Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
autoimmune hepatitis
antibody-mediated rejection
body mass index
chronic antibody-mediated rejection
cytomegalovirus
direct-acting antivirus
donation after (cardiac) circulatory death
donor-specific
Epstein-Barr virus
fibrosing cholestatic hepatitis
hepatitis B core antigen
hepatitis B immune globulin
hepatitis B surface antigen
hepatitis B virus
hepatitis C virus
human herpesvirus 6
human leukocyte antigen
isolated central perivenulitis
liver transplantation
mammalian target of rapamycin
nonalcoholic steatohepatitis
nodular regenerative hyperplasia
Organ Procurement and Transplant Network
primary biliary cholangitis
plasma cell hepatitis
primary sclerosing cholangitis
posttransplant lymphoproliferative disease
rejection activity index
Liver transplantation is an accepted therapeutic modality for end-stage liver disease and acute liver failure as well as for metabolic diseases such as familial amyloidotic polyneuropathy and cystinosis, in which the liver produces an abnormal protein while it remains undamaged by the disease process ( Fig. 38.1 , eSlide 38.1 , and Table 38.1 ). Considerable clinical advances have been made in the optimization of immunosuppression, control of infections such as cytomegalovirus (CMV) hepatitis, and prevention of recurrent diseases such as hepatitis B infection. Simultaneously, there is greater collective understanding of the pathologic processes involving the liver allograft. One-year patient survival rates after liver transplantation approach 90%, whereas overall 3-year, 5-year, and 10-year graft survival rates are 78%, 72%, and 60%, respectively (based on Organ Procurement and Transplantation Network [OPTN] data as of February 8, 2013). With the achievement of excellent short-term prognosis, current efforts are concentrated on improving long-term survival and well-being, which are mostly challenged by disease recurrence, side effects of prolonged immunosuppression, and de novo disease.
| Primary Diagnosis | 2002 (%) | 2012 (%) |
|---|---|---|
| Hepatitis C | 30.9 | 30.1 |
| Alcoholic liver disease | 22.7 | 23.9 |
| Other/Unknown | 24.7 | 25.7 |
| Cholestatic disease | 11.5 | 8.5 |
| Acute hepatic necrosis | 4.7 | 2.1 |
| Hepatitis B | 3.7 | 2.8 |
| Malignancy | 1.9 | 6.9 |
The success and therefore increasingly widespread practice of liver transplantation ensures that long-term care of graft recipients is not restricted to specialized tertiary institutions; an increasing number of pathologists in a wide variety of clinical settings encounter biopsies from liver allografts, which may either be clinically indicated or protocol biopsies performed at predetermined times ( Box 38.1 ). Because of an overall decrease in rejection rates and because of widespread availability, safety, and efficacy of novel direct-acting agents for hepatitis C virus (HCV), most transplant centers no longer perform protocol liver biopsies. Clinically indicated biopsies are performed to assess allograft dysfunction, rule out rejection, detect the presence and extent of recurrent disease, grade and stage recurrent viral hepatitis, or to exclude malignancy, a biliary or vascular problem. The diagnostic challenges of transplant pathology arise from modulation of the clinical, serologic, and histologic features of liver disease by immunosuppression, as well as overlap between the histologic features of various pathologic processes that do not infrequently occur simultaneously in the allograft. Knowledge of the status of immunosuppression and the original indication for transplantation is vital in making an accurate histologic diagnosis.
Clinically indicated biopsies
Elevation of liver tests
Signs and symptoms of liver disease
Imaging abnormalities of liver
Protocol biopsies
Time-zero biopsies (biopsies immediately after reperfusion)
Scheduled biopsies in patients transplanted for hepatitis C, nonalcoholic steatohepatitis, and autoimmune hepatitis
Biopsies in long-term survivors (annually or variably at 1, 3, 5, and 10 years)
Pathologic processes that are encountered in the liver allograft encompass preservation–reperfusion injury, hyperperfusion injury, rejection, infections, surgical complications, recurrent disease, and de novo diseases ( Box 38.2 ). Some of these processes occur within the first week of transplantation, others in the early posttransplant period (described variably as 3 to 6 months following transplantation), and others during the late posttransplant period (described variably as 3 months, 6 months, or 1 year after transplantation). In addition, the pathologist may be called on to assess suitability of a donor liver either by frozen section in the case of deceased donors or on paraffin-embedded liver tissue in the case of living donor transplantation.
Primary nonfunction
Preservation reperfusion injury
Hyperperfusion injury (small-for-size syndrome)
Rejection
Antibody-mediated rejection hyperacute, acute, chronic)
Acute (early) cellular rejection
Late cellular rejection
Central perivenulitis
“Idiopathic” chronic hepatitis
Plasma cell/de novo hepatitis
Chronic rejection
Central perivenulitis
Opportunistic infections
Epstein-Barr virus hepatitis
Cytomegalovirus hepatitis
Other viral hepatitides (adenovirus, human herpesvirus 6)
Surgical complications
Biliary strictures/ischemic cholangiopathy
Arterial thrombosis/stenosis
Venous thrombosis/stenosis
Recurrent diseases
De novo diseases
Hepatitis (viral, autoimmune, “idiopathic”)
Steatohepatitis (alcoholic, nonalcoholic)
De novo malignancy (posttransplant lymphoproliferative disease, hepatocellular carcinoma, nonhepatic carcinomas)
Architectural changes (nodular regenerative hyperplasia, undefined)
The process of donor selection aims to provide a functioning graft that will offer long-term graft and patient survival with minimal risk of transmitting disease. Histologic assessment constitutes an integral but not exclusive part of this process; in the case of deceased donors, frozen section evaluation of liver biopsies is not routinely performed because it may prolong the cold ischemia time, which is an important determinant of long-term complications such as bile duct strictures. However, the increasing use of extended criteria donors (described later) may increase the need for frozen section evaluation of donor livers. Living donor donation is an elective procedure; therefore, it does not suffer from similar time constraints, allowing evaluation of the donor liver on paraffin-embedded tissue. Some centers routinely perform liver biopsies as part of living donor evaluation, whereas others do so only when there is suspicion of underlying liver disease. Table 38.2 outlines the common indications for histologic evaluation of donor livers from deceased and living donors.
| Liver biopsy from deceased donor | Elevation of liver tests |
| Transaminases, bilirubin | |
| High gamma glutamyltransferase (>200 IU/L) | |
| Suspicion of fatty liver disease | |
| History of diabetes, obesity | |
| Fatty (yellow) liver at procurement | |
| History of alcohol use | |
| Positive serologies for viral hepatitis | |
| Anti-HCV antibody | |
| Anti-HBc antibody | |
| Findings during organ procurement Abnormal liver contour or color Suspicion of advanced fibrosis Focal liver lesion |
|
| Liver biopsy from living donor | Routine donor assessment |
| Any indication listed above for deceased donor | |
| Biopsy from other body sites | Focal lesion suspicious for malignancy |
Two liver core biopsies, one from the right lobe and one from the left lobe, are representative of the liver as a whole and adequate for assessment; there are no preferred sites in the right and left lobes for obtaining these biopsies. Although some centers also include an additional wedge biopsy in their protocol, a third biopsy is hardly necessary; wedge biopsies alone without accompanying core biopsies are not appropriate for donor liver assessment. The biopsies should be transported moistened in procurement fluid; transport in saline or on dry gauze or dry tissue paper is suboptimal because these techniques introduce artifacts and leach fat from tissues, respectively. The tissue should be frozen slowly; snap freezing in liquid nitrogen introduces freezing artifact ( Fig. 38.2 ).

The most significant factors affecting suitability of a liver for transplantation are steatosis, necrosis, inflammation, and fibrosis. Steatotic grafts are prone to more severe preservation–reperfusion injury than nonsteatotic grafts; therefore, macrovesicular steatosis, when present in significant amounts, is a well-recognized risk factor for primary nonfunction. The rate of primary nonfunction is 15% and that of delayed graft function is 35% when there is moderate (30% to 60%) macrovesicular steatosis. The rate is higher with severe (>60%) macrovesicular steatosis, whereas mild macrovesicular steatosis (<30%) does not affect the rate of primary nonfunction. Microvesicular steatosis usually accompanies macrovesicular steatosis, and unlike the latter, it does not adversely affect graft function. However, a donor liver that shows severe and exclusive microvesicular injury, especially in patients who may have demonstrated signs and symptoms of an energy utilization-depletion syndrome, should not be used for transplantation. An oil red O stain is not necessary for the assessment of steatosis.
An ideal donor is one who is younger than 40 years of age with trauma as the cause of death, donation after brain death, hemodynamic stability at the time of procurement, and less than 30% steatosis with no transmissible or underlying liver disease. The extreme shortage of donor organs has led to an expansion of criteria to include older donors and donation after circulatory (cardiac) death, as well as donors with positive hepatitis B and hepatitis C serologies. The use of livers from donors with hepatitis C for transplantation into recipients who also suffer from chronic hepatitis C greatly expands the donor pool without adverse effects on patient or graft survival ( Fig. 38.3 ) as long as there is no appreciable preexisting fibrosis in the donor liver. Livers from donors with chronic hepatitis C infection that show stage 2 fibrosis are not deemed suitable for LT. The pathology report of the liver biopsy from these donors should include the grade of interface and lobular activity, the presence and degree of necrosis, and the stage of fibrosis. Similarly, other extended criteria donor livers are increasingly used and appreciably expand the donor pool. Extended criteria include older donor age, prolonged cold ischemia time, donation after circulatory (cardiac) death, and presence of significant steatosis in the donor liver. The use of extended criteria donors necessitates minimization of operator-controlled factors, such as warm and cold ischemia times, to ensure long-term graft function without complications. Thus, it follows that except in the case of absolute contraindications, the role of the pathologist is not necessarily to comment on the suitability of grafts but to judiciously report histologic findings. The results of the biopsy interpretation are most often reported on standardized forms ( Fig. 38.4 ) provided by the local procuring organization, which seeks to assess the presence and degree of histologic features that suggest graft damage or the presence of significant donor liver disease.


Additional findings that may be noted on a liver biopsy include abnormalities of bile ducts, presence of hemosiderin pigment, and granulomas. Mild ductular reaction is a common finding in donor liver biopsies and does not by itself indicate underlying liver disease. Eligibility criteria are more stringent for living donors than deceased donors, and in a large number of centers liver biopsy is a routine mandatory component of donor evaluation. Most series of living donor liver biopsies have documented unsuspected findings in asymptomatic individuals without elevated liver function tests; these include steatosis, inflammation, iron deposition, cholestasis, perivenular sclerosis, granulomas, and sinusoidal vascular congestion.
Finally, in spite of all precautions and careful examination of liver biopsies, diseases may be inadvertently transmitted to the recipient; transmission of schistosomiasis, alpha-1 antitrypsin deficiency, and amyloidosis has been reported. Procurement surgery was aborted in two cases of living donor transplantation for Alagille syndrome when unsuspected paucity of intrahepatic bile ducts was diagnosed at frozen section in the donor livers; these donors had no preoperative signs or symptoms of Alagille syndrome. Primary nonfunction has been reported in grafts used from donors with hypertension. In these cases, evaluation of the failed grafts showed intimal thickening and luminal narrowing of medium-sized arteries. These arteries may not be present in peripheral liver biopsies but should be sought when there is a history of hypertension in the donor.
Rapid frozen section diagnosis may be required for tumors in the liver or other body sites; these may represent incidental tumors or unsuspected metastasis in patients with a known history of malignancy. Donor livers from patients with metastatic disease, malignant melanoma, lung carcinoma, colon carcinoma, and breast carcinoma carry high risks of transmission of malignancy and are not suitable for transplantation. However, despite utmost diligence in donor evaluation and procurement, transmission of malignancy through a donated liver may occur in rare cases. Livers from donors with nonmetastatic low-grade (grade 1 or 2) central nervous system tumors and renal cell carcinoma may be used with caution. Care is necessary to avoid excessive immunosuppression in these patients after transplantation because it dampens immune surveillance against malignant cells.
The processes of procurement, preservation, and reimplantation subject the liver allograft to physiologic stress and injury termed preservation–reperfusion injury , which may manifest with abnormal liver tests, histologic changes, or graft dysfunction. The three stressors of the allograft are warm ischemia, cold ischemia, and reperfusion. Warm ischemic injury results from poor perfusion of the liver while it is at body temperature and occurs before or during harvesting of the liver. Following harvesting, the liver is preserved in preservation solution at cold temperatures to minimize metabolism and energy use, subjecting it to cold ischemic injury. Rewarming of the graft before implantation subjects it to an added episode of warm ischemia, and finally, resumption of blood flow following vascular anastomosis subjects the graft to reperfusion injury. Warm ischemia causes damage to hepatocytes, whereas cold ischemia results in damage to endothelial cells and biliary epithelium. Reperfusion of the liver causes activation of Kupffer cells, which release reactive oxygen species, platelet-activating factor, and other cytokines, resulting in aggregation of neutrophils. The complement system is also activated by reoxygenation. All these factors contribute to microvascular dysfunction, which aggravates the endothelial and hepatocellular injury of cold and warm ischemia, respectively.
Warm ischemia time of less than 120 minutes and cold ischemia time of less than 10 to 12 hours are considered optimal for short-term and long-term graft survival. Minimizing warm ischemia time is especially important in donation after circulatory (cardiac) death (DCD), which is defined as liver donation after cessation of circulation is confirmed after a legally mandated “no touch” time period. An attempt is made to limit the warm ischemia time to 20 minutes, which in this context is the time from pronouncement of cardiac death to perfusion of the donor liver with preservation fluid. The risk of failure of DCD grafts increases by 6% for each hour of cold storage time.
Preservation–reperfusion injury may be mild and asymptomatic with minor elevations of aminotransferases, bilirubin, and gamma glutamyltransferase, which eventually normalize over a period of weeks to months. The amount of bile production soon after reperfusion is one of the most important parameters of preservation injury and good bile flow is a clinical indicator of good early graft function. Severe injury is manifested clinically by poor bile production, persistent elevation of serum lactate, and marked elevations of transaminases. The latter normalize in a few days and are followed by a prolonged cholestatic phase that resolves over many weeks to months. The extent of injury and resumption of normal liver function depend on the quality of the graft, ischemic times, and the medical condition of the recipient at transplantation.
Mild degrees of preservation and reperfusion damage are common in the liver allograft and are seen as hepatocyte swelling and cholestasis in the perivenular regions. Cellular swelling is caused by membrane and mitochondrial injury because of depletion of adenosine triphosphate secondary to ischemia, which is a major cause of cold ischemia preservation injury. Both hepatocyte swelling and cholestasis are reversible over several weeks ( Fig. 38.5A ). Detachment and rounding of hepatocytes accompanied by neutrophils and increased mitotic figures may be seen ( Fig. 38.5B ) ( eSlide 38.2 ). Severe damage is marked by the presence of confluent areas of necrosis, the extent of which determines prognosis ( Fig. 38.5C ). Necrosis may be more severe or extensive in subcapsular liver parenchyma and is not usually representative of the liver as a whole. Mild degrees of ductular reaction and small aggregates of neutrophils are common findings in time zero or reperfusion biopsies; the latter are not significant unless accompanied by hepatocyte necrosis.

Steatotic livers are more sensitive to reperfusion damage, which results in release of fat from damaged hepatocytes into the extracellular space; these extracellular and perisinusoidal fat globules compress the sinusoids and obstruct blood flow, leading to variable degrees of ischemic necrosis. Histologically, large fat droplets are seen that appear to be in sinusoids on a hematoxylin and eosin stain but are mostly in the Disse space on a trichrome stain. This lesion has been called lipopeliosis , a term that derives from the appearance of seemingly dilated sinusoids (peliosis) filled with fat ( Fig. 38.6 ) ( eSlide 38.3 ). As the lesion evolves, the fat droplets are surrounded by macrophages and mobilized from the lobule to the portal tracts, clearing within 4 weeks. The histology of the lesion thus depends on its stage of evolution.

The presence of significant parenchymal necrosis, or of spotty hepatocyte damage and necrosis, raises the possibility of ischemic injury as a result of hepatic artery occlusion ( Fig. 38.7 ). Parenchymal necrosis due to hyperacute rejection also occurs in the same time frame as reperfusion-preservation injury; however, the graft is intensely hemorrhagic, and vascular thrombi and necrosis are seen (discussed later). The presence of marked ductular reaction may raise suspicion of biliary tract obstruction, which can be excluded by imaging studies. The features of preservation–reperfusion injury do not resemble those of acute cellular rejection or recurrent hepatitis, which are not major considerations in this differential diagnosis.

Rejection of the liver represents the immunologic response of the host against foreign donor antigens located on the allograft. The response is usually targeted against biliary epithelium and endothelium of the hepatic veins, portal veins, and arteries; hepatocytes are not primary targets. Rejection is traditionally classified as hyperacute, acute, or chronic—terms that imply a classification based on the timing of rejection following transplantation ( Fig. 38.8 ). Although hyperacute rejection occurs in the immediate postoperative phase and most episodes of acute cellular rejection occur within the first few months of transplantation, the latter may occur years after transplantation, whereas chronic rejection has been reported within the first 3 months of transplantation. Therefore, these terms are best understood with reference to their underlying pathophysiologic processes, irrespective of whether they occur early or late in the posttransplant course. Acute rejection is a cellular process consisting of graft infiltration by a variety of inflammatory cells; chief among these are activated lymphocytes, also referred to as blastic lymphocytes. This process, which is T-cell mediated, is therefore more accurately termed cellular rejection, and as acute or late cellular rejection when occurring early or late, respectively, following transplantation. On the other hand, chronic rejection is not primarily a cellular process but an ischemic/fibrotic event manifested by progressive bile duct loss (ductopenic chronic rejection) and/or arterial damage and obstruction (arteriopathic chronic rejection). Therefore, acute or late cellular rejection can be treated using immunosuppressive drugs that abrogate the cellular immune response, whereas chronic rejection is not affected by these immunosuppressive drugs. However, newer therapeutic agents may attempt to “rescue” chronic rejection, especially when it is still evolving and is in the early stages. Thus, currently, immunosuppression may be attempted in both cellular as well as early chronic rejection, the latter with variable results. Although distinctive processes, cellular rejection and chronic rejection represent a continuum of immunologic damage; chronic rejection usually follows repeated, severe, or refractory cellular rejection. Thus, cellular rejection and chronic rejection are not mutually exclusive, and features of both may be present simultaneously in a biopsy.

In contrast to cellular rejection, hyperacute rejection results from a humoral immune response initiated by preformed antidonor antibodies in the recipient, which leads to rapid graft failure within hours (hyperacute humoral rejection) or days and even months (acute humoral rejection) following liver transplantation (LT). Less severe cases of humoral rejection that do not cause precipitous graft failure may clinically resemble acute cellular rejection. Most cases of humoral rejection are caused by preformed antibodies against ABO antigens.
Increasingly recognized is acute antibody-mediated rejection (AMR), which is primarily caused by human leukocyte antigen (HLA) class I and II antibodies and is rare, estimated to causing between 0.3% and 2% of rejection episodes. Acute AMR often occurs concomitantly with T cell–mediated acute rejection and shows overlapping features.
Humoral rejection may manifest either within a few hours or a few days of transplantation as hyperacute or acute humoral rejection, respectively. Vascularization of the newly implanted allograft results in normal bile production initially but also introduces preformed antibodies into the graft. AMR usually presents as unexplained allograft dysfunction, falling platelets and serum complement levels, as well as increased levels of circulating immune complexes in recipients with preformed donor-specific antibodies (DSA). Signs of microvascular injury on liver biopsy in addition to other characteristics commonly associated with rejection are noted. Bile production decreases, and the liver appears swollen and tender. There is progressive worsening of liver tests and eventually liver allograft failure ensues.
Acute cellular rejection occurs mostly, but not exclusively, between 5 and 30 days of transplantation. Many episodes of mild rejection are subclinical and detected by abnormal liver tests in asymptomatic patients. The biochemical profile of acute cellular rejection is characterized by elevations of alkaline phosphatase, bilirubin, and gamma glutamyltransferase, with minor elevation of transaminases. Peripheral leukocytosis and eosinophilia may be present. Patients with moderate and severe cellular rejection may complain of fever, abdominal pain, nausea, vomiting, and jaundice. Some patients may develop ascites as a result of liver swelling and increased hepatic pressures. The quantity of bile draining through a t-tube may decrease, and the bile may become pale and thin. However, the clinical and laboratory findings are variable and neither specific nor sensitive for the diagnosis of rejection, which is established on a liver biopsy. Although cellular rejection in the first 3 to 6 months of transplantation is related to a wide range of risk factors, cellular rejection occurring later in the course of transplantation is mostly related to inadequate immunosuppression either because of poor compliance or clinical indications such as infection or malignancy.
Better control of acute rejection and improvement of procurement techniques have dramatically decreased the incidence of chronic rejection, which now occurs in approximately 2% to 3% of cases. Of several factors associated with chronic rejection, the most common and most significant are related to inadequate immunosuppression and poorly controlled cellular rejection. CMV infection, older donor age, and long cold ischemia time are other risk factors strongly associated with an increased risk of chronic rejection. Chronic rejection usually occurs within the first year following transplantation, and the incidence does not necessarily increase with the period of follow up.
Clinically, chronic rejection is heralded by persistent jaundice or persistently elevated liver tests and usually follows several recurrent episodes of cellular rejection, a severe episode of cellular rejection, or refractory cellular rejection. The liver tests show a cholestatic profile with increasing elevations of bilirubin and alkaline phosphatase. Persistent alanine aminotransferase elevation is also present. Imaging studies may show strictures or peripheral pruning and narrowing of the hepatic arterial and biliary trees. The arterial changes result from foamy cell arteriopathy and peripheral ischemia, whereas the changes in the biliary tree reflect ischemic cholangiopathy secondary to arterial compromise.
Liver biopsies are not performed in the majority of cases of hyperacute humoral rejection because of rapidity of graft failure and severity of coagulopathy. The pathologist therefore mostly encounters a failed explanted liver, which is enlarged with a mottled and hemorrhagic external surface. The cut surface appears soft and shows hemorrhagic necrosis ( Fig. 38.9 ). Microscopically, widespread congestion, hemorrhage, and hepatocyte necrosis are seen accompanied by an intense neutrophilic infiltrate. Vessels of all sizes, including arteries, veins, and sinusoids, show thrombi and fibrinoid necrosis. Ischemic necrosis of bile ducts may be seen. Features of reperfusion injury may be seen in zone 3 hepatocytes. Less severe cases resemble biliary obstruction with portal edema, ductular reaction, and neutrophilic infiltrate; these changes are also seen in the early stages of severe injury.

Histologic features associated with acute AMR consist of endothelial cell hypertrophy, prominent portal eosinophils, and eosinophilic/monocytic venulitis. Severe acute AMR occurs in LT recipients who have preformed DSA with mean fluorescent intensities greater than 15000. Diffuse positive staining for C4d is currently required for diagnosis of acute AMR. Typically, C4d deposits are found in endothelial cells lining the portal veins and capillaries. C4d staining when noted early, as in the postperfusion biopsy, portends a poor prognosis ( Fig. 38.10 ). Immunostaining for C4d can now be performed on formalin-fixed paraffin-embedded tissue and no longer requires fresh frozen tissue. C4d deposition also occurs in typical acute cellular reaction and in a wide variety of other processes such as cholangitis in the absence of rejection; therefore, these conditions should be excluded and evidence of microvascular injury and graft dysfunction should be present before a definitive diagnosis of AMR is rendered. One study found that extensive versus focal staining and staining of the portal stroma are associated with high titers of DSA and worse outcomes, but these features are not useful in assessing efficacy of therapy.

Small-for-size syndrome (portal hyperperfusion syndrome) also manifests as hemorrhagic necrosis of the allograft in the early posttransplant period. This syndrome results from the inflow of more blood into an allograft than it can accommodate. Therefore, it occurs most commonly in reduced-size grafts, especially those that are less than 30% of the expected weight of the liver or less than 0.8% of the body weight of the recipient. The sudden inflow of blood into a small graft at portal pressures, which are still high in patients recently transplanted because of cirrhosis, causes endothelial injury and subsequent rupture of the thin-walled portal veins and sinusoids. Compensatory physiologic responses seek to limit blood flow into the allograft by constricting the hepatic artery; however, this results in ischemic injury to the hepatic parenchyma and the biliary tree. Small-for-size syndrome is manifested clinically as portal hypertension, ascites, encephalopathy, coagulopathy, and hyperbilirubinemia.
Histologically, the initial endothelial injury is seen as denudation of the endothelium of the portal veins and periportal sinusoids with severe congestion, thrombosis, and hemorrhage in the portal-periportal regions. Parenchymal ischemia results in infarcts, whereas ischemia to the biliary tree is manifested as bile duct necrosis, bile infarcts, ductular proliferation, and cholangitis. If the graft survives the initial insult, reparative changes result in endothelial hypertrophy, subendothelial edema, and intimal and luminal fibrosis. The late changes of ischemic cholangitis are manifested as biliary strictures, dilatation, and abscess formation. Most of the biliary changes occur in the hilar region and may not be seen in a peripheral biopsy, which, however, shows a characteristic triad of centrilobular microvesicular steatosis, centrilobular hepatocanalicular cholestasis, and ductular reaction (see Fig. 38.10 ).
Cellular rejection occurring in the early posttransplant period (acute cellular rejection) demonstrates a classic histologic triad of mixed portal inflammatory infiltrate, endothelitis, and bile duct injury. However, late cellular rejection may not display these typical findings. In addition, some cases of plasma cell hepatitis, central perivenulitis, and chronic hepatitis may represent histologic variants of late cellular rejection.
Acute cellular rejection is a portal-based inflammatory process that consists of three cardinal features: mixed portal inflammatory infiltrate, bile duct damage, and endotheliitis ( eSlide 38.4 ). The portal infiltrate consists of a mixture of acute and chronic inflammatory cells, including lymphocytes, eosinophils, and neutrophils ( Fig. 38.11 ). Plasma cells are rare in acute cellular rejection occurring in the early posttransplant period but may form an important constituent of the inflammatory response in late cellular reaction. Activated or blastic lymphocytes are sine qua non for the diagnosis of acute cellular rejection. These lymphocytes are large and contain prominent nucleoli and vesicular chromatin. Eosinophils and neutrophils are almost always present in variable numbers; the latter are usually seen adjacent to and infiltrating bile ducts. Eosinophils, although prominent, are by themselves not indicative of rejection and may occur with drug-induced hepatitis, autoimmune hepatitis (AIH), and viral hepatitis. Infiltration of biliary epithelium by lymphocytes (lymphocytic cholangitis) and neutrophils is a prominent feature of acute cellular rejection. This is accompanied by varying degrees of biliary epithelial damage manifested as cytoplasmic vacuolation, nuclear irregularity, nuclear overlapping, and nuclear hyperchromasia. A mild ductular reaction consisting of proliferating bile ductules with periductal neutrophilic infiltrate may be seen. Rarely, intraluminal neutrophils mimicking ascending cholangitis may be present. Endotheliitis is the third feature of rejection and is most prominent in portal venules but may be seen in hepatic venules. Endotheliitis of the hepatic arteries may also occur but is rare in the smaller peripheral arterioles sampled in core needle biopsies. Lymphocytes adhering to the endothelium do not constitute endotheliitis, which is identified by undermining and/or lifting up of the endothelium by the inflammatory cells (subendothelial inflammation).

Although rejection is a portal-based inflammatory process, parenchymal damage may be seen in severe acute cellular rejection and usually takes the form of parenchymal necrosis accompanying endotheliitis and inflammation of the hepatic venules; in severe rejection, these changes are seen in conjunction with severe changes in the portal tracts. These perivenular changes with or without accompanying portal tract changes are frequently seen in the late posttransplant period (6 months to 1 year following transplantation) ( eSlide 38.5 ). When unaccompanied by portal tracts changes, the term isolated central perivenulitis (ICP) is used; some of these cases represent a histologic variant of late cellular rejection (discussed in detail later). In some cases of severe acute cellular rejection, features of sinusoidal obstruction syndrome consisting of centrilobular congestion, perivenulitis, and hepatocyte necrosis may be seen and will clinically manifest with ascites.
Acute cellular rejection is graded as mild, moderate, and severe ( Table 38.3 ). The grade of rejection does not guide therapy, which is based on the presence or absence of rejection, except in patients who have received transplantation for chronic hepatitis C. Because avoiding unnecessary immunosuppression is vital to appropriate management of this group of patients, the preferred policy in most centers is to treat rejection only if it is moderate or severe and to carefully “watch” patients with mild rejection. In general, immunosuppression levels are optimized regardless of the degree of acute cellular rejection.
| Grade a | Criteria |
|---|---|
| Indeterminate b | Mild portal inflammatory infiltrate thought to represent rejection |
| Mild | Mild rejection infiltrate in a minority of portal tracts and confined within the portal spaces |
| Moderate | Rejection infiltrate, expanding most or all of the triads |
| Severe | Rejection infiltrate spills over into periportal parenchyma with moderate to severe central perivenulitis (inflammation and necrosis around the central venules) |
a Grading is carried out only after an unequivocal diagnosis of rejection has been established on global assessment of the liver biopsy.
b The term indeterminate is used when there is mild portal infiltrate thought to represent low-grade or early rejection, but the minimal diagnostic criteria for rejection are not met (at least two of three features [ie, mixed portal inflammatory infiltrate, bile duct damage, and endotheliitis] are required to make a definite diagnosis of acute cellular rejection). The term indeterminate is not intended to be used when it is uncertain whether the portal infiltrate represents rejection or some other pathologic process.
Each feature of acute cellular rejection can be individually scored and the sum totaled to derive a rejection activity index (RAI) ( Table 38.4 ). However, the RAI does not establish the diagnosis of rejection, which is based on a global assessment of the liver biopsy. Instead, the RAI is a tool for scoring and grading rejection once the diagnosis has been established. The RAI is most useful in clinical trials for comparison of therapeutic efficacy of various immunosuppressive drugs.
| Histologic Feature | Criteria | Score |
|---|---|---|
| Portal inflammation | Minority of portal tracts showing mostly lymphocytic inflammation | 1 |
| Portal tracts not expanded by infiltrate | ||
| Majority of portal tracts show mixed infiltrate of activated lymphocytes, neutrophils, and eosinophils | 2 | |
| Portal tracts expanded by infiltrate | ||
| Inflammatory infiltrate spills over into the periportal parenchyma in addition to 2 above | 3 | |
| Bile duct inflammation and damage | Minority of ducts show inflammatory infiltrate | 1 |
| Mild reactive epithelial changes such as increased nuclear-to-cytoplasmic ratio | ||
| Majority of ducts infiltrated by inflammatory cells | 2 | |
| Reactive epithelial changes more severe (nuclear pleomorphism, disordered polarity, cytoplasmic vacuolization) and present in more than an occasional duct | ||
| Majority of ducts infiltrated by inflammatory cells | 3 | |
| Severe reactive changes in most or all ducts | ||
| Venous endothelial inflammation | Minority of portal and/or hepatic venules show subendothelial lymphocytic infiltration | 1 |
| Majority of portal and/or hepatic venules show subendothelial lymphocytic infiltration | 2 | |
| Moderate to severe perivenular inflammation with hepatocyte necrosis in addition to 2 above | 3 |
Late cellular rejection occurring 6 months after transplantation does not show the classical features of acute cellular rejection described previously; as the time after transplantation increases, the reaction seems to be “toned down” or “muted.” Endotheliitis and bile duct damage may be neither prominent nor extensive, and the portal inflammatory infiltrate may not contain many blastic lymphocytes or eosinophils (see eSlide 38.5 ) ( Fig. 38.12 ). The presence of a chronic portal infiltrate without blastic lymphocytes, endotheliitis, and bile duct injury mimics chronic hepatitis and triggers serologic and other investigations to rule out various causes of chronic hepatitis; when these are negative, the biopsy may be labeled idiopathic chronic hepatitis (discussed in detail later) ( Fig. 38.13 ).


A characteristic pattern of late graft injury consists of an intense plasma cell infiltrate in portal tracts with variable interface hepatitis ( Fig. 38.14 ). The portal tract changes may be accompanied by central perivenulitis with a predominant plasma cell infiltrate. Alternatively, a plasma cell–rich perivenular infiltrate may be present without portal tract changes. This pattern of injury is variably referred to as de novo AIH or plasma cell hepatitis and is thought to represent late cellular rejection in at least some cases (discussed in detail later).

Thus, late cellular rejection may show at least three atypical histologic variants, namely, idiopathic chronic hepatitis (ICP), and de novo–autoimmune–plasma cell hepatitis. However, each of these patterns (discussed in detail later) may also represent other posttransplant diseases in the allograft, which need to be distinguished from rejection to ensure appropriate management of immunosuppression.
In the early postoperative period, elevation of liver enzymes may occur because of preservation–reperfusion injury, rejection, surgical complications, and infection. The microscopic features of these conditions (discussed under the appropriate sections) do not overlap with the histologic findings of rejection except in the case of infections. Infectious hepatitis, whether as a result of CMV, adenovirus or recurrent hepatitis C, may demonstrate portal inflammation and focal endotheliitis, features that may be confused with rejection ( Fig. 38.15 ). The distinction of these infectious hepatitides from cellular rejection is vital because the former require less, and the latter, more immunosuppression.

Two histologic features are most helpful in aiding this distinction. First, an infectious hepatitis shows lobular inflammation and necroinflammatory lesions, which are not prominent in cellular rejection. Second, endotheliitis and bile duct damage are present in the majority of portal tracts in cellular rejection, whereas they tend to be focal in the infectious hepatitides. Coexistence of significant lobular inflammation with endotheliitis and bile duct damage in more than 50% of portal tracts suggests the presence of both rejection and an infectious hepatitis; this is not entirely surprising, because viral infections are known triggers of rejection.
The histologic features of late cellular rejection overlap with those of recurrent or de novo hepatitides. The distinction can hardly ever be established by histology alone and requires correlation with serologic findings and recent history of immunosuppression.
The ischemic/fibrotic process of chronic rejection affects the bile ducts and the arteries of the liver allograft, giving rise to ductopenic and arteriopathic rejection, respectively. Ductopenia affects the small interlobular bile ducts, whereas arterial changes affect medium-sized arteries, including the main hepatic artery and its lobar branches. Ductopenic rejection can be visualized on a liver biopsy, but arterial changes are rarely seen on biopsy material, which samples the peripheral parenchyma and does not include arterial branches of large caliber. The peripheral liver sampled in such a case may demonstrate the consequences of arterial occlusion in the form of perivenular necrosis and cell loss ( Fig. 38.16 ). Rarely, a medium-sized artery may be included in the sample of a core needle biopsy and will thus be diagnostic of foam cell arteriopathy.

In the early stages of chronic rejection, the small interlobular bile ducts are infiltrated by inflammatory cells, chiefly lymphocytes. This is accompanied by epithelial damage in the form of nuclear loss, nuclear irregularity, and cytoplasmic eosinophilia with flattening and attenuation of the biliary epithelium ( Fig. 38.17 ). These ducts show immunohistochemical positivity for p21, a marker of senescence. Cellular and canalicular cholestasis are seen, and ductular reaction is minimal, if present at all ( eSlide 38.6 ). As the ducts atrophy and disappear as a result of progressive damage, the portal infiltrate recedes while cholestasis worsens. Besides cellular and canalicular cholestasis, bile is seen within Kupffer cells, and clusters of bile-laden foamy macrophages may be seen within the lobules. At low power, the biopsy has a dull, dusky appearance. The diagnosis of ductopenia requires absence of ducts in 50% of portal tracts in a biopsy that contains at least 10 portal tracts; this requirement avoids misinterpreting physiologic absence of ducts, which occurs in about 7% of portal tracts. An immunohistochemical stain for the biliary keratins, K7 or K19, is helpful in assessing the degree of duct loss. ( Fig 38.17D ) However, evolving or early ductopenic rejection can be suggested, even when bile ducts are not lost in such significant numbers, if there is evidence of epithelial damage as described previously. This allows for appropriate therapy when the process is still potentially reversible ( Table 38.5 ).

| Structure | Early | Late |
|---|---|---|
| Small bile ducts | Degenerative and atrophic changes in the majority of ducts (cytoplasmic eosinophilia, irregular nuclear spacing, nuclear loss, and overlapping hyperchromasia leading to epithelial attenuation) | Bile duct loss in at least 50% of portal tracts in a biopsy that contains at least 10 portal tracts |
| Bile duct loss in less than 50% of portal tracts | ||
| Central veins | Variable combinations of intimal/luminal inflammation, necrosis of zone 3 hepatocytes, and mild perivenular fibrosis | Focal obliteration with variable inflammation |
| Severe fibrosis including bridging fibrosis | ||
| Hepatic arterioles | Loss of hepatic arterioles in less than 25% of portal tracts | Loss of hepatic arterioles in more than 25% of portal tracts |
| Lobular parenchyma | Spotty necrosis of hepatocytes (“transition” hepatitis) | Marked cholestasis; clusters of foamy macrophages |
| Large perihilar hepatic arteries | Intimal inflammation and focal foam cell deposition without luminal compromise | Luminal narrowing by subintimal foam cells |
| Fibrointimal proliferation, sometimes with calcifications | ||
| Large perihilar bile ducts | Periductal inflammation | Mural fibrosis |
Arterial changes consist of the presence of foamy macrophages, which are usually subintimal but may be located in any layer of the arterial wall. These cells cause variable degrees of luminal occlusion ( Fig. 38.18A ). Rarely, the walls may appear calcified or ossified and occasionally clusters of foamy cells may be present within sinusoids ( Fig. 38.18B ). The peripheral liver demonstrates effects of arterial compromise in the form of perivenular necrosis of hepatocytes. Features of arteriopathic rejection may often be associated with cholestasis and bile duct damage, which is thought to result from ischemic damage to the biliary tree secondary to the vasculopathy of rejection ( Fig. 38.17 ) ( eSlide 38.6 ).

The differential diagnosis of ductopenic rejection includes other causes of biliary damage such as ischemic cholangitis, bile duct strictures, and recurrent sclerosing cholangitis. The diagnosis is established by a combination of radiologic findings and recent history of immunosuppression. Patients with chronic rejection often have a history of repeated episodes of cellular rejection, refractory cellular rejection, or severe cellular rejection. The absence of ductular reaction is a useful histologic clue that favors the diagnosis of chronic ductopenic rejection over other causes of biliary damage listed earlier, all of which show variable, but usually prominent, ductular reaction.
Although chronic AMR (cAMR) has not been fully characterized, it has been shown that LT recipients who have high titers of DSA have greater fibrosis progression and persistent chronic inflammation as compared with those who do not have DSAs. Sinusoidal and perivenular fibrosis have also been associated with presence of DSA as have portal inflammation, portal venopathy, and interface hepatitis ( Box 38.3 ).
Early acute cellular rejection
Steroid-resistant rejection
Chronic ductopenic rejection
Plasma cell hepatitis
Idiopathic hepatic fibrosis progression
Idiopathic biliary abnormalities
Portal venopathy and nodular regenerative hyperplasia
Sinusoidal fibrosis and centrilobular necrosis with intrahepatic thromboses
A recently proposed cAMR score identifies patients with DSAs at highest risk for subsequent allograft loss despite normal or near-normal liver enzymes. The cAMR score includes interface activity, lobular inflammation, portal fibrosis, portal venopathy, sinusoidal fibrosis, and HCV virus status. Portal venopathy and portal collagenization, as well as sinusoidal fibrosis are attributed to complement activation as evidenced by C4d deposition. The cAMR scoring system highlights the necessity of testing for serum DSA when unusual patterns of fibrosis such as sinusoidal and perivenular fibrosis or portal vein collagenization are seen on biopsy. In addition to a high cAMR score, the diagnosis of cAMR, the presence of serum DSA, and exclusion of other causes that may have similar injury patterns.
Various therapeutic strategies are currently used to prevent hyperacute rejection in ABO-incompatible grafts, which are often used in living donor liver transplantation. These consist of anti-CD20 antibodies, intravenous immunoglobulins, plasma exchange, and splenectomy. Management of AMR consists of plasmapheresis, cyclophosphamide, and intravenous immunoglobulins in an effort to salvage the graft. Treatment of cellular rejection involves increasing immunosuppression; this may translate into increasing levels of maintenance immunosuppressive drugs in mild cases or steroid bolus therapy in severe rejection. Current maintenance therapy consists of calcineurin inhibitors, primarily tacrolimus, and occasionally cyclosporine if tacrolimus is not deemed suitable. Acute cellular rejection responds well to an increase of immunosuppression, but late cellular rejection is less responsive and in many cases portends the onset of chronic rejection. Medical therapy may be effective in the early stages of chronic rejection as it seeks to rescue the graft from progressive damage. Retransplantation remains the only option for end-stage chronic rejection.
Histologic features of rejection, in the absence of clinical symptoms or even biochemical abnormalities, occur frequently in protocol biopsies during the first few weeks after transplantation. A pooled analysis of 15 studies reported histologic rejection in 67% of 1566 otherwise asymptomatic individuals; of these, approximately one-third had normal biochemical function. On follow up, 14% of patients with histologic rejection and normal liver tests developed biochemical graft dysfunction, and 4% progressed to steroid-resistant rejection and chronic rejection. However, because the majority of patients did not develop graft dysfunction, current practice does not favor treatment of incidentally discovered histologic rejection.
Central perivenulitis is a term used for necroinflammatory lesions that affect the central (hepatic) venules and surrounding perivenular hepatocytes. The lesion consists of variable degrees of inflammation, endotheliitis, hepatocyte necrosis, and perivenular fibrosis. The inflammatory infiltrate consists of variable combinations of plasma cells, lymphocytes, and eosinophils. The preferential centrilobular localization of inflammatory processes reflects the presence of potent antigen-presenting cells such as dendritic cells in this region. These cells may present antigens in viral and autoimmune hepatitis as well as during rejection.
Central perivenulitis with concomitant portal tract changes occurs soon after transplantation as well as in the late posttransplantation period (see eSlide 38.5 ). The portal changes, along with clinical and laboratory findings, provide a clue to the underlying pathologic process, which may represent acute cellular rejection, chronic viral hepatitis, or AIH ( Fig. 38.19 ). Drug-induced hepatitis may rarely show a similar histologic picture. When occurring by itself without accompanying portal tract inflammation, the lesion is termed ICP ( Fig. 38.20 ). It is not infrequent for ICP to precede portal tract changes.
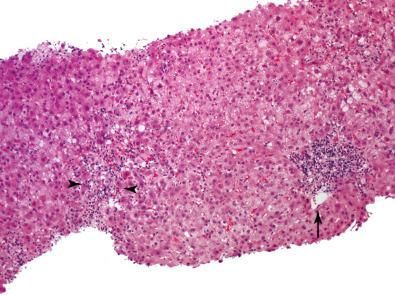

ICP occurs most often 6 months to 1 year after transplantation in approximately one-third of both adult and pediatric transplant recipients. Most patients have had prior episodes of acute rejection, many with central perivenulitis, either isolated or in conjunction with portal tract changes. In a minority of patients with ICP, the ICP evolves to de novo AIH or recurrent AIH. ( Fig. 38.21 ) and recurrent primary biliary cholangitis (PBC), whereas most others remain idiopathic. In the absence of treatment, almost half of all patients progress to perivenular fibrosis and a few to ductopenia ; these adverse outcomes are common in patients with severe central perivenulitis, whereas minimal or mild cases may resolve spontaneously ( Table 38.6 ). The incidence of perivenular fibrosis is less common in centers that treat these lesions with immunosuppressive drugs.

| Criteria | ||
|---|---|---|
| Grade | Inflammation | Necrosis |
| Minimal/indeterminate | Few central veins involved | Patchy perivenular necrosisNo confluent necrosis |
| Mild | Most central veins involved | Patchy perivenular necrosisNo confluent necrosis |
| Moderate | Most central veins involved | Patchy confluent necrosisNo bridging necrosis |
| Severe | Most central veins involved | Confluent necrosis involving most central veinsCentral to central bridging necrosis |
The resolution of ICP with immunosuppression and lower incidence of perivenular fibrosis in these patients are strong indicators that ICP represents a histologic variant of cellular rejection. It is further believed that ICP indicates transition from acute to chronic rejection. However, the diagnosis of rejection in a case of ICP necessitates exclusion of viral hepatitides, which is aided by a review of the serum levels of viral nucleic acids and recent history of immunosuppression. The diagnosis of rejection can be reliably made only when the majority, or at least 50%, of central veins are involved; a localized reaction involving only a minority of central veins is not indicative of rejection ( Fig. 38.22 ).

Clinically, patients may be asymptomatic or there may be mild elevations of liver tests. In contrast to portal-based rejection, transaminases are elevated more than the biliary enzymes. Some patients may miss clinical detection altogether and present with ascites, raising clinical suspicion of Budd-Chiari syndrome or veno-occlusive disease.
A subcategory of patients with central perivenulitis may show a predominant plasma cell infiltrate; this may also occur as an isolated lesion in association with a plasma cell–rich portal infiltrate. Some of these patients represent recurrent AIH in the allograft. When the lesion occurs in patients not transplanted for AIH, the terms de novo AIH and plasma cell hepatitis have been used. This lesion is discussed later in the chapter.
Perivenular hepatocyte necrosis without accompanying inflammation indicates vascular ischemia that may result from arteriopathic chronic rejection (see eSlide 38.6 ; and Fig. 38.16 ). Perivenular hepatocyte necrosis due to venous outflow obstruction is accompanied by sinusoidal dilatation, congestion, and hemorrhage; there is no inflammation ( eSlide 38.7 ) ( Fig. 38.23 ). Similar changes are seen in some patients who suffered severe acute cellular rejection. In addition, features of venous outflow obstruction unaccompanied by inflammation are also seen with azathioprine toxicity.

Recurrent disease is a frequent cause of long-term graft dysfunction; almost all primary diseases recur in the liver allograft, although they do so with varying incidence and progress at variable rates. Some, such as PBC, pursue a slow course stretching over decades, thus providing the recipient with a new lease of life, whereas others, such as recurrent hepatitis C, progress to cirrhosis within 5 years in approximately one-third of allograft recipients. However, in spite of the very real risk of recurrent disease, liver transplantation remains the only practical therapeutic option for patients with end-stage liver disease. Therefore, the emphasis is currently placed on identifying pretransplant and posttransplant factors predictive of recurrence and timely and accurate diagnosis of recurrent disease and its appropriate management.
The diagnosis and management of recurrent disease is challenging for several reasons. Clinical, serologic, and histopathologic features of recurrent disease are often modulated by immunosuppression, which means that they may not provide the same diagnostic value as they do in nontransplanted patients. Furthermore, the clinical and histologic features overlap with those of other complications of transplantation, and not infrequently, several processes may occur simultaneously. The course and progression of recurrent disease may differ from that in nontransplanted patients and among individual patients. In addition, variability in diagnostic criteria, methods for detection of recurrence (protocol versus clinically indicated biopsies), immunosuppression protocols, and the length of the follow-up period explain the variability in recurrence rates for individual diseases reported in different studies ( Table 38.7 ).
| Disease | Incidence (%) | Diagnosis Based on | Similarity of Histologic Findings to Nontransplanted Liver |
|---|---|---|---|
| HCV | 60–100 | Liver biopsy HCV RNA does not accurately correlate with histology |
Findings similar to nontransplanted liver Acute hepatitis more often detected in transplanted patients because of routine monitoring; subclinical in nontransplanted individuals and does not undergo biopsy ( Fig. 38.24 and eSlide 38.8 ) Chronic hepatitis: lymphoid aggregates and germinal centers less common, ductular reaction more frequent than in nontransplanted liver ( Fig. 38.25 and eSlide 38.9 , eSlide 38.10 ) Fibrosing cholestatic hepatitis, a severe form of recurrent hepatitis C, unique to graft ( Fig. 38.26 and eSlide 38.11 ) |
| HBV | <10 | Serum viral DNA, serum viral antigens Serum DNA used to monitor recurrence and therapeutic effectiveness |
Findings similar to nontransplanted liver ( Fig. 38.27 and eSlide 38.12 ) Fibrosing cholestatic hepatitis unique to graft in patients not on antiviral prophylaxis ( Fig. 38.28 ) |
| AIH | 30 | Combination of clinical, histologic, and serologic findings ( Box 38.4 ) Criteria of International Autoimmune Hepatitis Group not applicable Yearly protocol biopsies recommended for early detection because histologic changes may predate biochemical abnormalities |
Findings similar to nontransplanted liver ( Fig. 38.29 and eSlide 38.13 ) Central perivenulitis may be initial lesion of recurrent disease ( Fig. 38.21 ) |
| PBC | 20–30 | Liver biopsy Most often detected by protocol biopsies AMA and liver tests not sensitive or specific |
Findings similar to nontransplanted liver ( Figs. 38.30 and 38.31 and eSlide 38-14 ) Chronic hepatitis and isolated central perivenulitis may represent initial lesion of recurrence; typical lesions may emerge on subsequent biopsies |
| PSC | 30 | Cholangiogram (nonanastomotic strictures >90 days after transplantation) Requires exclusion of competing causes ( Box 38.5 ) |
Findings similar to nontransplanted liver ( Fig. 38.32 and eSlide 38.15 ) May be mimicked by other types of biliary strictures such as ischemic cholangitis ( Fig. 38.33 ) |
| NASH | 20–40 | Liver biopsy Yearly protocol biopsies recommended for early detection |
Findings similar to nontransplanted liver ( Fig. 38.34 and eSlide 38.16 ) |
| ALD | 5–30 a | Liver biopsy Serum carbohydrate deficient transferrin Blood alcohol levels |
Findings similar to nontransplanted liver ( Fig. 38.35 ) |
a Variability in incidence rates reflects variability in definitions and diagnostic criteria. When defined as any alcohol use, the incidence of recidivism is 30%. When only heavy drinking is considered (> 8 drinks or 200 g of alcohol per week), the incidence is 5% to 8%.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here