Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
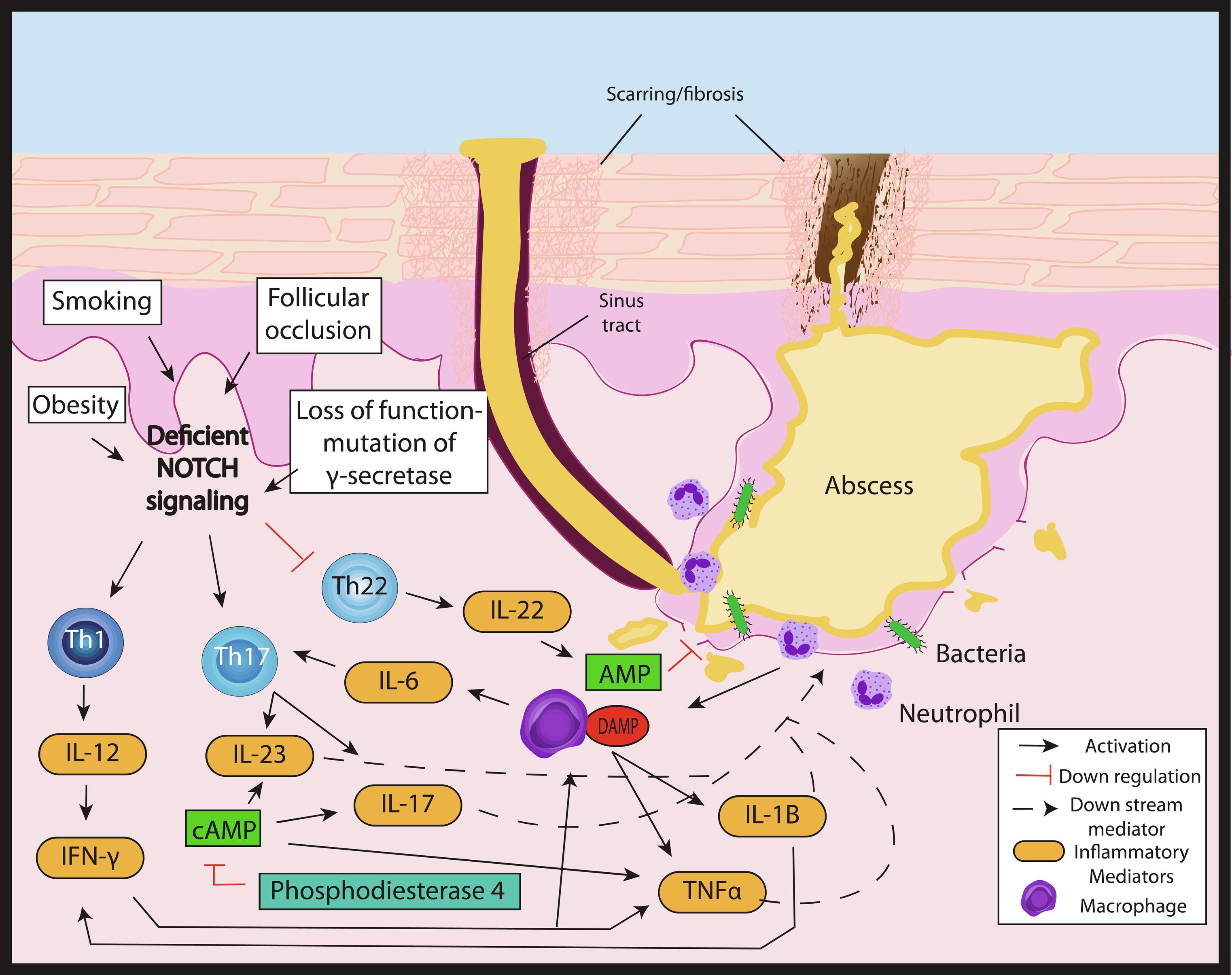
Hidradenitis suppurativa (HS) is a chronic inflammatory disorder with a complex, multifactorial, and incompletely elucidated pathophysiology. Among relevant host factors are a number of important inflammatory pathways, including increased production of oxygen free radicals, enhanced expression of toll-like receptors and release of pro-inflammatory cytokines, increased tumor necrosis factor (TNF)-α expression, activation of the interleukin-23 (IL-23)/T helper-17 (TH-17) pathway, overproduction of interleukin-1 (IL-1), and others ( Fig. 18.1 ). Historically, antibiotic and surgical treatments were the mainstay of HS therapy, but in the last decade a growing number of biologic therapies which modulate these inflammatory pathways have become available and have been investigated for treatment of moderate-to-severe HS. The appropriate candidate for biologic therapy has moderate or severe HS (correlating with Hurley stage II or III) and has either failed appropriate non-biologic therapy (e.g., 3 months or longer of doxycycline) or has inflammatory HS, which would benefit from the use of a biologic as initial therapy. This chapter will review in detail the evidence for these targeted therapeutics, including adverse effects, monitoring recommendations, treatment pearls, and considerations for successful treatment of HS in the setting of relevant patient comorbidities ( Table 18.1 ).
| Biologics and Small Molecules | Mechanism of Action | Comorbidity Use (FDA-approved for use in below conditions) | Contraindication | Efficacy Data and Level of Evidence | Major Adverse Events | Additional Notes |
|---|---|---|---|---|---|---|
| Adalimumab | Fully humanized IgG1 monoclonal antibody against TNF-α | FDA approved for use in: Rheumatoid arthritis (RA), psoriatic arthritis (PsA), ankylosing spondylitis (AS), Crohn's disease and ulcerative colitis (IBD), psoriasis (PsO), hidradenitis suppurativa (HS) uveitis | Demyelinating conditions such as multiple sclerosis (MS); congestive heart failure (CHF); Lupus (SLE) | A, I |
|
|
| Infliximab | IgG1 monoclonal antibody against TNF-α | IBD, RA, AS, PsO, PsA | MS; Relative contraindication: CHF and SLE | B, II |
|
|
| Anakinra | IgG1 monoclonal antibody IL-1-α and 1β inhibitor | RA, Cryopyrin-Associated Periodic Syndromes (CAPS) | Known hypersensitivity to E. coli -derived proteins, Anakinra | B, II |
|
|
| Canakinumab | IgG1 monoclonal antibody to IL-1β inhibitor | CAPS, Muckle-Wells (MW), Familial Cold Autoinflammatory Syndrome (FCAS) | Known hypersensitivity to Canakinumab | C, III |
|
|
| Ustekinumab/IL12/23 inhibitors | IgG1 monoclonal antibody binds to p-40 subunit of both IL-12 and IL-23 | PsO, PsA, IBD | Known hypersensitivity to Ustekinumab | B, II |
|
|
| Guselkumab/p19 inhibitors | IgG1 monoclonal antibody that binds the P19 subunit of IL-23 | PsO | None | C, III |
|
|
| Secukinumab, Ixekizumab/IL17 inhibitors | IgG1 monoclonal antibody that binds IL-17A | PsA | Known hypersensitivity to Secukinumab, Ixekizumab; IBD | C, III |
|
|
| Tofacitinib and janus kinase (JAK) inhibitors | JAK 3 inhibitor | RA, PsA, UC | Patients with severe hepatic impairment, known hypersensitivity | C, III |
|
|
Adalimumab is the first FDA-approved medication for the treatment of HS and is thus the first-line agent for treatment of moderate or severe HS which has failed to respond to non-biologic therapies. Efficacy was established with publication of the Pioneer I and II trials in 2016. Pioneer I and II demonstrated a modest but significant improvement in disease for roughly half of patients. The two trials included anti-TNF treatment-naive patients with Hurley Stage II or III disease and at least 3 inflammatory nodules. Treatment included a loading dose followed by maintenance dosing at 40 mg weekly. HS Clinical Response, termed the HiSCR, was defined as a 50% reduction in inflammatory nodule count. HiSCR was achieved in 41.8% of patients in the adalimumab arm of Pioneer I and 58.9% of patients in the adalimumab arm of Pioneer II, compared to 26% and 27.6% of patients in the placebo arms, respectively. Health-related quality-of-life measures also improved significantly among patients in the treatment arm, including better pain numeric rating scale scores and improvements in Dermatology Life Quality Index (DLQI) measures. Based on this and subsequent data, adalimumab has become the mainstay of therapy for patients with moderate-to-severe HS following antibiotic failure.
Pioneer I and II demonstrated efficacy in HS through the use of higher dosages than were previously standard for other inflammatory conditions; psoriasis, rheumatoid arthritis, inflammatory bowel disease (IBD), and psoriatic arthritis all utilize every-other-week maintenance dosing of adalimumab. However, in studies comparing weekly with every-other-week dosing of adalimumab for HS, patients receiving every-other-week dosing (or decreasing to every-other-week dosing after initial response to weekly dosing) experienced reduced efficacy. Standard treatment based on these and other trials has established the need for a loading dose of 160 mg at week 0, followed by 80 mg at week 2, and then continued use of 40 mg weekly thereafter beginning at day 28.
While adalimumab has clear efficacy for the treatment of HS, outcomes from the PIONEER trials were also notable for the number of non-responders. Roughly half of PIONEER I and PIONEER II patients did not achieve HiSCR (50% disease response) with the use of weekly adalimumab. Patients experiencing a partial response to therapy at week 12 frequently achieve HiSCR by week 36 with continued therapy, but those who do not show at least a partial response by week 12 are unlikely to benefit from continued treatment and should be considered for alternative therapies. Among responders, continued weekly dosing of adalimumab is considered the most effective strategy, yet those who achieve HiSCR generally continue to have flares and waxing and waning disease activity.
Patients with inadequate response to therapy with adalimumab frequently benefit from a switch to infliximab. While infliximab has not been studied in a randomized controlled trial for HS comparable in size to those for adalimumab, it nevertheless offers an evidence-based alternative for treatment of moderate or severe HS, as outlined in the European and American treatment guidelines and a Cochrane Review. Infliximab is an anti-TNF-α therapy administered intravenously in a weight-based fashion. Loading doses at weeks 0, 2, and 6 are followed by maintenance dosing every 4 to 8 weeks thereafter. Randomized controlled trials of Infliximab have demonstrated significant disease improvement over placebo; however, sample sizes are small.
Weight-based dosing is a potential advantage of infliximab, as is the ability to titrate the dose to effect. In general, higher doses of the medication may be more effective for more severe, inflammatory HS. Published data support the need for dose escalation in most patients (64%; 34/52), with treatment response and achievement of stable dosing generally occurring at 10 mg/kg every 6 to 8 weeks. Other data suggest most (71%; 17/24) patients receiving 7.5 mg/kg every 4 weeks (after loading) achieve a good (HS PGA 0 to 2) clinical response, while dose escalation to 10 mg/kg every 4 weeks is often (50%; 6/12) successful in those who do not.
Etanercept, 50 mg injected subcutaneously weekly or twice weekly, was studied in two small prospective studies versus placebo and failed to yield significant disease improvement. Based on available evidence, it is not recommended for treatment of HS.
Evidence for the use of golimumab in HS is limited to two case reports, one positive and one negative. Use of certolizumab is anecdotal. Despite a lack of data, these agents may be useful in some cases, particularly in the setting of a comorbid condition (e.g., rheumatoid arthritis, IBD) or a prior intolerance of alternative therapies.
Injection site reactions are a common complication of adalimumab use, characterized by erythematous patches and plaques, generally round and edematous, arising minutes to hours following drug administration. Reactions generally occur within the first month of treatment and last 3 to 5 days after each exposure. They are thought to constitute a type I hypersensitivity reaction and should be treated as such. Reactions can be treated with topical steroids, antihistamines, nonsteroidal antiinflammatory drugs (NSAIDs) or acetaminophen, and ice. Some severe reactions cause significant pain and edema and may require medication discontinuation. In these cases, a switch to a citrate-free formula of adalimumab can help control injection site reactions while sparing the patient the need to switch to an alternative therapy.
Infusion reactions (IR) to infliximab are well-documented given the drug’s extensive use in other rheumatologic, dermatologic, and gastrointestinal conditions. IRs can be divided into early reactions, which occur peri-infusion, and late reactions, which occur 24 hours following infusion. Immediate IRs include symptoms such as pruritus, dyspnea, flushing, and headache, as well as anaphylaxis. These reactions are thought to be mediated via host activation of complement, IgE, and mast-cell degranulation in the setting of the TNF immune globulin. Mild infusion reactions such as transient flushing, myalgia, and pruritus are managed with attenuation of infusion rate, while moderate reactions such as fever, urticaria, and hypertension are treated with infusion interruption, oral antihistamine and acetaminophen therapy, and resumption of infusion at slower rates. Severe IRs, including bronchospasm and hypotension, are managed promptly with typical anaphylaxis response and discontinuation of the infusion. Patients can be re-challenged with test doses and graded drug infusion if infliximab is deemed warranted. Because immediate IRs are common, infliximab is generally ordered with as needed medication available and may be given following pre-medication with corticosteroids, antihistamines, and anti-pyretics. Montelukast may help prevent some respiratory symptoms. In the case of severe IRs with intended re-challenge, input from allergy/immunology may help guide premedication protocols. While these premedication protocols are routinely employed in the setting of infliximab, evidence for their use is limited.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here