins. The intima consists of endothelium, basal lamina, and reticular fibers. The media consists of a very thin layer of circular smooth muscle and few collagen fibers. The adventitia, the thickest of all the layers, consists of both collagen and elastic fibers.
The large size veins consist of veins central to the axillary or inguinal crease, superior vena cava (SVC), IVC, renal veins, hepatic veins, and azygos veins. The intima is similar to the medium size veins. Interestingly, a tunica media is lacking in most of the large veins, with the exception of the gravid uterus and pulmonary veins. A thick adventitia makes up the greater part of the thickness of the wall. This layer is rich in elastic fibers and longitudinally oriented collagen. The IVC is exceptional in that its adventitia contains scattered longitudinal bundles of smooth muscle. Large size veins, similar to large arteries, get their nutrient blood supply from very small penetrating vessels called vasa vasorum.
Valve leaflets are a thin fold of the intima, with a thin layer of collagen and a network of elastic fibers that extend toward the intima of the vessel wall. Anatomically, valves are generally bicuspid in structure and are more numerous in the veins of the lower extremity, where the force of gravity is greatest. In general, small and large size veins have no valves. The wall of the vessel becomes thinner and slightly expanded just above the attachment of each valve cusp, creating a cavity called the valve sinus . When competent, the valves are forced shut by the weight of the column of blood above and the manner in which this column interacts with the valve sinus. This interaction causes valvule coaptation, preventing venous reflux ( Fig. 45.1 ).
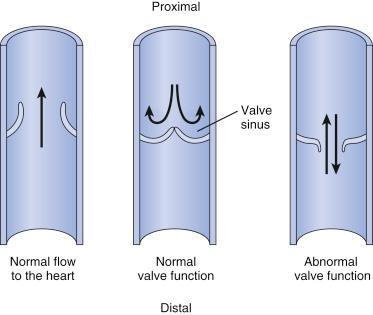
Only medium size veins have valves, as their predominant function is to ensure the antegrade flow of blood. This includes flow both peripheral to central and superficial to deep, with unidirectional valves preventing retrograde flow of blood away from the heart
The venous system has a large capacity, accounting for approximately two-thirds of the systemic blood volume. Veins, because of their unique vascular structure, can undergo a large change in volume with minimal change in transmural pressure. This characteristic is called venous capacitance .
Because of their low elastic tissue content and collagen dense adventitia, veins are actually stiffer than arteries when compared at the same distending pressure. A person standing at rest has significantly elevated venous pressures at the level of the feet and calves, accumulating a large volume of blood in the lower extremities. The calf muscles augment venous return by working as a hydraulic pump. During walking or exercise, calf muscle contraction pushes the accumulated blood toward the heart, decreasing the venous pressures close to zero. The venous pressure remains low even during calf muscle relaxation. At this time, the unidirectional venous valves prevent backflow of blood, but allow antegrade flow towards the heart. It is imperative that the venous valves are competent for the calf pump system to work efficiently. A normal pump system reduces venous pressures and volume in the exercising muscle, increases venous return, and improves arterial perfusion.
Incompetent valves in the saphenous system permit reflux of blood from the central veins to the peripheral veins. The superficial veins dilate once the volume of retrograde flow exceeds their capacity; this ultimately leads to poor coaptation and venous valve incompetence. This valve incompetence can occur in the deep and superficial systems, creating a standing column of blood with a constant increase in pressure. The transmission of an elevated venous pressure into the superficial system leads to the characteristic clinical sequalae known as varicose veins.
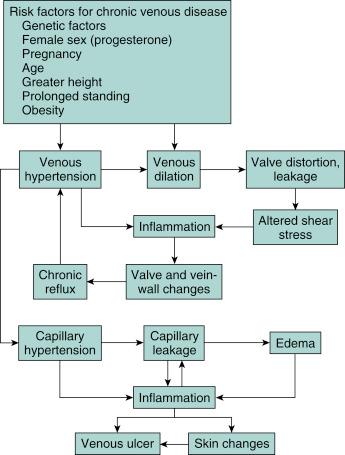
Chronic venous insufficiency is a significant problem in the United States, affecting as much as 25 to 40 million people. Venous valve incompetence is central to the underlying venous hypertension that appears to underlie most or all signs of chronic venous disease. Chronic venous disease afflicts a younger segment of the population, and the morbidity of edema, leg pain, and ulceration may result in lifestyle alterations, loss of work, and frequent hospitalizations. The prevalence of venous ulcerations is not restricted to the elderly, but certainly increases with age. It has been estimated that venous ulcers have had a major negative economic impact, with the loss of approximately 2 million working days, and incur treatment costs of approximately $3 billion dollars per year in the United States. The chief clinical manifestations of chronic venous disease are aching, leg pain, heaviness, a sensation of swelling, itching, cramping, and restless legs. Chronic venous disease can be graded according to the descriptive clinical, etiologic, anatomical, and pathophysiological (CEAP) classification, which provides an orderly framework for communication and decision making ( Table 45.1 ). The pathophysiology of chronic venous disease in regard to its clinical expression has been well described, involving venous valve incompetence, structural changes in the vein wall (manifest as hypertrophy), and the role of elevated venous pressure and shear stress. There has been new progress made in understanding the pathophysiology of the skin changes of chronic venous disease. These studies have emerged to validate that chronic inflammation has a key role in the skin changes of chronic venous disease. Support for the role of chronic inflammation in chronic venous disease has come to be known as the microvascular leukocyte-trapping hypothesis, where elegant studies have shown elevated numbers of macrophages, T lymphocytes, and mast cells in skin biopsy specimens from lower limbs affected by chronic venous disease. The chronic inflammatory state in patients with chronic venous disease is related to the skin changes that are typical of the condition. Increased expression and activity of metalloproteinase (MMP, especially MMP2) has been reported in lipodermatosclerosis, venous leg ulcers, and wound fluid from nonhealing ulcers.
| Clinical Classification | |
| C 0 | No visible or palpable signs of venous disease |
| C 1 | Telangiectasias, reticular veins, malleolar flares |
| C 2 | Varicose veins |
| C 3 | Edema without skin changes |
| C 4 | Skin changes ascribed to venous disease (e.g., pigmentation, venous eczema, lipodermatosclerosis) |
| C 4a | Pigmentation or eczema |
| C 4b | Lipodermatosclerosis or atrophie blanche |
| C 5 | Skin changes as defined above with healed ulceration |
| C 6 | Skin changes as defined above with active ulceration |
| S | Symptomatic, including ache, pain, tightness, skin irritation, heaviness, and muscle cramps, and other complaints attributable to venous dysfunction |
| A | Asymptomatic |
| Etiologic Classification | |
| Ec | Congenital |
| Ep | Primary |
| Es | Secondary (postthrombotic) |
| En | No venous cause identified |
| Anatomic Classification | |
| As | Superficial veins |
| Ap | Perforator veins |
| Ad | Deep veins |
| An | No venous location identified |
| Pathophysiologic Classification | |
| Pr | Reflux |
| Po | Obstruction |
| Pr,o | Reflux and obstruction |
| Pn | No venous pathophysiology identifiable |
The treatment of chronic venous disease has evolved considerably over the past few years and is aimed at preventing venous hypertension, venous reflux, and chronic inflammation ( Fig. 45.2 ). Compression stockings remain the initial therapy for controlling venous hypertension, and are often the initial treatment before undertaking an interventional procedure such as endovenous laser ablation (EVLA). Endovenous ablation procedures utilizing laser energy or radio frequency are now available to treat venous reflux, and are becoming a common part of the armamentarium used to combat chronic venous disease. As such, this has become an area of interest of those “vascular minded” interventional cardiologists.
EVLA is a percutaneous technique that uses laser energy to ablate incompetent superficial veins. The axial veins are the primary target for this therapy and include the great saphenous vein (GSV), small saphenous vein (SSV), and accessory saphenous veins (ASVs).
The other alternative approaches to the treatment of chronic venous disease include radiofrequency energy, liquid and foam sclerotherapy, and open surgical management. A review of these other modalities is beyond the scope of this chapter and, as such, we will focus on EVLA.
The indications for EVLA are the same as other vein ablation therapies, including radiofrequency ablation, sclerotherapy, and open surgical stripping. The use of open surgical vein stripping for symptomatic varicose veins has become less as these other percutaneous options have evolved.
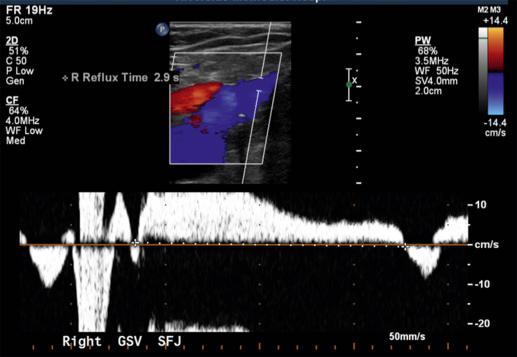
Venous ablation is indicated in patients with symptoms and signs of venous disease that persist in spite of a trial of medical management. This constellation of symptoms and clinical findings would include chronic leg pain and achiness, recurrent stasis cellulitis, venous stasis ulcers, and preulcerative skin changes. From a physiologic standpoint, documenting reflux in the target vein (i.e., retrograde flow >0.5 seconds) is essential before entertaining EVLA ( Fig. 45.3 ). In addition, the patient’s symptoms should directly relate to the incompetent veins being treated.
Absolute contraindications for EVLA would include patients with acute deep vein thrombosis (DVT) and pregnancy, due to the risk of developing a new DVT. We prefer to wait a minimum of 6 weeks after delivery in pregnant patients before performing EVLA.
Relative contraindications to EVLA for a given vein segment include
Chronic or recurrent phlebitis in the target vein, since formation of synechiae in the vein can prevent passage of the laser sheath. This decision should be based on duplex ultrasound screening.
Passage of the device may not be possible in severely tortuous venous segments.
Due to the concern of skin burns, the target vein should ideally be at least 1 cm deep to the skin dermis after the tumescent anesthesia is applied.
Aneurysms of the GSV are at a greater risk of procedural failure. In general, an aneurysmal vein segment of greater than 2.5 cm is not ideal for EVLA.
As patients may present with “mixed” arterial and venous disease, patients with lower extremity venous ulcers should be evaluated for peripheral arterial disease. These patients should at a minimum have a lower extremity ankle/brachial index (ABI) measurement to ensure the adequacy of the arterial circulation. If the patient is found to have peripheral arterial disease, especially if the ABI is 0.7 or less, the arterial disease should be addressed prior to treating the venous disease.
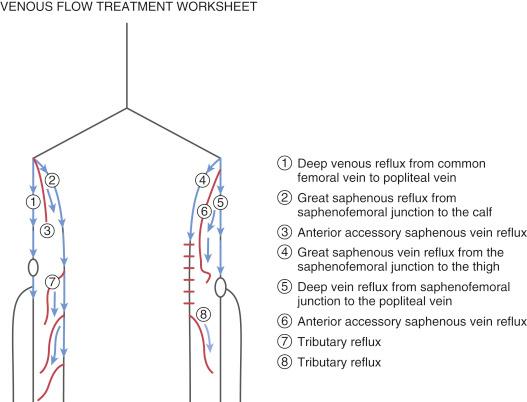
The preoperative evaluation should include a comprehensive history and physical exam, and obtaining noninvasive vascular laboratory studies. A venous duplex ultrasound of the affected lower extremity should evaluate the deep veins, GSV, ASVs, if present, and SSV for patency, as well as the presence of reflux (see Fig. 45.3 ). Ideally, a “treatment roadmap” should be drawn out for every patient that illustrates the target vein, important perforators that may require foam sclerotherapy, and anatomic details such as vein tortuosity that may influence the procedural approach ( Fig. 45.4 ).
Aspirin, antiplatelet agents, and nonsteroidal antiinflammatory drugs (NSAIDs) are sometimes discontinued to limit postoperative bruising. The decision to withhold these medications must be made on a case-by-case assessment of risk/benefit in individual patients.
For patients who are on full dose anticoagulation, cessation of anticoagulation is not necessary prior to EVLA. Perioperative anticoagulation with low molecular weight heparin or warfarin does not appear to affect the success of EVLA.
If cessation is selected, warfarin may be temporarily discontinued or bridging anticoagulation initiated with heparin or low-molecular-weight heparin. If the patient is on a direct oral anticoagulant (DOAC), and cessation is felt to be clinically indicated for EVLA, the DOAC can be withheld for 24 hours before EVLA.
Antibiotic prophylaxis is generally not needed prior to EVLA. However, when EVLA is combined with vein excision, antibiotics should be administered to decrease the risk of surgical site infection.
EVLA is a brief outpatient procedure with an overall low risk of DVT. DVT prophylaxis is needed only for high-risk patients.
Considerations for adjunctive procedures should be based on the preprocedure history and physical, and the venous duplex “treatment roadmap” done prior to EVLA. The adjunctive procedures may include percutaneous phlebectomy and/or foam sclerotherapy at the same time as EVLA, or in a staged fashion.
Patients are typically prescribed knee high/thigh high compression stockings (20 to 30 mm Hg), which they should obtain prior to the day of the scheduled procedure. However, the type of compression prescribed and the length of time in compression may vary widely, and is ultimately up to the treating physician.
A prescheduled appointment should be made for follow-up duplex examination 2 to 3 days after EVLA to assess for DVT.
On the day of the procedure, the veins to be ablated can be marked (i.e., marked with indelible ink) with duplex ultrasound to aid in the administration of tumescent anesthesia, but it is generally not necessary. This may be useful when first getting started using EVLA.
EVLA can be performed in a clinic or office or other outpatient surgery setting with oral diazepam and local anesthesia with or without supplemental venous procedures. The dose and type of oral sedative should be made on a case-by-case basis, based on the patient’s comorbid medical conditions. If moderate sedation is used, a nurse should be dedicated to monitoring the patient’s level of consciousness, heart rate, blood pressure, and oxygen saturation. Local tumescent anesthesia is always required, even if the patient undergoes general anesthesia in an operating room. It is beyond the scope of this chapter to provide specific instructions on how to perform EVLA. There are numerous textbooks now available on both the techniques and technology of EVLA.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here