Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Tuberculosis remains one of the most important diseases in the world. Although the DNA of the microorganism responsible for tuberculosis has been recovered from ancient Egyptian mummies, the profound increase in the incidence of the disease occurred in conjunction with the Industrial Revolution. The overcrowding, poor living conditions, and lack of effective therapy created perfect conditions for the effective transmission of the airborne tuberculous bacillus. Currently, the life expectancy of infected individuals, as well as in the transmission of disease, has been substantially impacted in resource-rich countries wherein health care and the public health systems are in force and effective antimycobacterial therapeutics are available. The opposite, however, is the case in resource-poor countries. In addition, the HIV/AIDS epidemic, like the Industrial Revolution of the past, has created ideal conditions for the transmission of tuberculosis. Individuals with HIV, who do not have the benefit of highly active antiretroviral therapy, cannot mount a sufficient T-cell based granulomatous response to contain the infection. This results in progressive disease with a large load of mycobacteria, which increases the likelihood of airborne transmission. Unfortunately, these individuals often live in resource-limited countries, where the burden of HIV is high and there is limited access to health care and adequate therapy. Therefore, although tuberculosis still occurs in resource-rich countries, it is a significant problem in resource-limited countries, particularly in areas with a high prevalence of HIV. The world is in the midst of a renewed epidemic of tuberculosis according to the World Health Organization (WHO). Fortunately, significant efforts are being made worldwide, which are having measurable impacts. The favorable trends are that new cases of tuberculosis fell at a rate of 2% between 2010 and 2011, and the mortality rate has decreased 41% since 1990. Although these trends are encouraging, a review of the burden of the disease is staggering; an estimated 8.7 million new cases of tuberculosis were reported in 2011, with 13% HIV coinfection, and an overall 1.4 million deaths due to disease. The executive summary for the annual Global Tuberculosis Report contains the most updated information on this project.
There are several agents in the Mycobacterium tuberculosis complex, including M. tuberculosis, M. bovis, M. microti, M. africanum, M. pinnipedii, and M. caprae. Of these, M. tuberculosis causes the majority of disease in humans. The other members of the M. tuberculosis complex are predominantly animal pathogens, but they may also occasionally cause human disease.
The molecular biology of M. tuberculosis has been studied extensively. It is one of the microorganisms for which the whole genome has been sequenced. This extensive knowledge of the genetic composition of M. tuberculosis has been important for the development of a number of assays for the rapid detection and characterization of the M. tuberculosis complex. These include several nucleic acid amplification assays, DNA sequencing tests, and molecular typing procedures. These assays, some of which have been approved for use by the U.S. Food and Drug Administration (FDA), afford the detection of M. tuberculosis in smear-positive (and, in one instance, smear-negative) respiratory specimens. DNA sequencing applications allow the interrogation of genetic targets containing taxonomically important information, which in turn allows for the identification of mycobacteria and, depending on the target, the differentiation of members of the M. tuberculosis complex from the nontuberculous mycobacteria. The most commonly used target, which resides within the 16S ribosomal RNA gene, is conserved for the entire M. tuberculosis complex. Therefore, in the rare instances when it is necessary for clinical reasons to differentiate M. bovis from M. tuberculosis, then either an alternate genetic target must be sequenced or differentiation would need to be achieved using traditional biochemical testing.
The genetic targets and the most common mutations associated with resistance to the most frequently used antimycobacterial agents have also been identified. These may be most thoroughly assessed through DNA sequencing, but reverse hybridization assays and assays that employ molecular beacons have also been used to detect genetic determinants of resistance. In addition to identification and detection of resistance determinants, molecular methods have also been used to strain-type organisms, which has proven critical for molecularly mapping the spread of isolates, the most important of which are multidrug-resistant isolates.
The clinical signs and symptoms of tuberculosis vary greatly depending on the stage of disease, the anatomic site affected, and the immunologic status of the host. The most common route of transmission is via inhalation, and thus the lungs are the typical site of primary infection. Individuals may be unaware of the primary infection, particularly when it is adequately controlled by the immunologic response. In contrast, the immunologically compromised host, and occasionally others for unknown reasons, can develop locally progressive disease or disseminated (i.e., miliary) tuberculosis following the primary infection.
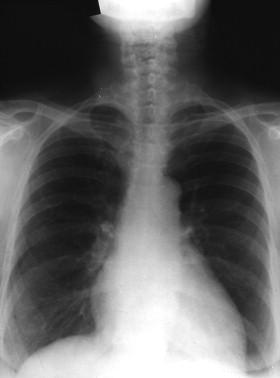
In most individuals, primary infection results in transient mycobacteremia followed by the development of a successful immunologic response and containment or destruction of the infecting bacteria within necrotizing or caseous granulomas. Primary infection is usually in the midregion of the lung and may be evidenced radiologically with calcification in the associated hilar lymph nodes ( Figure 20-1 ) (i.e., a Ghon complex). Patients who have successfully contained the primary infection will be positive when evaluated by either a skin test or a blood test (i.e., a interferon-gamma release assay) for the presence of latent tuberculosis.
Reactivation disease may later occur in individuals of advancing age or those with other immunocompromising conditions. Infection with HIV, as well as a number of immunosuppressive medications, has also been associated with reactivation disease. Further information regarding HIV-associated mycobacterial diseases is covered elsewhere in this text. The most common site for reactivation disease is the apical aspect of the lungs. The reason for reactivation in this location is a matter of substantial debate. The traditional view is that the apex is the most highly aerated portion of the lung and is therefore the preferential site for replication of an obligate aerobe. Dissemination and subsequent reactivation may also occur in extrapulmonary sites, such as the ends of long bones. This is most likely a result of hematogenous spread during the mycobacteremic stage of primary infection.
The signs and symptoms of disease include nonspecific constitutional symptoms such as weight loss, anorexia, fatigue, fever, night sweats, and chills. Patients with progressive disease will develop a productive cough with mucopurulent or bloody sputa containing acid-fast bacilli. Physical examination may disclose decreased fremitus and dullness when there is pleural involvement and a tuberculous empyema. Whispered pectoriloquy may be appreciated with large lesions and over areas of consolidation.
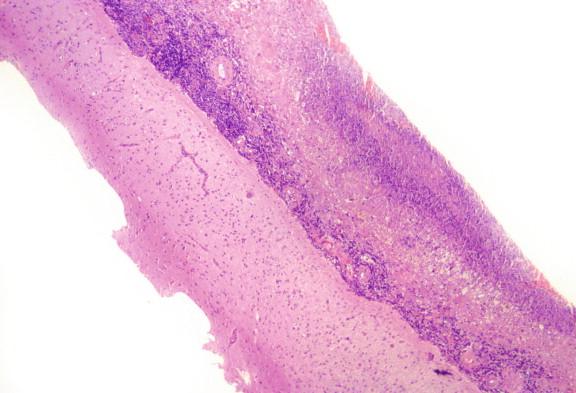
Patients who develop reactivation in the spine or long bones may present with pain as the lesion expands ( Figure 20-2 ). Patients with tuberculous meningitis usually have the most pronounced involvement in the basilar meninges ( Figure 20-3 ). Nonspecific findings such as intermittent headache and low-grade fever may herald the event. With progression, the headache becomes protracted and there may be cranial nerve involvement and other focal neurologic signs, as well as vomiting and confusion.
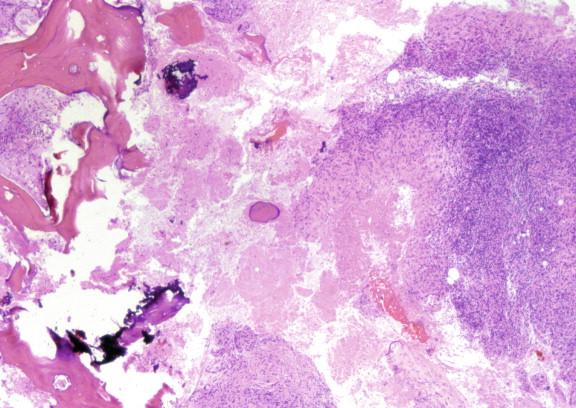
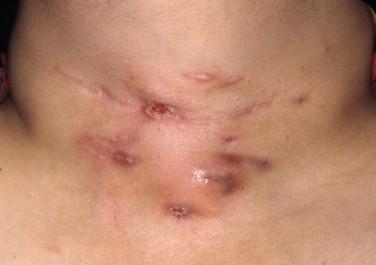
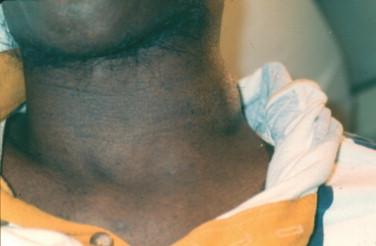
Lymphadenopathy is present in patients with progressive and disseminated disease ( Figure 20-4 ). Draining sinus tracts may develop from the affected lymph node chain ( Figure 20-5 ). Many other areas may become involved in the course of disease, such as the peritoneum and the pericardium, and the signs and symptoms of infection in these sites are not specific to tuberculosis and similar to other infections in these locations. Biopsies/aspirates and cultures with corresponding acid-fast staining and histo- or cytopathology are required to identify the etiologic agent of infection.

The diagnosis of tuberculosis often begins with the elucidation of an appropriate clinical history and an assessment of the clinical findings. These, in conjunction with distinctive radiologic findings, are usually enough to identify a patient who needs to be placed in respiratory isolation until definitive laboratory findings become available. The additional tests to be performed include an immunologic assessment of the patient to assess previous exposure, acid - fast staining of respiratory specimens, culture, and in some instances molecular diagnostic studies.
The tuberculin skin test is the traditional method for the determination of an existing immunologic response to M. tuberculosis and closely related mycobacteria. This is largely being replaced by cellular-based assays. These assays detect gamma-interferon that is produced by the patient’s T cells, if there has been previous exposure to the antigens of M. tuberculosis. The advantages of this assay include fewer false positives for patients who have been given the Bacille de Calmette et Guérin (BCG) vaccine (extensively used worldwide) and far less cross-reactivity (i.e., fewer false positives) in patients who have had infections caused by a nontuberculous mycobacterium. Another practical advantage is that only a single blood specimen is required, which eliminates the need for a repeat visit. In comparison, the tuberculin skin test requires a second visit to assess the patient’s response to the injection. The disadvantage of the gamma-interferon assays is a higher direct cost, as the test components are more expensive. This, however, may be less of a disadvantage overall when the cost to the system is considered, because the individuals being tested must return for a second visit, potentially necessitating a missed day from work, and a nurse or other health care professional is required to evaluate the injection site. Regardless, it is important to note that both the tuberculin skin test and gamma-interferon assays do not differentiate latent from active disease and may be falsely negative in the immunocompromised patients.
The acid - fast staining of sputa and other respiratory specimens is critical, as it may be the first definitive evidence of mycobacteria available in respiratory specimens. The acid-fast stain result also has implications for communicability, since patients with tuberculosis and acid-fast bacillus-positive respiratory specimens have a greater potential to transmit the organisms than acid-fast bacillus (AFB) smear–negative patients. Although smear-negative patients have a decreased transmission rate, it is not zero. Laboratories should be using fluorochrome stains for the screening of respiratory specimens, as these are more sensitive and improve efficiencies in the laboratory. Positive results may be confirmed by a traditional Ziehl-Neelsen stain, which is the stain commonly used on histologic and cytologic specimens suspected of containing mycobacteria.
The definitive diagnosis of tuberculosis has traditionally required the isolation of M. tuberculosis from a clinical specimen. This is usually a respiratory specimen, but could be tissue, urine, or a fluid aspirate in patients with extrapulmonary disease. Specimens from nonsterile sources are decontaminated prior to media inoculation to prevent overgrowth from normal microbiota. A combination of broth and solid media is the standard for culture. The broth medium facilitates quicker recover and, therefore, more rapid identification, whereas the solid medium allows for an assessment of colony morphology and the detection of different mycobacterial populations. Once a culture isolate is obtained, it has traditionally been identified using biochemical reactions and biophysical profiling. This, however, has largely been replaced by molecular methods, such as DNA sequencing and, more recently, mass spectrometry.
It is important to perform susceptibility testing on culture isolates. Multidrug-resistant tuberculosis (MDR TB) is present in many parts of the world and is spreading. These strains can spread to nonendemic regions as readily as an infected individual can board an airplane. The detection of MDR TB is particularly important for patient care, as several drugs used simultaneously are needed to successfully treat these patients. Once MDR TB is detected and the resistance profile known, then the treatment regimen may be tailored. The detection of MDR TB is also important for public health practitioners, because they will track, and in some instances prophylactically treat, contacts. Traditionally, antimicrobial susceptibility testing is performed using culture-based methods in which the organism is exposed to different levels of antibiotics and observed for growth. However, the molecular determinants of resistance to the drugs most commonly used to treat patients with tuberculosis have been determined. Assays for these determinants are not commonly used in routine practice but are available to specialized reference laboratories. The use of these as well as molecular methods for the detection of tuberculosis will likely increase as these methods become more established.
The radiologic features, as with the clinical presentation, depend on the stage of the disease. The immunocompetent individual may not seek medical attention during the acute infection. However, the calcified focus of initial infection and calcified foci within the hilar lymph nodes (see Figure 20-1 ) (i.e., the Ghon complex) will serve as subsequent evidence of initial infection that may be seen on chest roentgenogram.
Patients who develop locally progressive disease may have radiologic findings of a lobar pneumonia. Those who develop miliary tuberculosis will develop multiple, small nodular foci characteristic of this disease. The size of these nodules is similar to that of a millet seed, from which the name of the condition is derived. Reactivation of tuberculosis occurs most commonly in the upper lobes of the lungs and is characterized by a nodular enhancement with eventual cavity formation. Although these findings may be demonstrated on a chest roentgenogram, they are better demonstrated with CT or MRI scans.
Tuberculous meningitis, which is caused by dissemination of the tuberculous bacillus to this location, has radiologic features similar to fungal meningitis caused by the dimorphic fungal pathogens that are endemic to North America. These infections produce predominantly a basilar meningeal thickening and enhancement that are appreciated on high-resolution scans.
Radiologic tools are also important in the assessment of reactivation disease that occurs in the bones. The lesions of Pott disease (tuberculosis involving the spine) are readily identified by radiologic methods, but culture is necessary to exclude other causes of vertebral osteomyelitis. Similarly, the lesions that occur in long bones may be mistaken for a neoplastic process, and therefore culture and histopathologic examination is an important component of the workup.
The gross findings are dependent on the stage of disease and the organ involved. The gross findings of lymph nodes with a Ghon complex are similar to other causes of nodal calcification, such as remote histoplasmosis. There with be a gritty feel when the scalpel sections
Tuberculosis is a systemic disease that is caused by Mycobacterium tuberculosis. Pulmonary manifestations are most common, but disseminated disease, meningitis, and skeletal disease are well known.
There is a worldwide epidemic of tuberculosis, which is exacerbated by the HIV epidemic.
There were an estimated 8.7 million new cases of tuberculosis in 2011, 13% of which were in hosts who were coinfected with HIV, with overall deaths from disease totaling 1.4 million.
The World Health Organization (WHO) and others have implemented treatment and control strategies that are producing favorable trends: new cases of tuberculosis fell at a rate of 2% between 2010 and 2011, and the mortality rate has decreased 41% since 1990.
Tuberculosis is spread primarily through a respiratory route. Transmission is facilitated in resource-poor areas with overcrowding, poor health care facilities, and public health systems, and among individuals with immunocompromising conditions. Advanced HIV infection facilitates the spread of tuberculosis, because these individuals cannot produce the granulomatous response necessary to contain the infection, which results in multibacillary disease that potentiates transmission.
Radiologic studies are important for demonstrating evidence of remote infection (i.e., a Ghon complex), primary progressive disease, miliary spread, and reactivation disease with cavitation. Similar studies are important for characterizing the basilar meningitis and skeletal disease that can be caused by M. tuberculosis.
Necrotizing and non-necrotizing granulomas characterize the inflammatory response to M. tuberculosis.
Mycobacteria stain red (i.e., are positively stained) with the Ziehl-Neelsen stain and also stain with the Fite stain, are gram positive, and are detected with the GMS stain.
Molecular tools may be used to differentiate M. tuberculosis from the nontuberculous mycobacteria.
The prognosis is favorable when there is limited disease (e.g., latent tuberculosis), the patient is otherwise healthy, there is access to appropriate medical care, and the infecting strain is susceptible to antituberculous drugs. The prognosis is worsened with any of the following factors: there is extensive or disseminated disease, the patient is immunocompromised or has other significant comorbid conditions, there is inadequate access to health care, or the infecting strain is resistant to antituberculous drugs.
through the node and when the cut surface is scraped with the edge of the scalpel blade.
The portions of the lung affected by locally progressive primary disease will be firm, more so than the consolidated areas of a bacterial pneumonia. The cut section will demonstrate white-to-tan nodules that coalesce. The nodules of miliary tuberculosis in the lung and other organs are obvious and distributed throughout the parenchyma. These nodules are firm when cut and do not demonstrate the gritty consistency of calcification, unlike similarly sized nodules in the liver and spleen of patients with remote histoplasmosis.
The established granulomas may become quite large, and when present within a lymph node chain, produce lymphadenopathy (see Figure 20-4 ). Similar lesions are present in the lungs prior to cavitation. The cut surface demonstrates white-to-tan, creamy-to-friable material ( Figure 20-6 ). The repeated rounds of inflammatory response may be responsible for the “onion skin” or concentric rings that may be seen ( Figure 20-6 ).
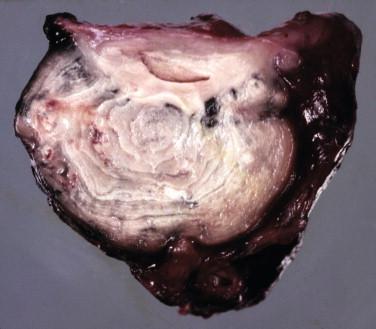
The lesion produced by reactivated disease in the upper lobes of the lungs is most impressive. It usually consists of a large cavity with a firm border. Upon sectioning, the cavitary nature of the lesion will become apparent, with copious amounts of necrotic debris in the center of the cavity. The walls of the lesions are firm, but inner aspects are friable at the junction with the cavity and may appear ragged. Old tuberculosis cavities remain following successful treatment and may contain a saprophytic fungus ball.
The lesions associated with disseminated tuberculosis consist of large granulomas with central necrosis in many organs. These have a white cut surface on gross examination with central white, friable material. The surrounding parenchyma may be significantly compressed by the nodules.
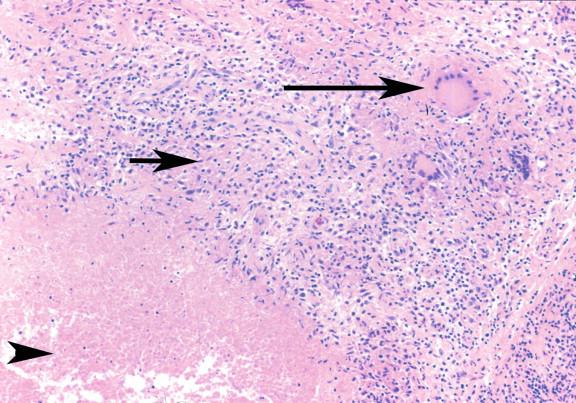
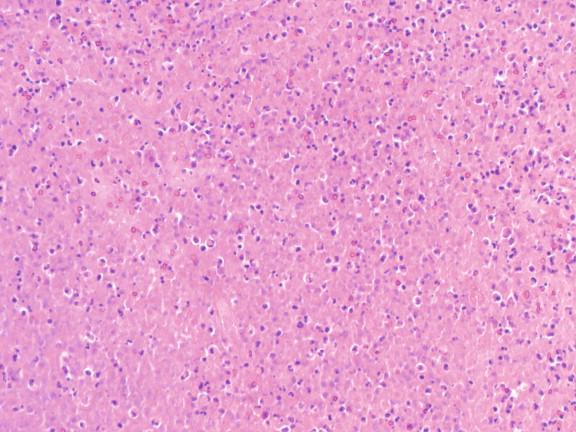
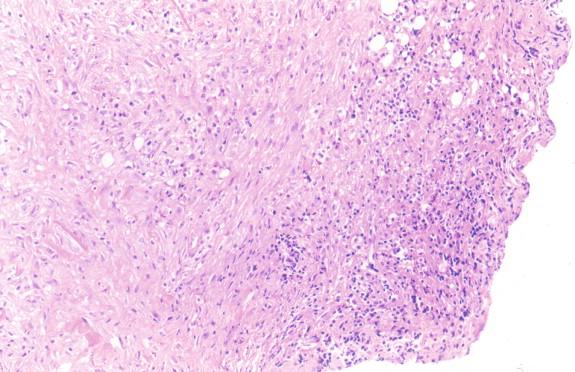
Tuberculosis is characterized by necrotizing or caseating granulomas ( Figure 20-7 ). Non-necrotizing granulomas may also be present ( Figure 20-8 ). The cellular components of the granulomas consist of activated histiocytes, multinucleate giant cells, and infiltrating lymphocytes. These will surround areas of caseous necrosis in the necrotizing granulomas. In areas of rapid progression, there may be simply necrosis, neutrophils, and neutrophilic debris ( Figure 20-9 ). Immunocompromised hosts who lack the ability to produce granulomas may form a fibrous/inflammatory pseudotumor ( Figures 20-10 through 20-12 ).

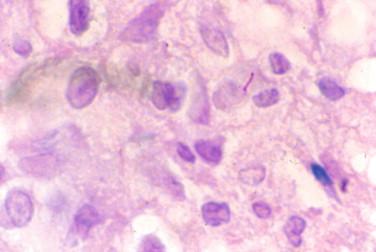
Although not always discernible, the tuberculous bacillus may be demonstrated using an acid-fast stain, such as Ziehl-Neelsen ( Figure 20-13 ). Although these may be seen at 400 × magnification, oil immersion microscopy at 500 × or 1000 × magnification provides increased sensitivity. This higher resolution is warranted when the inflammatory response is as described earlier, particularly if the fungal stain is negative and the etiology of disease remains uncertain. Mycobacterium tuberculosis will appear as a red bacillus in the Ziehl-Neelsen stain and may have a beaded appearance. Mycobacteria may also be detected in the Gomori methenamine silver (GMS) stain commonly used for fungi. When bacillary forms are present in the GMS stain and the aforementioned histopathologic findings are present, then the Ziehl-Neelsen stain should be extensively studied. Mycobacteria may sometimes be detected with the Gram stain, although this is not the stain of choice for these microorganisms. Mycobacteria are gram positive and disclose the beaded appearance mentioned previously.
Depend on the stage of disease, organ involved, and immunologic status of the host.
Lymph nodes associated with a Ghon complex are calcified, and there is a gritty feel when the scalpel sections through the node.
The portions of the lung affected by locally progressive primary disease will be firm, even more so than the consolidated areas of a bacterial pneumonia. The cut section will demonstrate white-to-tan nodules that coalesce.
The nodules of miliary tuberculosis in the lung and other organs are small nodules reminiscent of “millet seeds” distributed throughout the parenchyma.
Large, noncavitary nodes may be seen that are white to tan and filled with creamy to friable material. The repeated rounds of inflammation may produce an “onion skin” appearance of concentric rings.
Reactivated disease in the upper lobes of the lungs is most impressive and usually consists of a large cavity with a firm border. There are copious amounts of necrotic debris in the center of the cavity. The walls of the lesions are firm, but inner aspects are friable at the junction with the cavity and may appear ragged. Old tuberculosis cavities remain following successful treatment and may contain a saprophytic fungus ball.
The lesions associated with disseminated tuberculosis may consist of large granulomas with central necrosis in many organs. These have a white cut surface on gross examination with central white, friable material. The nodules may significantly compress the surrounding parenchyma.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here