Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Obstructive lung diseases consist of a heterogeneous group of chronic respiratory illnesses characterized by airway obstruction and air trapping ( Box 20.1 ). The most common causes include chronic obstructive pulmonary disease (COPD), which includes emphysema and chronic bronchitis, followed by asthma and bronchiectasis. Bronchiectasis is discussed in Chapter 6 . Constrictive or obliterative bronchiolitis is a less common cause of obstructive lung disease. Patients with obstructive lung diseases often present with nonspecific symptoms such as dyspnea, particularly during exertion, and cough. Pulmonary function tests (PFTs) characteristically show disproportionate reduction in the forced expiratory volume in 1 second (FEV 1 ) compared with the forced vital capacity (FVC) as reflected in the decreased FEV 1 /FVC ratio.
Emphysema
Chronic bronchitis
Asthma
Bronchiectasis
Constrictive bronchiolitis
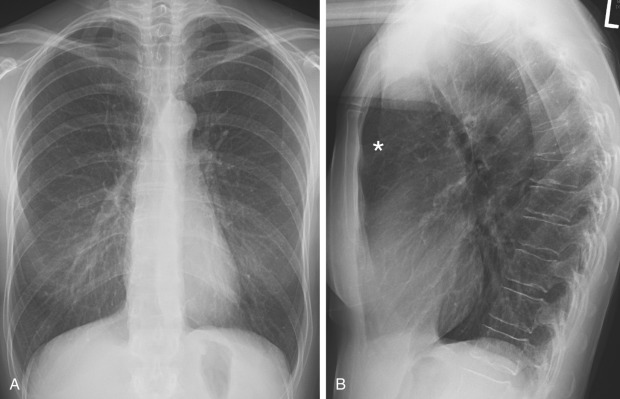
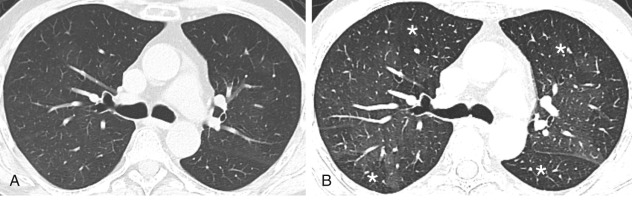
The presence of air trapping is common to all obstructive lung diseases, which in its mild form is difficult to detect on plain radiography. When severe, air trapping can be detected radiographically as hyperinflation with more than 6 anterior or 10 posterior ribs visualized above the diaphragm at the midclavicular line ( Fig. 20.1 ). Hyperinflation is also characterized by a low, flat diaphragm, particularly on the lateral view. The diaphragm is considered to be flattened when the highest level of the dome is less than 1.5 cm above a straight line drawn between the costophrenic junction and the vertebral phrenic junction. The angle formed by the diaphragm and the anterior chest wall is often 90 degrees or greater compared with the acute angle seen with a normal upwardly curved diaphragm. Another criterion of overinflation is a widened retrosternal air space greater than 2.5 cm in diameter. The cardiac silhouette tends to be long and narrow. High-resolution computed tomography (HRCT) has significantly improved our ability to image morphologic abnormalities associated with obstructive lung diseases, particularly in emphysema and bronchiectasis. On inspiratory CT, air trapping manifests as mosaic attenuation of the lungs, with areas involved by air trapping manifesting as hypoattenuation compared with the normal lung parenchyma. Air trapping can be distinguished from mosaic attenuation caused by vascular and interstitial lung disease on expiratory CT images where the hypoattenuating areas caused by air trapping are accentuated ( Fig. 20.2 ) (see Chapter 17 ).
Chronic obstructive pulmonary disease includes chronic bronchitis, which is defined in clinical terms, and emphysema, which is defined anatomically. Although asthma is also characterized by airflow obstruction, it differs from COPD in its pathogenic and therapeutic responses. Asthma is therefore considered a separate clinical entity from COPD, and it is discussed separately. However, there is some overlap between these conditions, and some patients have COPD and asthma. COPD is predominantly a disease of smokers, although only about 15% of smokers develop disabling airflow obstruction.
The chest radiograph is an important imaging modality in the assessment of patients with COPD. However, it has limitations in the detection and differential diagnosis of obstructive airway disease. It is relatively insensitive in the detection of early emphysema, and it is frequently normal for patients with pure chronic bronchitis and asthma. HRCT has improved our ability to differentiate between emphysema and bronchitis. CT also can delineate functional abnormalities such as air trapping and decreased perfusion.
Emphysema is a condition of the lung characterized by abnormal, permanent enlargement of the airspaces distal to the terminal bronchiole, accompanied by destruction of their walls without obvious fibrosis ( Box 20.2 ).
Cigarette smoking
Dyspnea
Chronic airflow obstruction
Permanent enlargement of the airspaces distal to the terminal bronchiole and destruction of their walls
Centrilobular (central lobule)
Panlobular (entire lobule)
Paraseptal (distal lobule, subpleural)
Radiography
Overinflation
Low, flat diaphragm
Increased retrosternal clear space
Vascular deficiency shown by areas of irregular lucency
Bullae
Computed tomography
Centrilobular
Multiple, small areas of low attenuation without walls
Upper lobes > lower lobes
Panlobular
Fewer and smaller vessels
Lower lobes
Paraseptal
Subpleural and along fissures
Thin walls
Single row
Emphysema is defined anatomically and pathologically. Emphysema may occur without detectable chronic airway obstruction. Mild degrees of emphysema are frequently found in smokers at autopsy. Widespread and severe emphysema is usually associated with a history of cigarette smoking, chronic airflow obstruction, and dyspnea. The airflow obstruction can be measured with PFT by a diminution of the FEV 1 or the ratio of the FEV 1 to the FVC. Lung volumes increase in emphysema as a result of hyperinflation with increases in the total lung capacity (TLC), functional residual capacity, and residual volume, with a concomitant decrease in vital capacity as the emphysema becomes more severe. The loss of the internal surface area of the lung and of the alveolar capillary bed, two components of emphysema, is reflected in a decrease in the diffusing capacity of the lung for carbon monoxide (D LCO ).
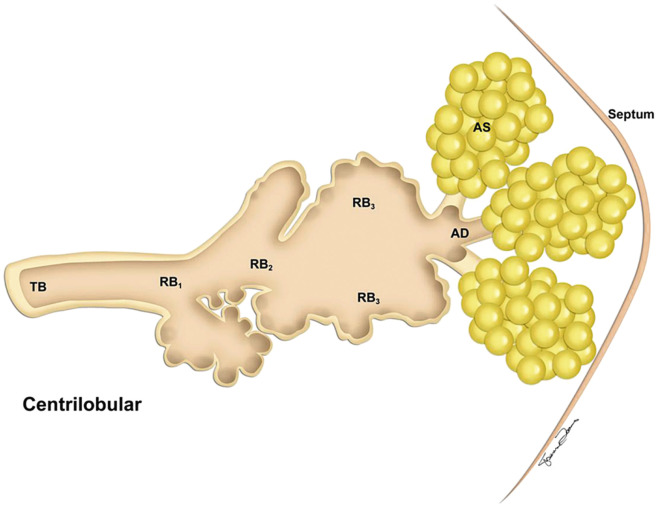
The four anatomically defined types of emphysema are centrilobular (centriacinar), panlobular (panacinar), paraseptal (distal acinar), and paracicatricial (irregular) emphysema. The acinus is the air-exchanging unit of the lung, and it is located distal to the terminal bronchiole. It includes the respiratory bronchioles, alveolar ducts, alveolar sacs, and alveoli ( Fig. 20.3 ). Although this classification relies on the relationship of emphysema to the acinus, acini cannot be resolved on HRCT, and it may be more useful for the radiologist to consider the types of emphysema relative to their location at the lobular level. Centrilobular, panlobular, and paraseptal emphysema often can be distinguished morphologically, but as emphysema becomes severe, distinction among the three types becomes more difficult.
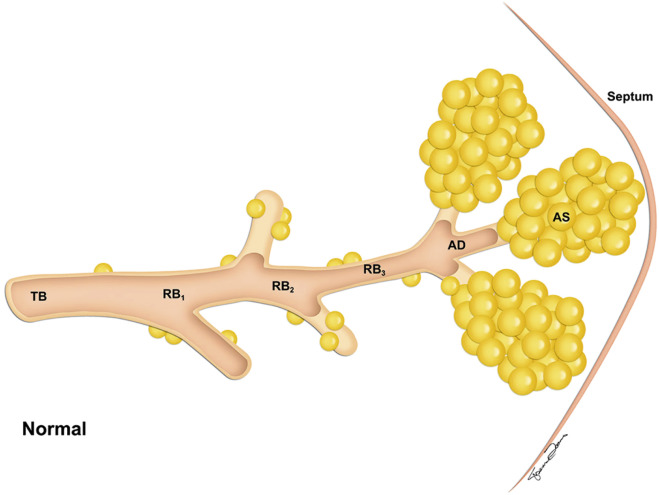
Centrilobular emphysema affects predominantly the respiratory bronchioles in the central portion of the secondary pulmonary lobule ( Fig. 20.4 ). It is usually identified in the upper lung zones, and it is associated with cigarette smoking.
Panlobular emphysema involves all of the components of the acinus and therefore involves the entire lobule ( Fig. 20.5 ). It is classically associated with α 1 -antitrypsin deficiency (AATD), although panlobular emphysema may also be seen without AATD in smokers, in older adults, and distal to bronchial and bronchiolar obstruction. Thurlbeck described this entity as a “diffuse simplification of the lung structure with progressive loss of tissue until little remains but the supporting framework of vessels, septa, and bronchi.” α 1 -Antitrypsin (AAT) is a potent inhibitor of neutrophil elastase, which is released as part of an inflammatory pathway triggered by smoking. In smokers with genetic mutations resulting in deficiency of AAT, unchecked activities of neutrophil elastase cause alveolar destruction at an accelerated rate compared with those normal with a AAT level.
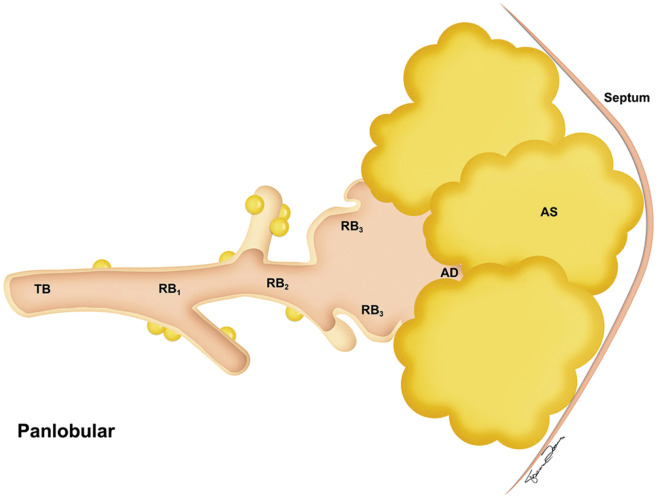
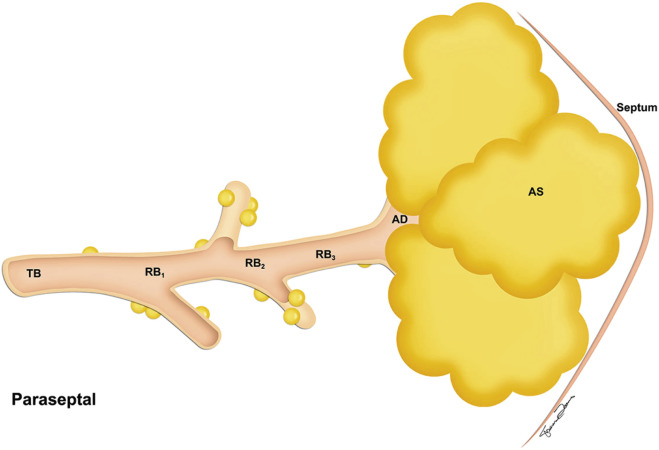
Paraseptal emphysema is characterized by involvement of the distal part of the secondary lobule (i.e., alveolar ducts and sacs), and it therefore occurs in a subpleural location ( Fig. 20.6 ). It can be seen in the periphery of the lung adjacent to the chest wall and along the interlobular septa and the fissures. Paraseptal emphysema, which can be an isolated phenomenon in young adults, is associated with spontaneous pneumothorax without other evidence of restriction in lung function. However, it can also be seen in older patients with centrilobular emphysema.
Paracicatricial or irregular emphysema refers to abnormal airspace enlargement associated with pulmonary fibrosis. This is most frequently a localized phenomenon and not necessarily associated with smoking or airflow obstruction.
Emphysema can be diagnosed by standard radiography when the disease is severe. If the lungs are mildly affected by emphysema, the chest radiograph is usually normal. Only about half of cases of moderately severe emphysema are diagnosed radiologically. The standard radiograph is not considered a reliable tool for diagnosing and quantitating emphysema. However, certain radiographic signs are accurate in the diagnosis of emphysema. The first sign is overinflation of the lungs. Similar radiographic findings are seen in patients with severe asthma, but the signs of overexpansion abate with clinical improvement. In emphysema, they persist. The second major sign of emphysema is a rapid tapering and attenuation of pulmonary vessels accompanied by irregular radiolucency of affected areas ( Fig. 20.7 ). Although this is an important radiographic finding, it is subjective and difficult to detect before the disease is severe. Localized lucent areas, particularly if they are surrounded by consolidation, may be apparent in the periphery of the lungs.
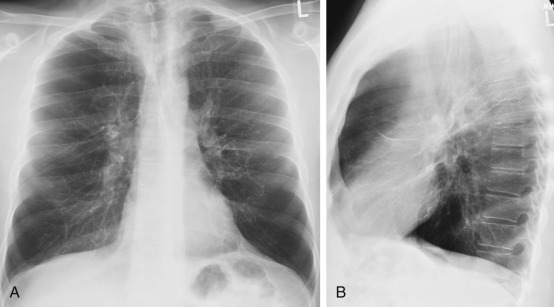
Bullae may occur in emphysema ( Figs. 20.8 and 20.9 ). A bulla is a sharply demarcated area of emphysema that is 1 cm or more in diameter and that has a wall less than 1 mm thick. Evidence of bullae should be sought on standard radiographs to support the diagnosis of emphysema. Bullae reflect only locally severe involvement and do not necessarily mean that the disease is widespread.
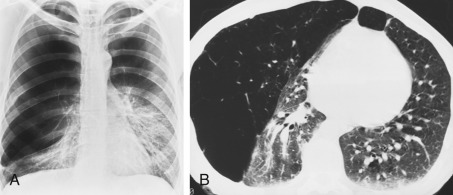
Computed tomography is superior to chest radiography in showing the presence, extent, and severity of emphysema. CT has high sensitivity and specificity for emphysema. It is also possible with CT to distinguish among the anatomic types of emphysema (see Box 20.2 ).
Centrilobular emphysema is characterized on CT by the presence of multiple small round areas of abnormally low attenuation that are several millimeters to 1 cm in diameter and distributed throughout the lung, usually with an upper lobe predominance ( Fig. 20.10 ). The centrilobular location of these lucencies can sometimes be recognized. The lucencies tend to be multiple, small, and “spotty.” Classically, the areas of low attenuation of centrilobular emphysema lack visible walls. A central dot, representing the central bronchovascular bundle, is sometimes visible. As the disease becomes more severe, the areas of lung destruction become more confluent, and the centrilobular distribution may no longer be recognizable. The HRCT appearance can then closely simulate panlobular emphysema.
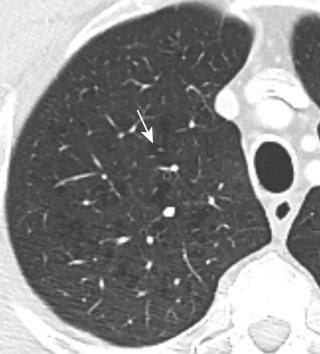
Panlobular emphysema is characterized on CT by the presence of fewer and smaller-than-normal pulmonary vessels ( Fig. 20.11 ). It is almost always more severe in the lower lobes but may appear diffuse. When it is advanced, extensive lung destruction can be identified, and the associated paucity of vessels is readily detectable. However, in moderate disease, increased lucency of the lung parenchyma and a limited, slight decrease in the caliber of the pulmonary vessels may be more difficult to recognize. When lower lobe–predominant panlobular emphysema is identified, the possibility of AATD should be raised. The disease can be confirmed by genetic and blood tests, and treatments with smoking cessation and possible AAT augmentation therapy can be initiated.
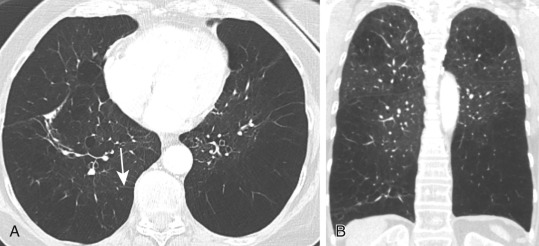
Paraseptal emphysema ( Fig. 20.12 ) is easily detected by CT. The appearance is that of multiple areas of subpleural emphysema, often with visible thin walls that correspond to interlobular septa. The emphysema is localized in the subpleural zones and along the interlobar fissures. When larger than 1 cm, areas of paraseptal emphysema are most appropriately called bullae . Subpleural bullae are manifestations of paraseptal emphysema, although they may be seen in all types of emphysema. They are often associated with spontaneous pneumothorax.
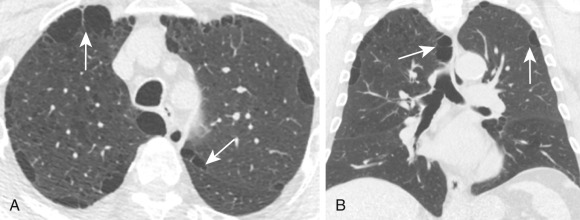
Paracicatricial or irregular emphysema ( Fig. 20.13 ) is focal emphysema that is usually found adjacent to parenchymal scars in diffuse pulmonary fibrosis, sarcoidosis, and pneumoconioses, particularly when progressive, massive fibrosis is present. It is usually recognized on CT when the associated fibrosis is identified.
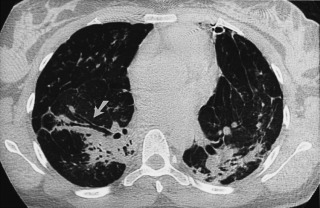
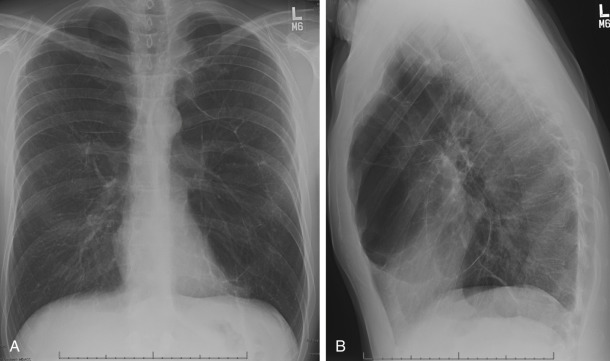
Bullous emphysema does not represent a specific pathologic entity but refers to the presence of emphysema associated with large bullae. Bullae can develop in association with any type of emphysema, but they are most common in paraseptal emphysema and centrilobular emphysema. A bulla is a sharply demarcated area of emphysema that is more than 1 cm in diameter and that possesses a well-defined wall less than 1 mm thick. Bullae occasionally can become quite large and may be rather focal and may result in atelectasis of normal adjacent lung; occasionally, they are not associated with diffuse emphysema. Large bullae may result in compromised respiratory function. This syndrome has been referred to as bullous emphysema, vanishing lung syndrome, and primary bullous disease of the lung (see Fig. 20.8 and Fig. 20.14 ). This entity may occur in young men and is characterized by large progressive upper lobe bullae. Most are smokers, but the entity may occur in nonsmokers.
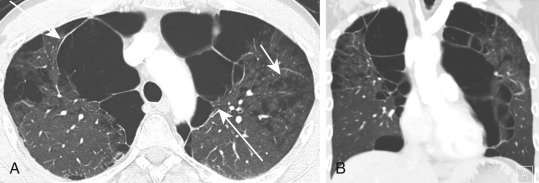
The diagnosis of emphysema is usually based on a combination of clinical features, a smoking history, and compatible pulmonary function abnormalities. However, HRCT may be useful in diagnosing patients whose clinical findings suggest another disease process, such as interstitial lung disease or pulmonary vascular disease. For these patients, HRCT can be valuable in detecting the presence of emphysema and in excluding other abnormalities in the chest. More recently, combined pulmonary fibrosis and emphysema is increasingly recognized in smokers. The presence of both obstructive and restrictive lung diseases can mask the degree of airway obstruction on PFT, which often shows disproportionately impaired D LCO , and CT is helpful in uncovering the abnormalities. With increasing utilization of chest CT for various clinical indications, such as evaluating traumatic injuries and screening for lung cancer, emphysema is sometimes incidentally detected before clinical symptoms become apparent.
With its excellent capability in showing the distribution of emphysema, CT plays an important role in the preoperative evaluation of patients with severe emphysema who are selected for lung volume–reduction surgery (LVRS). This surgery typically involves the resection of target areas of emphysema to reduce lung volume, restore elastic recoil, and improve ventilation–perfusion matching. In selected patients, this procedure has improved respiratory mechanics, diminished oxygen requirement, reduced dyspnea, increased exercise capacity, improved the quality of life, and extended survival. Based on the results of the National Emphysema Treatment Trial, which randomized patients to LVRS or to maximal medical therapy, patients with upper lobe–predominant emphysema are candidates for this procedure.
Various bronchoscopic techniques have been introduced as an alternative to LVRS. Most of these methods attempt to occlude the conducting airways to regions of severe emphysema by using a variety of techniques, including endobronchial airway sealants, coils, and one-way valves. The goal of most of these approaches is to achieve atelectasis of the targeted lung segments, resulting in nonsurgical volume reduction. Similar to LVRS, these methods are designed to treat patients with heterogeneous distribution of emphysema rather than diffuse disease. In contrast, airway bypass is a new bronchoscopic technique under investigation for treating patients with homogeneous distribution of emphysema. This method aims to decrease hyperinflation by placing shunts between the lung parenchyma and central airways, creating new conducting expiratory airways to bypass high-resistance, collapsing airway segments. CT plays an important role in preprocedural delineation of distribution of emphysema and patient selection. The presence of incomplete fissures should be reported because collateral ventilation through bridging lung parenchyma can prevent successful collapse of the targeted lobe with endoscopic lung reduction. CT is also useful for evaluation of procedural effectiveness and complications.
Emphysema is an independent risk factor for lung cancer, and about 5% of patients evaluated by CT before LVRS had incidentally detected lung cancer. When evaluating patients with emphysema, it is important to report incidental lung nodules and recommend appropriate imaging follow-up.
Several HRCT methods are available to quantitate the degree and severity of emphysema. The most common quantitative method is lung densitometry analysis. This technique provides histograms of lung attenuation values and calculates the area of lung occupied by pixels within a predetermined range of attenuation values. A percentage of low-attenuation area under -950 Hounsfield units has been shown to have the strongest associations with microscopic and macroscopic emphysema. Previous studies have shown that visual and quantitative CT methods for estimating severity of emphysema correlate well with findings on pathologic specimens of emphysematous lungs. However, quantitative methods have been shown to provide more objective and reliable assessment than visual scoring.
Although not routinely employed, postprocessing of CT data using a minimum-intensity projection technique, which highlights the lowest attenuation voxels on CT, has been shown to enhance the detection of mild emphysema ( Fig. 20.15 ). This method may help to improve the detection of mild emphysema when conventional CT images are equivocal.
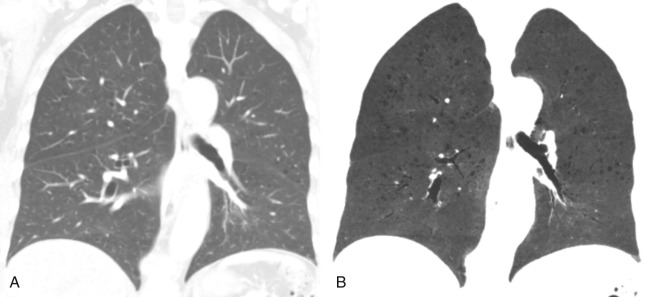
Several entities should be considered in the differential diagnosis of emphysema on HRCT. The first is honeycombing, which occurs in pulmonary fibrosis and is characterized by areas of subpleural cystic lesions that somewhat mimic the appearance of paraseptal emphysema. However, honeycomb cysts are usually smaller; occur in several layers along the pleural surface, often localized in the lung bases; and are associated with other findings of fibrosis. Paraseptal emphysema is often associated with bullae, and the areas of emphysema are larger and occur in a single layer. They predominate in the upper lobes without evidence of fibrosis.
The second feature to be considered in the differential diagnosis of emphysema is pneumatoceles. A pneumatocele is a thin-walled, gas-filled space within the lung that usually is associated with a prior pneumonia or a pulmonary laceration. It can be identical to a bulla on CT. However, the association with previous infection or trauma should suggest the diagnosis.
The third entity is cystic lung disease. Multiple thin-walled lung cysts can be seen in a variety of disorders, such as lymphangioleiomyomatosis and Langerhans cell histiocytosis. These cysts usually can be differentiated from centrilobular emphysema because the walls of cysts are more distinct and appear larger. When the lucency can be clearly identified as within the center of the pulmonary lobule, it is diagnostic of centrilobular emphysema. Cystic bronchiectasis can be readily differentiated from lung cysts and emphysema based on connectivity with the proximal airways and a constant relationship with its accompanying pulmonary artery.
The fourth possibility is bronchiolitis obliterans. Constrictive bronchiolitis, a disease of the small airways described in more detail later in this chapter, can result in increased lung volume and oligemia that is similar to that of panlobular emphysema. However, it usually has a patchy distribution, which is an important distinguishing feature.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here