Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Upper tract urothelial carcinoma (UTUC) refers to malignant changes of the uroepithelial cells lining the urinary tract anywhere from the renal calyces to the ureteral orifice.
Urothelial carcinoma is relatively common worldwide, and its incidence has a marked geographic variation. Urothelial carcinoma may be located in the lower urinary tract; the bladder or urethra, or the upper urinary tract; renal calyces, renal pelvis and ureter. Bladder cancers (BCas) account for 90%–95% of urothelial carcinomas and are the second most common malignancy of the urogenital tract after prostate cancer. UTUCs are uncommon and account for only 5%–10% of urothelial carcinomas. Urothelial carcinomas account for 90% of tumours of the renal pelvis; other epithelial cell types include squamous cell carcinoma, adenocarcinoma, carcinosarcoma, small cell carcinoma and papilloma.
The accuracy of incidence and mortality statistics for renal pelvic UTUC in the USA is limited by the fact that UTUC and renal cell carcinoma are grouped together for reporting purposes. The estimated annual incidence of UTUCs in Western countries is approximately 1–2 new cases per 100,000 inhabitants. Pyelocalyceal tumours are about twice as common as ureteral tumours. In 17% of cases a concurrent bladder cancer is present. Recurrence of disease in the bladder occurs in 22%–47% of UTUC patients and recurrences in the contralateral upper tract are observed in 2%–6%.
The natural history of UTUCs is different from that of bladder cancer; 60% of UTUCs are invasive at diagnosis compared with only 15% of bladder tumours.
Hereditary cases of UTUC are linked to hereditary non-polyposis colorectal carcinoma (HNPCC), which should be suspected if the patient is below 60 years of age or has a history of an HNPCC-type cancer.
UTUC has a peak incidence in patients in their 70s and 80s. UTUC is three times more prevalent in men than in women, predominantly because of the higher rates of smoking and occupational exposure to carcinogens among men.
Chemical carcinogens predispose the entire urothelium to the development of multifocal urothelial carcinoma by an effect sometimes referred to as field change of the urothelium. Smoking is the principal risk factor for UTUC, increasing the relative risk nearly three-fold. Occupational exposure is most commonly caused by aromatic amines found in certain chemicals used in many industries—e.g. dyes, textiles, rubber, chemicals, petrochemicals and coal—and is responsible for ‘amino tumours’. Chemicals such as benzidine and β-naphthalene were banned in the 1960s in most industrialised countries. Usually a UTUC caused by occupational exposure to aromatic amines is also associated with a bladder tumour. The average duration of exposure needed for a UTUC to develop is 7 years, but the latency following exposure can be up to 20 years.
UTUCs resulting from the drug phenacetin have almost disappeared after the product was withdrawn in the 1970s. UTUC is very common in patients and families afflicted with Balkan endemic nephropathy (BEN). The incidence is highest in Croatia, Bosnia, Bulgaria, Romania and Serbia. Aristocholic acid (AA), contained in Aristolochia fangchi and Aristolochia clematitis , plants endemic to the Balkans, is thought to play a role in the aetiology of UTUC. AA is the cause of Chinese herb nephropathy (aristocholic nephropathy or AAN), which is a rapidly progressive interstitial fibrosing renal disease with frequent urothelial malignancies. A high incidence of UTUC has also been described in Taiwan, especially in the population of the southwest coast, representing 20%–25% of urothelial carcinomas in the region.
Most urothelial carcinomas of the renal pelvis are of high histological grade and present in advanced stages, whereas most bladder cancers present as low grade and stage.
In the region of 40% of renal pelvis urothelial carcinomas can show unusual atypical features and metaplastic phenomena. Squamous cell carcinoma of the upper urinary tract represents less than 10% of upper tract malignancies. This is usually associated with chronic inflammation, most often associated with urolithiasis. Other rare histological variants include micropapillary carcinoma, sarcomatoid carcinoma and lymphoepithelioma.
Prognostic factors predict the disease outcome, including the chance of recovery or recurrence. Prognostic factors are significant and important in clinical practice as they can guide selection of treatment and the timing of surveillance. Identification of prognostic factors, determining which patients can benefit from endoscopic versus radical nephroureterectomy (RNU), neoadjuvant or adjuvant chemotherapy and/or lymphadenectomy, is urgently needed. The recent European Association of Urology (EAU) guidelines identify several prognostic factors that should be taken into account when a patient is diagnosed with an upper tract urothelial cancer. An older age at diagnosis is associated with decreased cancer-specific survival. Smoking at the time of diagnosis is associated with increased recurrence rates and increased mortality and smoking cessation improves cancer control. Ureteral and multifocal tumours are associated with poorer outcomes compared with tumours that are confined to the renal pelvis. Other adverse prognostic factors include a delay in resection of greater than 12 weeks after diagnosis, higher American Association of Anaesthesiologists (ASA) grade and raised body mass index (BMI). Age, sex and ethnicity are no longer considered to have prognostic significance.
The most important prognostic factors are tumour stage and grade. Lymphovascular invasion in the primary tumour and lymph node involvement are both independent predictors of survival. Positive tumour margins, extensive tumour necrosis and sessile growth pattern are all associated with worse outcomes.
Risk factors for bladder recurrence following RNU include patient factors (smoking history, chronic renal disease, previous bladder tumour), tumour-specific factors (positive preoperative cytology, ureteral location, multifocal disease, necrosis) and treatment-related factors (laparoscopic approach, extravesical bladder cuff removal, positive surgical margins).
UTUC stage can be difficult to ascertain clinically and by imaging in the preoperative setting. Alternatively, the concept of low- and high-risk tumours can be used to stratify patients into local or radical treatment arms. Low-risk UTUC can be defined by unifocal disease, tumour size less than 2 cm, low-grade cytology, low-grade ureteroscopic biopsy and no invasion on imaging. Multifocal or recurrent urothelial cancer, tumour greater than 2 cm, high-grade cytology or biopsy and variant histology are deemed high-risk UTUC. Pathological staging of UTUC is by the 2017 tumour, node, metastasis (TNM) system ( Table 31.1 ).
| T—Primary Tumour | |
| T0 | No evidence of primary tumour |
| Ta | Non-invasive papillary carcinoma |
| Tis | Carcinoma in situ |
| T1 | Tumour invades subepithelium |
| T2 | Tumour invades muscularis |
| T3 | Tumour invades beyond muscularis into perirenal or periureteric fat or into renal parenchyma |
| T4 | Tumour invades adjacent organs or through the kidney into perinephric fat |
| N—regional lymph nodes | |
| N0 | No regional lymph node spread |
| N1 | Spread to single lymph node <2 cm |
| N2 | Spread to single lymph node >2 cm or multiple lymph nodes |
| M—distant metastasis | |
| M0 | No distant metastasis |
| M1 | Distant metastasis present |
Multiple treatment options are available for the management of patients with UTUC. Open radical nephroureterectomy (ONU) with excision of an ipsilateral bladder cuff is the reference standard surgical treatment for UTUC in patients with a normal contralateral kidney, regardless of the location of tumour in the upper tract. Nephroureterectomy can be performed by an open, laparoscopic (LNU) or hand-assisted laparoscopic technique (HALNU). Resection of the distal ureter and its orifices is performed because it is a part of the urinary tract with especially high risk of recurrence and this resection has been reported to be beneficial.
Laparoscopic nephroureterectomy (LNU) is increasingly used in the management of UTUC.
Several studies have compared perioperative outcomes between ONU and LNU. LNU generally results in less blood loss, shorter hospital stay, decreased analgesic intake, a shorter interval to oral intake and decreased interval to convalescence, with no significant difference in the rate of perioperative complications.
Regarding oncological outcomes of ONU compared with LNU, emerging data support at least equivalent outcomes for LNU and ONU. In a prospective randomised trial, Simone et al. showed improved disease-specific and metastasis-free survival for ONU in patients with stage T3 tumours and high-grade disease. A recent systematic review from the EAU synthesised data from 42 studies and concluded that oncological outcome is likely equivalent in most cases but may be poorer in LNU compared with open RNU when the bladder cuff is excised laparoscopically and in patients with locally advanced (pT3/4) or high-grade disease. This review also notes that randomised studies assessing robotic nephroureterectomy are required. Port-site metastases following laparoscopic surgery for UTUC have been reported, which is another factor to be considered.
Traditionally, endoscopic management of UTUC was reserved for patients with solitary kidneys, bilateral synchronous tumours, baseline renal insufficiency, or comorbid diseases that preclude abdominal surgery. Recently, the use of endoscopic treatment via the retrograde ureteroscopic approach for patients with UTUC, normal renal function and low-grade, low-volume disease has been advocated, so long as patients are able to accept strict surveillance protocols. Opponents of ureteroscopic management argue that any delay in surgery might adversely affect prognosis but there is evidence suggesting that ureteroscopic biopsy/ablation before surgery does not adversely affect the outcomes compared with immediate surgery. Studies with longer follow-up are needed to assess the oncological efficacy of ureteroscopic treatments of UTUC.
Topical immunotherapy, in addition to endoscopic management, can be administered directly through a ureteral catheter or antegradely via a nephrostomy tube. Topical immunotherapy with bacille Calmette–Guérin (BCG) and chemotherapy with mitomycin C have a clear role in patients with bladder urothelial carcinoma, and two randomised controlled trials have demonstrated that intravesical chemotherapy (mitomycin C, pirarubicin) decreases bladder tumour recurrence after RNU for UTUC. This is recommended in the treatment algorithm advocated by the EAU.
The advantages of lymphadenectomy (LND) in the setting of UTUC remain a topic of debate. There is much clinical data supporting lymphadenectomy for bladder cancer but to date evidence with respect to lymphadenectomy for UTUC is mixed. Kondo et al. reported that preoperative imaging accurately identified lymph node-positive patients in less than 50% of cases. This means that some patients might be understaged and undergo suboptimal treatment without lymphadenectomy. A recent systematic review carried out by the EAU examined the best-available evidence (eight retrospective studies and one retrospective/prospective non-randomised study) regarding the impact of lymphadenectomy at the time of RNU on patient outcome in terms of survival and recurrence. This systematic review concluded that template-based and complete LND improves cancer-specific survival in patients with high-stage (>pT2) disease of the renal pelvis and reduces the risk of local recurrence. The impact of LND in the setting of ureteral tumours remains uncertain.
Chemotherapeutic regimens for patients with advanced UTUC are based on those used for bladder urothelial carcinoma, of which cisplatin-based regimens are the most commonly used. In a large multicentre retrospective study on 140 patients, with locally advanced, node-positive disease and metastases, adjuvant chemotherapy with cisplatin-based regimens did not have any effect on overall or cancer-specific survival compared with the patients who did not undergo chemotherapy.
For patients with suspected UTUC found by diagnostic imaging, ureteroscopic biopsy provides histopathological confirmation and information as to the histological grade of the UTUC, which is one of the best predictors of pathological stage, tumour recurrence and overall survival. The diagnostic accuracy of ureteroscopic biopsy for UTUC is high but recent work has shown that ureteroscopic biopsy of upper tract lesions tends to underestimate the tumour grade when compared with the grade determined by histopathological analysis of the nephroureterectomy specimen. If ureteroscopic biopsy underestimates tumour grade, this may go some way in explaining the failure of some endoscopic procedures for management of UTUC. A high grade at biopsy could be used as supporting radical surgery.
Diagnostic imaging plays a significant role in diagnosing and staging of UTUC. Previously, multiple imaging investigations were common with ultrasound, excretory urography and retrograde pyelography. Currently, computed tomography urography (CTU) is the optimal imaging technique for imaging UTUC, with reported sensitivity of 67%–100% and specificity of 93%–99% in the diagnosis of UTUC. Magnetic resonance urography (MRU) is advocated for use in patients for whom CTU is contraindicated or less desirable. Clinical presentation of UTUC is usually with haematuria, and so the imaging techniques used must not only be highly accurate for diagnosing UTUC but also be able detect other diseases responsible for haematuria.
CTU is now the primary imaging method for evaluating the upper urinary tracts. The superiority of CTU over both ultrasound and excretory urography for the detection of UTUC has now been well established. Current Royal College of Radiologists (RCR) guidelines recommend ultrasound as the initial imaging investigation of choice in patients with low risk of urothelial malignancy, with CTU recommended for those at high risk.
American College of Radiology (ACR) guidelines recommend CTU as the first-line imaging investigation for frank haematuria or persistent microscopic haematuria for all patients except those in whom the cause can be attributed to renal tract calculi, renal parenchymal disease, infection, vigorous exercise or menstruation. The American Urological Association recommend that once benign disorders have been excluded clinically, the initial investigation of asymptomatic microscopic haematuria should include CTU. In summary, current guidelines recommend CTU for all patients in whom a benign cause of haematuria (either frank or microscopic) has not been established.
For patients in whom iodinated contrast medium is contraindicated (previous contrast agent reaction, renal impairment etc.), or in whom ionising radiation is best avoided (child, pregnancy, etc.), MR urography is the investigation of choice. Technical considerations for MRU will be discussed below.
Overutilisation of high-technology imaging services such as multidetector CT, MR imaging and positron emission tomography (PET)/CT is defined as performing imaging procedures that are unlikely to improve patient outcome. There is a danger that CTU may be overutilised; it may be used for patients without an appropriate clinical indication. The use of referral guidelines that incorporate appropriateness criteria based on objective clinical evidence would make an important contribution to reducing overutilisation.
There is currently no consensus on the optimum technique for CTU. There are multiple different protocols varying in the number of boluses of intravenous (IV) contrast agent used (single, double or triple bolus), and on the number of CT acquisitions. In practice, the technique can be refined, depending on the clinical indication, technical capabilities of the local CT equipment and local radiologist/ urologist preference.
The single-bolus protocol is optimised for patients with haematuria at high risk of UTUC, renal masses and stones. This protocol consists of a single contrast injection and three series of image acquisition: an unenhanced series optimised for detecting stones; a nephrographic series optimised for detecting renal masses; and an excretory series optimised for detecting UTUC.
The double-bolus (or ‘split-bolus’) protocol involves a slightly lower radiation dose than the single-bolus protocol by combining the nephrographic and excretory phases. It uses less contrast medium than the single-bolus protocol for demonstration of the nephrographic and excretory phases and so may not produce such an intense nephrogram for demonstration of renal cell carcinoma or excretory series for diagnosing UTUC. If the unenhanced series is excluded, and the enhanced nephrographic and excretory phases are acquired in one series, a low-dose protocol is achieved which is commonly used for the follow-up of patients with known bladder cancer or UTUC. A recent trial of double-bolus CTU with 192 patients randomised to either 30% volume at first bolus and 70% at second bolus, 70% and 30%, or 50% and 50% and different delay times of 8, 10, 12 or 14 minutes for the excretory phase image acquisition demonstrated the optimal renal and ureteric enhancement occurred when 70% of the contrast agent was administered in the first bolus and images were taken at 8 minutes.
The triple-bolus protocol is used for the assessment of living related kidney donors and patients undergoing percutaneous nephrolithotomy. Although use of a triple-bolus protocol for investigating haematuria has been described, it is currently not recommended for routine use because the volume of contrast agent available for the excretory phase is reduced by bolus splitting, which is likely to compromise diagnostic accuracy.
Many studies have evaluated techniques aimed at promoting complete opacification of the urinary tract during CTU, to optimise the contrast between urine and urothelial disease. Currently there is no consensus regarding the optimum technique. A number of manoeuvres have been suggested that are often interrelated in a complex fashion so that adjustment of one will impact on the others. Such manoeuvres include oral administration of water, intravenous furosemide or saline to encourage urine flow, abdominal compression, varying the timing of the image acquisition, acquiring a second series of images in the excretory phase, increasing the volume of contrast injected and encouraging patient movement (walking or log rolling on the CT table) to promote mixing of contrast agent and urine.
In practice, most patients do not require these manoeuvres to achieve optimum opacification of the upper tracts. At the authors’ institution, the practice is to perform a split-bolus protocol, following a period of oral hydration prior to the scan. The combined nephrographic and excretory phase is performed with the patient in the prone position to optimise contrast filling of the ureters, and the patient is encouraged to sit and/or stand between the first and second bolus. The images are checked prior to the patient leaving the scanner, as opacification can be significantly delayed in patients with obstructed urinary tracts. Further, delayed acquisitions may be required in such cases.
Dual-energy CT (DECT) has received much attention recently and applications in various settings are emerging. The main potential benefit of using DECT for CTU is the ability to obtain virtual unenhanced CT to assess for calculi in the setting of haematuria, without the need to perform a separate unenhanced CT, thereby potentially decreasing patient radiation dose. It has been suggested that the virtual unenhanced CT should be generated from the nephrographic phase scan to reduce false-negative and false positive studies that may be caused by the presence of iodinated contrast material in the renal excretory system during the urographic phase. Another interesting concept is a single-phase dual-energy double-bolus technique which condenses a traditionally three-phase CTU into a single-phase acquisition. The unenhanced images are generated virtually from the dual-energy attenuation information and the spilt bolus allows simultaneous renal parenchymal and excretory system enhancement. Recent studies have demonstrated the ability of this technique to detect stones and UTUC with a high degree of accuracy.
Continuous audit of opacification is required to maintain the standard of CTU from day to day. All users of CTU should employ comparable scoring systems. In one such scoring system, the urinary tract is divided into four anatomical components (collecting system and renal pelvis; abdominal ureter; pelvic ureter; and bladder), and each is given a score of 0 (no opacification), 1 (partial opacification) or 2 (complete opacification). The bladder is scored slightly differently, with 0 representing no opacification, 1 is used for regions of interest within the bladder at less than 100 HU and 2 corresponds to all regions of interest in the bladder over 100 HU. Only by using a similar scoring system across different centres will it be possible to accurately compare different CTU techniques.
There has been much recent interest in radiation dose optimisation strategies. There are many ways to reduce radiation exposure from CT while maintaining diagnostic accuracy. The most effective way to reduce radiation exposure is to not perform the examination. Scrutiny of examination appropriateness is essential. Once the decision has been made to perform CT, there are many available strategies to reduce radiation exposure; these include the use of size-dependent protocols, automated tube current modulation, reduction of the number of passes, reduction in duplicate coverage, reduction of mAs and kVp where possible, optimisation of IV contrast medium administration and possible use of external shielding. Radiation exposure can also be reduced with better post-processing methods to decrease noise, smoother kernels for reconstruction, reconstruction at larger slice thickness and iterative reconstruction.
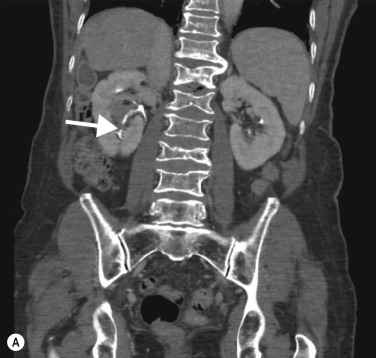
UTUC arising in the kidney most commonly presents as an infiltrating mass arising centrally in the collecting system of the kidney, with preservation of the renal contour ( Figs 31.1 and 31.2 ). There are a number of lesions that can mimic UTUC and these are listed in Table 31.2 .
| 1 | Clot |
| 2 | Debris |
| 3 | Kink of the ureter (accentuated by respiration) |
| 4 | Fibroepithelial polyp |
| 5 | Injury to the ureter following passage of a stone, stent placement or ureteroscopy |
| 6 | Ureteritis cystica |
| 7 | Flow artefact following furosemide administration or layering effects |
| 8 | Unusual-looking papilla, a normal variant |
| 9 | Nephrogenic adenoma |
| 10 | Amyloid |
| 11 | Renal cell carcinoma |
| 12 | Lymphoma |
| 13 | Vascular impression |
| 14 | Inflammation, fibrosis |
Ultrasound is less sensitive than CT for detecting UTUC in the kidney but, when visible, it presents as a mass in the region of the renal sinus which may be hypoechoic, isoechoic or hyperechoic. It usually causes hydronephrosis or calyceal dilatation. Most UTUCs are hypovascular and therefore lack of detectable vascularity on Doppler interrogation of the mass does not exclude UTUC. Ureteric transitional cell carcinoma (TCC) is more difficult to diagnose with ultrasound given poor visualisation of the ureters in most cases. If present, it is usually seen as focal ureteric thickening with proximal hydroureter and hydronephrosis.
Intravenous pyelography has been superseded by CTU. Retrograde pyelography may be performed during cystoscopy and ureteroscopy. Signs on retrograde pyelography include irregular, serrated filling defect in one or more calyces, ‘stipple sign’ or contrast filling the interstices of the mass, and ‘phantom calyx’ or unopacified calyx due to obstruction of the calyceal infundibulum by tumour. In the ureter, UTUC usually manifests as a focal stricture, which may be smooth or irregular, eccentric or circumferential. The ‘champagne glass sign’ or ‘goblet sign’ refers to luminal dilatation distal to a tumour caused by slow-growing tumour, expanding the lumen of the ureter.
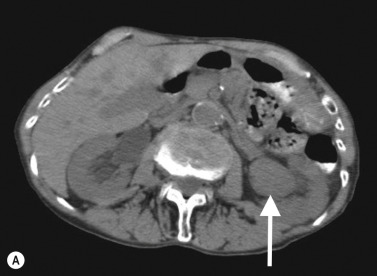
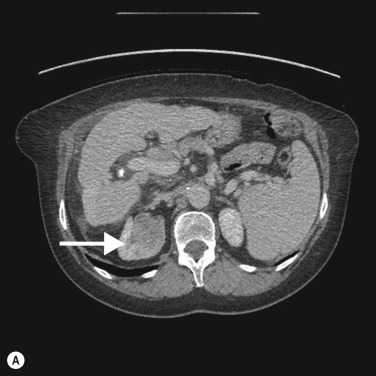
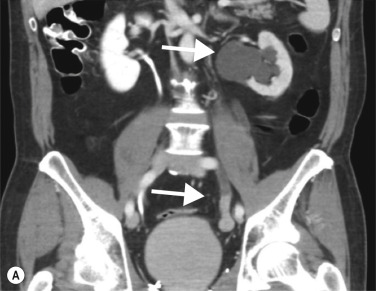
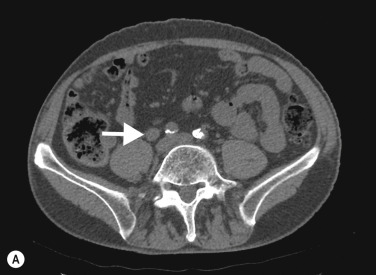
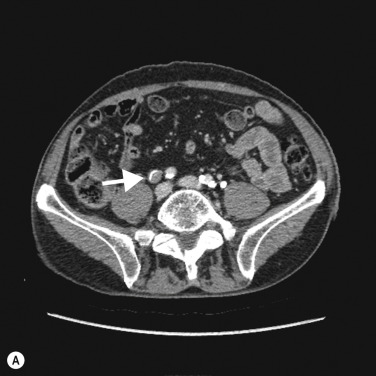
On CTU, UTUC in the kidney usually presents as a hypovascular, infiltrative mass in the pelvicalyceal region ( Fig. 31.3 ), with loss of normal calyceal contrast opacification in the excretory phase. Invasion of the renal sinus fat and parenchyma may be seen, but the renal contour is usually preserved. In the ureter there is usually eccentric or circumferential wall thickening with hydroureter and hydronephrosis ( Figs 31.4–31.6 ). Obstruction usually occurs early due to the small luminal diameter of the ureter. Most UTUC occurs in the distal third of the ureters. Care should be taken to look for multiple lesions, and contralateral tumours are found in 2%–5% of cases. Also, assessment of the regional lymph node basin for lymphadenopathy is necessary and evaluation for other metastatic sites.
MRU can be used as an alternative to CTU in certain situations, such as when iodinated contrast medium is contraindicated due to previous reactions or renal impairment, pregnancy and in the paediatric population. While its use in characterisation of renal parenchymal lesions has been well studied, there is less evidence for the use of MRU in the detection and characterisation of urothelial lesions. Currently, MRU is considered to be inferior to CTU for this purpose, and it is poorly sensitive for calculous disease. However, it is useful for detecting the presence and level of urinary tract obstruction and can be helpful in determining the cause.
A combination of T 2 weighted sequences and gadolinium-enhanced sequences should be used for optimal detection of UTUC. Caution is advised regarding the use of IV gadolinium in patients with glomerular filtration rate (GFR) less than 30 mL/min due to the theoretical risk of nephrogenic systemic fibrosis. The sensitivity of gadolinium-enhanced MRU for tumours less than 2 cm is 75%. MRU has certain advantages such as high contrast resolution and lack of ionising radiation. Limitations of MRU include its cost, length of time required to perform and presence of imaging artefacts. Emerging techniques such as diffusion-weighted imaging (DWI) have shown promise in the detection and follow-up of UTUC.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here