Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
During the fifth week of gestation, the urinary collecting system begins development as an outpouching from the mesonephric duct known as the ureteral bud. Contact between the bulbous portion of the ureteral bud and the metanephric blastema induces this portion of the bud to branch and develop into the renal pelvis, calyces, and collecting tubules. Meanwhile, the stalk of the ureteral bud develops into the ureter. The segment of distal mesonephric duct located between the ureteral bud and developing bladder, known as the common nephric duct, undergoes apoptosis, bringing the distal ureter into contact with the developing bladder. The ureteral orifice then migrates cranially and laterally as the bladder tissue surrounding the ureteral insertion expands and differentiates.
Ultrasound is the first-line imaging technique in pediatric uroradiology. Unfortunately, the ureter can generally only be visualized when it is sufficiently filled with urine, and even then only the proximal and distal aspects are typically seen. Using the kidney as an acoustic window from a posterior or lateral approach, the ureteropelvic junction (UPJ) can usually be identified in well-hydrated patients with a tapering of the pelvis toward the proximal ureter. To visualize the ureterovesical junction (UVJ) and distal ureter, sufficient fluid distension of the urinary bladder is mandatory so that it can serve as a window to the retrovesical space. Once the bladder is sufficiently filled, the normal ureter can be followed superiorly for several centimeters. Further visualization up to the iliac vessels or higher is only possible in cases with a significantly dilated ureter and sufficient sonographic access.
Sonographic evaluation of the urine inflow jet from the ureteral orifice into the urinary bladder can provide useful information about the patency and peristaltic activity of the ureter, as well as the position and function of the UVJ. This ureteral jet can sometimes be seen on gray-scale ultrasound as a rush of echoes from the ureteral ostium into the bladder lumen, but it is more easily seen with color Doppler sonography. Sonographic visualization of this jet can be limited with patient dehydration or when the specific gravity of ureteral urine and bladder urine are similar after recent voiding.
Modern multidetector computed tomography (CT) can rapidly acquire high spatial resolution images of the entire ureter and surrounding structures. In pediatric uroradiology, CT can be useful for evaluation of severe trauma, urolithiasis and its complications, inflammatory disease, suspected obstructing crossing vessels, and tumors. For evaluation of urolithiasis, noncontrast enhanced CT is typically performed, but for other indications, opacification of the ureteral lumen with contrast is often beneficial. This can be achieved either with delayed, excretory phase imaging after a single contrast material injection or with a split-bolus CT urography technique in which the renal parenchyma and ureter are opacified with contrast material simultaneously. CT angiography can be used to assess vascular anatomy. Although a useful technique, CT does impart ionizing radiation and often requires iodinated contrast, which carries its own inherent risks. Thus CT should be used only when necessary, and protocols should be tailored to minimize radiation exposure.
Diuretic renography is a well-established nuclear medicine technique for assessment of renal function and urinary tract drainage, where serial image acquisitions are obtained after intravenous injection of 99m-technetium-mercaptoacetyltriglycine ( 99m Tc-MAG3) or 99m Tc-diethylene triamine pentaacetic acid ( 99m Tc-DTPA) both before and after diuretic stimulation. With this technique, ureteral drainage can be visualized and quantified in a standardized fashion providing excellent functional information. Limitations and drawbacks include patient exposure to ionizing radiation, limited assessment of urinary tract drainage in the setting of poor renal function, and poor anatomic resolution.
Magnetic resonance urography (MRU) can provide excellent spatial resolution and functional information in a single study without the use of ionizing radiation or iodinated contrast material. In children, MRU can be particularly useful in the investigation of congenital genitourinary anomalies and hydronephrosis. With MRU, anatomic assessment of the entire ureter can be obtained without the need for intravascular contrast material by using a variety of T2-weighted pulse sequences. Postdiuretic contrast-enhanced MRU (so-called functional MRU) allows for functional assessment by using fast T1-weighted gradient recalled echo pulse sequences to image at multiple time points after contrast material administration, allowing assessment of the arterial, corticomedullary, nephrographic, and excretory phases. Downsides of this technique at present include availability, cost, inability to image patients with certain implantable devices, need for sedation or anesthesia for many pediatric patients, and risk of nephrogenic systemic fibrosis if a linear gadolinium contrast agent is administered to patients with severe kidney disease. There is also growing concern about detectable deposition of gadolinium in body tissues, but at present long-term effects remain uncertain.
There is a spectrum of ureteral obstruction ranging from partial obstruction without any clinical consequence to severe obstruction that can threaten renal function. Most ureteral obstructions occur at the UPJ or UVJ. However, obstructive lesions between these two points can occur as a result of congenital and acquired causes.
Congenital ureteral valves are transverse folds of mucosa and variable smooth muscle that are typically cusp-shaped or diaphragm-like in configuration. They are by definition associated with upstream obstructive uropathy and a normal downstream urinary tract, without evidence of other cause for obstruction. They are often associated with other urinary tract anomalies ( Fig. 118.1 ). Neonates and infants may have physiologic ureteral folds that can mimic ureteral valves, but the former are not associated with obstruction and typically disappear during the first years of life ( Fig. 118.2 ). Imaging will demonstrate dilation of the upstream ureter and renal collecting system with abrupt transition at the level of obstruction. The fold may be visible as a membrane-like filling defect when the entire ureter is opacified with contrast material or on heavily T2-weighted noncontrast MRU sequences. Renal scintigraphy will demonstrate impaired collecting system drainage suggesting obstruction. Treatment consists of resection of the valve-containing ureteral segment and ureteral end-to-end anastomosis.
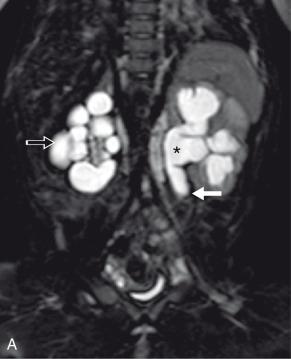
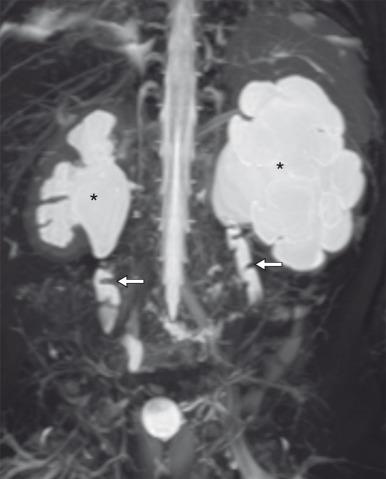
A congenital ureteral stricture is a discrete segment of congenital narrowing along the length of the ureter that does not involve the UPJ and UVJ. It is most commonly seen in the middle third of the ureter, typically where the ureter crosses the common iliac vessels, and is therefore often referred to as a “midureteral stricture” ( Fig. 118.3 ). However, it can also occur in the proximal or distal ureter. Unlike with ureteral valves, there are no mucosal folds present, and at pathology, a smooth funnel-shaped narrowing of the lumen is seen. The etiology is uncertain, with possibilities including focal abnormalities of ureteral wall musculature or relative focal ischemia during development. Association with multicystic dysplastic kidney (MCDK), either ipsilateral or contralateral to the involved ureter, is not uncommon. Detection on prenatal imaging has been reported, but differentiating a midureteral stricture from the more common UPJ or UVJ obstruction is challenging. On postnatal imaging, findings will often be similar to those of ureteral valves, with distinction between the two entities generally made at pathology ( e-Fig. 118.4 ). Most congenital ureteral strictures require surgical management, but patients with preserved function and no symptoms may be managed conservatively.
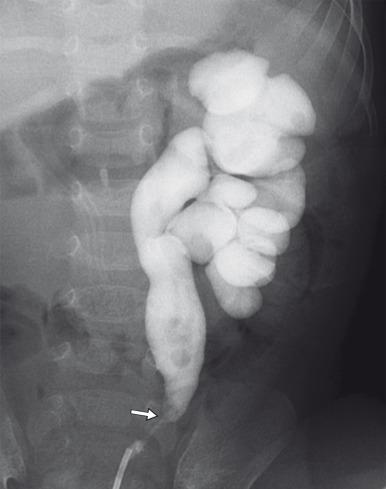
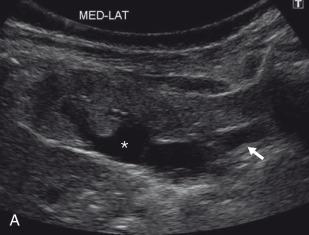
A retrocaval or circumcaval ureter is an uncommon anomaly more often seen in males in which the right ureter passes posterior to the inferior vena cava (IVC), emerges between the cava and the aorta, and then curves around and in front of the cava to return to its normal position in the pelvis ( e-Fig. 118.5 ). This anomaly results from anomalous development of the infrarenal IVC from the more laterally located right posterior cardinal vein rather than the more typical right supracardinal vein. At least some degree of ureteral obstruction is common. However, patients are often asymptomatic, and the abnormality may be discovered on imaging for other reasons. The anomaly may be demonstrated with contrast-enhanced CT, but MRU can establish the diagnosis and assess the degree of functional obstruction. Surgery is performed when the patient is symptomatic or when there is decreased function of the affected kidney, and consists of division of the retrocaval ureter and ureteroureteric or ureteropelvic anastomosis anterior to the IVC.
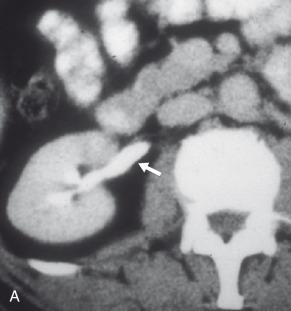
Crossing renal arteries and veins have been associated with urinary tract obstruction, particularly intermittent obstruction manifesting as recurrent renal colic and hydronephrosis. These vessels typically cross anterior to the UPJ or proximal ureter and may be branches of the renal artery or vein, or accessory vessels ( Fig. 118.6 ). The crossing vessel may be demonstrated with either contrast-enhanced CT or MR angiography. Normal vascular impressions can be seen on imaging but are not associated with secondary findings of obstruction. When a crossing vessel is identified in a patient with clinical and imaging findings suggesting intermittent obstruction and there is no intrinsic UPJ obstruction identified at surgery, a vessel transposition can be performed. When a crossing vessel is felt to be potentially aggravating an underlying intrinsic UPJ obstruction, a dismembered pyeloplasty is performed.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here