Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Hemostasis occurs in response to vessel injury. The clot is essential both for the prevention of blood loss and the initiation of the wound repair process. When there is a lesion present in the blood vessel, the response is rapid, highly regulated, and localized. If the process is not balanced, abnormal bleeding or nonphysiological thrombosis, can result. In cardiovascular disease, the formation of abnormal thrombus at the area of an atherosclerotic plaque results in significant morbidity and mortality. This chapter will focus on normal mechanisms of hemostasis with specific attention to the role of the platelet in the process, as well as the coagulation cascade, and fibrinolytic mechanisms, as a basis for understanding how abnormalities in these processes can lead to thrombotic and hemorrhagic disorders.
Platelets are anucleate cells that are produced by megakaryocytes in the bone marrow. Once they have traversed from the bone marrow to the general circulation, their lifespan is approximately 10 days. They function mainly to limit hemorrhage after trauma resulting in vascular injury. Normally in the vasculature, platelets are in a resting state and only become activated after exposure to a stimulus leads to a shape change and a release reaction that causes the platelets to export many of its biologically important proteins. Some of the agonists that can initiate this response include thromboxane A 2 , adenosine diphosphate (ADP), thrombin, and serotonin. In areas of vascular injury, platelets are attracted to the impaired site by collagen through binding with von Willebrand factor via the glycoprotein Ib/V/IX (GPIb/V/IX) complex. This initial binding results in platelet activation with a subsequent feedback mechanism in which ADP, thrombin, and thromboxane A 2 further activate the platelets and recruit additional platelets to the area. The complex firmly binds the platelet to the area of injury so that there is no disruption by the high shear forces of turbulent blood flow that occur with vessel disruption. This amplification of the response is essential to form a hemostatic plug, and represents the first stage in the hemostatic process. When von Willebrand factor is not present, hemostatic abnormalities result with deficiencies leading to von Willebrand disease, which can be associated with severe bleeding. Hemostasis issues also arise when the platelet receptor complex GPIb/V/IX is mutated, resulting in the inability for von Willebrand to bind, a disorder termed Bernard-Soulier syndrome.
Additional platelet aggregation occurs through the activation of G-protein–coupled receptors with the final pathway relying on the glycoprotein IIb/IIIa complex, which is the main receptor for platelet aggregation and adhesion. Fibrinogen tethers glycoprotein IIb/IIIa complexes on different platelets stabilizing the clot. The integral role of this receptor is manifest in Glanzmann thrombasthenia, a disorder in which fibrinogen binding is impaired leading to mucocutaneous bleeding episodes that occur spontaneously.
Recently, the role of the platelet has been expanded to include a subset of platelets termed procoagulant platelets. The main characteristic of this subtype of platelets is the presence of phosphatidylserine (PS) on the plasma membrane presenting a surface for binding of tenase and the prothrombinase complexes, thereby upregulating coagulation. These platelets have been characterized as ballooning platelets or as containing the mitochondrial permeability transition pore. This subgroup of platelets is dependent on collagen as the main agonist that regulates their phenotype making them ideal to accelerate hemostasis at the site of vessel injury.
The vascular endothelium is essential to this hemostatic process as this is the cellular site at which the regulation and initiation of coagulation begins. Endothelial cells modulate vascular tone, generate mediators of inflammation, and provide a resistant surface that allows for platelets to experience laminar flow with minimal shear. The endothelial cells regulate hemostasis through the release of a number of inhibitors of platelets and of inflammation. The vascular endothelium is essential for regulating uncontrolled platelet activity through mechanisms of inhibition including the arachidonic acid-prostacyclin pathway, the l -arginine-nitric oxide pathway, and the endothelial ectoadenosine diphosphatase (ecto-ADPase) pathways ( Table 4.1 ).
| Prohemostatic | Antihemostatic |
|---|---|
| Circulating | |
| α 2 -Antiplasmin Thrombin Thrombin-activatable fibrinolysis inhibitor (TAFI) |
Antithrombin III Protein C Protein S Tissue factor pathway inhibitor (TFPI) |
| Endothelium-Derived | |
| Plasminogen activator inhibitor-1 (PAI-1) Tissue factor (TF) von Willebrand factor (vWF) |
Ecto-ADPase/CD39 Heparan sulfate Nitric oxide (NO) Prostacyclin (PGI 2 ) Thrombomodulin Tissue plasminogen activator (tPA) Urokinase plasminogen activator (uPA) |
Nitric oxide (NO) is produced constitutively by endothelial cells via an endothelial isoform of nitric oxide synthase (eNOS) in a process that is dependent on the conversion of l -arginine to l -citrulline. Vascular tone is regulated by NO as it controls smooth muscle cell contraction. It also inhibits platelets directly, thereby blocking platelet aggregation through the stimulation of guanylyl cyclase and cyclic guanosine-monophosphate (cGMP) formation and the inhibition of platelet phosphoinositol-3-kinase (PI-3 kinase). NO functions by decreasing the intracellular Ca 2 + level through cGMP, which inhibits the conformational change in glycoprotein IIb/IIIa suppressing fibrinogen’s ability to bind to the receptor, thereby attenuating platelet aggregation.
Prostacyclin, which is synthesized in the endothelial cells from arachidonic acid through cyclooxygenase-1 or -2 (COX-1 and COX-2)-dependent pathways, inhibits platelet function through increasing cyclic adenosine-monophosphate (cAMP). This is essential for aspirin’s ability to diminish platelet function through the acetylation of platelet COX-1 at serine 529.
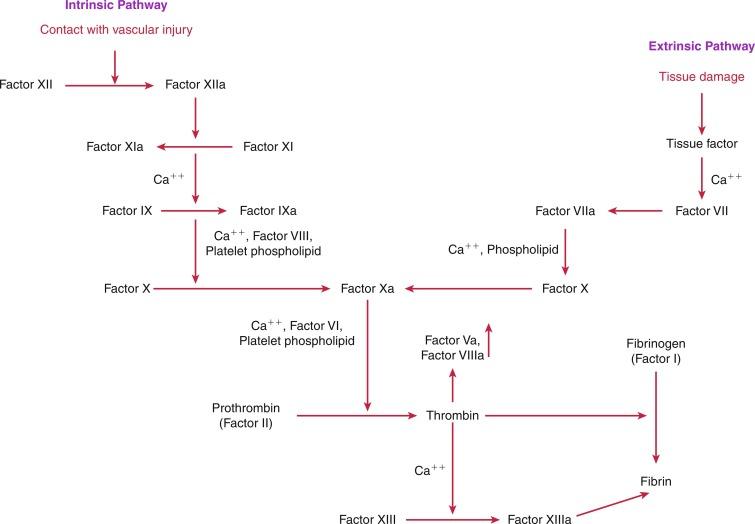
The last pathway important in modulation of the vascular endothelium’s interaction with platelets is the endothelial ecto-ADPase pathway, which impairs ADP-mediated platelet activation. By hydrolyzing ADP, this enzyme inhibits the critical state of platelet recruitment to a growing aggregate, thereby limiting thrombus formation. Once the platelet aggregate has been stabilized by fibrin with red cells to the vessel wall, the next stage of hemostasis involves activation of the highly regulated coagulation cascade ( Fig. 4.1 ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here