Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Radiofrequency ablation (RFA) has become the standard of care for catheter ablation of cardiac arrhythmias. Other ablative technologies, including cryoablation and lasers, are variably used depending on the anatomic features of the tissue being targeted. Common to all is the requirement of contact between an intracardiac catheter and a destructive energy of physical nature. The historic evolution of catheter ablation, however, commenced with the concept of delivering a cytotoxic agent through the arterial vasculature supplying the targeted myocardium, which was pioneered in Zipes’ laboratory, , using a variety of alcohols. The approach was largely superseded by RFA for most clinical uses, given its consistency, safety, and efficacy.
Currently, the most common type of alcohol used for this purpose is ethanol, a highly cytotoxic agent with multiple other uses in humans. Intravascular ethanol injections can target arrhythmogenic foci that are deep seated in the myocardium and are not amenable to catheter ablation from either the endocardium or the epicardium using contact-based physical forms of ablative energy.
Ethanol (CH 3 CH 2 OH) is a short-chain alcohol, water-soluble compound that rapidly crosses the cell membranes. When cells are exposed to high concentrations, ethanol solubilizes the cell membranes and alters the tertiary protein structures, leading to immediate cell destruction. , Most of the fluid membranes, including those that are low in cholesterol, are easily solubilized by ethanol. Ethanol interferes with the packing of molecules in the phospholipid bilayer of the cell membrane, thus increasing membrane fluidity. The nonoxidative metabolism of alcohol with the formation of phosphatidylethanol and fatty acid alcohol ester disrupt intracellular signal transduction. Additionally, degradation of proteins and mitochondrial injury are other mechanisms associated with the cytotoxicity of short-chain alcohols. Protein degradation occurs via the production of reactive oxygen species such as superoxide anions, lipid peroxyl radicals, and alcohol radicals. A variety of mechanisms may account for the biochemical alterations of mitochondria induced by alcohols, which include decreases in mitochondrial adenosine triphosphatase (ATPase) activity.
The cytotoxicity of ethanol at intravascular concentrations can be attributed to various mechanisms. Ethanol, when injected at slower rates (at which it is assumed to undergo some degree of dilution) produces denaturation of erythrocytes; activation of the coagulation cascade and thrombosis; aberrations in phospholipid and fatty acid metabolism; changes in the cellular redox state; disruption of energy state; and an increase in reactive metabolites. The optimal ethanol concentration and rate of infusion for arrhythmia ablation are controversial and have yet to be clarified. Additionally, vascular damage with sclerosis of the injected vessel follows routinely after infusion and therefore with intraarterial infusions, tissue ischemia and infarction of the injected territory are expected to play a role in ethanol’s therapeutic effect.
Ethanol was first reported in the anesthesiology literature for the purposes of pain control, using a transsphenoidal injection approach for ethanol-induced hypophysectomy. Although the exact mechanisms of neuroadenolysis of the pituitary gland for cancer pain control remained unclear, autopsy studies revealed necrosis of the gland. Ethanol-induced neurolysis remains a valuable therapeutic strategy for intractable cancer pain, and it is under this indication that it is labeled in the manufacturer’s package insert. Tissue ablation effects were also confirmed in renal tissue, when absolute ethanol injected into the renal artery at a rate approximating renal arterial blood flow was distributed rapidly to the renal parenchyma, causing cell necrosis. ,
These effects led to the therapeutic use of ethanol to treat isolated metastatic lesions of the kidney, liver, and other organs by selective intraarterial ethanol injection. ,
Potential complications of ethyl alcohol injection include temporary or irreversible organ failure and pulmonary or systemic embolization of nontarget tissues with high concentrations of ethyl alcohol. Although complications are rare for noncardiac use, ventricular fibrillation (VF) developed after renal artery embolization with absolute ethyl alcohol in 3 of 11 dogs, presumably because of spillover into the circulation.
The most common myocardial ablative use of ethanol remains transcoronary interventricular septal ablation in hypertrophic obstructive cardiomyopathy (HCMP). The procedure requires 95% to 96% ethanol to be injected into a septal branch, which supplies blood flow to the hypertrophied myocardium in the left ventricular (LV) outflow tract (LVOT). Ethanol (0.5–3 mL) is infused depending on a septal artery width or interventricular septum (IVS) thickness in the zone of the mitral-septal contact.
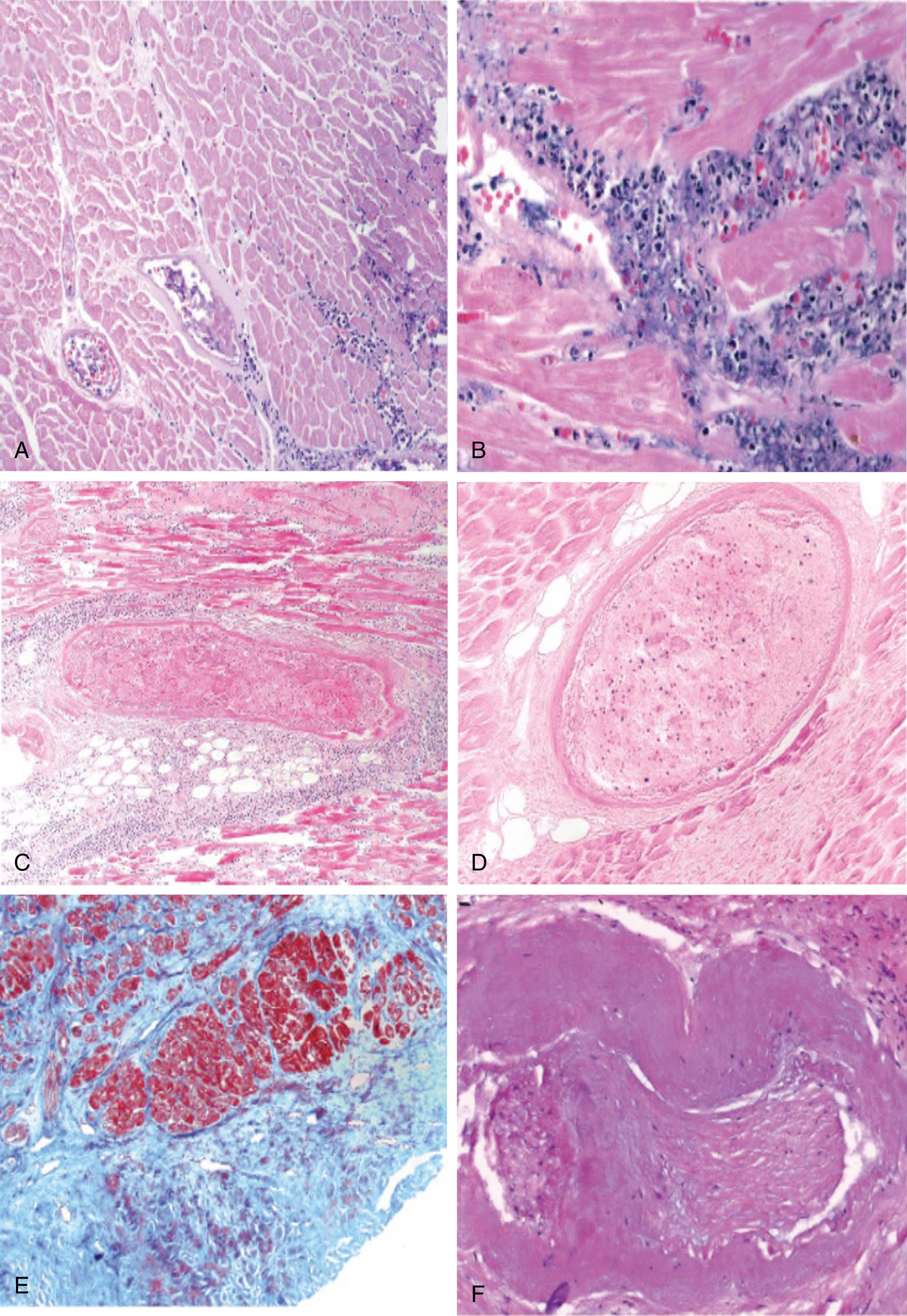
Relatively few human pathologic data are available on the effects of alcohol on the myocardium when given intracoronary or directly. In dogs, intracoronary injection of 100% ethanol caused myocardial necrosis involving greater than half the thickness of the LV wall. The resultant necrosis was focal or patchy and nonconfluent. Inoue et al. speculated that ethanol may have induced thrombus formation even in the absence of intimal injury. Injection of lower concentrations of ethanol caused similar patterns of necrosis without intraluminal thrombus. Injection of 100% ethanol into the canine coronary venous circulation caused myocardial necrosis. Transventricular injection of 60% ethanol in dogs produced coagulative necrosis and hemorrhage, with a narrow border zone consisting of myocytes with neutrophilic infiltration. After 1 month, there was dense fibrosis with mild to moderate inflammation. In the context of septal ablation for HCMP, Arrazaghi et al. observed replacement fibrosis with islands of myocytes in a myectomy specimen obtained 2.5 years after the procedure. Baggish et al. observed, early after alcohol septal ablation, coagulative necrosis of both the myocardium and the arteries, with marginating neutrophils at the periphery of the infarct. In distinction to the pathology of spontaneous myocardial infarction (MI), however, the septal branches showed endothelial and smooth-muscle necrosis, and luminal occlusion with necrotic debris and “fixed” blood. Fixation of blood and direct toxicity to the vessel wall may be unique components of an ethanol-induced tissue injury ( Fig. 132.1 ). The toxicity of ethanol to the vasculature, including the microvasculature, may preclude myocardial protection by collateral blood flow, ensuring the efficacy of ethanol in causing myocardial necrosis. Late after ablation, necrotic myocardium is replaced by a permanent scar, and granulation tissue is interspersed with islands of atrophic myocardium.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here