Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Injury severity and the spectrum of injuries continue to evolve, resulting in greater and different challenges for the trauma surgeon. High-energy blunt trauma, as well as increased availability of military firearms with greater wounding capacity, have resulted in an increase in complex injuries involving multiple organs. Improvements in prehospital care and trauma resuscitation have allowed moribund patients to reach the hospital alive but in extremis. Damage control surgery, with its focus on treating the life-threatening injuries and delaying definitive repair until the metabolic and physiologic perturbations have been corrected, has reduced mortality rates in this subset of injured patients.
Damage control management has three separate and distinct phases. The first phase includes prompt resuscitation with blood products, rapid hemorrhage control, and the minimizing the duration of contamination from hollow viscus injuries. Packing is utilized to tamponade bleeding, and temporary wound management strategies are employed. The second phase, which occurs in the intensive care unit (ICU), involves rewarming, correction of coagulopathy, and restoration of oxygen delivery with blood components while minimizing crystalloid solutions. Once normal physiology has been established, the third phase involves definitive operative management of the patient’s injuries.
The goal of a damage control procedure is to preserve the life or a limb of a patient with devastating injuries in the setting of ongoing hemorrhagic shock. Most commonly applied with exsanguinating abdominal injuries, damage control approaches can also be used to successfully manage life- and limb-threatening thoracic and orthopedic injuries, respectively.
Damage control procedures attempt to truncate surgical intervention prior to the development of irreversible physiologic derangement. Uncontrolled hemorrhage leading to hypotension results in global ischemia. Delay in surgical control of the blood loss results in the lethal triad of hypothermia, coagulopathy, and acidosis. Although each of these individual complications is potentially life-threatening, the combination of all three results in an exponential increase in mortality. Damage control surgery, by abbreviating the initial surgical intervention and correction of the lethal triad, has reduced mortality rates.
Hypothermia is defined as a core temperature of less than 35° C. Cooling of the core temperature by radiant heat loss begins in the prehospital setting and continues after arrival to the emergency department (ED) during the primary and secondary surveys. During a damage control operation, aggressive resuscitation with unwarmed fluids and evaporative loss from exposed peritoneal or pleural surfaces contribute to continued loss of body temperature and should be minimized. The incidence of clinical hypothermia after laparotomy for traumatic injuries is 57%. Hypothermia has clearly been shown to increase the risk of mortality. The mortality rate increases significantly with a core temperature less than 34° C, and is uniformly fatal with a core temperature less than 32° C.
Hypothermia has systemic effects, including a negative impact on cardiovascular performance (decreased heart rate and cardiac output, increased systemic vascular resistance), renal function (decreased glomerular filtration rates), and the central nervous system (depressed mental status). The coagulation cascade is prolonged in a linear fashion with reduction of core temperature, as clot formation relies on a series of temperature-dependent enzymatic reactions. Studies have demonstrated hypothermia-related increases in prothrombin time, thrombin time, and partial thromboplastin time (PTT). Hypothermia also affects platelet function, leading to sequestration in the portal circulation and prolonged bleeding times.
Metabolic acidosis occurs after hemorrhagic shock due to the switch from aerobic to anaerobic metabolism during periods of hypoperfusion. The detrimental effects of acidosis include depressed myocardial contractility and a diminished response to inotropic medications. Acidosis predisposes the myocardium to ventricular dysrhythmias. In patients with a traumatic brain injury, it can contribute to intracranial hypertension. Finally, acidosis has been shown to worsen coagulopathy by independently prolonging PTT, decreasing factor V activity, and contributes to the development of disseminated intravascular coagulation.
Even in the absence of hypothermia and acidosis, coagulopathy develops secondary to dilutional effects when massive resuscitation is composed primarily of crystalloid and packed red blood cells without clotting factor replacements. Massive transfusion protocols (MTP) aim to prevent this dilutional coagulopathy by providing matched ratio product resuscitation that includes fresh frozen plasma and platelets. Coagulopathy may also be exacerbated when there is significant tissue injury. This results in exposed tissue factor, which activates the clotting cascade, resulting in further consumption of clotting factors.
A systematic approach to the initial management of the injured patient has been promulgated by the Advanced Trauma Life Support course of the American College of Surgeons Committee on Trauma. The primary and secondary surveys as described in this course allow the rapid identification of life-threatening injuries and enable the surgeon to prioritize subsequent operative management of the unstable trauma patient. Patients with exsanguinating hemorrhage (class IV hemorrhagic shock) should be expeditiously transported to the operating room. In patients with obvious ongoing resuscitation requirements, a large-bore central venous catheter should be placed for rapid volume resuscitation. Given the multiple factors that predispose these patients to coagulopathy, early consideration should be given to the initiation of the MTP with administration of matched ratios of coagulation factors (fresh frozen plasma and cryoprecipitate) and platelets in addition to packed red blood cells while minimizing crystalloids.
Resuscitative endovascular balloon occlusion of the aorta (REBOA) is a tool that can be deployed for temporary hemostasis while in the trauma bay or the operating theatre to reduce blood loss until direct control is obtained. In certain settings, it can be used in place of an ED thoracotomy. Using the Seldinger technique, an intra-aortic balloon is inserted into the aorta via the common femoral artery. The balloon can be inflated at one of three zones of the aorta ( Fig. 1 ). The decision on where to place the REBOA is dependent on the suspected source of bleeding: zone 1 (between left subclavian artery and celiac artery) for any abdominal bleeding or zone 3 below the renal arteries and above the aortic bifurcation for suspected pelvic bleeding. Placement and inflation of the balloon in zone 2 is not recommended.
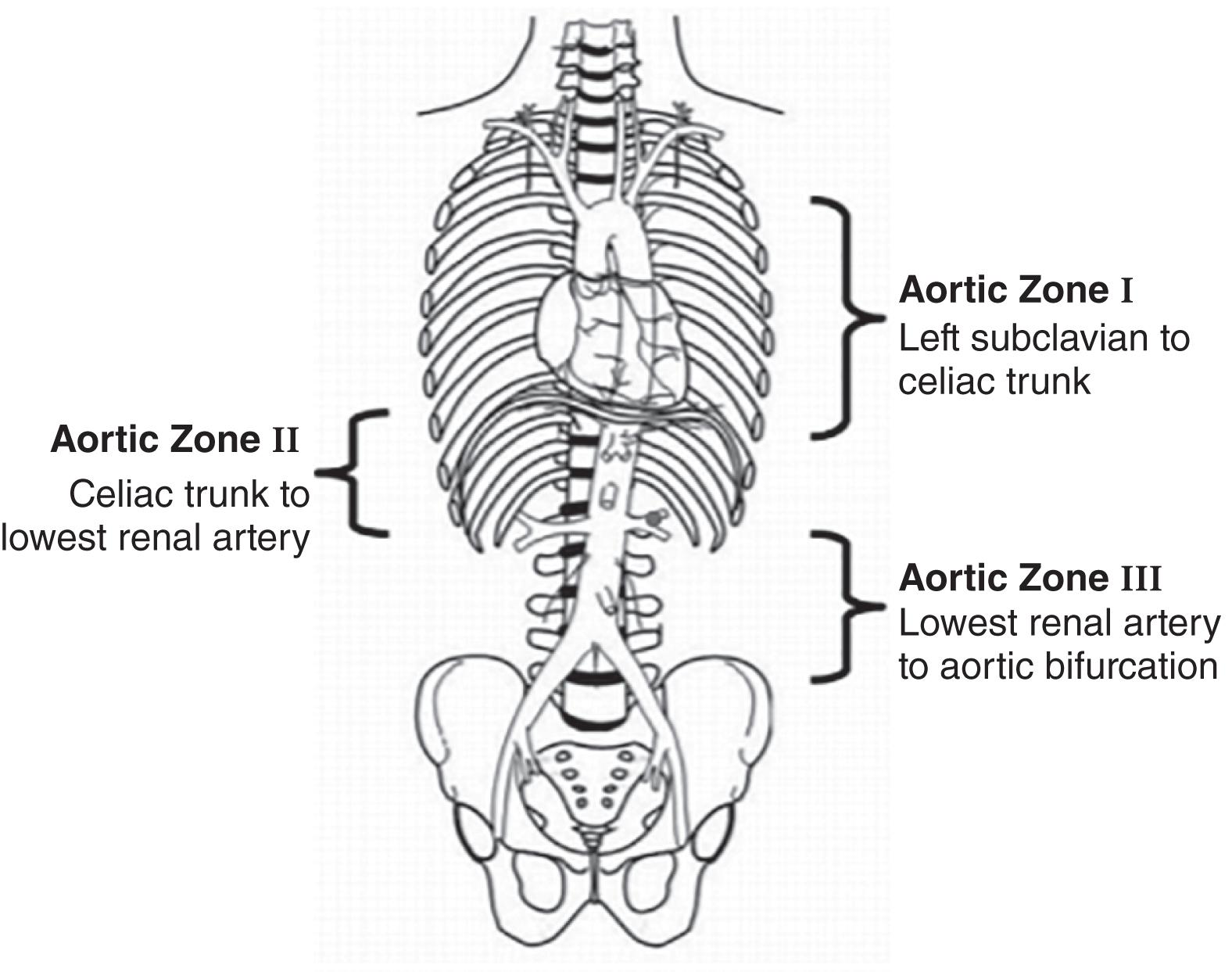
Use of the REBOA is controversial. Brenner et al state that the placement of REBOA can be done quickly and efficiently to prevent hemorrhage as an alternative to cross-clamping the aorta after a resuscitative thoracotomy without an increase in mortality. In their experience, REBOA allowed for a minimally invasive way to stabilize the patient long enough to transport to the operating theater. Even though that study did not show an increase in morbidity, Bellal et al showed that an increase in REBOA use leads to increasing acute kidney injury and lower leg amputations as well a possible increase in mortality. Currently, the primary indication for placement of REBOA is noncompressible hemorrhage below the diaphragm (pelvic fracture), for patients in hemorrhagic shock.
The decision regarding the initiation of damage control strategies should be made early in the operative course based on the patient’s physiologic parameters, body temperature, and intravascular volume status. As described by Moore et al, the original indications for a damage control strategy included an inability to achieve hemostasis due to ongoing coagulopathy, a technically difficult or inaccessible major venous injury, a time-consuming procedure in the setting of under resuscitated shock, and a need to address other life-threatening injuries. The earliest descriptions of what subsequently became known as damage control surgery involved patients with major hepatic injuries, in whom the placement of perihepatic packing and staged operative management resulted in decreased morbidity and mortality rates. Expanded indications include hemodynamically unstable patients with either high-energy blunt torso trauma or multiple penetrating injuries or any trauma patient presenting in shock with hypothermia and coagulopathy ( Table 1 ). More recently, Roberts et al, further defined preoperative and intraoperative indications for damage control surgery that result in improved patient outcomes ( Table 2 ). Major liver and vascular injuries remain the most common triggers for performing a damage control operation.
| Hemodynamic instability due to hemorrhagic shock |
| Presenting coagulopathy or hypothermia |
| Major abdominal vascular injury with or without associated visceral injury |
| Complex hepatic injuries with or without associated visceral injury |
| Multicavitary injuries with hemorrhagic shock |
| Multisystem trauma with competing management priorities |
| Metabolic acidosis with pH < 7.3 |
| Massive transfusion requirements |
| Preoperative Indications | Intraoperative Indications |
|---|---|
| Cardiac arrest | Abdominal vascular injury + major abdominal solid or hollow organ injury |
| Concomitant severe TBI | Major pancreatic or duodenal injury |
| Injury Severity Score > 25 | Involvement of multiple anatomic regions requiring surgery |
| Hypotension: systolic BP < 90 mm Hg | Prolonged surgery > 90 minutes |
| Hyperthermia: core temperature <34° C | Estimated blood loss > 5 L |
| Acidosis: BD > 15 mmol/L or lactate > 5 mmol/L | Massive resuscitation: >10 units PRBCs |
| Coagulopathy or hyperfibrinolysis on rTEG | Persistent hypotension: systolic BP < 90 mm Hg |
| Massive resuscitation: >10 units PRBCs | Persistent hyperthermia: core temperature <34° C |
| Acidosis: pH < 7.2 |
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here