Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Three main histologic patterns of injury are encountered in the setting of airways disease; minimal change, nodules, and fibrosis. Minimal change is the classic pattern encountered in the setting of small airways disease or constrictive bronchiolitis from any etiology. In this setting it is important to examine the bronchovascular bundle for subtle histologic evidence of injury, including loss of associated airways. Nodules, especially in a bronchovascular distribution, can be a strong clue for an airway or inhalational injury process. Finally, many airways diseases can result in centrilobular fibrosis and fibrosis can be the dominant pattern of injury at scanning magnification.
Nonneoplastic diseases affecting the trachea and bronchi are commonly related to infections, inhalation exposure or, more rarely, to developmental anomalies and systemic diseases; they may also involve the larynx ( Box 9.1 ). The effect of these diseases is usually luminal obstruction or collapse, whereas they rarely present as circumscribed nodular lesions or marked dilation of the tracheal and bronchial lumen.
Asthma
Bronchitis related to:
Infection
Inhalational injury (including smoking)
Collagen vascular disease
Aspiration
Miscellaneous inflammatory diseases (e.g., inflammatory bowel disease)
Bronchiectasis
Bronchocentric granulomatosis
Mucoid impaction of the bronchi
Allergic bronchopulmonary fungal disease
Plastic bronchitis
Fistulas
Congenital bronchial cartilage deficiency
Relapsing polychondritis
Broncholithiasis, bronchostenosis
Miscellaneous conditions (tracheobronchial amyloid, tracheobronchomegaly, tracheomalacia, tracheobronchopathia osteochondroplastica)
Nonneoplastic pathology of the trachea can be divided broadly into extrinsic and intrinsic diseases. Among extrinsic diseases are certain infections, such as rhinoscleroma (caused by Klebsiella rhinoscleromatis ) ( Fig. 9.1 ) and COVID-19 (caused by SARS-CoV-2) that frequently present acute tracheobronchitis independent from intubation or superimposed pneumonia, , and systemic autoimmune diseases that affect cartilage (e.g., granulomatosis with polyangiitis, formerly called “Wegener granulomatosis” , ). These diseases may cause tracheal obstruction or collapse and may also involve the larynx, bronchi, or both. Diseases intrinsic and unique to the trachea are few; they usually also involve the large bronchi and are mainly of congenital and/or metabolic origin. The following section describes three of these entities: tracheobronchial amyloidosis, tracheobronchomalacia, and tracheobronchopathia osteochondroplastica. The last of these may be rare but deserves mention because it can be encountered incidentally during bronchoscopy.
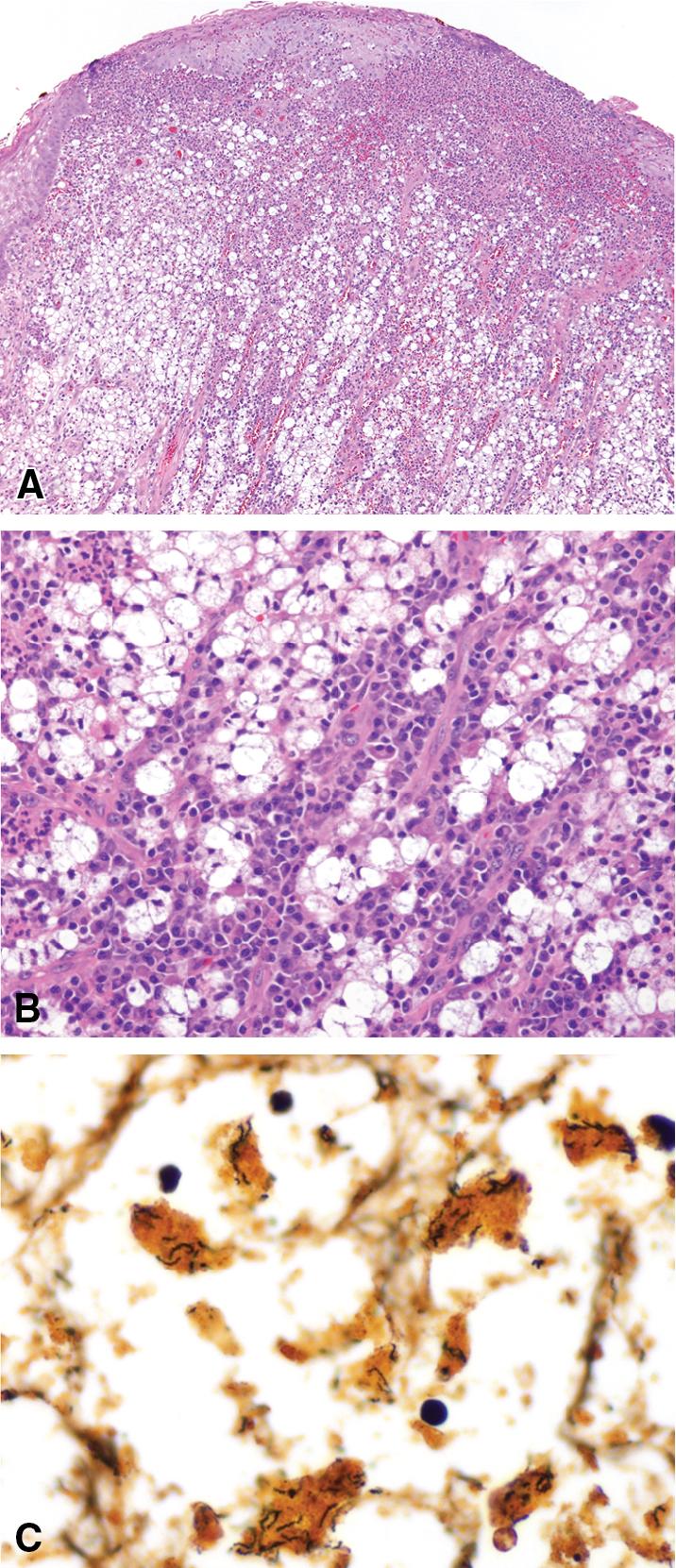
Tracheobronchial amyloidosis (TBA) is the least frequent form of pulmonary amyloidosis and represents a localized, idiopathic form of AL amyloidosis, which is characterized by amyloid deposition in various segments of the tracheobronchial tree. , The alveolated parenchyma is typically not involved; rare cases of tracheal amyloidosis with laryngeal involvement have been described. ,
TBA typically presents in middle-aged males (2:1 predilection vs. females) and the most common symptoms are: dyspnea, cough, wheezing (sometimes misdiagnosed as asthma), and occasionally hemoptysis, but patients could also be asymptomatic. , TBA determines narrowing of the airways, which may cause recurrent pneumonias and parenchymal atelectasis, or rarely it may present as yellowish tumor-like nodules bleeding on touch. Three patterns of involvement have been described: proximal, mid-, and distal airway disease. Bronchoscopy with transbronchial biopsy is the gold standard to obtain definite diagnosis, while computed tomography (CT) is helpful to determine the extent of the disease. Thoracic imaging may show circumferential airway thickening, tracheal stenosis, calcified extraluminal deposits or mediastinal lymphadenopathy. Pulmonary function tests usually show decreased airflows in proximal airway disease, whereas patients with distal airway disease have normal airflow. , , , TBA can be associated with tracheobronchomegaly, a rare disorder characterized by marked dilation of the trachea and bronchi. On histopathologic examination, amyloid deposits are usually seen in the submucosa and may involve the entire tracheal or bronchial wall, occurring in irregular masses or sheets ( Fig. 9.2 ). The deposits are composed of homogeneous amorphous eosinophilic extracellular material that shows apple-green birefringence with polarized light when stained with Congo red. Amyloid may be accompanied by multinucleated foreign-body type giant cells, calcification, ossification, or plasma cells ( Fig. 9.3 ). Small submucosal vessels are involved in most cases. Of the three major types of amyloid, amyloid light chain (AL), serum amyloid A (AA), and transthyretin, the AL type is the most common in tracheobronchial amyloid deposits. TBA is typically an organ-limited disease; however, systemic amyloidosis should be ruled out through serum and urine electrophoresis tests for monoclonal proteins and echocardiography. Lysozyme amyloidosis, a rare autosomal dominant hereditary systemic amyloidosis involving the airway, has been described in patients coming from Piedmont (Italy). The prognosis is variable with a mortality rate from 30% to 50% after 5 to 10 years. Therapy is limited to debulking procedures and stent placement for localized lesions, although some evidence suggests that radiation therapy and cryotherapy may provide more definitive treatment. ,
The key histopathologic features of TBA are:
Submucosal and perivascular deposits of amorphous eosinophilic material.
Foreign-body giant-cell reaction.
Calcification, ossification, or lymphoplasmacytic infiltrate.
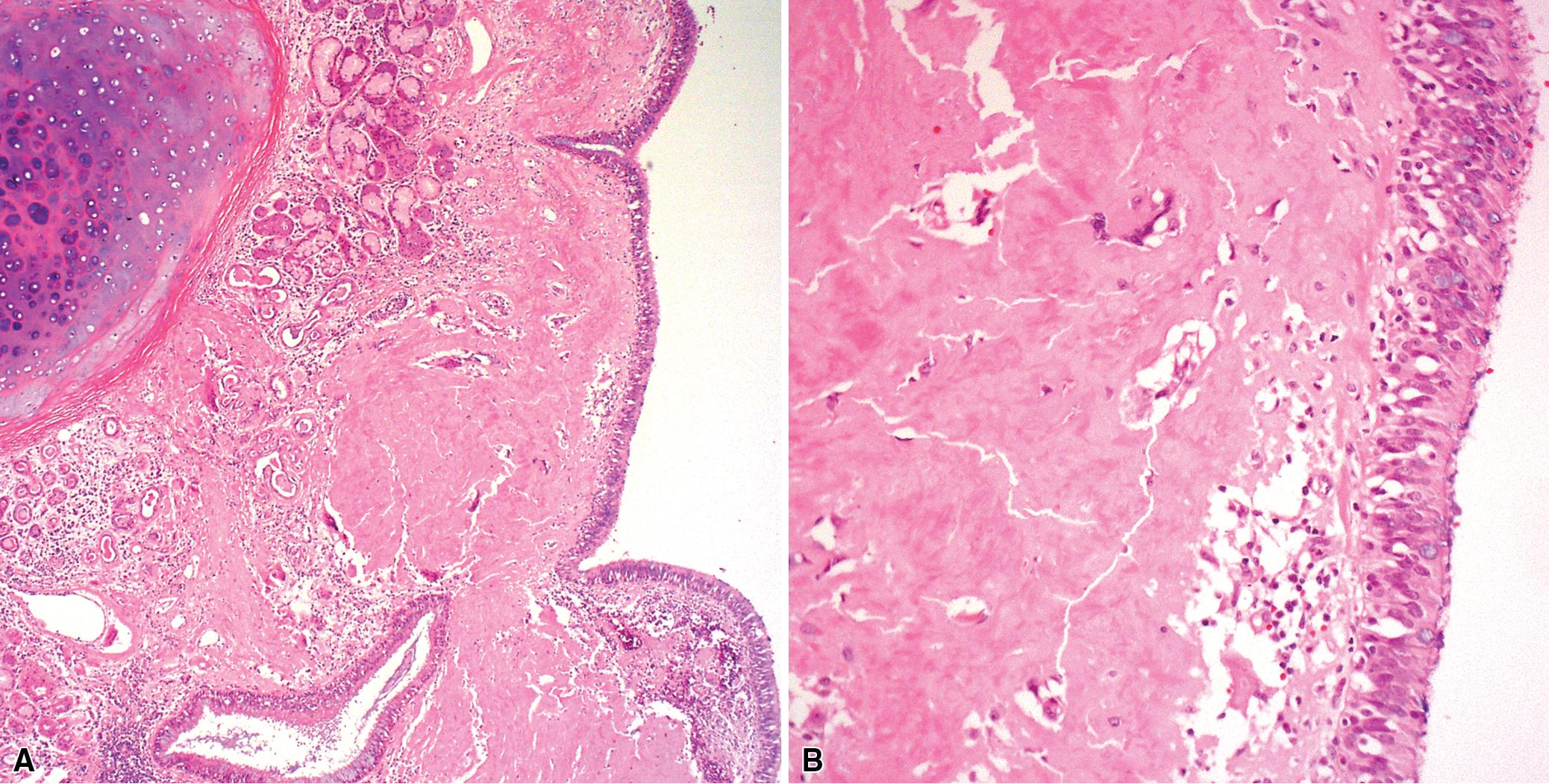
Tracheobronchomalacia (TBM) is a term used to encompass a number of conditions characterized by a decrease in the structural rigidity of the trachea and conducting airways caused by intrinsic tracheal weakness, some forms of tracheal deformations or extrinsic compression. Such changes are manifested clinically by excessive expiratory collapse of the central airways owing to increased flaccidity or redundancy of the posterior membranous airway wall or to weakness of the supporting cartilage. The clinical presentation of TBM includes a range of non-specific respiratory symptoms such as barking cough with expiratory rhonchi or inspiratory stridor, recurrent or persistent pneumonias due to reduced clearance of secretions, exercise intolerance, hypoxic episodes, or apneic events exacerbated by any conditions that increase the intrathoracic pressure such as coughing, crying, Valsalva maneuvers, feeding, forced expiration, or lying supine. The diagnosis should be suspected clinically; however, the most accurate diagnosis is reached through direct visualization of the tracheobronchial tree by flexible or rigid endoscopy, and three-phase dynamic bronchoscopy is considered the gold standard. There are no standard criteria for the diagnosis, but generally more than 50% dynamic narrowing in the airway lumen with forced exhalation or coughing is considered necessary to diagnose TBM. Although typically a disease process of the central airways, TBM has been shown to frequently have associated air trapping on high-resolution CT (HRCT) scan, an indicator of small airway disease (SAD). ,
Both children and adults may be affected. In children, the disease is most often related to prematurity and the requirement for prolonged mechanical ventilation. In these patients, TBM appears to be self-limited. However, TBM is also the most frequent congenital tracheal anomaly, affecting 1:2100 children and often associated with other congenital anomalies or syndromes. In adults, TBM is seen in the middle-aged and elderly and is a progressive condition, commonly seen in association with chronic obstructive pulmonary disease (COPD). Other reported associations in adults include prolonged intubation, vascular anomalies, and exposure to mustard gas. TBM may complicate TBA and rheumatoid arthritis. TBM has been reported in the literature in up to 50% of patients with relapsing polychondritis, a systemic disease affecting the cartilaginous tissue in several organs, including the airways, and can be the only manifestation of the disease. , Pathologically, the tracheal wall is soft as a result of inflammatory destruction of the cartilage ( Fig. 9.4 ). Over time, the cartilage is replaced by fibrous tissue, accompanied by inflammatory infiltration and reparative vascular proliferation.
Treatment is variable and depends on symptom severity, with conservative therapy appropriate for mildly symptomatic cases (nebulizer treatments, minimal use of inhaled corticosteroids, gastroesophageal reflux disease [GERD] therapy and continuous positive airway pressure [CPAP]), and more invasive therapy (including tracheobronchoplasty, anterior and posterior tracheobronchopexy and anterior and posterior aortopexy) indicated for severely symptomatic cases for reduction of expiratory tracheal collapse and symptomatic improvement. , , In the absence of treatment, severely symptomatic TBM can lead to significant respiratory morbidity and may rarely be fatal.
The key histopathologic features of TBM are:
Inflammatory and fibrous replacement of the airway cartilage plates.
Granulation tissue with vascular proliferation.
Tracheobronchomegaly (TBMe), also known as Mounier–Kuhn syndrome , is a rare disorder characterized by marked dilation of the trachea and central bronchi with thinning of the wall and loss of elastic fibers. The age of patients varies greatly from 18 months to 86 years old, there is a strong male predominance (8:1), and most of the patients are smokers. A recently proposed classification scheme grouped the affected individuals according to clinical features, as follows: type 1A is infants who developed TBMe consequent to fetal tracheal occlusion; type 1B is infants and children who developed TBMe after prolonged intubation; type 2 individuals developed TBMe following recurrent pulmonary infections (2A) or pulmonary fibrosis (2B); type 3 represents TBMe with evidence of extra-pulmonary elastolysis; and type 4 groups patients without clear predisposing factors. The disease is sometimes associated with congenital connective tissue diseases (CTDs) such as Ehlers-Danlos syndrome, cutis laxa, Marfan syndrome, and Kenny-Caffey syndrome, as well as rheumatologic disorders like ankylosing spondylitis or rheumatoid arthritis. , The diagnostic gold standard is the CT, by measuring the diameter of the trachea (>27 mm in males; >23 mm in females). A suggestion to classify cases of TBMe according to anatomical involvement has been made: type 1 is characterized by symmetrical diffuse enlargement of both trachea and bronchi; in type 2, which is the most common, the enlargement is more eccentric with pronounced diverticula and abrupt change to normal bronchial size; in type 3, the diverticula may extend into more distal bronchi.
TBMe is associated with impaired dynamic function, in particular dynamic collapse, and patients may present with varying degrees of recurrent infection, breathlessness, hemoptysis, and dyspnea. The most common respiratory complications are bronchiectasis, TBM, and emphysema, while the most common non-respiratory comorbidity is GERD, which increases the risk of aspiration.
Treatment is aimed at reducing infections (vaccinations, antibiotics) and improving airway clearance (mucolytics, chest physiotherapy).
The key histopathologic features of TBMe are:
Thinning of the muscular wall of the affected airway.
Atrophy of longitudinal muscle and loss of elastic fibers in the trachea and main bronchi.
Saccular diverticula between cartilages and bulging and spindle-shaped dilatation in the posterior walls of the trachea may be seen.
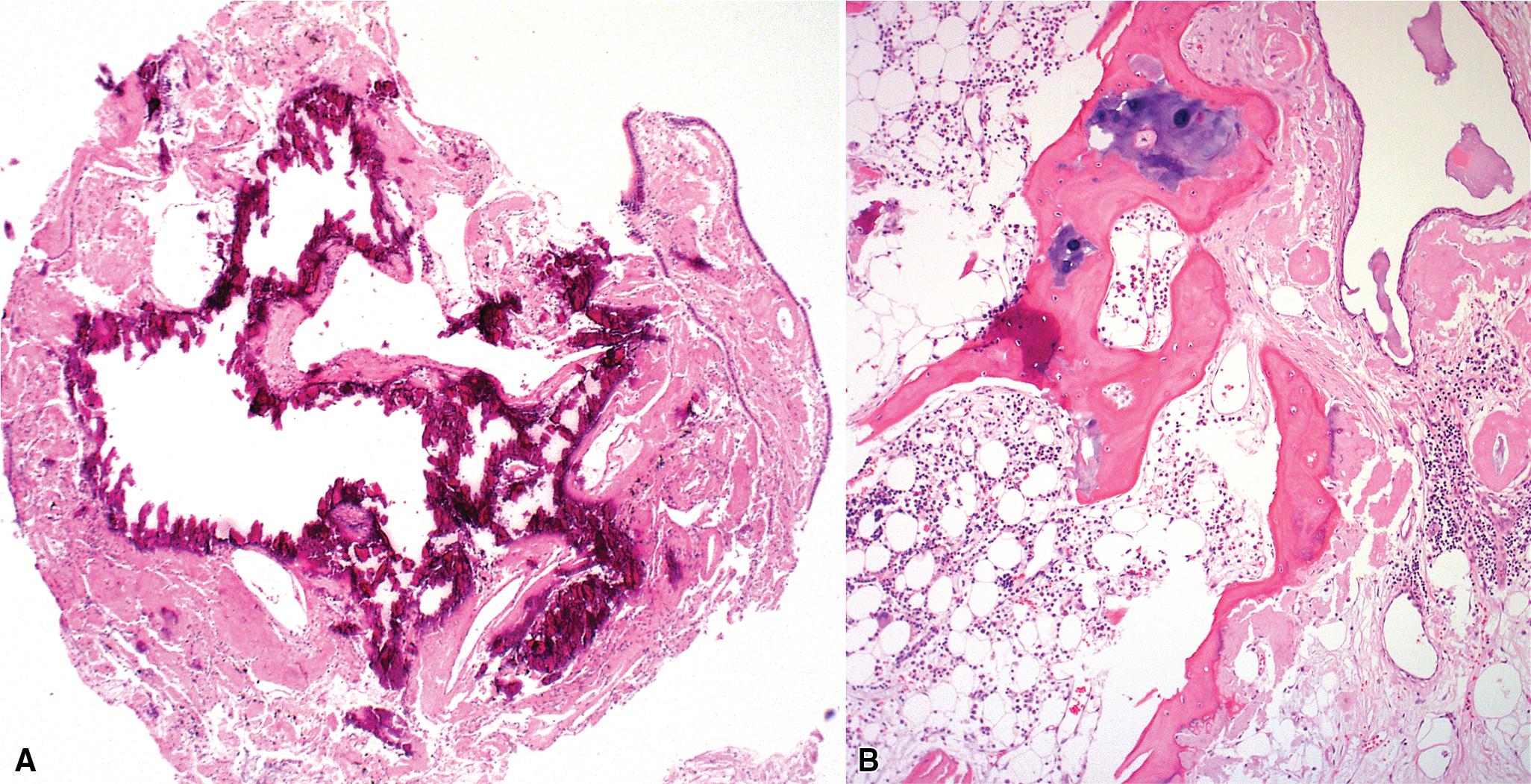
Tracheobronchopathia osteochondroplastica (TBO) is a rare, idiopathic disease characterized by the presence of cartilaginous or osseous submucosal nodules that bulge into the lumen of the trachea and bronchi ( Fig. 9.5 ) with sparing of the posterior membranous portion. The etiology and pathogenesis are unknown. The disease affects adults more commonly than children, with a predilection for males. Most cases are asymptomatic and are most often diagnosed incidentally during intubation or bronchoscopy. A minority of patients may experience cough, dyspnea, and wheezing which can be misdiagnosed as asthma, hemoptysis, or recurrent infections.
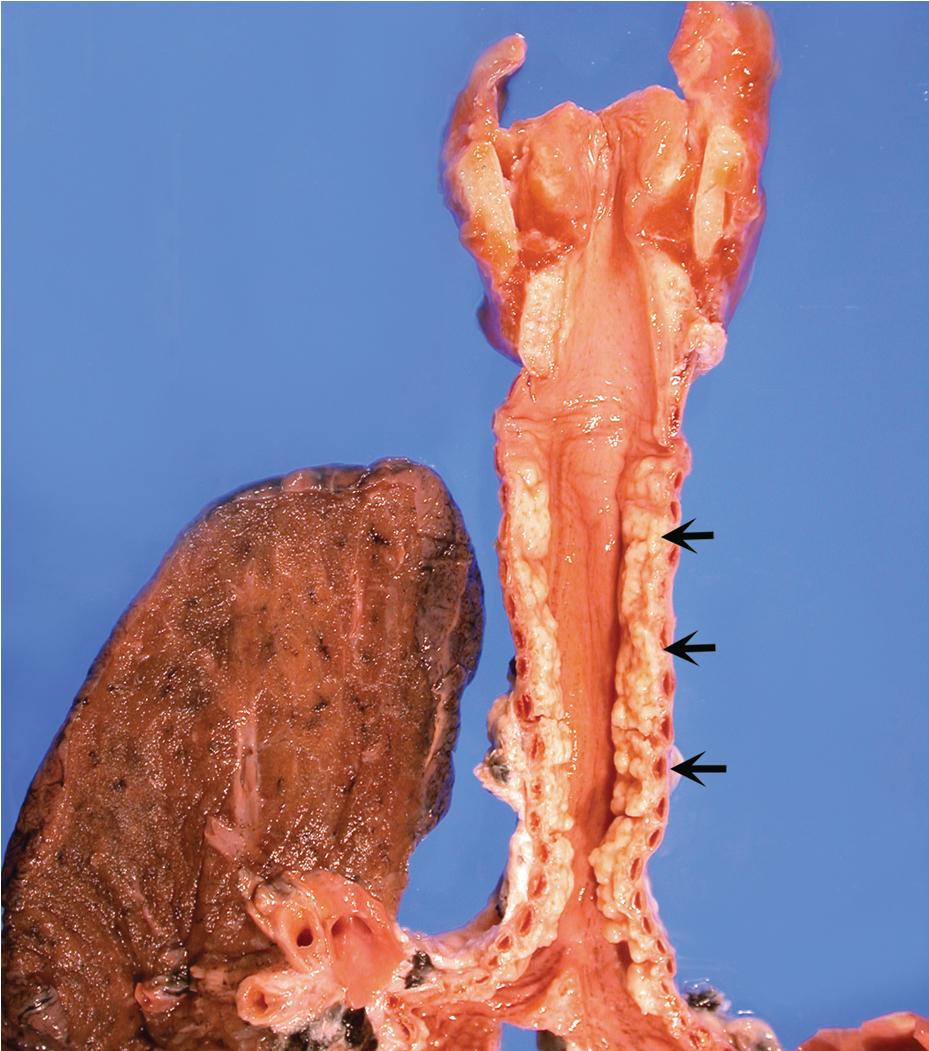
The bronchoscopic appearance of nodular, sessile, diffuse, and hard excrescences of 1 to 3 mm described as “stalactite cave,” “rock-garden,” or “cobble-stone” with sparing of the posterior membranous wall is usually diagnostic, and biopsy is seldom, if ever, required. In the rare bronchoscopic biopsy showing the cartilaginous or ossified lesions of TBO ( Fig. 9.6 ), the differential diagnosis includes ossified amyloid deposits, relapsing polychondritis and calcification due to ageing. Treatment is rarely required except in cases of severe airway obstruction where bronchoscopic dilation may be indicated.
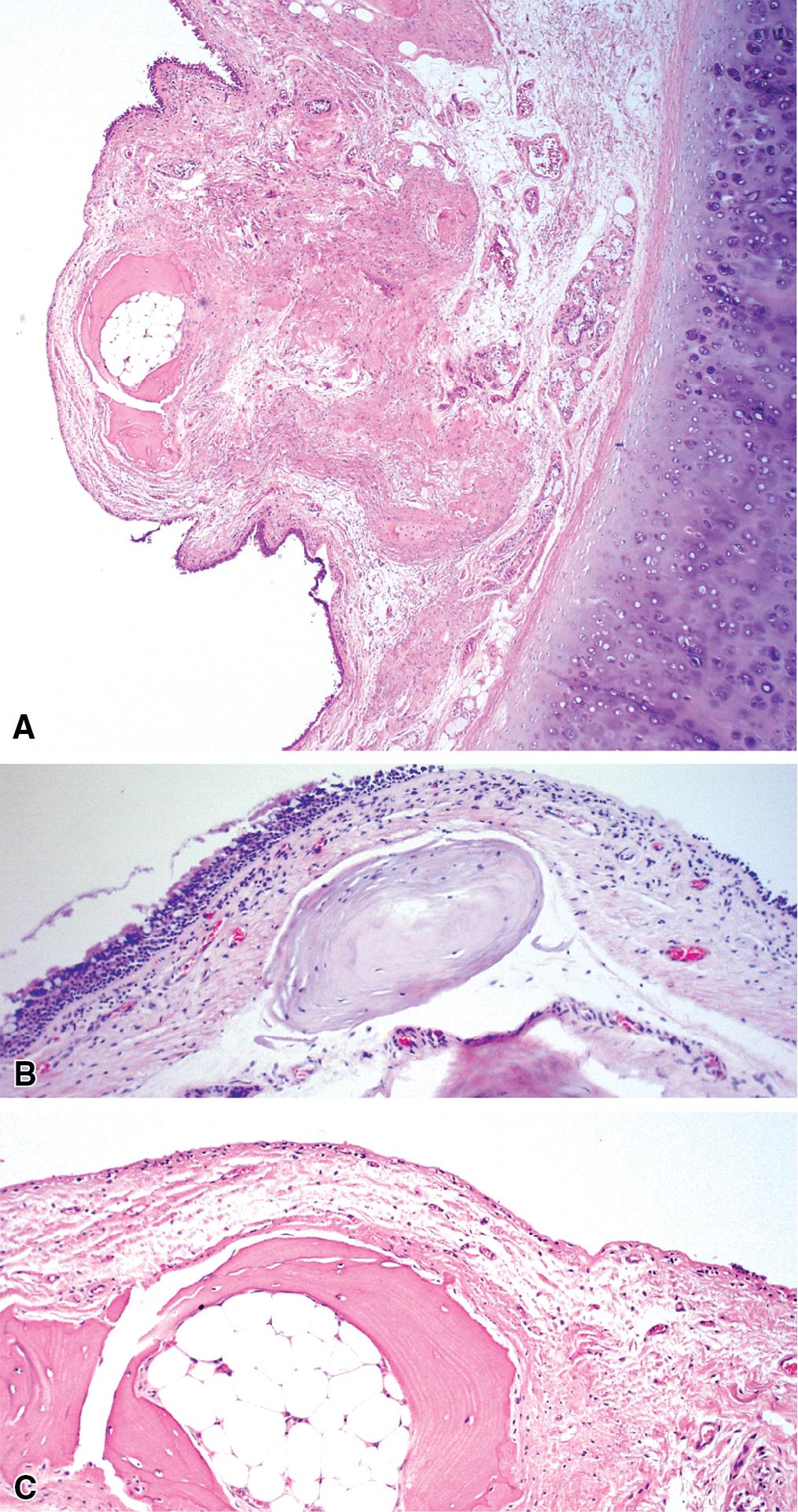
The key histopathologic features of TBO are:
Tracheal and bronchial submucosal nodules composed of metaplastic cartilage and bone (with or without bone marrow), with sparing of the posterior membranous wall of the trachea.
The nodules may have a rim of fibrous tissue continuous with the perichondrium of the cartilaginous plates.
Erosion/ulceration or metaplastic changes (squamous metaplasia) of the overlying mucosa may be seen.
The conducting airways begin at the carina and extend distally in the lung to the level of the noncartilaginous bronchioles. Although many diffuse lung diseases involve the large airways secondarily, some diseases may affect primarily the bronchi and are presented here. Asthma-associated diseases, typically observed in the large airways, are discussed later in the final section of this chapter. Cystic fibrosis and ciliary disorders are not discussed here because detailed expositions can be found in other sources. ,
Inflammatory conditions of the large airways are frequently seen in small biopsies and cytologic specimens but are rarely specific: in practice, the presence of acute or chronic inflammation in endobronchial biopsies demands a descriptive diagnosis ( Fig. 9.7 ). It is best to avoid the use of the term chronic bronchitis in reference to such inflammatory changes involving respiratory mucosa because it carries a clinical implication regarding the diffuse nature of the disease and specific clinical manifestations. When bronchitis occurs as a direct result of respiratory infection, necrosis of the mucosa may be present (acute necrotizing bronchitis). In the postinfection period, residual chronic inflammatory changes may be the dominant findings. Unfortunately the list of possible etiologic disorders associated with chronic inflammation in bronchial mucosa is quite long ( Box 9.2 ) and sufficiently diverse as to not be useful in narrowing the clinical differential diagnosis. For this reason, a careful search for other, more specific findings is always in order (e.g., vasculitis, granulomas, necrosis, tumor).
Infections
Aspecific reaction at the periphery of a localized process (tumor, abscess, etc.)
Hypersensitivity reactions
Collagen vascular disease
Allergic bronchopulmonary fungal disease
Bronchiectasis
Marginal zone lymphoma (of mucosa-associated lymphoid tissue [MALT])
Drug reactions
Toxic inhalation
COPA syndrome
Normal mucosa-associated lymphoid tissue (MALT)
Bronchiectasis is defined as a permanent dilation of the bronchi, often attended by acute and chronic inflammation. Conceptually, bronchiectasis can be considered the end result of any number of conditions that damage the airway wall and result in weakening over time and dilation.
In a significant percentage of cases no specific cause can be identified. , Among recognizable causes of bronchiectasis are infections, primary ciliary dyskinesia, immunodeficiency, cystic fibrosis, rheumatoid arthritis, inflammatory bowel disease and graft-versus-host disease. An intriguing implication of Helicobacter pylori in the pathogenesis of bronchiectasis has been raised in some studies. In specific conditions such as cystic fibrosis, bronchiectasis may dominate the pulmonary pathology. Bronchiectasis outside the setting of cystic fibrosis is often perceived to be rare in developed nations, but it remains an important cause of chronic suppurative lung disease in the developing world. The decline in hospital admissions for pediatric bronchiectasis in the developed nations has been noted since the 1950s and has been attributed to improvements in sanitation and nutrition, introduction of childhood immunization (particularly against pertussis and measles), and the early and frequent use of antibiotics.
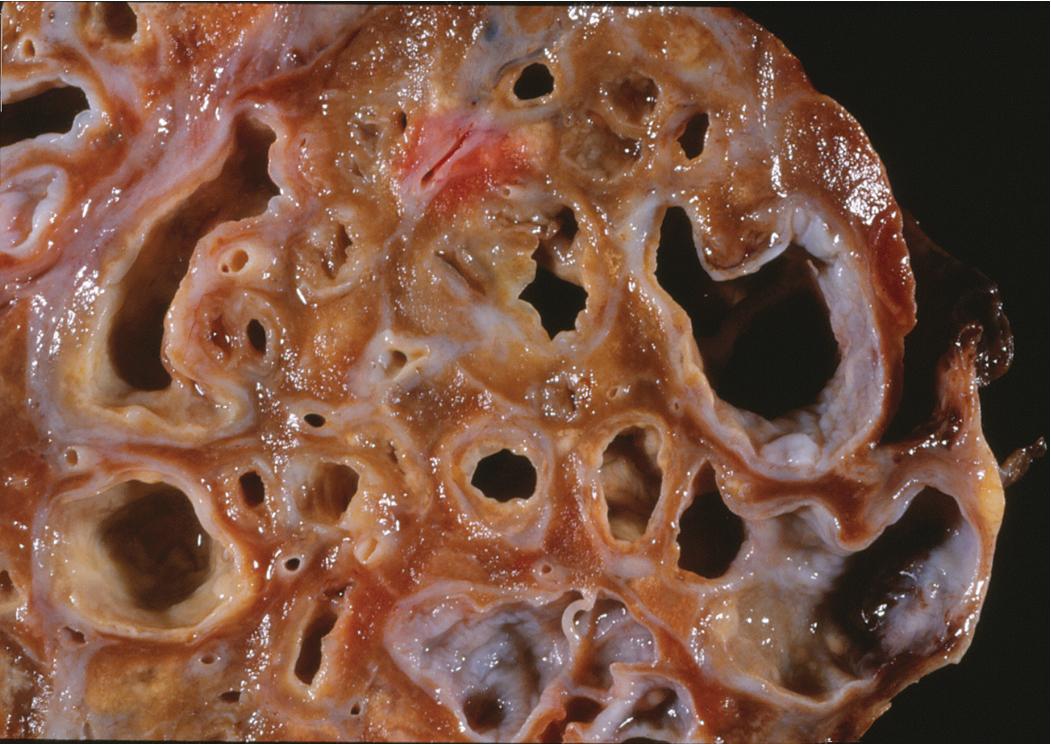
Bronchiectasis is a radiologic diagnosis in the living patient but has distinctive features in resected lungs ( Fig. 9.8 ) and lobes and sometimes in surgical biopsies. The historical classification of bronchiectasis divided the process into three types: saccular (progressive dilation of the bronchi from central to peripheral), varicose (combined dilation and constriction), and cylindrical (uniform dilation). Today most cases of bronchiectasis are diagnosed with great sensitivity by HRCT. The HRCT findings in bronchiectasis are summarized in Box 9.3 .
Lack of bronchial tapering
Presence of visible bronchi within 1 cm of the costal pleura
Visible bronchial lumens abutting mediastinal pleura, bronchi seen as horizontal parallel lines (“tram tracks”)
Bronchial diameter exceeding adjacent pulmonary diameter (“signet ring sign”)
Irregular bronchial dilatation
Clustered thin-walled cystic spaces with or without air-fluid levels
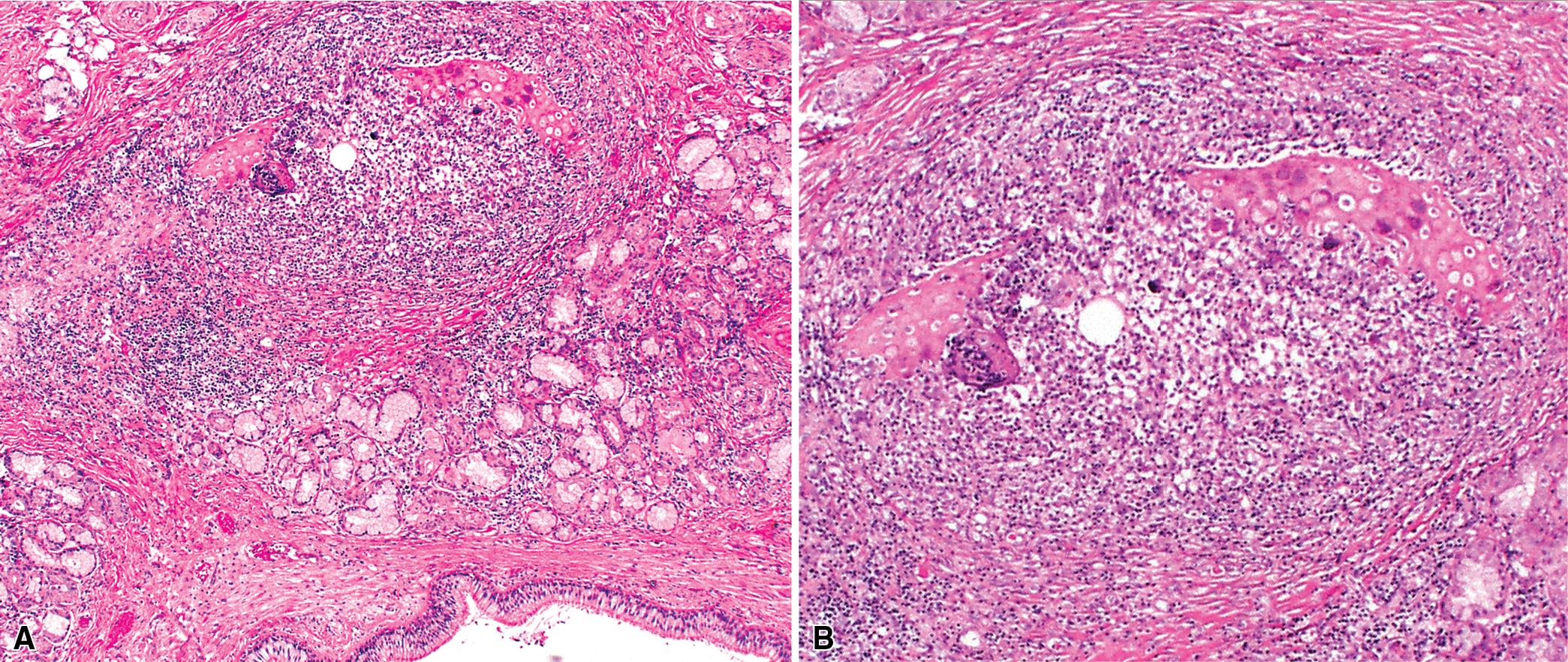
Presenting signs and symptoms include cough with production of purulent sputum, fever, shortness of breath, and occasionally hemoptysis. No age or sex predilection has been noted, and the disease tends to run a course of recurrent exacerbations, sometimes with superimposed infection. The gross findings in bronchiectasis have been well described in autopsy series. For the histopathologist, acute and chronic inflammatory changes dominate the bronchoscopic biopsy picture ( Fig. 9.9 ). These findings are not specific; however, bioptic specimens may be useful for ancillary laboratory studies (such as microbiologic investigations or ultrastructural studies in search of ciliary abnormalities). In most instances, even surgical lung biopsy findings reflect only downstream secondary manifestations of obstruction or infection occurring more proximally. Lymphoid hyperplasia surrounding the large airways may occur in bronchiectasis and bronchoscopically derived biopsies may sample such hyperplastic lymphoid tissue, raising concern for lymphoproliferative disease.
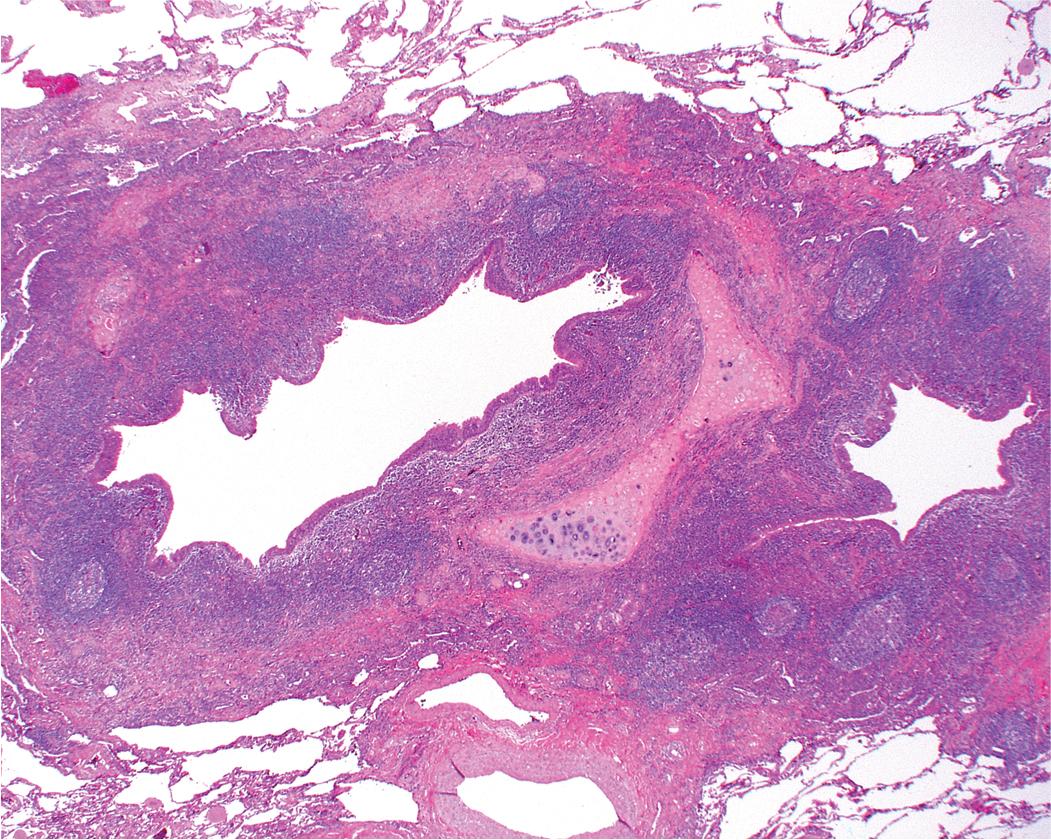
Middle lobe syndrome (MLS) is included in this disease category because the disorder is fundamentally one of chronic large airway obstruction with secondary bronchiectasis, bronchitis, and bronchiolitis-associated parenchymal changes that arise as additional downstream effects of these airway abnormalities.
MLS is characterized by recurrent or chronic atelectasis and collapse of the middle lobe of the right lung but can also involve the lingual of the left lung. There are two forms of MLS: obstructive and nonobstructive. Obstructive MLS is usually caused by endobronchial lesions or extrinsic compression of the middle lobe bronchus, frequently caused by hilar lymphadenopathy or tumors causing postobstructive atelectasis and pneumonitis. In the nonobstructive type of MLS, the middle lobe bronchus is patent, but several anatomical as well as functional factors make it susceptible to collapse. They include: the narrow diameter and long length of the middle lobe bronchus, its acute take-off angle from the intermediate bronchus and the relative anatomical isolation of the middle lobe and lingula. The above-mentioned factors may create a poor condition for drainage of secretions and lead to subsequent atelectasis when inflammation and/or edema due to lower respiratory tract infections intervene. The swelling of peribronchial lymph nodes due to granulomatous infections such as typical and atypical mycobacterial infection, histoplasmosis or other fungal infections; sarcoidosis; lymph node metastases or mediastinal tumors may cause the compression of the bronchus in extrinsic obstructive type of MLS. Tumors, both benign (hamartomas, polyps, adenomas) and malignant, (primary lung cancers and metastases) may be responsible of intrinsic obstruction. Other less common causes of obstruction of the right middle lobe bronchus include aspirated foreign objects (particularly in children), broncholiths that erode endobronchially from adjacent calcified lymph nodes, inspissated mucus associated with cystic fibrosis or allergic bronchopulmonary aspergillosis and cardiomegaly. In clinical practice, the disease occurs more frequently in adults and often without an identifiable obstructing lesion. The affected population consists predominantly of middle-aged or elderly women, and the condition has been referred to as Lady Windermere syndrome when correlated with intentional long-term cough suppression and isolation of Mycobacterium avium complex . Some patients are asymptomatic with incidental detection of the disease; however, most of the patients complain of chronic cough with sputum, hemoptysis, chest pain, dyspnea, and fever caused by recurrent chronic pneumonitis. When MLS occurs in children, it is often seen in asthmatic patients. Typical HRCT findings of MLS are: bronchiectasis in more than half of cases, atelectasis, consolidation and patchy infiltrates involving the right middle lobe or lingula. Chest HRCT is also informative in evaluating bronchial patency, lymph node enlargement and calcifications or other causes of extrinsic compression of the middle lobe airway. ,
In surgical lung biopsies of the right middle lobe or lingula, bronchiectasis, advanced remodeling, extensive bronchiolocentric inflammatory infiltrates and mucostasis, when present together, should always suggest the possibility of MLS ( Fig. 9.10 ). The presence of granulomas in the context of MLS should raise suspicion for colonization by atypical mycobacterial species, and acid-fast organisms may be identified in granulomas. Removal of any obstructing lesions, whether neoplasm or other, in early stages of the disease process may avoid the need for lobectomy. Patients with non-obstructive MLS usually respond to medical therapy (bronchodilators, mucolytics, vaccination, and broad-spectrum antibiotics). ,
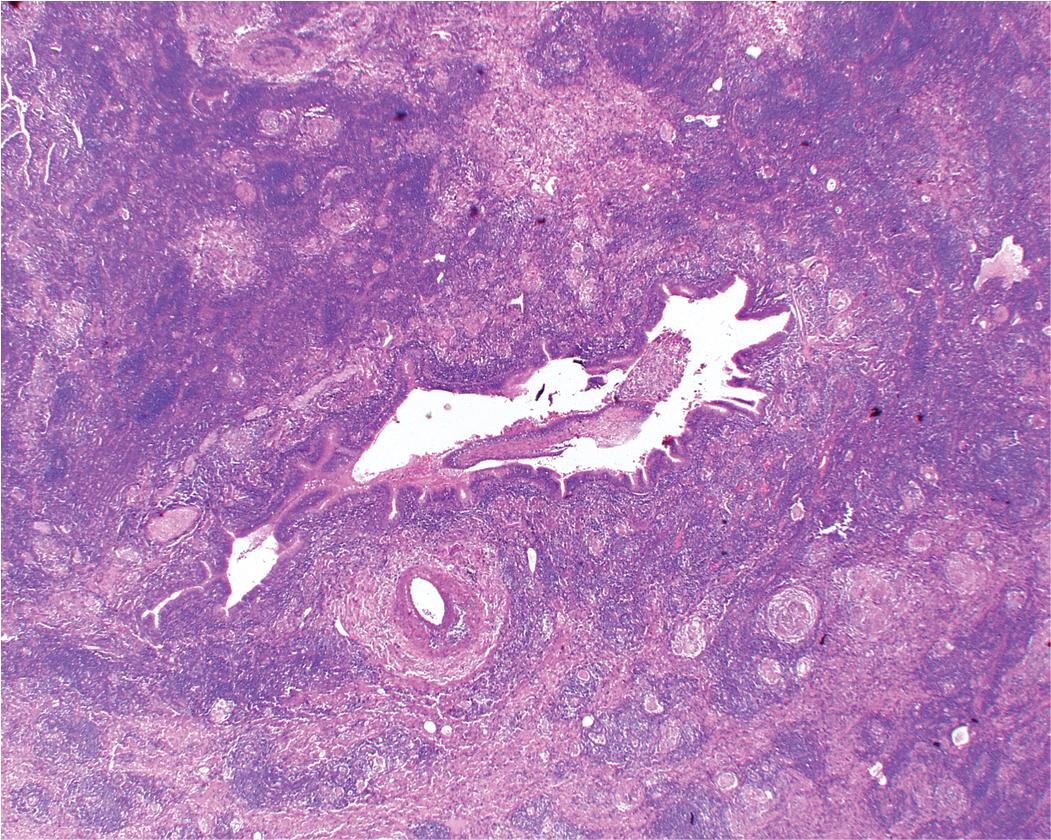
The key histopathologic features of MLS are:
Bronchiectasis, follicular bronchiolitis, bronchial lithiasis.
Parenchymal organizing pneumonia, abscess, atelectasis.
Granulomatous inflammation with or without necrosis suggesting a mycobacterial (mainly due to Mycobacterium avium-intracellulare ) or fungal infection for which special stains are mandatory.
Interstitial fibrosis, honeycomb changes, pleural fibrosis.
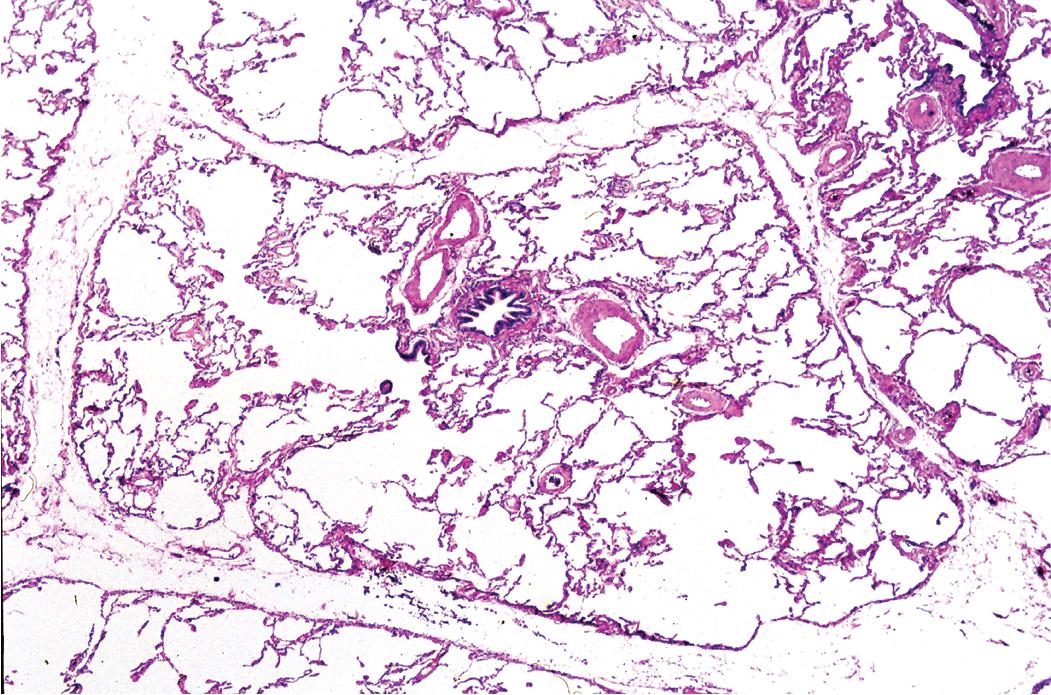
The small airways of the lung include the small bronchi, with a diameter less than 2 to 3 mm, and the membranous bronchioles (terminal and respiratory bronchioles). Terminal bronchioles have a diameter of less than 1 mm and are just proximal to the respiratory bronchioles, the first airways that have alveoli budding from their walls. The terminal bronchioles are located in the center of the pulmonary lobule and, like all conducting airways, are always accompanied by a pulmonary artery branch ( Fig. 9.11 ). The morphology of bronchioles in surgical specimens is modified by smooth muscle contraction, which causes prominent narrowing of the lumen with epithelial folding; this may give a false impression of constrictive bronchiolitis or other SAD, including asthma. A useful criterion to evaluate bronchiolar morphology in cross-sectional views is that the respective diameters of the bronchiole and artery are normally approximately equal ( Fig. 9.12 A). The lumen of the terminal bronchiole is uniform in longitudinal histologic sections with minor variations in size ( Fig. 9.12 B). From a radiologic standpoint, the anatomy of the small airways is straightforward: simply, they are not visible even in a HRCT scan. Whereas the large airways are distinctly seen as black, roundish, or linear elements alongside their companion arteries, the small airways are not.
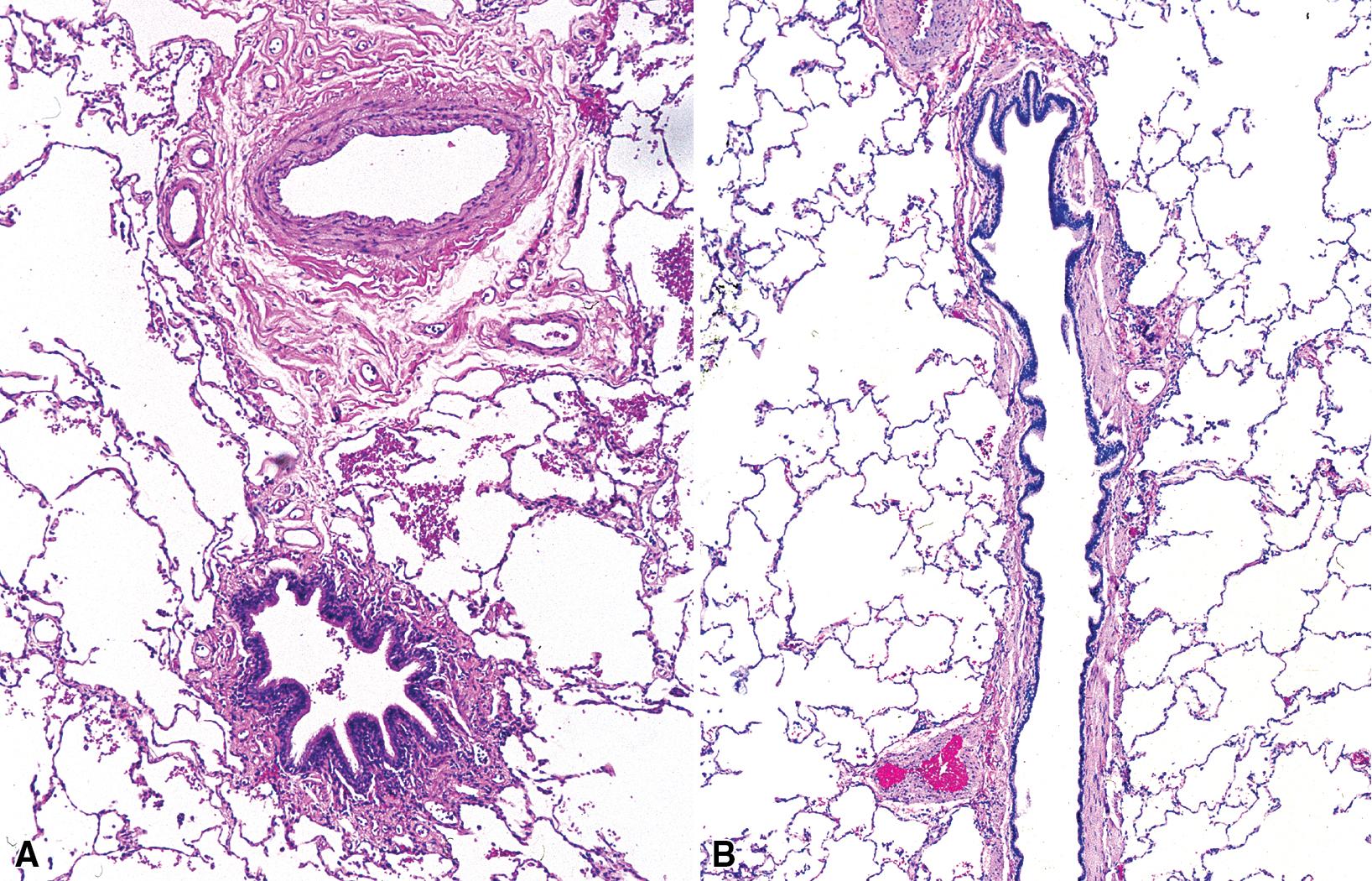
The small airways play an important role in air distribution and flow but lack the rigid structure of the bronchi to protect them from collapse during exhalation, especially when affected by disease. This anatomic condition is at the base of the main functio nal manifestation of SAD, which is obstruction due to collapse, especially during expiration.
The small airways can be affected secondarily by inflammatory diseases that involve primarily the bronchi, alveoli, or both, or by primary diseases that selectively involve these delicate anatomic structures. No single classification of bronchiolar disorders has been widely accepted, and many classification schemes have been proposed based on cause and underlying diseases, radiologic features, histopathologic findings, or some combination of these parameters. Ryu and colleagues have suggested a classification of bronchiolar disorders in three groups: primary bronchiolar disorders, bronchiolar involvement in interstitial lung diseases, and bronchiolar involvement in large airway disease ( Box 9.4 ). Rice and Nicholson proposed an integrated classification, which, though similarly distinguishing between disorders where bronchioles are primarily or secondarily affected, also includes a distinction between diseases with nonspecific/suggestive/specific histologic changes, and whether the secondary involvement is associated with proximal airway disease or diffuse interstitial lung disease ( Box 9.5 ).
Respiratory bronchiolitis
Acute bronchiolitis
Constrictive (obliterative) bronchiolitis
Follicular bronchiolitis
Diffuse aspiration bronchiolitis
Diffuse panbronchiolitis
Mineral dust airway disease
Miscellaneous forms
Histopathology features depending on the underlying ILD
Histopathology features depending on the underlying ILD
Without specific histologic features
Cellular changes (acute/acute and chronic/chronic)
Fibrotic changes (peribronchiolar/intraluminal/constrictive)
With histologic features suggestive of diseases
Follicular bronchiolitis
Eosinophilic bronchiolitis
Granulomatous bronchiolitis
Mineral dust airway disease
With specific histologic features
Diffuse panbronchiolitis (DPB)
Diffuse idiopathic neuroendocrine cell hyperplasia (DIPNECH)
Neuroendocrine cell hyperplasia of infancy (NEHI)
Other
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here