Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Myelinated brain white matter, as in adults, appears hyperintense relative to gray matter on T1-weighted MR images and hypointense on T2-weighted images. In nonmyelinated brain, this pattern is reversed. Myelinization of the infant brain occurs in a predictable pattern, beginning in the brainstem and cerebellum and progressing to the posterior limb of the internal capsule, optic pathways, and parietal lobes. This pattern of change occurs from caudal to cephalad, dorsal to ventral, and central to peripheral. Myelinization should appear complete on MRI by 24 months but may be incomplete in the terminal zones up to approximately 4 to 5 years of age and in rare cases until the first decade of life.
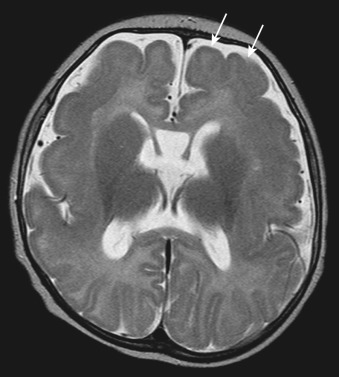
Normal neuronal migration occurs during fetal brain development as neurons migrate from the germinal matrix to the cortex along radial glial fibers. Partial or total arrest of this process leads to a migrational anomaly. These anomalies include the following:
Focal cortical dysplasia (subtle subcortical high T2-weighted signal intensity and blurring of the gray-white matter junction).
Heterotopias (abnormal gray matter found anywhere along glial migration pathways to the cortex).
Schizencephaly (a cleft lined by gray matter extending from the outer cortex to the [lateral] ventricles).
Polymicrogyria (too many small nodular gyri).
Pachygyria (abnormally thickened gyri).
Lissencephaly (complete absence of gyri).
Children with CNS migrational anomalies typically present with developmental delay or seizures ( Figure 89-1 ).
Holoprosencephaly refers to incomplete differentiation of the fetal prosencephalon (forebrain) into separate hemispheres. Holoprosencephaly may manifest as alobar , resulting in one posteriorly located monoventricle (versus the normally separated two lateral ventricles) and fusion of the thalami; semilobar , with two hemispheres posteriorly but not anteriorly; or lobar, with only mild frontal abnormalities. The middle interhemispheric variant has been more recently described and consists of abnormal midline hemispheric connection in the posterior frontal and parietal lobes. The mildest form along the continuum is thought to include septo-optic dysplasia, which is characterized by absence of the septum pellucidum and optic nerve hypoplasia.
The appearance of massively dilated ventricles may be secondary to holoprosencephaly, hydranencephaly, or hydrocephalus. Hydranencephaly results from massive necrosis of the cerebral hemispheres and may be secondary to vascular disease with bilateral internal carotid occlusion, infection, or trauma. In contrast to hydranencephaly, there is a thin residual remnant of cerebral cortex applied to the inner table of the calvaria seen with severe hydrocephalus. The most common causes of prenatal hydrocephalus include Arnold-Chiari malformation II, intracranial hemorrhage, aqueductal stenosis, and Dandy-Walker malformation.
The germinal matrix lies in the caudothalamic groove. This is a highly vascular region of the prenatal and premature brain and is prone to hemorrhage, with rupture of the thin venules subsequent to decreased perfusion or oxygenation. Morbidity and mortality may be predicted based on the grade of the hemorrhage:
Grade I hemorrhage is confined to the germinal matrix.
Grade II hemorrhage extends into the adjacent lateral ventricle.
Grade III hemorrhage expands into and dilates the adjacent lateral ventricle.
Grade IV hemorrhage is in the adjacent parenchyma, probably due to hemorrhagic venous infarction.
US of the premature infant's head through the anterior fontanelle is the imaging modality of choice for the diagnosis and follow-up of germinal matrix hemorrhage.
Periventricular leukomalacia (PVL) is characterized by ischemia in an end arterial distribution (in the watershed regions of white matter that surround the ventricles). US may demonstrate an increase in periventricular echogenicity soon after the initial insult. Cystic change in these regions can be seen later in the subacute stage, as the white matter begins to be resorbed. MRI demonstrates abnormal periventricular signal intensity and volume loss. Before 28 weeks of gestation, the developing brain does not display a leukomalacic response, and only volume loss results. This volume loss may take the form of dilated ventricles and expanded extra-axial spaces or porencephalic cysts (expanded cystlike dilations from the lateral ventricles that may reach the cortex). A porencephalic cyst can be differentiated from schizencephaly by noting that, in the former entity, there is no gray matter lining the cyst cavity.
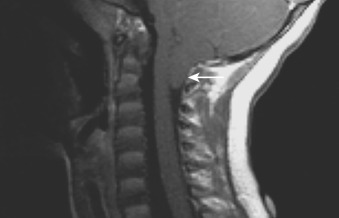
Chiari I malformation involves herniation of the peg-like cerebellar tonsils into the foramen magnum (>5 mm in patients <15 years old) ( Figure 89-2 ), and is associated with syringomyelia.
Chiari II malformation is more severe, involving herniation of the medulla and vermis and elongation and downward displacement of the brainstem. This malformation is virtually always associated with a meningomyelocele. Prenatal US may show crowding of the posterior fossa (“banana” sign appearance of the cerebellar hemispheres as they wrap around the brainstem). There may also be kinking of the medulla, agenesis of the corpus callosum, polygyria, hydrocephalus, and colpocephaly (disproportional enlargement of the posterior horns of the lateral ventricles).
Chiari III malformation involves herniation of contents of the posterior fossa through the occiput or upper cervical canal via a bony defect (i.e., akin to an encephalocele).
The corpus callosum develops front to back except for the rostrum (anterior genu, body, splenium, and rostrum last). This is important because an in utero insult may result in destruction of part of the corpus callosum. If posterior portions of the corpus callosum are present and anterior portions are not, this means that they were present at one point and were destroyed. If posterior portions are absent instead, this may mean that they did not develop.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here