Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
A variety of congenital and developmental anomalies, inflammatory and infectious diseases, and benign and malignant neoplasms occur in the mediastinum in the pediatric population. Up-to-date knowledge and a practical diagnostic approach combined with a clear understanding of the characteristic imaging appearances of these conditions can facilitate prompt diagnosis and optimal patient management. This chapter reviews the underlying etiology, imaging findings, treatment, and follow-up of commonly encountered congenital and acquired anomalies and abnormalities that occur within the mediastinum in the pediatric population.
Pneumomediastinum occurs more frequently in infants than in older children. Affected pediatric patients may present with a sensation of retrosternal fullness, dysphagia, sore throat, chest pain, or dyspnea.
Pneumomediastinum may be spontaneous or iatrogenic. Spontaneous pneumomediastinum results from a sudden forceful increase in intraalveolar pressure such as forceful inhalation or the Valsalva maneuver. In this instance, an alveolus can rupture, allowing gas under pressure into the low-pressure pulmonary interstitial compartment. Subsequently, the air then travels via the peribronchovascular space medially toward the hilum, which opens into the mediastinum. Occasionally, the air dissects along the lymphatics as well and extends to the visceral pleura, where a concomitant pneumothorax may occur. Iatrogenic pneumomediastinum may result from chest, abdominal, and cardiac surgery; endotracheal intubation; or cardiac catheterization. Secondary pneumomediastinum can occur after foreign body ingestion, trauma to the neck or chest, or any disruption of the tracheobronchial tree or esophagus (e.g., Boerhaave syndrome).
It is not uncommon for pneumomediastinum and pneumothorax to coexist. This phenomenon sometimes can be attributed to a common mechanism of air leak, or the pneumothorax may arise as a result of a pneumomediastinum. In addition, anatomic communications between the mediastinum and the peritoneal cavity exist. As such, intraabdominal air can dissect superiorly into the mediastinum and vice versa. Rarely, air within the mediastinum can enter the spinal canal, termed pneumorrhachis.
Air within the mediastinum typically displaces the pleura and lung laterally ( Fig. 58.1 ). It may decompress into the superior mediastinum and dissect along fascial planes into the subcutaneous tissues of the neck and retropharynx ( e-Fig. 58.2 ). In infants and younger children, air within the mediastinum can displace the thymus superiorly to produce the “spinnaker sail” sign (see Fig. 58.1 ).
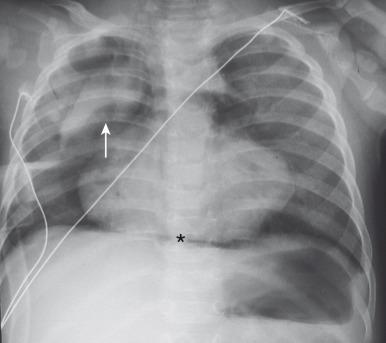
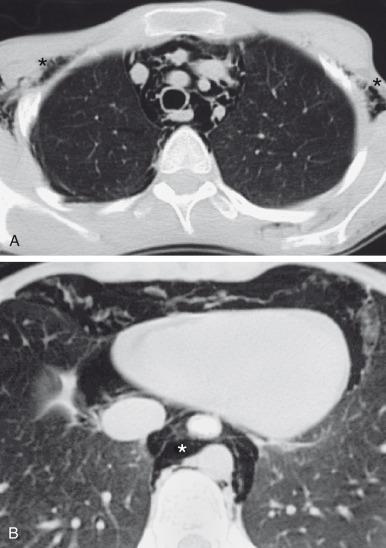
Occasionally, only a sliver of curvilinear radiolucency is seen adjacent to the cardiac border, often on the left, outlining the aortic arch and descending aorta. When air is adjacent to the pulmonary artery (typically on the right), the “ring around the artery” sign is seen on the lateral view. The “continuous diaphragm sign” results from air interposed between the pericardium and the diaphragm. Although pneumomediastinum is usually anterior in location, posterior mediastinal air collections can also occur, typically toward the right in the infra-azygous space ( e-Fig. 58.3 ). Posterior pneumomediastinum may occur secondary to barotrauma but should raise concern for iatrogenic and especially tracheal or esophageal injury.
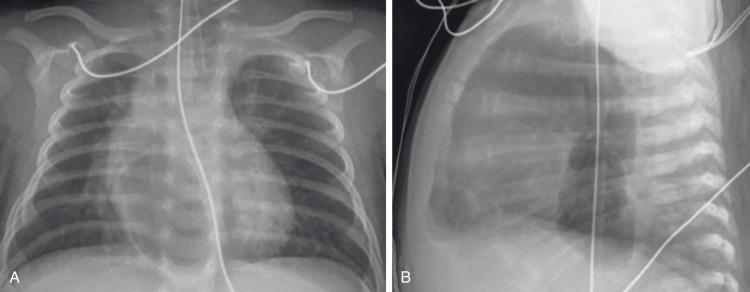
A large pneumomediastinum may be confused with a pneumothorax, especially when viewed on a supine radiograph. When equivocal, a decubitus radiographic view may be helpful because the mediastinal air does not move, whereas the pneumothorax rises nondependently.
Pneumomediastinum may mimic pneumopericardium. In contrast to pneumomediastinum, pneumopericardium does not lift the thymus or outline the aortic arch as the air is contained by pericardium. Pneumopericardium is almost always seen in association with pneumomediastinum except after open-heart surgery.
Treatment of pneumomediastinum is aimed at its underlying cause. In most cases, management is supportive. If there is concern for esophageal rupture, an esophagram using water-soluble contrast may be performed, and surgical consultation should be obtained. However, esophageal rupture in children is rare and, in most uncomplicated cases of pneumomediastinum, no further imaging beyond the diagnostic chest x-ray is necessary. Very rarely, pseudotamponade, laryngeal compression, tension pneumomediastinum, tension pneumothorax, or mediastinitis occur and require surgical intervention.
Mediastinal hemorrhage in children may occur as a result of blunt trauma (usually venous bleeding) or iatrogenic causes related to central catheter placement or cardiothoracic procedures. However, mediastinal hemorrhage in children may occur spontaneously in the context of hemophilia, in which case the hemorrhage may be retropharyngeal and dissect into the mediastinum. Rare cases of neonatal thymic hemorrhage have been reported, possibly related to vitamin K deficiency.
Although imaging findings may be nonspecific, the possibility of mediastinal hemorrhage should be considered when mediastinal widening, blurring of the aortic margin, deviation of a nasoenteric tube, and/or left apical “capping” are present on chest radiographs. When evaluating mediastinal widening on chest radiographs, careful attention must be paid to the technique. For example, a portable anteroposterior chest radiograph may exaggerate the size of the mediastinum. Therefore in equivocal cases, confirmation with a posteroanterior view or a cross-sectional imaging study such as computed tomography (CT) may be necessary. On CT, mediastinal fluid with a Hounsfield unit greater than water (approximately 20–80 HU) suggests mediastinal hemorrhage ( Fig. 58.4 ).
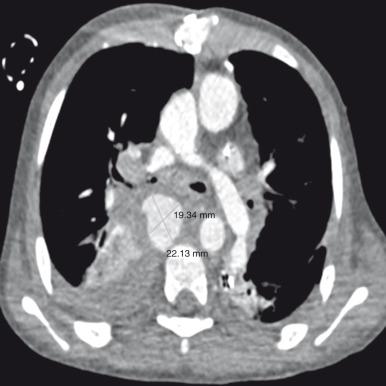
When mediastinal hemorrhage is considered on chest radiographs, further imaging studies such as echocardiography and/or contrast-enhanced CT using CT angiographic technique with multiplanar and three-dimensional reconstruction is warranted. With rapid clinical deterioration, urgent surgical exploration is an option to avoid delaying proper treatment.
Infections arising de novo in the mediastinum are rare in the pediatric population. Mediastinal infections can be classified into acute infection and chronic fibrosing mediastinitis.
Acute mediastinal infection in children is most often iatrogenic, related to endoscopic surgery, esophageal instrumentation, and cardiac surgery. Chronic fibrosing mediastinitis typically results from tuberculosis or Histoplasmosis infections in the pediatric population.
In the past, esophageal perforation and spread of infection from other sites into the mediastinum were relatively more common. Affected pediatric patients typically present with chest pain, fever, and an elevated white blood cell count.
Plain radiographic findings of acute mediastinitis are nonspecific. However, mediastinal widening, obliteration of normal mediastinal contours, and displaced or narrowed trachea should suggest acute mediastinitis in the appropriate clinical setting. The more specific radiologic imaging finding of acute mediastinitis is the presence of gas within the mediastinum, which can be better evaluated with CT. CT also can show complications from acute mediastinitis such as mediastinal abscess formation or empyema ( Fig. 58.5 ).
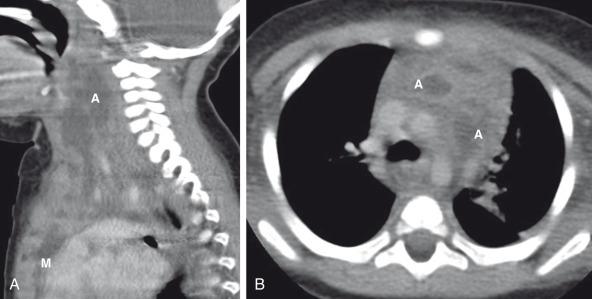
Acute mediastinitis is treated aggressively with a combination of antibiotics and surgical irrigation. Localized mediastinal abscess formation can be managed with either surgical or percutaneous abscess drainage. Recently, a combination of continuous irrigation and vacuum-assisted postthoracotomy closure has been used and shown to be safe in children.
Chronic fibrosing mediastinitis, also known as sclerosing mediastinitis, is rare in the pediatric population. It is a condition characterized by abnormal proliferation of dense acellular collagen and fibrous tissue. Although it is most frequently attributed to the sequelae of granulomatous infection such as Mycobacterium tuberculosis or Histoplasmosis infection, it also can arise as an idiopathic condition or as the sequelae of autoimmune disease, radiotherapy, or drugs such as methysergide and metoprolol. Chronic fibrosing mediastinitis is also associated with retroperitoneal fibrosis, sclerosing cholangitis, and Riedel thyroiditis. Affected pediatric patients often present with respiratory distress related to airway narrowing, dysphagia due to esophageal compromise, and/or facial and neck swelling resulting from obstruction of the superior vena cava.
Widening of the mediastinum with a lobular paratracheal and/or subcarinal mass that may be calcified is a typical finding on chest radiographs. CT imaging findings of chronic fibrosing mediastinitis can be categorized into two patterns: focal or diffuse. The focal form is more likely to calcify ( Fig. 58.6 ). The diffuse type demonstrates a more diffusely infiltrating appearance without calcification.
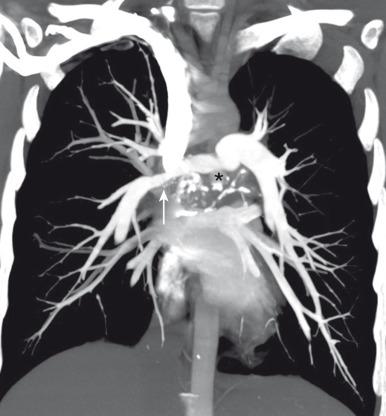
Although no consensus or widely accepted guidelines currently exist for the treatment of chronic fibrosing mediastinitis, systemic antifungal or corticosteroid treatment, surgical resection, and local therapy for complications are the current management options. Surgical resection may be necessary for symptomatic pediatric patients who have extensive chronic fibrosing mediastinitis that results in either obstruction or compression of mediastinal structures such as central airways, the esophagus, or large vessels.
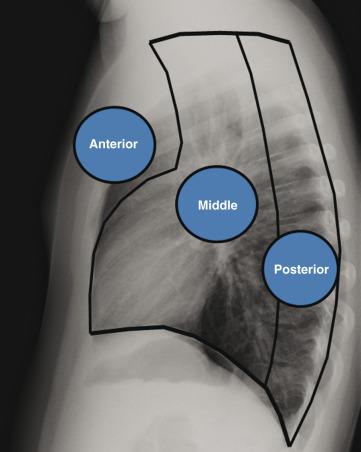
The mediastinum is the most common location of chest masses in the pediatric population. Mediastinal masses in infants and children can be benign or malignant neoplasms, congenital anomalies, infections, vascular malformations, or pseudomasses. As with adult patients, it is useful to localize the mediastinal mass to one of three mediastinal compartments (anterior, middle, or posterior) ( Fig. 58.7 ). However, such a system of compartmentalizing the mediastinum may have shortcomings. Specifically, the borders of the anterior, middle, and posterior mediastinum, which are defined by anatomic landmarks as assessed on a lateral radiograph of the chest, do not have true fascial planes. Recently, the International Thymic Malignancy Interest Group has published a similar three compartmental approach but is based on CT anatomy, allowing for anatomic delineation defined, in part, by pericardial and pleural reflections. Regardless of the technique or specific classification used, the practice of assigning a mediastinal mass to a specific mediastinal compartment is useful by enabling one to formulate a manageable differential diagnosis, and effectively to direct further imaging workup.
The most common anterior mediastinal masses in children are thymic lesions, lymphoma, lymphatic malformations, and germ cell tumors.
Arising from the third pharyngeal pouch, the thymus serves as the site of T-cell maturation. It begins a process of caudal and ventromedial elongation during the seventh and eighth week of gestation whereby the two sides fuse at about the level of the aortic arch. Absence or hypoplasia of the thymus is a component of DiGeorge syndrome, with an incidence of 1 per 2,000 to 4,000.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here