Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The authors thank Dr. Peter Libby, co-author of this chapter in editions 6 to 10, for his contributions and mentorship.
![]() Additional content is available online at Elsevier eBooks for Practicing Clinicians
Additional content is available online at Elsevier eBooks for Practicing Clinicians
Peripheral artery disease (PAD) generally refers to acute or chronic obstruction of the arteries supplying the lower or upper extremities that, when severe, results in downstream ischemia and potentially tissue loss. , Most often caused by atherosclerosis, PAD may also result from thrombosis, embolism, vasculitis, fibromuscular dysplasia (FMD), or entrapment. The term peripheral vascular disease is less specific because it encompasses a group of diseases affecting blood vessels that include other atherosclerotic conditions, such as renal artery disease and carotid artery disease, as well as vasculitides, vasospasm, venous thrombosis, venous insufficiency, and lymphatic disorders.
The primary morbidity in patients with PAD relates to limb symptoms and ischemic limb complications including intermittent claudication, chronic critical limb ischemia (CLI), acute limb ischemia (ALI), and tissue loss. The majority of patients with PAD suffer from functional limitation even though a minority report typical claudication. This limb morbidity impacts quality of life and independence and, in its severe forms, associates with increased mortality. , In addition to limb morbidity, patients with atherosclerotic PAD have a heightened risk of major adverse cardiovascular events (MACEs) similar to patients with prior myocardial infarction or stroke, and this risk is increased in patients with symptomatic disease in multiple territories, called polyvascular disease.
Due to lower rates of public recognition relative to coronary artery disease (CAD) and stroke as well as underrecognition of atypical claudication symptoms, PAD is underdiagnosed. A telephone survey of more than 2500 people found that only 26% knew about PAD relative to more than 65% who were familiar with coronary heart disease, stroke, and heart failure and only 14% recognized that PAD could lead to amputation. Even in patients with a diagnosis of PAD, preventive therapies are underused overall and relative to patients with CAD. These observations may explain in part why rates of CLI and related hospitalizations are stable or increasing. Clinicians treating patients with PAD not only must be skilled in application of strategies to reduce systemic ischemic risk but also must know how to characterize the severity of limb disease and use therapies to optimize function and reduce the risk of tissue loss. This chapter provides a framework for the diagnosis and management of patients with PAD.
The prevalence of PAD varies according to the population studied, the diagnostic method used, and whether symptoms are included to derive estimates. Most epidemiologic studies have used a noninvasive measurement, the ankle-brachial index (ABI), to diagnose PAD. The ABI is the ratio of ankle to brachial systolic blood pressure (SBP) (see later). The prevalence of PAD based on abnormal ABI values ranges from approximately 6% in persons 40 years and older to 15% to 20% in those 65 years and older ( eFig. 43.1 ). , Claims data suggest that the annual incidence and prevalence of PAD are 2.69% and 12.02%, respectively. PAD affects some 8 to 10 million individuals in the United States and more than 200 million people worldwide. The prevalence of PAD is greater in men than in women in most studies. Taking into consideration the total number of women and men in the U.S. population, however, there are more women than men with PAD. , Blacks have a higher prevalence of PAD than non-Hispanic whites, as well as higher rates of amputation.
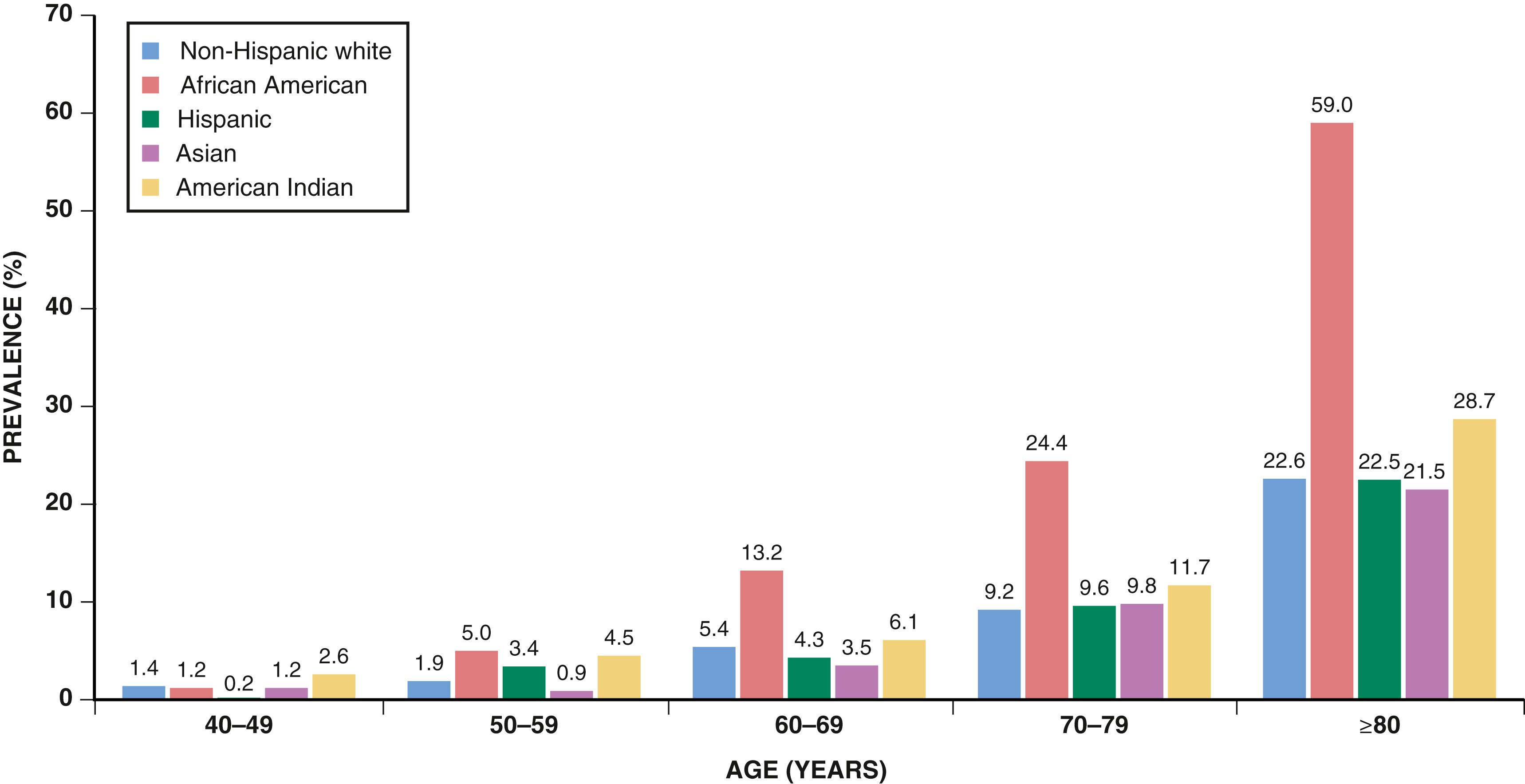
Questionnaires specifically designed to elicit symptoms of intermittent claudication can serve to assess the prevalence of symptomatic disease in these populations. Overall, the estimated prevalence of claudication ranges from 1.0% to 4.5% in a population older than 40 years. , , The prevalence and incidence of claudication increase with age and are greater in men than in women in most studies. Estimates vary by age and sex but generally indicate that 10% to 30% of patients with PAD have claudication. The incidence of CLI is approximately 0.35%, and the prevalence is 1.33% in an insured population, with an annualized incidence of CLI in patients with known PAD of 11.08%. , , , The incidence of ALI in patients with PAD is estimated to be approximately 1% to 2% per year, with higher rates in patients with a history of lower extremity revascularization (LER). The incidence of amputation ranges from 112 to 250 per million per year.
The well-known modifiable risk factors associated with coronary atherosclerosis also contribute to atherosclerosis of the peripheral arteries; however, the impact of risk predictors is not identical (see Chapter 25 ). Risk factors are listed in Table 43.1 . For example, cigarette smoking has an age- and sex-adjusted odds ratio (OR) of 3.94 for PAD and 1.66 for coronary heart disease. Smoking, type 2 diabetes mellitus (DM), hypertension, and hypercholesterolemia account for about 75% of the risk of developing PAD. Other risk factors include inflammation as measured by C-reactive protein (CRP), chronic kidney disease (CKD), diet, and sedentary lifestyle. Data from several observational studies indicate a twofold to fourfold increase in the prevalence of PAD in current smokers in comparison to never smokers, with smoking cessation associated with better outcomes. In the Women’s Health Study, the hazard ratio for incident symptomatic PAD in smokers of more than 15 cigarettes per day was 17 (95% confidence interval [CI] 11 to 27); the risk decreased following smoking cessation.
| Risk Factor | Odds Ratio (95% CI) |
|---|---|
| Cigarette smoking | 4.46 (2.25–8.84) |
| Diabetes mellitus | 2.71 (1.03–7.12) |
| Hypertension | 1.75 (0.97–3.13) |
| Hypercholesterolemia | 1.68 (1.09–2.57) |
| Hyperhomocysteinemia | 1.92 (0.95–3.88) |
| Chronic kidney disease | 2.00 (1.08–3.70) |
| Insulin resistance | 2.06 (1.10–4.00) |
| C-reactive protein | 2.20 (1.30–3.60) |
Diabetes and metabolic syndrome similarly associate strongly with incident PAD. , Diet also associates with incident PAD. Patients with DM often have extensive and severe PAD and a greater propensity for distal disease and arterial calcification. Involvement of the femoral and popliteal arteries resembles that in nondiabetic persons, but distal disease affecting the tibial and peroneal arteries occurs more frequently. Among patients with PAD, diabetic patients are more likely than nondiabetic patients to have CLI or to undergo an amputation, and microvascular disease may be a compounding problem to conduit artery disease in this population. , In addition, concomitant DM independently associates with increased MACE risk in patients with PAD even after adjusting for other risk factors.
Abnormalities in lipid metabolism associate with PAD. Elevations in total or low-density lipoprotein (LDL) cholesterol increase the risk for PAD and claudication in most studies. , , Hypertriglyceridemia predicts risk for PAD when considered as an independent variable, but its effect diminishes when adjusted for other lipid fractions. , In addition, lipoprotein(a) associates with both incident PAD as well as higher risk of adverse limb outcomes and amputation in patients with PAD.
The pathobiology of PAD involves inflammation, as does atherosclerosis in other tissue beds; however, inflammation may be increased in PAD patients beyond that observed in stable CAD. , High concentrations of fibrinogen associate with an increased risk of developing PAD, most likely a reflection of increased inflammation rather than a procoagulant effect. Levels of leukocyte adhesion molecules and characteristics of leukocyte-platelet aggregates correlate with the development and extent of PAD. Levels of CRP, monocytes, and lipoprotein-associated phospholipase A 2 in peripheral blood are independently associated with PAD, consistent with a role of innate immunity and chronic inflammation in its pathogenesis. , Inflammation provides the mechanistic link between many of the common risk factors for atherosclerosis and the pathophysiologic processes in the arterial wall that lead to PAD (see also Chapter 24 ).
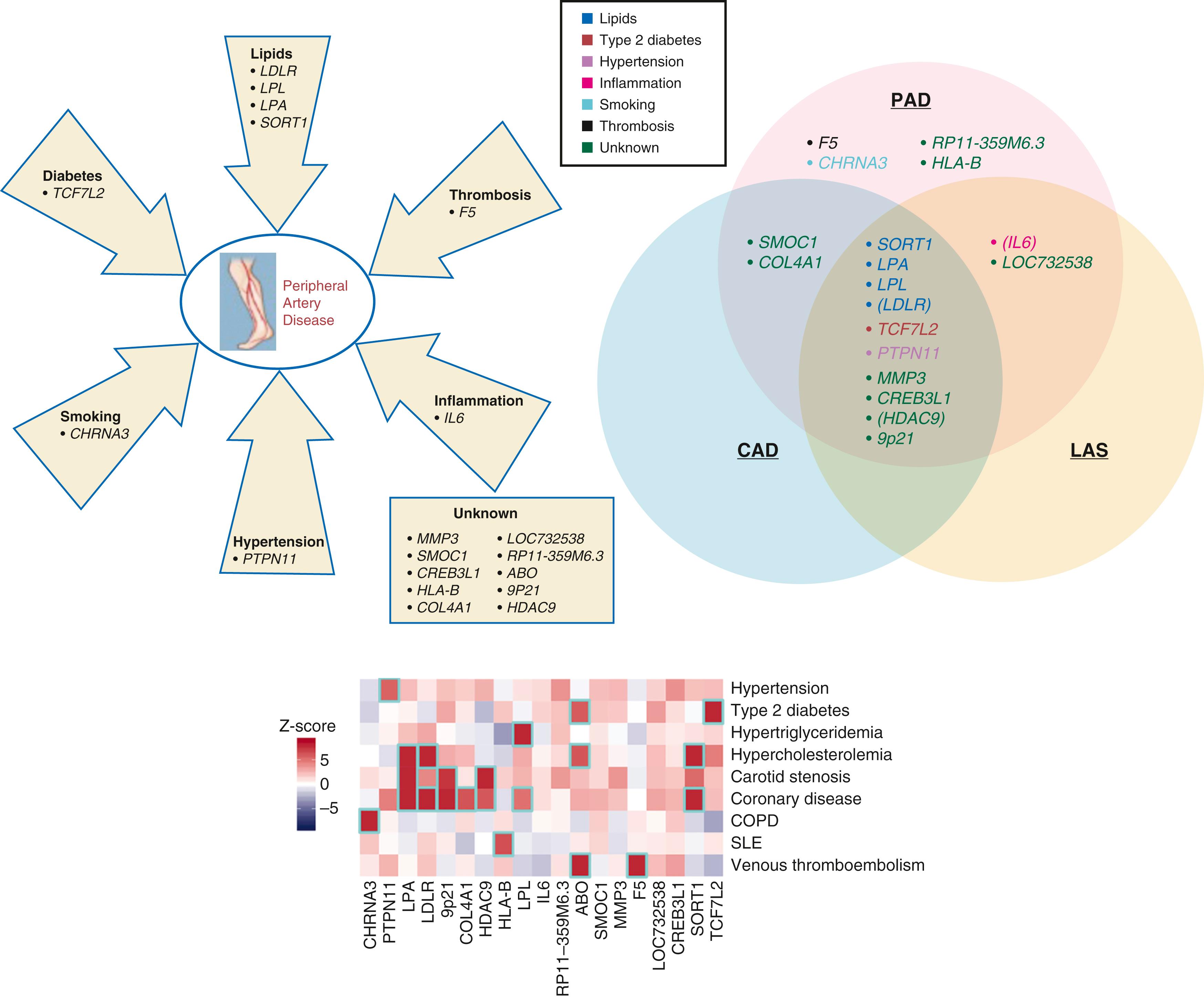
Genetic factors affect both incident PAD and adverse outcomes in PAD. Monogenic diseases relating to lipid metabolism and homocysteine associate with PAD. In addition, genome-wide association studies (GWASs) have identified several loci associated with PAD including 9p21 and DAB2 interaction protein (DAB21P) ( eFig. 43.2 ). A large study involving the Million Veteran Program and replicated in the UK biobank identified 19 autosomal dominant loci including four variants that appeared specific for PAD (relative to coronary or cerebrovascular disease) including F5 p.R506Q, known to be associated with thrombosis risk.
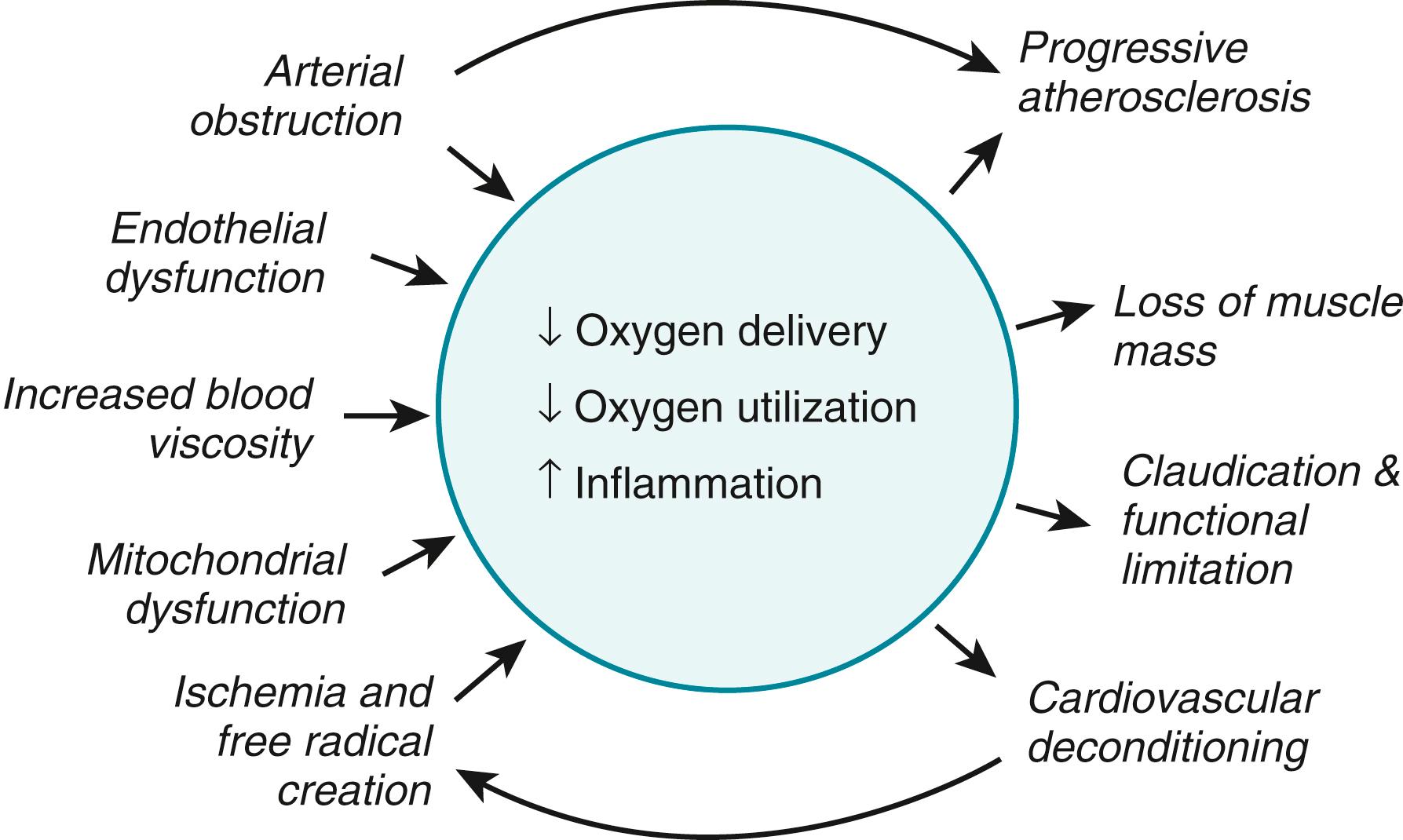
Intermittent claudication results from an oxygen (O 2 ) supply-demand mismatch analogous to angina in patients with stable angina. Impairment in O 2 delivery capacity coupled with dysfunction in O 2 extraction and utilization at the muscular level result in ischemic pain through activation of local sensory receptors by accumulation of lactate or other metabolites ( Fig. 43.1 ). Patients with intermittent claudication may have single or multiple occlusive lesions in the arteries supplying the limb. Blood flow and leg O 2 consumption are normal at rest, but the obstructive lesions limit blood flow and O 2 delivery during exercise such that the metabolic needs of the exercising muscle outstrip the available supply of O 2 and nutrients. Patients with CLI typically have multiple occlusive lesions such that even the resting blood supply cannot meet the nutritional needs of the limb, leading to rest pain and tissue loss. In addition, there is considerable heterogeneity in patterns of occlusive disease including medial artery calcification and a predominance of below knee disease particularly in patients with diabetes and/or CKD. In addition, there is increasing evidence that thrombosis plays a key role not only in the pathophysiology of ALI but also in the development of chronic CLI, suggesting a complex interplay between progressive atherosclerosis and subacute thrombosis leading to worsening ischemia. ,
Flow through an artery is directly related to perfusion pressure and inversely related to vascular resistance. Stenoses reduce flow through the artery ( Fig. 43.2 ), as described in the Poiseuille equation:
where ΔP is the pressure gradient across the stenosis, r is the radius of the residual lumen, η is blood viscosity, and l is the length of the vessel affected by the stenosis. As the severity of a stenotic lesion increases, flow falls progressively. The pressure gradient across the stenosis increases in a nonlinear manner, thus emphasizing the importance of a stenosis at high blood flow rates. Usually, a blood pressure (BP) gradient exists at rest if the stenosis reduces the diameter of the lumen by more than 50% because as distorted flow develops, kinetic energy is lost. A stenosis that does not cause a pressure gradient at rest may cause one during exercise when blood flow increases because of the higher cardiac output and decreased vascular resistance. Consequently, as flow through a stenosis increases, distal perfusion pressure drops. As the metabolic demand of exercising muscle outstrips its blood supply, local metabolites (including adenosine, nitric oxide [NO], potassium [K + ], and hydrogen ion [H + ]) accumulate, and peripheral resistance vessels dilate. Perfusion pressure then drops further because the stenosis limits flow. In addition, intramuscular pressure rises during exercise and may exceed the arterial pressure distal to an occlusion and halt blood flow. Flow through collateral blood vessels can usually meet the resting metabolic needs of skeletal muscle tissue at rest but does not suffice during exercise.
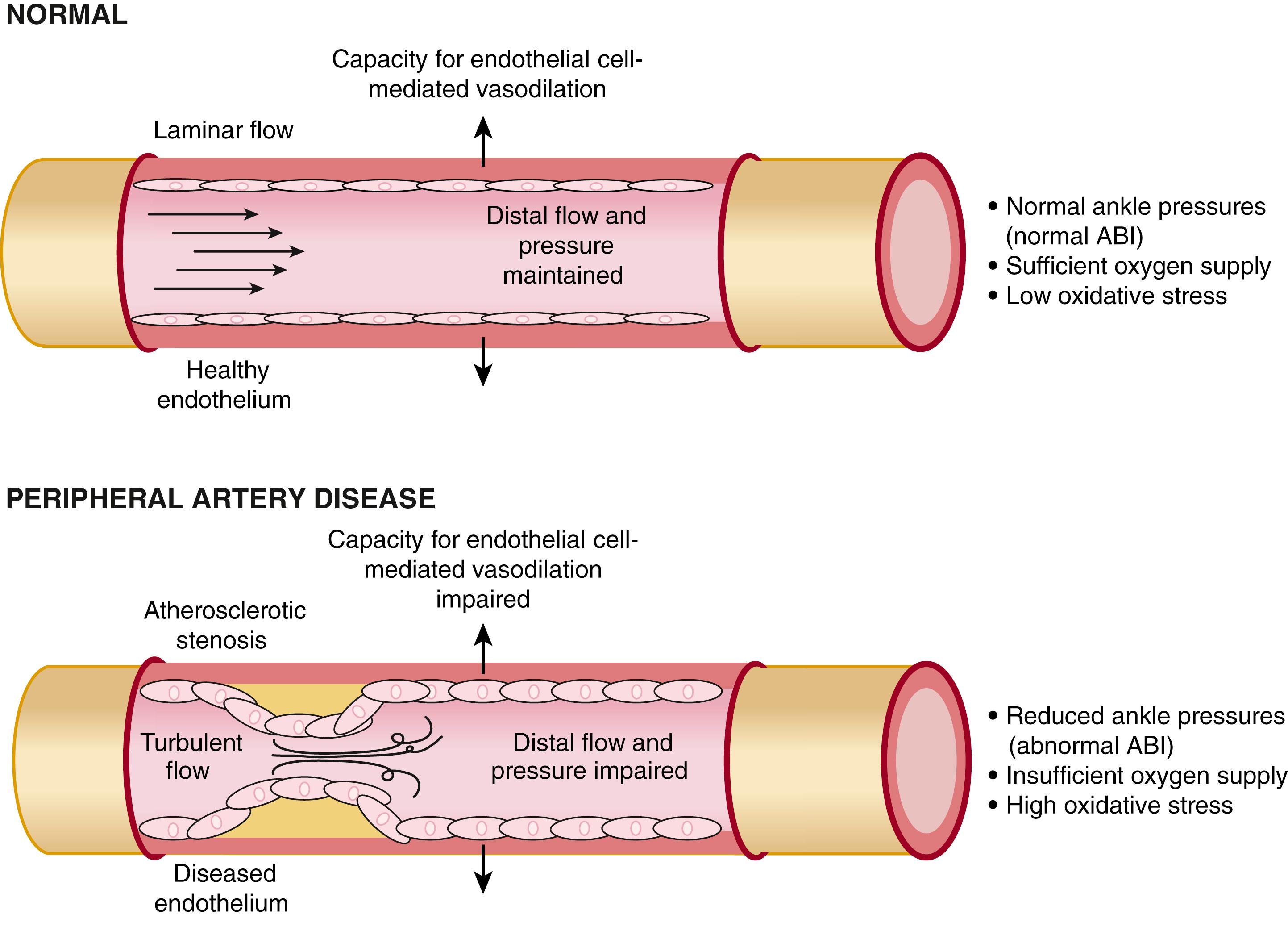
Functional abnormalities in vasomotor reactivity may also interfere with blood flow. Patients with peripheral atherosclerosis have reduced vasodilator capability of both conduit and resistance vessels. Normally, arteries dilate in response to pharmacologic and biochemical stimuli, such as acetylcholine, serotonin, thrombin, and bradykinin, as well as in response to shear stress induced by increases in blood flow. This vasodilator response results from the release of biologically active substances from the endothelium, particularly NO. The relaxation of a conduit vessel that occurs after a flow stimulus, such as that induced by exercise, may facilitate the delivery of blood to exercising muscles in healthy persons. The atherosclerotic femoral arteries and calf resistance vessels of patients with PAD have impaired endothelium-dependent vasodilation in response to flow or pharmacologic stimuli. This failure of vasodilation might prevent an increase in blood supply to exercising muscle because endothelium-derived NO can contribute to hyperemic blood flow after an ischemic stimulus.
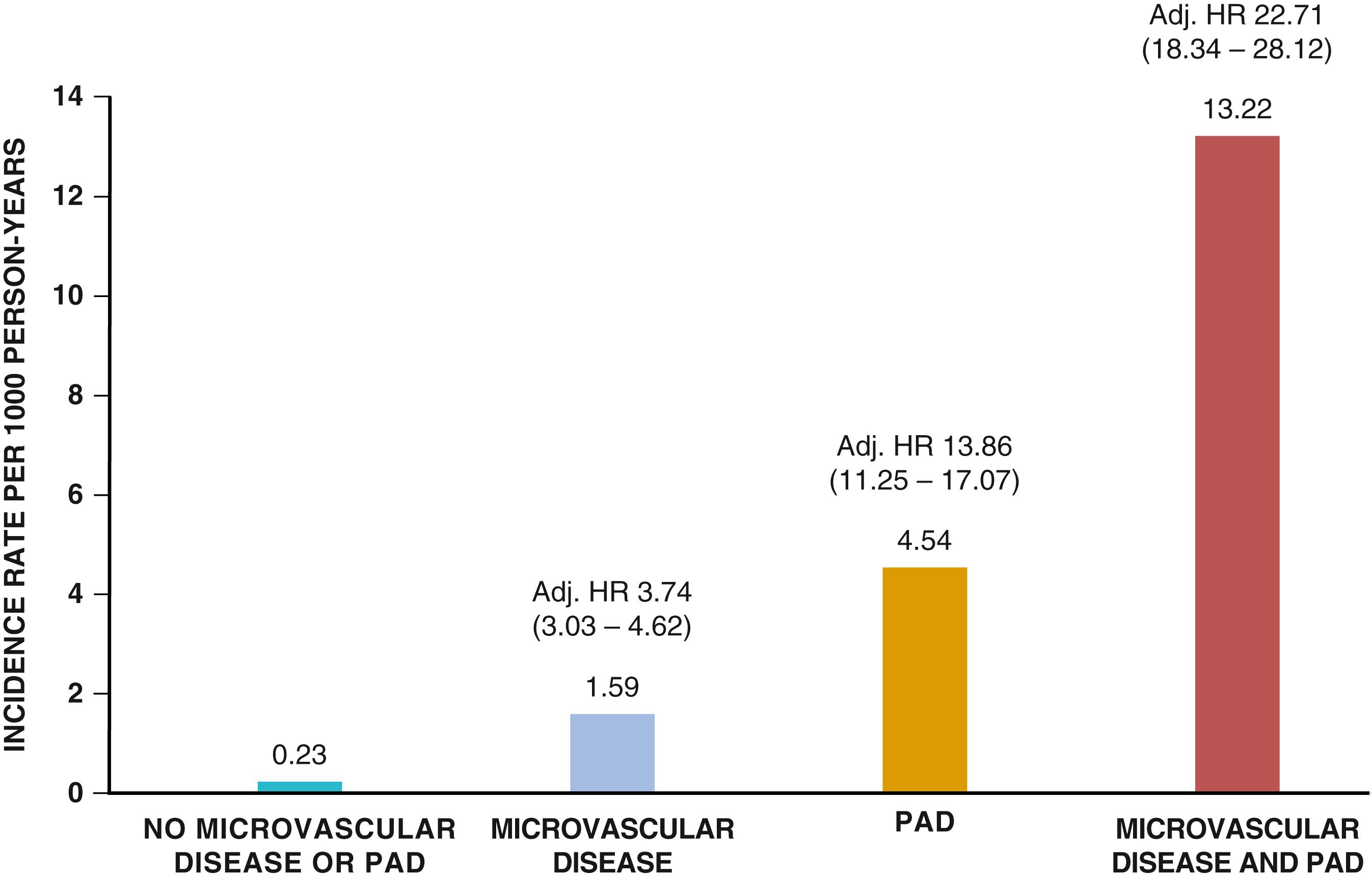
Abnormalities in the microcirculation also contribute to the pathophysiology of CLI. Patients with severe limb ischemia have a reduced number of perfused skin capillaries. Observational studies have observed that microvascular disease associates with amputation independently of conduit artery disease or concomitant diabetes ( eFig. 43.3 ). Other potential causes of decreased capillary perfusion in CLI include reduced red blood cell deformability, increased leukocyte adhesiveness, platelet aggregates, fibrinogen, microvascular thrombosis, excessive vasoconstriction, and interstitial edema. Intravascular pressure may also decrease because of precapillary arteriolar dilation secondary to locally released vasoactive metabolites.
Electrophysiologic and histopathologic examination has found evidence of partial axonal denervation of skeletal muscle in legs affected by PAD. Type I, oxidative slow-twitch fibers are preserved, but type II, glycolytic fast-twitch fibers are lost in the skeletal muscle of patients with PAD. The loss of type II fibers correlates with decreased muscle strength and reduced exercise capacity. In skeletal muscle distal to PAD, the shift to anaerobic metabolism occurs earlier during exercise and persists longer after exercise. Patients with claudication have increased lactate release and accumulation of acylcarnitines during exercise and slowed O 2 desaturation kinetics, indicative of ineffective oxidative metabolism. Moreover, mitochondrial respiratory activity and phosphocreatine and adenosine triphosphate (ATP) recovery time are delayed in the calf muscles of PAD patients, as assessed after submaximal exercise by 31 P magnetic resonance spectroscopy. ,
The American College of Cardiology/American Heart Association (ACC/AHA) Guideline recommendations for clinical assessment and testing are outlined online in Tables 43G.1 to Table 43G.3 and Table 43G.6 . ![]()
The cardinal symptoms of PAD include limb pain either with exercise (intermittent claudication) or at rest. The term claudication is derived from the Latin claudicare , “to limp.” Intermittent claudication refers to a pain, ache, sense of fatigue, or other discomfort that occurs in the affected muscle group with exercise, particularly walking, and resolves with rest. The location of the symptom is often related to the site of the most proximal stenosis. Buttock, hip, or thigh claudication typically occurs in patients with obstruction of the aorta and iliac arteries. Calf claudication is caused by femoral or popliteal artery stenoses. The gastrocnemius muscle consumes more oxygen during walking than other muscle groups in the leg do and thus causes the most frequent symptoms reported by patients. Ankle or foot claudication occurs in patients with tibial and peroneal artery disease. Similarly, stenoses of the subclavian, axillary, or brachial arteries may cause shoulder, biceps, or forearm claudication, respectively. The symptoms should resolve several minutes after cessation of effort. Episodic calf or thigh pain that occurs during rest, such as nocturnal cramps, should not be confused with claudication and are not symptoms of PAD. The history obtained from persons reporting claudication should note the walking distance, speed, and incline that precipitate claudication. Such baseline assessment serves to evaluate disability and provides an initial qualitative measure with which to determine stability, improvement, or deterioration during subsequent encounters with the patient. PAD may result in functional limitations beyond those caused by pain. Patients with PAD walk more slowly and have less walking endurance than do patients without PAD.
Several questionnaires serve to assess the presence and severity of claudication. The Rose Questionnaire was developed initially to diagnose both angina and intermittent claudication in epidemiologic surveys. It queries whether pain develops in either calf with walking and whether the pain occurs at rest, while walking at an ordinary or hurried pace, or on walking uphill. Several modifications of this questionnaire have been developed, including the Edinburgh Claudication Questionnaire and the San Diego Claudication Questionnaire, both of which are more sensitive and specific than a physician’s diagnosis of intermittent claudication based on walking distance, walking speed, and nature of the symptoms. Another validated instrument, the Walking Impairment Questionnaire, asks a series of questions and derives a point score based on walking distance, walking speed, and nature of the symptoms. , ,
Symptoms resembling limb claudication occasionally result from nonatherosclerotic causes of artery obstruction ( Table 43.2 ), including arterial embolism; vasculitides such as thromboangiitis obliterans (TAO), Takayasu arteritis, and giant cell arteritis; aortic coarctation; FMD; irradiation; endofibrosis of the external iliac artery; and extravascular compression as a result of arterial entrapment or an adventitial cyst (see Chapter 97 ). Several nonvascular causes of exertional leg pain should be considered in the differential diagnosis of intermittent claudication. Lumbosacral radiculopathy resulting from degenerative joint disease, spinal stenosis, and herniated discs can cause pain in the buttock, hip, thigh, calf, or foot with walking, often after very short distances or even with standing. This symptom has been called neurogenic pseudoclaudication. Lumbosacral spine disease and PAD both preferentially affect the elderly population and thus may coexist in the same individual. Positional changes in symptoms or attenuation of pain while walking stooped forward, such as with a shopping cart, are suggestive of a neurogenic rather than a vascular cause of symptoms. Arthritis of the hips and knees also provokes leg pain with walking. Typically, the pain is localized to the affected joint and can be elicited on physical examination by palpation and range-of-motion maneuvers. Exertional compartment syndrome most often occurs in athletes with large calf muscles; increased tissue pressure during exercise limits microvascular flow and results in calf pain or tightness. Symptoms improve after cessation of exercise. Rarely, skeletal muscle disorders such as myositis can cause exertional leg pain. Glycogen storage disease type V, also known as McArdle syndrome, in which skeletal muscle phosphorylase is deficient, can cause symptoms mimicking the claudication of PAD. Patients with chronic venous insufficiency sometimes report leg discomfort with exertion, a condition designated venous claudication. Venous hypertension during exercise increases arterial resistance in the affected limb and limits blood flow. In the case of venous insufficiency, elevated extravascular pressure caused by interstitial edema further diminishes capillary perfusion. Peripheral edema, venous stasis pigmentation, and occasionally venous varicosities demonstrated on physical examination may provide clues for this unusual cause of exertional leg pain.
| Vascular Causes |
| Atherosclerosis Thrombosis Embolism Vasculitis Thromboangiitis obliterans Takayasu arteritis Giant cell arteritis Aortic coarctation Fibromuscular dysplasia Irradiation Endofibrosis of the external iliac artery Extravascular compression Arterial entrapment (e.g., popliteal artery entrapment, thoracic outlet syndrome) Adventitial cysts |
| Nonvascular Causes |
| Lumbosacral radiculopathy Degenerative arthritis Spinal stenosis Herniated disc Arthritis Hips, knees Venous insufficiency Myositis Glycogen storage disease type V (McArdle syndrome) |
Symptoms or tissue loss occur at rest in patients with CLI. Typically, patients complain of pain or paresthesias in the foot or toes of the affected extremity. This discomfort worsens with leg elevation and improves with leg dependency, as might be anticipated by the effect of gravity on perfusion pressure. The pain can be particularly severe at sites of skin fissuring, ulceration, or necrosis. Frequently, the skin is very sensitive, and even the weight of bedclothes or sheets elicits pain. Patients may sit on the edge of the bed and dangle their legs to alleviate the discomfort. Patients with neuropathy, however, can experience little or no pain despite the presence of severe ischemia.
CLI and digital ischemia can result from arterial occlusions from a variety of etiologies in addition to atherosclerosis, including conditions such as TAO, vasculitides such as systemic lupus erythematosus (SLE) or scleroderma, vasospasm, atheromatous embolism, and acute arterial occlusion secondary to thrombosis or embolism (see later). Acute gouty arthritis, trauma, and sensory neuropathy such as that caused by DM, lumbosacral radiculopathies, and complex regional pain syndrome (previously known as “reflex sympathetic dystrophy”) can cause foot pain. Leg ulcers also occur in patients with venous insufficiency or sensory neuropathy, particularly that related to diabetes. These ulcers appear to be distinct from those caused by PAD. The ulcer of venous insufficiency usually localizes near the medial malleolus and has an irregular border and a pink base with granulation tissue. In general, they produce milder pain than those caused by PAD. Neurotrophic ulcers occur at sites of pressure or trauma, usually on the sole of the foot. These ulcers are deep, frequently infected, and not generally painful because of the loss of sensation. In patients with PAD, concomitant diabetes associates with increased risk of ulcer and tissue loss, and the etiology is multifactorial including chronic ischemia and infection in the setting of impaired perfusion, microcirculatory disease, and increased susceptibility to infection.
A complete CV examination includes palpation of the peripheral pulses, inspection of the extremities, including the feet, and auscultation of accessible arteries for bruits. Pulse abnormalities and bruits increase the likelihood of PAD. , , Readily palpable pulses in healthy individuals include the brachial, radial, and ulnar arteries in the upper extremities and the femoral, popliteal, dorsalis pedis, and posterior tibial arteries in the lower extremities. The aorta can also be palpated in thin people. A decreased or absent pulse indicates diminished pressure from a more proximal stenosis. For example, a normal right femoral pulse but absent left femoral pulse suggests the presence of left iliofemoral arterial stenosis. A normal femoral artery pulse but absent popliteal artery pulse would indicate a stenosis in the superficial femoral artery or proximal popliteal artery. A palpable popliteal artery pulse with absent dorsalis pedis or posterior tibial artery pulses indicates disease of the anterior and posterior tibial arteries, respectively.
Bruits are often a sign of accelerated blood flow velocity and flow disturbance at sites of stenosis. A stethoscope should be used to auscultate the supraclavicular and infraclavicular fossae for evidence of subclavian artery stenosis; the abdomen, flank, and pelvis for evidence of stenoses in the aorta and its branch vessels; and the inguinal region for evidence of femoral artery stenoses. Pallor can be elicited on the soles of the feet of some patients with PAD by performing a maneuver in which the feet are elevated above the level of the heart and the calf muscles are exercised by repeated dorsiflexion and plantar flexion of the ankle. The legs are then placed in the dependent position, and the time until the onset of hyperemia by evident rubor and venous distention is measured. Each of these variables depends on the rate of blood flow, which in turn reflects the severity of stenosis and adequacy of collateral vessels.
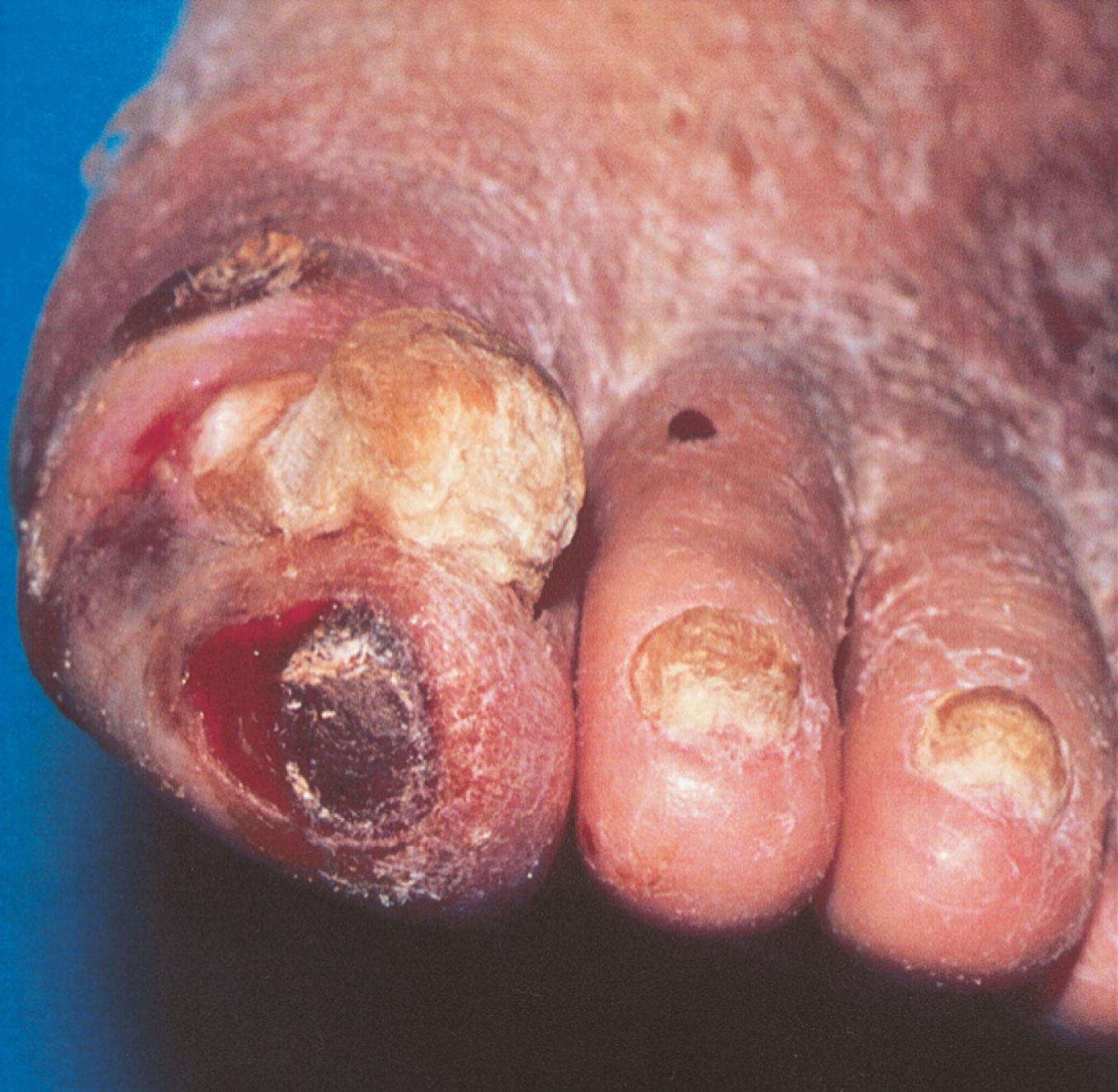
The legs of patients with chronic aortoiliac disease may show muscle atrophy. Additional signs of chronic low-grade ischemia include hair loss, dystrophic, thickened and brittle toenails, smooth and shiny skin, and atrophy of the subcutaneous fat of the digital pads. Patients with severe limb ischemia have cool skin and may also have petechiae, persistent cyanosis or pallor, dependent rubor, pedal edema resulting from prolonged dependency, skin fissures, ulceration, or gangrene. The ulcers caused by PAD typically have a pale base with irregular borders and usually involve the tips of the toes or the heel of the foot or develop at sites of pressure ( Fig. 43.3 ). These ulcers vary in size and may be as small as 3 to 5 mm.
Classification of patients with PAD depends on the severity of the symptoms and abnormalities detected on physical examination. Categorization of the clinical manifestations of PAD helps to characterize risk and provides a basis for the types and intensity of therapeutic intervention. Fontaine described one widely used scheme that classifies patients into one of four stages, progressing from asymptomatic to CLI ( Table 43.3 ), a term first used in 1982. Several professional vascular societies have adopted the Rutherford scale, a contemporary, more descriptive classification that includes asymptomatic patients, three grades of claudication, and three grades of CLI ranging from rest pain alone to minor and major tissue loss ( Table 43.4 ). , ,
| Stage | Symptoms |
|---|---|
| I | Asymptomatic |
| II | Intermittent claudication |
| IIa | Pain free, claudication walking >200 m |
| IIb | Pain free, claudication walking <200 m |
| III | Rest and nocturnal pain |
| IV | Necrosis, gangrene |
| Grade | Category | Clinical Description |
|---|---|---|
| 0 | Asymptomatic | |
| I | 1 | Mild claudication |
| 2 | Moderate claudication | |
| 3 | Severe claudication | |
| II | 4 | Ischemic rest pain |
| 5 | Minor tissue loss: nonhealing ulcer, focal gangrene with diffuse pedal ulcer | |
| III | 6 | Major tissue loss extending above the transmetatarsal level, functional foot no longer salvageable |
The term CLI is also in evolution and it has been modified by the Global Vascular Guidelines writing group to “chronic limb-threatening ischemia” or CLTI. The definition of CLTI is “advanced PAD with rest pain, gangrene, or ulceration of greater than 2 weeks.” These guidelines also endorse the WIFI (Wound, Ischemia, and Foot Infection) classification, which acknowledges and evaluates the multifactorial nature of wounds in patients with PAD including wound characteristics, infection, and ischemia ( Table 43.5–43.7 ). The WIFI classification is associated with outcomes and has been validated in observational cohorts. The pathobiologic complexity implicit in the definition of CLTI and the WIFI classification reflects that tissue loss in PAD is often not driven by acute ischemia but rather the complex interplay between chronic ischemia and infection, particularly in patients with diabetes ( eFig. 43.4 ).
| Grade | Ulcer | Gangrene |
|---|---|---|
| 0 | No ulcer | No gangrene |
| Clinical description: ischemic rest pain (requires typical symptoms + ischemia grade 3); no wound | ||
| 1 | Small, shallow ulcer on distal leg or foot; no exposed bone, unless limited to distal phalanx | No gangrene |
| Clinical description: minor tissue loss. Salvageable with simple digital amputation (1 or 2 digits) or skin coverage | ||
| 2 | Deeper ulcer with exposed bone, joint, or tendon; generally not involving the heel; shallow heel ulcer, without calcaneal involvement | Gangrenous changes limited to digits |
| Clinical description: major tissue loss salvageable with multiple (3 or more) digital amputations or standard transmetatarsal amputation with or without skin coverage | ||
| 3 | Extensive, deep ulcer involving forefoot and/or midfoot; deep, full-thickness heel ulcer ± calcaneal involvement | Extensive gangrene involving forefoot and/or midfoot; full-thickness heel necrosis ± calcaneal involvement |
| Clinical description: extensive tissue loss salvageable only with a complex foot reconstruction (nontraditional transmetatarsal, Chopart, or Lisfranc amputation); flap coverage or complex wound management needed for large soft tissue defect. | ||
| Grade | Ankle Brachial INDEX | Ankle Systolic Pressure | Toe Pressure, TcPo 2 |
|---|---|---|---|
| 0 | ≥ 0.80 | >100 mm Hg | ≥ 60 mm Hg |
| 1 | 0.60–0.79 | 70–100 mm Hg | 40–59 mm Hg |
| 2 | 0.40–0.59 | 50–70 mm Hg | 30–39 mm Hg |
| 3 | ≤ 0.39 | <50 mm Hg | <30 mm Hg |
| Clinical manifestation of infection | SVS Grade | Infection severity |
|---|---|---|
| No symptoms or signs of infection | 0 | Uninfected |
| Infection present but involving the skin and subcutaneous tissue only | 1 | Mild |
| Local infection involving structures deeper than skin and subcutaneous tissues | 2 | Moderate |
| Local infection with signs of systemic inflammatory response syndrome | 3 | Severe |
Patients with signs or symptoms suggestive of PAD should have testing to confirm the diagnosis and to characterize the distribution and severity of disease. In patients with risk factors, clinicians should be aware of atypical symptoms, perform a vascular physical examination, and perform diagnostic testing in those with a history or examination suggestive of PAD. Many patients with PAD are asymptomatic or have unrecognized symptoms, and appropriate testing should be considered in high-risk individuals after careful history taking. , , One randomized trial of men aged 65 to 74 years found that screening for PAD, as well as hypertension and abdominal aortic aneurysm, had a mortality benefit.
Measurement of SBP along sequential segments of each extremity is one of the simplest noninvasive measures for ascertaining the presence and severity of stenoses in the peripheral arteries. In the lower extremities, pneumatic cuffs are placed on the upper and lower portions of the thigh, on the calf, above the ankle, and often over the metatarsal area of the foot. Similarly, for the upper extremities, pneumatic cuffs are placed on the upper part of the arm over the biceps, on the forearm below the elbow, and at the wrist. SBP at each respective limb segment is measured by first inflating the pneumatic cuff to suprasystolic pressure and then determining the pressure at which blood flow occurs during deflation of the cuff. The onset of flow is assessed by placing a Doppler ultrasound flow probe over an artery distal to the cuff. In the lower extremities, it is most convenient to place the Doppler probe on the foot over the posterior tibial artery, because it courses inferior and posterior to the medial malleolus, or over the dorsalis pedis artery on the dorsum of the metatarsal arch. In the upper extremities the Doppler probe can be placed over the brachial artery in the antecubital fossa or over the radial and ulnar arteries at the wrist.
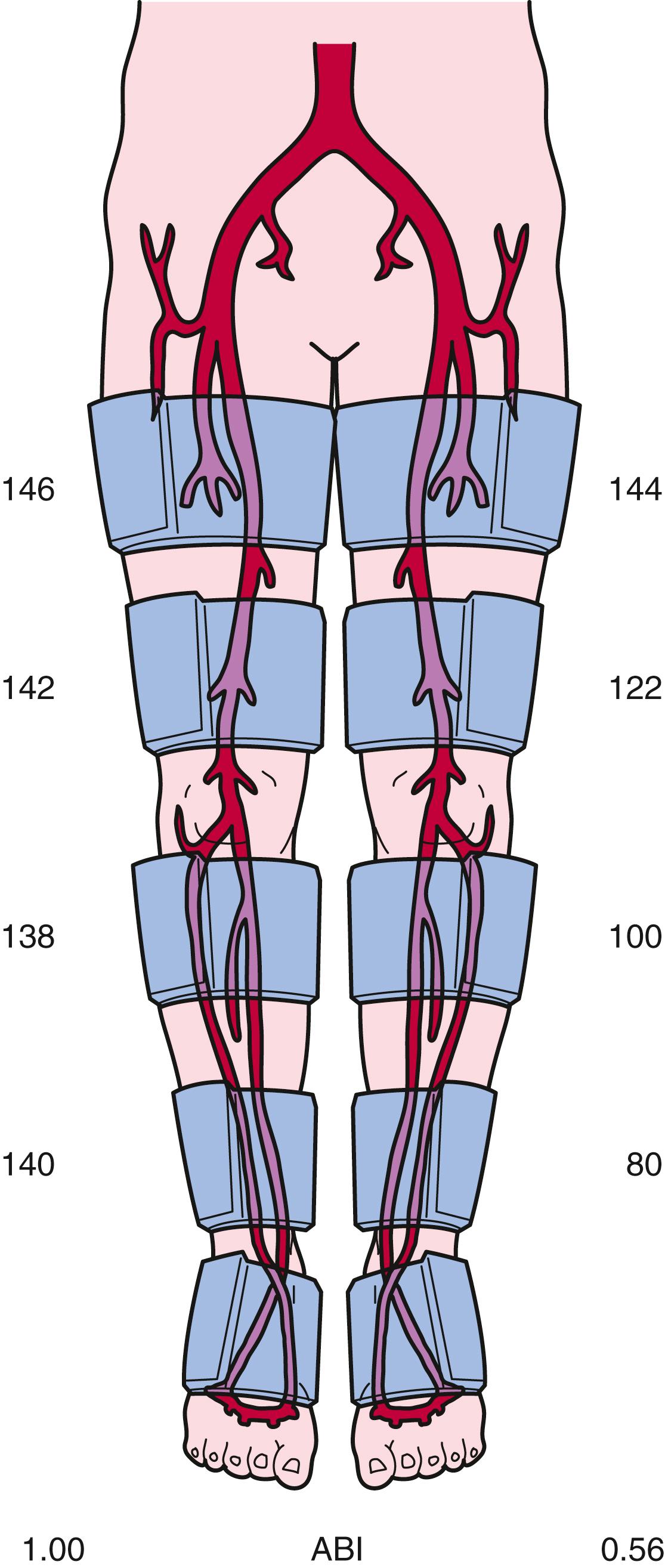
Left ventricular contraction imparts kinetic energy to blood, which is maintained throughout the large- and medium-sized vessels. SBP may be higher in the more distal vessels than in the aorta and proximal vessels because of amplification and reflection of BP waves. A stenosis can cause loss of pressure energy because of increased frictional forces and disturbance of flow at the site of the stenosis. Approximately 90% of the cross-sectional area of the aorta must be narrowed before a pressure gradient develops. In smaller vessels, such as the iliac and femoral arteries, a 70% to 90% decrease in cross-sectional area will cause a resting pressure gradient sufficient to decrease SBP distal to the stenosis. Taking into consideration the precision of this noninvasive method and the variability in BP during even short periods, a BP gradient in excess of 20 mm Hg between successive cuffs is generally used as evidence of arterial stenosis in the lower extremity, whereas a gradient of 10 mm Hg indicates a stenosis between sequential cuffs in the upper extremity. SBP in the toes and fingers is approximately 60% of SBP at the ankle and wrist, respectively, because pressure diminishes further in the smaller distal vessels. Fig. 43.4 provides examples of leg segmental BP measurements in a patient with left calf claudication. In the right leg, there are no pressure gradients between the upper and lower parts of the thigh or between the calf and ankle. In the left leg, pressure gradients between the upper and lower parts of the thigh, between the lower part of the thigh and calf, and between the calf and ankle indicate stenoses in the superficial femoral and popliteal arteries and in the tibioperoneal arteries.
Determination of the ABI is a simplified application of leg segmental BP measurements that can readily be used at the bedside. This index is the ratio of SBP measured at the ankle to SBP measured at the brachial artery. , A pneumatic cuff placed around the ankle is inflated to suprasystolic pressure and subsequently deflated while the onset of flow is detected with a Doppler ultrasound probe placed over the dorsalis pedis and posterior tibial arteries, thus denoting ankle SBP. Brachial artery SBP can be assessed in a routine manner with either a stethoscope to listen for the first Korotkoff sound or a Doppler probe to listen for the onset of flow during cuff deflation. The normal ABI range is 1.00 to 1.40. An ABI value of 0.91 to 0.99 is borderline, and an ABI of 0.90 or less is abnormal. , An ABI of 0.90 or lower has a specificity of 83% to 99% and a sensitivity of 69% to 73% in detecting stenoses greater than 50%. , The sensitivity of an ABI less than 1.0 approaches 100%. The ABI is often used to gauge the severity of PAD. Patients with symptoms of leg claudication often have an ABI ranging from 0.5 to 0.8, and patients with CLI usually have an ABI lower than 0.5. A low ABI is associated with shorter walking distance and lower speed. Less than 40% of patients whose ABI is lower than 0.40 can complete a 6-minute walk. , , In patients with skin ulcerations, an ankle SBP less than 55 mm Hg predicts poor ulcer healing. Leg BP recordings are not reliable in patients with calcified vessels, as might occur in those with DM or renal insufficiency. Inflation of the pneumatic cuff cannot compress the calcified vessel; the Doppler probe consequently indicates continuous blood flow, even when the cuff pressure exceeds 250 mm Hg. An ABI higher than 1.40 indicates a noncompressible artery, and the test is not informative for either confirming or excluding PAD. In this case, a toe-brachial index (TBI) may be informative, with a ratio of 0.70 or higher reflecting normal perfusion pressure. ,
Treadmill exercise testing can be used to evaluate the clinical significance of peripheral artery stenoses and provide objective evidence of the patient’s walking capacity. The claudication onset time is when symptoms of claudication first develop, and the peak walking time occurs when the patient can no longer continue walking because of severe leg discomfort. This standardized and more objective measure of walking capacity supplements the patient’s history and provides a quantitative assessment of the patient’s disability, as well as a metric for monitoring therapeutic interventions. Treadmill exercise protocols use a motorized treadmill that incorporates fixed or progressive speeds and angles of incline. A fixed workload test usually maintains a constant grade of 12% and a speed of 1.5 to 2.0 mph. A progressive, or graded, treadmill protocol typically maintains a constant speed of 2 mph while the grade gradually increases by 2% every 2 to 3 minutes. Repeated treadmill test results have better reproducibility with progressive than with constant-grade protocols.
Treadmill testing can determine whether arterial stenoses contribute to the patient’s symptoms of exertional leg pain. During exercise, blood flow through a stenosis increases as vascular resistance falls in the exercising muscle. According to the Poiseuille equation, described previously, the pressure gradient across the stenosis increases in direct proportion to flow. Thus, ankle and brachial SBP is measured during resting conditions before treadmill exercise, within 1 minute after exercise, and repeatedly until baseline values are reestablished. Normally, the BP increase that occurs during exercise should be the same in both the upper and the lower extremities, with a constant ABI of 1.0 or greater being maintained. In the presence of peripheral artery stenoses, however, the ABI decreases because the BP increase observed in the arm is not matched by a comparable increase in ankle BP. A 25% or greater decrease in the ABI after exercise in a patient whose walking capacity is limited by claudication is considered diagnostic and implicates PAD as a cause of the patient’s symptoms. This provocative test should be considered in patients with risk factors and symptoms suggestive of vascular claudication but with normal resting ABI, as may occur in those with proximal disease. ,
The pulse volume recording graphically illustrates the volumetric change in a segment of the limb that occurs with each pulse. Plethysmographic instruments, typically using strain gauges or pneumatic cuffs, can transduce volumetric changes in the limb, which can be displayed on a graphic recorder. These transducers, placed strategically along the limb, record the pulse volume in its different segments, such as the thigh, calf, ankle, metatarsal region, and toes, or the upper part of the arm, forearm, and fingers. The normal pulse volume contour depends on both local arterial pressure and vascular wall distensibility and resembles a BP waveform. It consists of a sharp systolic upstroke rising rapidly to a peak, a dicrotic notch, and a concave downslope that drops off gradually toward the baseline. The contour of the pulse wave changes distal to a stenosis, with loss of the dicrotic notch, a slower rate of rise, a more rounded peak, and a slower descent. The amplitude becomes lower with increasing severity of disease, and the pulse wave may not be recordable at all in a critically ischemic limb. Segmental analysis of the pulse wave may indicate the location of an arterial stenosis, which probably resides in the artery between a normal and an abnormal pulse volume recording. The pulse volume wave also provides information about the integrity of blood flow when BP measurements cannot be obtained accurately because of noncompressible vessels.
Continuous-wave and pulsed-wave Doppler systems transmit and receive high-frequency ultrasound signals. The Doppler frequency shift caused by moving red blood cells varies directly with the velocity of blood flow. Typically, the perceived frequency shift is between 1 and 20 kHz and is within the audible range of the human ear. Therefore, placement of a Doppler probe along an artery enables the examiner to hear whether blood flow is present and the vessel is patent. Processing and graphic recording of the Doppler signal permit a more detailed analysis of the frequency components.
Doppler instruments can be used without or with gray-scale imaging to evaluate an artery for the presence of stenoses. The Doppler probe is positioned at approximately a 60-degree angle over the common femoral, superficial femoral, popliteal, dorsalis pedis, and posterior tibial arteries. The normal Doppler waveform has three components: a rapid forward-flow component during systole, a transient flow reversal during early diastole, and a slow anterograde component during late diastole. The Doppler waveform becomes altered if the probe is placed distal to an arterial stenosis and is characterized by deceleration of systolic flow, loss of the early diastolic reversal, and diminished peak frequencies. Arteries in a limb with critical ischemia may not show any Doppler frequency shift. As with pulse volume recordings, change from a normal to an abnormal Doppler waveform as the artery is interrogated more distally suggests the location of a stenosis.
Duplex ultrasound imaging provides a direct, noninvasive means of assessing both the anatomic characteristics of peripheral arteries and the functional significance of arterial stenoses. The methodology incorporates gray-scale B-mode ultrasound imaging, pulsed Doppler velocity measurements, and color coding of the Doppler shift information ( Fig. 43.5 ). Real-time ultrasonographic scanners emit and receive high-frequency sound waves, typically ranging from 2 to 10 MHz, to construct an image. The acoustic properties of the vascular wall differ from those of the surrounding tissue, thus enabling them to easily be imaged. Atherosclerotic plaque may be present and visible on gray-scale images. Pulsed-wave Doppler systems emit ultrasound beams at precise times and can therefore sample the reflected ultrasound waves at specific depths to enable the examiner to determine blood cell velocity within the lumen of the artery. By positioning the pulsed Doppler beam at a known angle, the examiner can calculate blood flow velocity according to the following equation:
where Df is the frequency shift, V is the velocity, F is the frequency of the transmitted sound, θ is the angle between the transmitted sound and the velocity vector, and C is the velocity of sound and tissue. For optimal measurements, the angle of the pulsed Doppler beam should be less than 60 degrees. With color Doppler, the frequency shift information within the entire field sampled by the ultrasound beam can be superimposed on the gray-scale image. This approach provides a composite real-time display of flow velocity within the vessel.
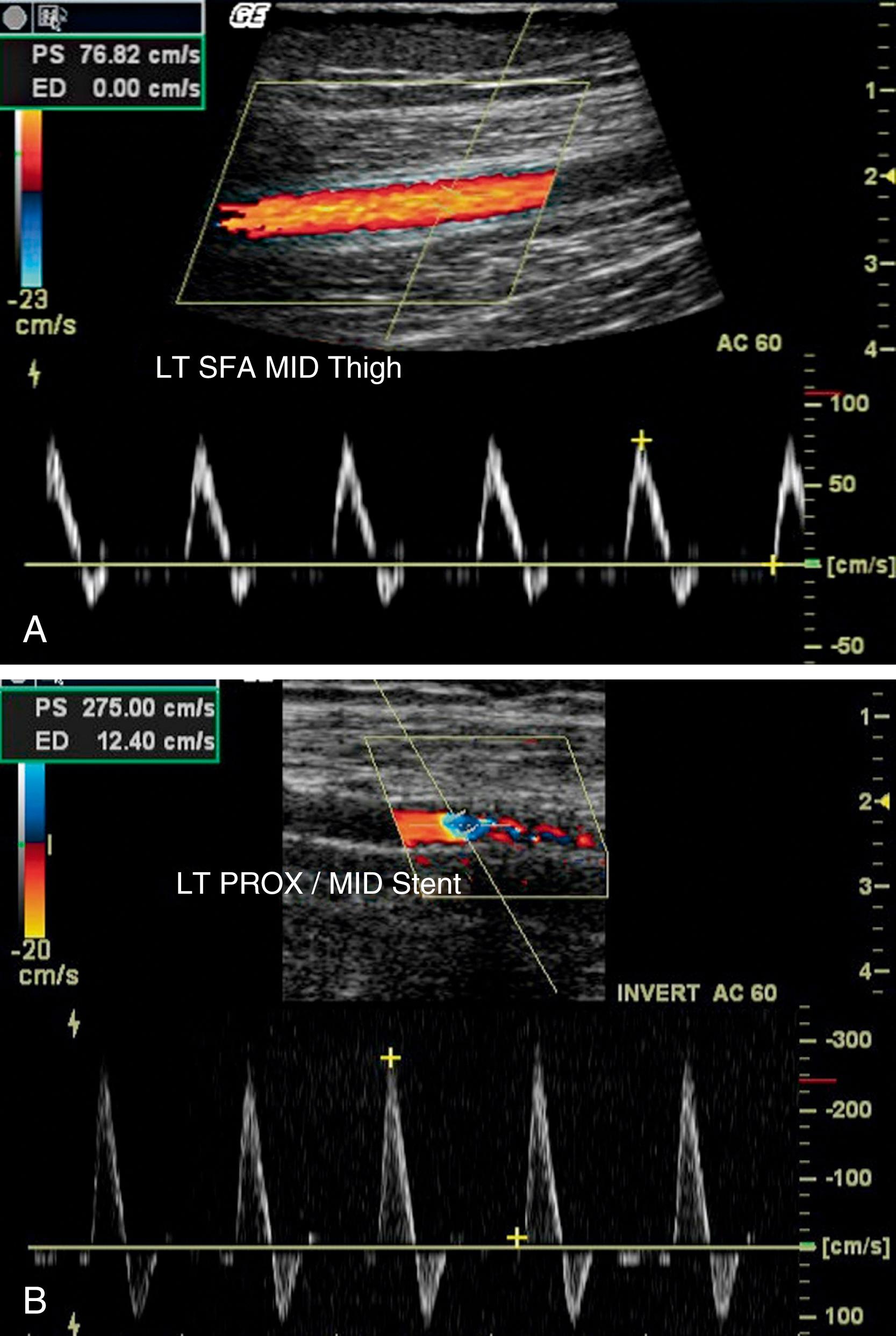
Color-assisted duplex ultrasound imaging is an effective means of localizing peripheral arterial stenoses ( Fig. 43.6 ). Normal arteries have laminar flow, with the highest velocity occurring at the center of the artery. The corresponding color image is usually homogeneous with relatively constant hue and intensity. In the presence of an arterial stenosis, blood flow velocity increases through the narrowed lumen. As the velocity increases, there is progressive desaturation of the color display, and flow disturbance distal to the stenosis causes changes in hue and color. Pulsed Doppler velocity measurements can be made along the length of the artery and particularly at areas of flow abnormalities suggested by the color images. A twofold or greater increase in peak systolic velocity at the site of an atherosclerotic plaque indicates a 50% or greater stenosis (see Fig. 43.6 ). A threefold increase in velocity suggests a 75% or greater stenosis. An occluded artery generates no Doppler signal. Duplex ultrasound imaging for identification of sites of arterial stenosis has approximately 89% to 99% specificity and 80% to 98% sensitivity. , Measurement of sequential peak systolic velocities enables evaluation of restenosis of peripheral stents or bypass grafts and to determine the need to consider reintervention to preserve vessel patency. Assessment of peak systolic velocities postendovascular intervention also may be used to determine hemodynamic improvement.
Magnetic resonance angiography (MRA) can visualize the aorta and peripheral arteries noninvasively. Resolution of the vascular anatomy with gadolinium-enhanced MRA approaches that of conventional contrast-enhanced digital subtraction angiography (DSA). Comparison of MRA with intra-arterial DSA found a sensitivity of 95% and a specificity of 96% for detecting segmental stenotic and occlusive lesions. , MRA currently has its greatest usefulness in the evaluation of symptomatic patients to assist in decision making before endovascular and surgical intervention or in patients at risk for renal, allergic, or other complications during conventional angiography.
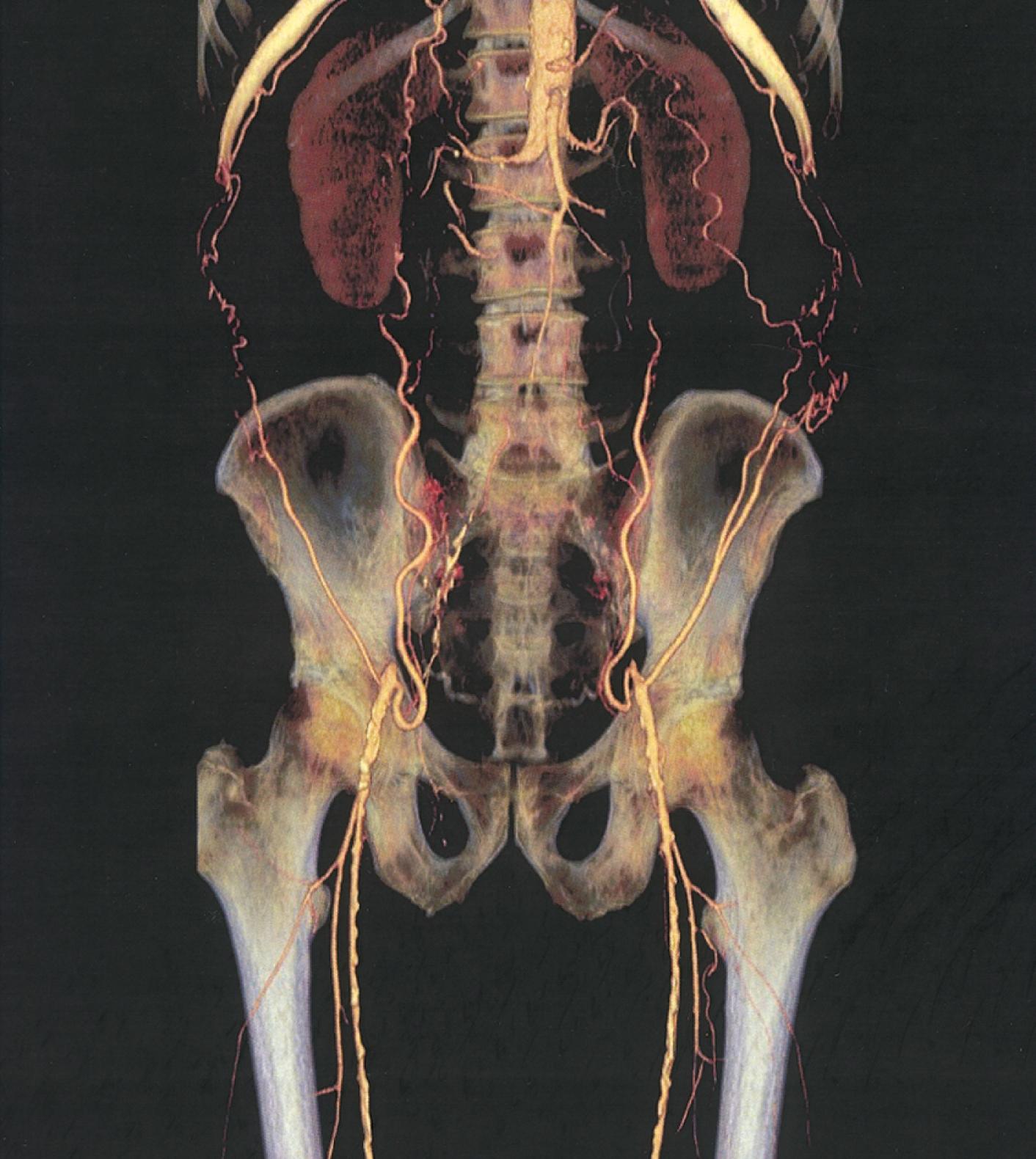
Computed tomography (CT) permits imaging of peripheral arteries with excellent spatial resolution during a relatively short time and with limited amounts of radiocontrast material ( Fig. 43.7 ). Image reconstructions in three dimensions permit rotation to optimize visualization of arterial stenoses. Compared with conventional contrast-enhanced angiography, the sensitivity and specificity for stenoses greater than 50% or occlusion reported for computed tomographic angiography (CTA) using multidetector technology are 95% and 96%, respectively. , CTA offers advantages over MRA because it can be used in patients with stents, metal clips, and pacemakers, although it has the disadvantage of requiring radiocontrast material and ionizing radiation.
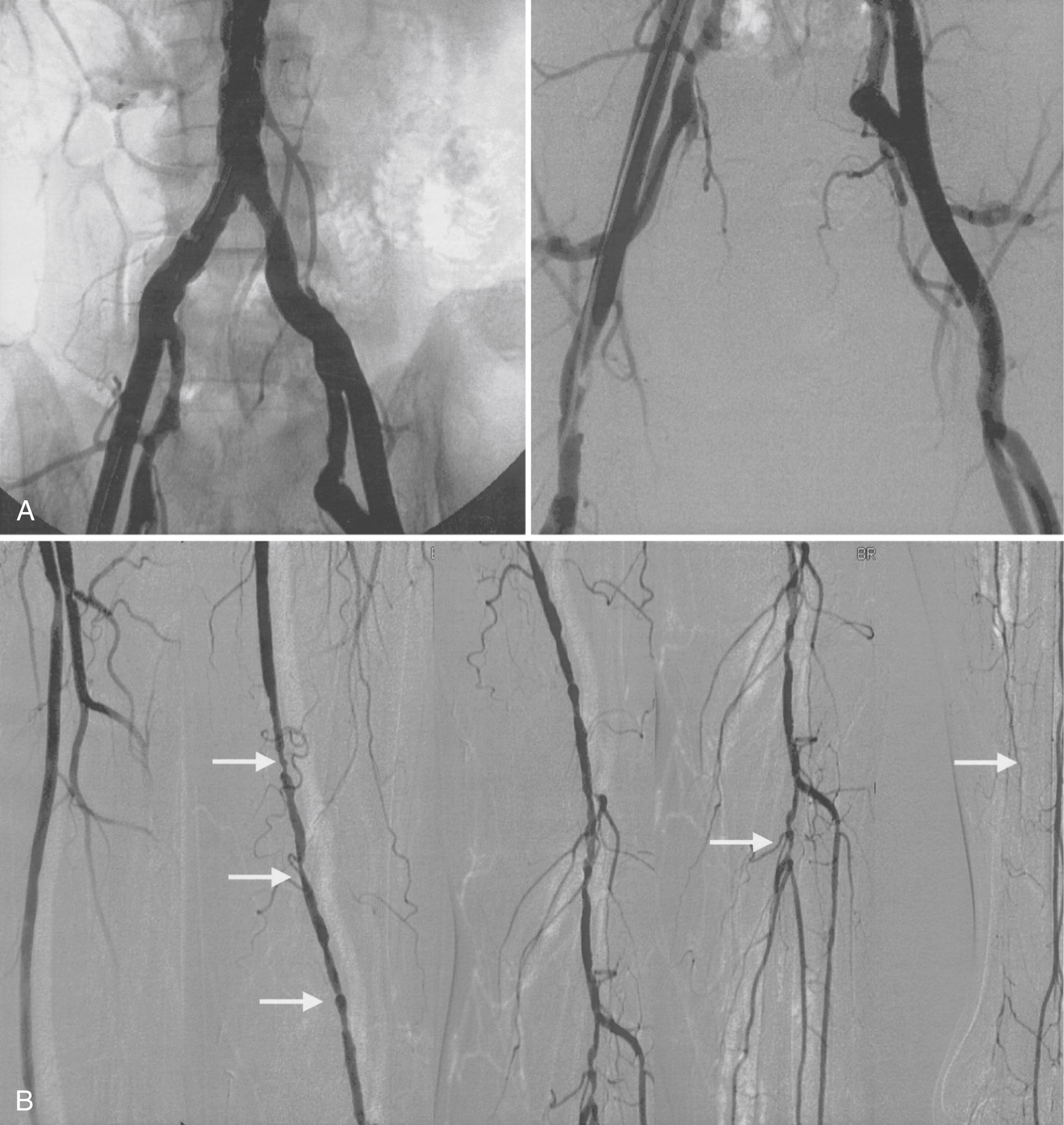
Conventional angiography can aid in evaluation of the arterial anatomy before a revascularization procedure. It still has occasional usefulness when the diagnosis is in doubt. Most contemporary angiography laboratories use digital subtraction techniques after intra-arterial administration of contrast material to enhance resolution. Injection of the contrast material into the aorta permits visualization of the aorta and iliac arteries, and injection of contrast material into the iliofemoral segment of the involved leg permits optimal visualization of the femoral, popliteal, tibial, and peroneal arteries ( Fig. 43.8 ).
Other measures of flow and perfusion are available to assess flow and perfusion in PAD. One such technology is near-infrared fluorescence imaging (NIR), used also for imaging retinal vessels, which uses near-infrared laser light to produce fluorescence of an intravenously injected dye. Detection and measurement of this fluorescence evaluates perfusion. Meta-analyses report a sensitivity of 67% to 100% and specificity of 72% to 100% for diagnosing PAD. Indications for using NIR include diagnosis, monitoring in revascularization, assessment for potential amputation, and visualization of perfusion. NIR may be helpful in selected circumstances and is technique used in research. Transcutaneous oxygen pressure measurement (TcpO 2 ), contrast-enhanced ultrasound (CEUS), and scintigraphy has utility primarily in research, particularly in patients with PAD and diabetes. The use of TcpO 2 has been adopted particularly in patients with CLI to assess the likelihood of healing after amputation, particularly where toe wounds or forefoot amputation may prohibit toe pressure measurement. A normal TcPO 2 is above 55 mm Hg with levels below 30 mm Hg associated with low probability of wound healing. Studies have reported a sensitivity of 98% and a specificity of 44% for predicting resolution of limb salvage problems. Venous occlusion plethysmography (VOP) has been around for more than 100 years and can quantify limb blood flow and hemodynamic compromise in PAD. In addition, data are emerging supporting positron emission tomography (PET) both to understand plaque characteristics as well as perfusion and muscle function.
Patients with PAD have, by definition, a heightened risk of severe ischemic limb complications as well as loss of function. In addition, by nature of their underlying atherosclerotic disease, which frequently affects multiple territories (called polyvascular disease), they have risk for adverse CV events with rates as high or higher than patients with CAD and no PAD ( Fig. 43.9 ). Overall, PAD associates with high rates of CV complications and mortality, with a strong inverse relationship between ABI (excluding noncompressibility) and mortality for both men and women. The specificity of an abnormal ABI in predicting future CV events is approximately 90%.
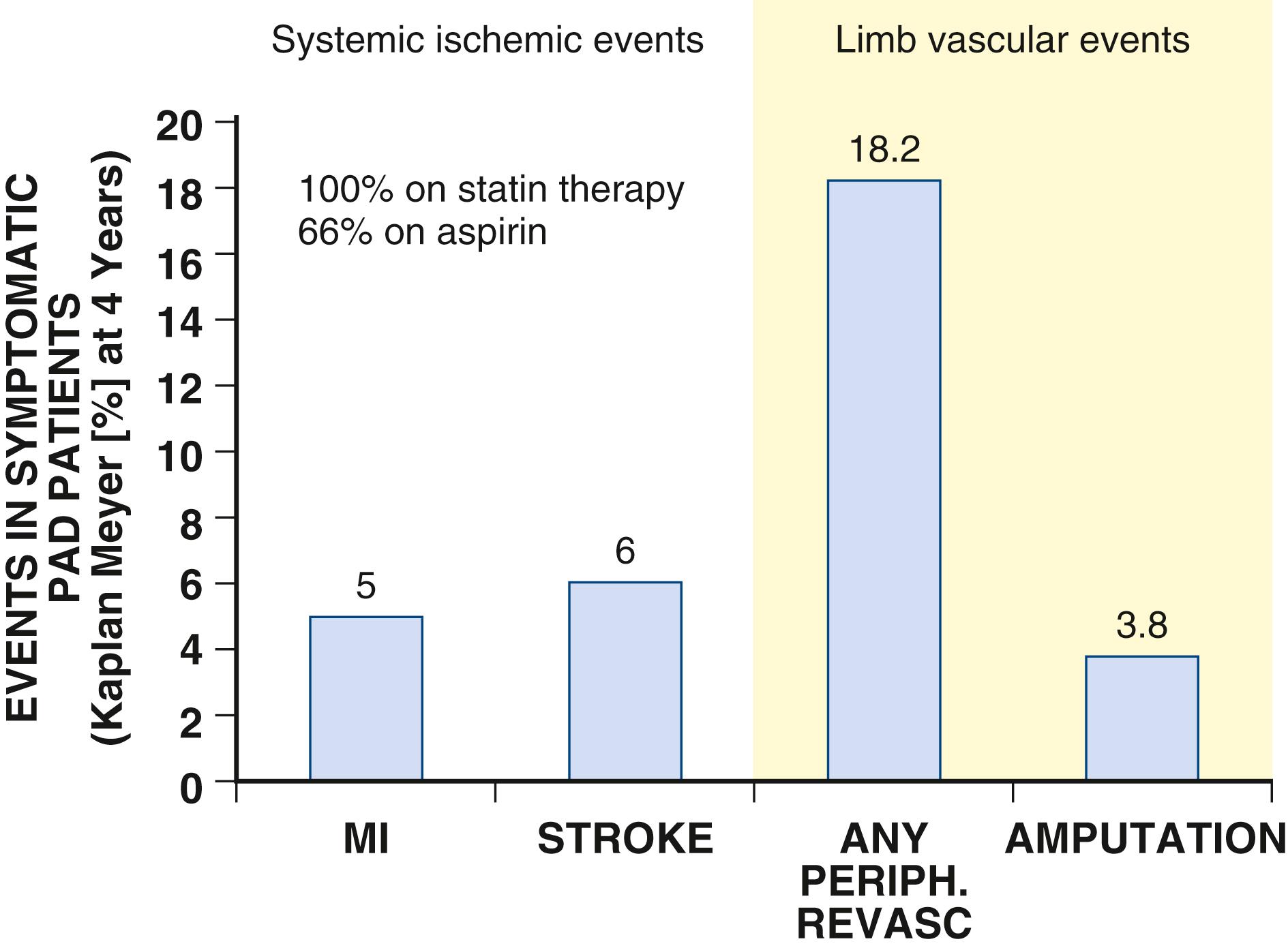
Worsening symptoms develop in approximately 25% of patients with claudication, and over 3 years, approximately 20% will require an intervention to improve lower extremity perfusion (see Fig. 43.9 ). Moreover, loss of mobility occurs more often in patients with PAD than in those without PAD, even in patients without classic symptoms of claudication. Both smoking and DM independently predict progression of disease. , Those with DM have at least a 12-fold higher likelihood of amputation than nondiabetic persons. The risk for limb loss is higher in PAD patients with CLI in whom revascularization fails or is not feasible and approximates 40% by 6 months.
Due to complex comorbidities and advanced age of many patients with PAD, causes of death are as likely to be non-CV as they are CV. Patients with PAD have heterogeneous risk profiles. Those patients with multiple symptomatic territories, or polyvascular disease, particularly involving the coronary bed have a very high risk of MACEs even beyond those with CAD alone. , , Patients with PAD and prior myocardial infarction have a particularly poor CV prognosis, with a 3-year risk of CV death, myocardial infarction, or stroke approaching 20%, reflecting a 60% increase in risk relative to those with prior myocardial infarction but no PAD ( eFig. 43.5A and B ). , Given the systemic manifestations of atherosclerosis, many patients with PAD have coronary atherosclerosis; however, in recent clinical trials of patients with PAD only a third have symptomatic CAD. ,
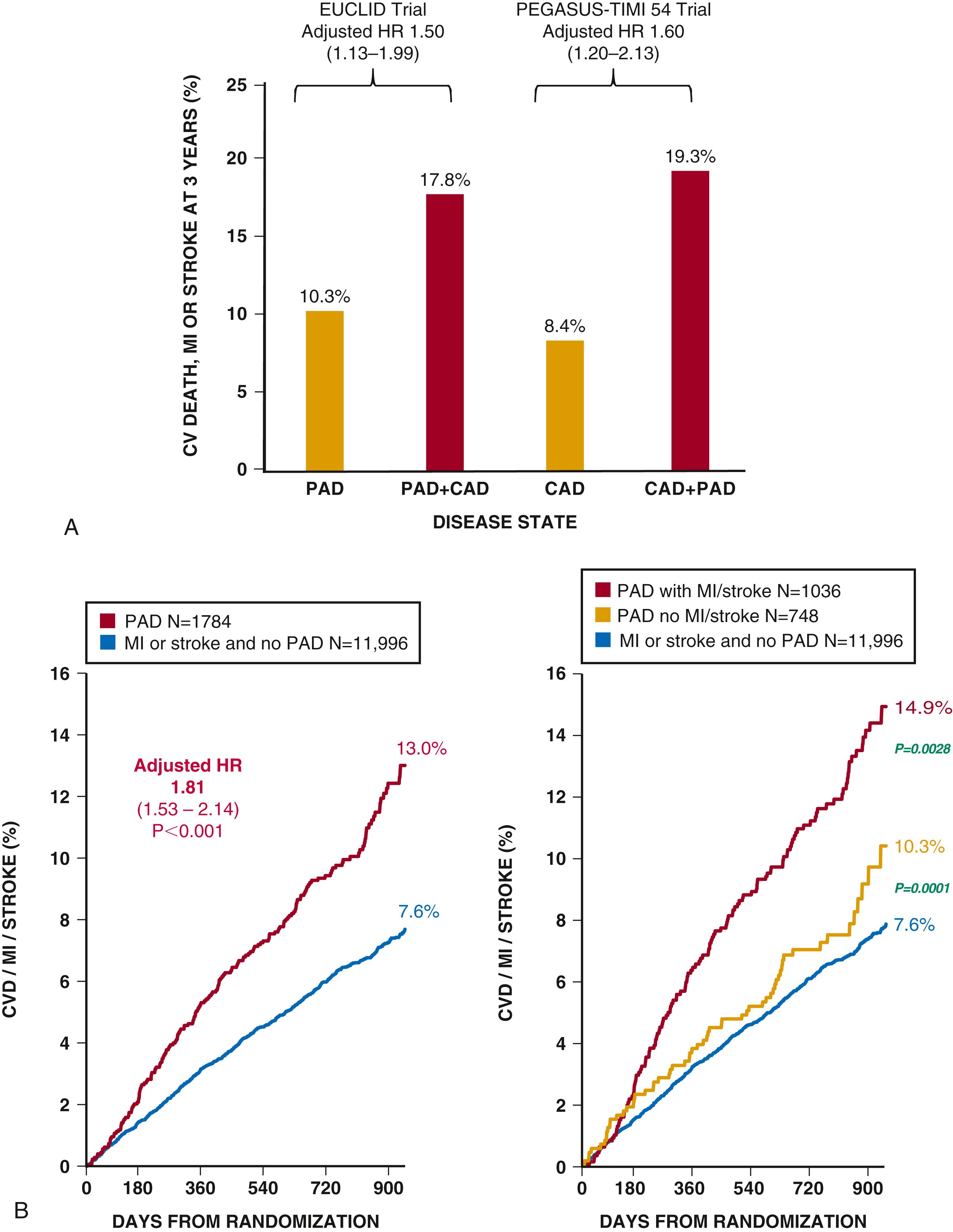
Patients with PAD who have had severe enough symptoms to necessitate revascularization appear to be at particularly high risk of future limb complications such as ALI, even long-term after intervention. , , , Those with the most severe symptoms of PAD, notably those with CLI, have the worst prognosis with high rates of amputation and mortality. Patients with PAD and concomitant DM have increasing rates of amputation in spite of lower rates of myocardial infarction and stroke over the same period. , , It is also increasingly recognized that the etiologies of amputation in those with PAD and DM are often multifactorial, including infection and ischemia.
Prognosis in PAD is heterogenous based on geography, socioeconomic factors and race. Black patients with PAD face higher amputation rates. In addition, PAD incidence and hospitalizations are associated with socioeconomic status. Disparities in socioeconomic factors and health care delivery are important factors in the prognosis of patients with PAD.
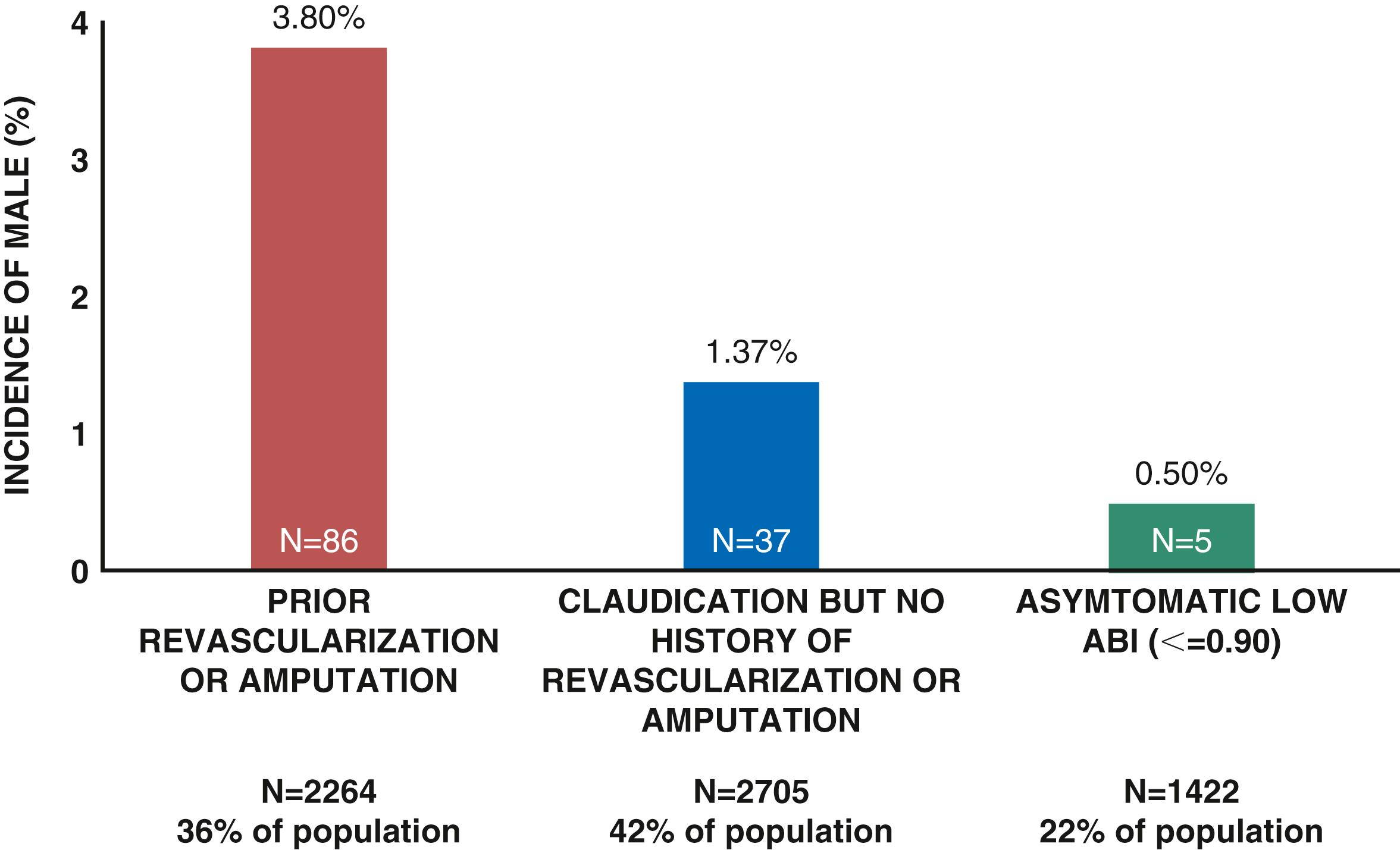
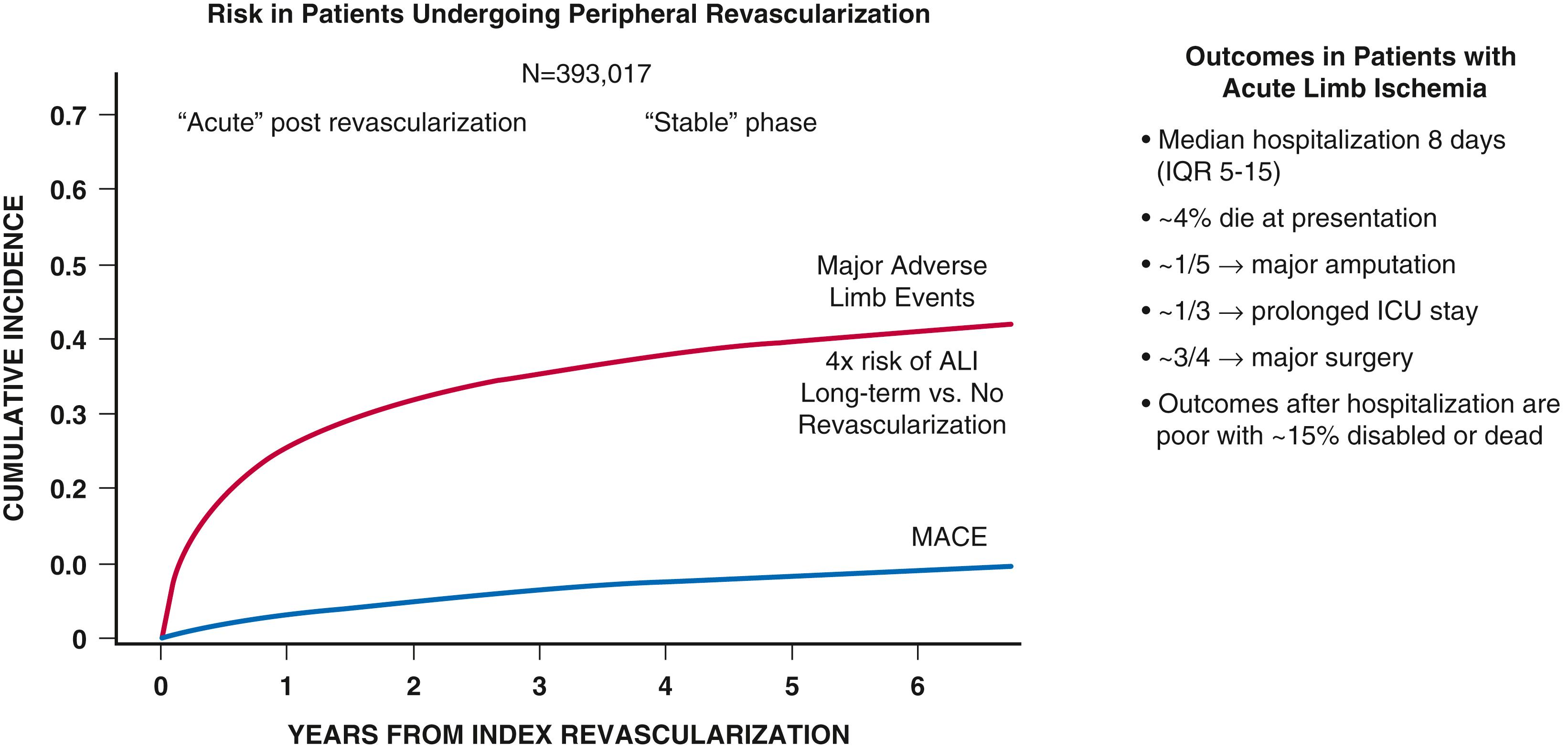
![]() The ACC/AHA Guidelines for Medical Management of PAD are summarized in online Table 43G.1 to Table 43G.8 . The goals of treatment in PAD are to improve quality of life and function as well as to reduce the risk of ischemic limb and CV complications. Therapeutic considerations therefore include modification of risk factors by alterations in lifestyle and use of pharmacologic therapy to reduce the risk for adverse limb outcomes as well as CV events, such as myocardial infarction, stroke, and death, There has been increasing appreciation of the frequency and morbidity of major adverse limb outcomes (MALEs) including ALI and major amputation, as well as the role of medical therapies in reducing these risks. Increasing data suggest heterogeneity in the risk of MALEs in patients with PAD with the greatest risk in those with prior LER or amputation ( eFig. 43.6 ). , , , , This risk appears particularly high in the acute postrevascularization setting ( eFig. 43.7 ). , Unfortunately, secondary prevention in PAD is underused, particularly when compared with other manifestations of atherosclerosis such as CAD. Screening programs demonstrating improved outcomes may in part result in greater use of preventive strategies through increased diagnosis, education, and awareness.
The ACC/AHA Guidelines for Medical Management of PAD are summarized in online Table 43G.1 to Table 43G.8 . The goals of treatment in PAD are to improve quality of life and function as well as to reduce the risk of ischemic limb and CV complications. Therapeutic considerations therefore include modification of risk factors by alterations in lifestyle and use of pharmacologic therapy to reduce the risk for adverse limb outcomes as well as CV events, such as myocardial infarction, stroke, and death, There has been increasing appreciation of the frequency and morbidity of major adverse limb outcomes (MALEs) including ALI and major amputation, as well as the role of medical therapies in reducing these risks. Increasing data suggest heterogeneity in the risk of MALEs in patients with PAD with the greatest risk in those with prior LER or amputation ( eFig. 43.6 ). , , , , This risk appears particularly high in the acute postrevascularization setting ( eFig. 43.7 ). , Unfortunately, secondary prevention in PAD is underused, particularly when compared with other manifestations of atherosclerosis such as CAD. Screening programs demonstrating improved outcomes may in part result in greater use of preventive strategies through increased diagnosis, education, and awareness.
| Cor | Indication | Loe | |
|---|---|---|---|
| I |
|
B-NR | |
|
B-NR | ||
|
B-NR | ||
| Cor | Indication | Loe |
|---|---|---|
| I |
|
C-EO |
|
C-EO | |
| IIa |
|
B-R |
|
C-LD | |
| IIb | The effectiveness of duplex ultrasound for routine surveillance of infrainguinal prosthetic bypass grafts in patients with PAD is uncertain. | B-R |
| COR | Indication | LOE |
|---|---|---|
| I |
|
B-NR |
|
C-LD | |
|
B-NR | |
|
B-NR | |
|
B-NR | |
|
C-EO | |
| IIa |
|
B-NR |
|
B-NR | |
|
B-NR | |
|
B-NR | |
|
C-EO | |
|
B-NR | |
| III |
|
B-NR |
|
B-R |
| Cor | Indication | Loe |
|---|---|---|
| I |
|
A |
|
A | |
|
A | |
|
A | |
|
A | |
|
B-NR | |
|
C-EO | |
|
A | |
|
C-EO | |
| IIa |
|
C-EO |
|
A | |
|
B-NR | |
| IIb |
|
B-R |
|
B-R | |
|
C-LD | |
|
B-R | |
|
B-R | |
| III |
|
A |
|
B-R | |
|
B-R | |
|
B-R |
| Cor | Indication | Loe |
|---|---|---|
| I |
|
A |
|
B-R | |
| IIa |
|
A |
|
A |
| Cor | Indication | Loe |
|---|---|---|
| I |
|
C-LD |
|
C-LD | |
| IIa |
|
C-LD |
|
C-EO | |
|
C-EO |
| COR | Indication | LOE |
|---|---|---|
| I |
|
B-NR |
|
C-EO | |
|
B-R | |
|
C-LD | |
|
A | |
|
A | |
|
A | |
| IIa |
|
A |
|
B-R | |
|
B-NR | |
|
C-LD | |
|
B-R | |
|
B-NR | |
|
C-LD | |
| IIb |
|
C-LD |
|
B-NR | |
| III |
|
B-NR |
|
B-NR | |
|
B-R |
| Cor | Indication | Loe |
|---|---|---|
| I |
|
B-NR |
|
C-LD | |
| IIb |
|
B-NR |
|
C-LD | |
| III |
|
B-R |
Symptoms of claudication improve with exercise and selected pharmacotherapies. Optimal management of CLI often includes endovascular interventions or surgical reconstruction to improve blood supply and maintain limb viability. Revascularization can aid some patients with disabling symptoms of claudication that persist despite exercise therapy and pharmacotherapy. ,
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here