Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Naegleria fowleri is a free-living ameba that causes primary amebic meningoencephalitis (PAM). , It is the only known species of the genus Naegleria pathogenic to humans amongst over 40 other identified species. Two other species, N. australiensis and N. italica, are known to cause disease in experimental animals. ,
The organism is a facultative pathogen that occurs worldwide in soil and warm freshwater ponds, hot springs, streams, rivers, pools, and tap water. Notably, it is not found in saltwater. The parasite is thermophilic and thermotolerant, proliferates in naturally and artificially warmed waters, and has optimum activity at 37°C but can grow at temperatures as high as 45°C. As a free-living ameba, Naegleria do not have a known intermediate host.
The life cycle includes 3 phases: trophozoite (the infective stage), flagellate, and cyst phases ( Fig. 269.1 ). The trophozoite measures 10–25 μm, has a distinctive limacine (slug-like) pattern of locomotion, and has ≥1 ectoplasmic pseudopods ( Fig. 269.2 ). Naegleria is the only genera of free-living ameba that can transform into a nonreproductive, nonfeeding flagellate form in response to unfavorable environmental conditions. , The flagellate can in turn spontaneously revert to the trophozoite. Cysts are resistant to environmental stresses, double-walled, and approximately 9 μm in diameter, with a central nucleus and pores through which the trophozoites excyst. The genome of N. fowleri was sequenced in 2014.
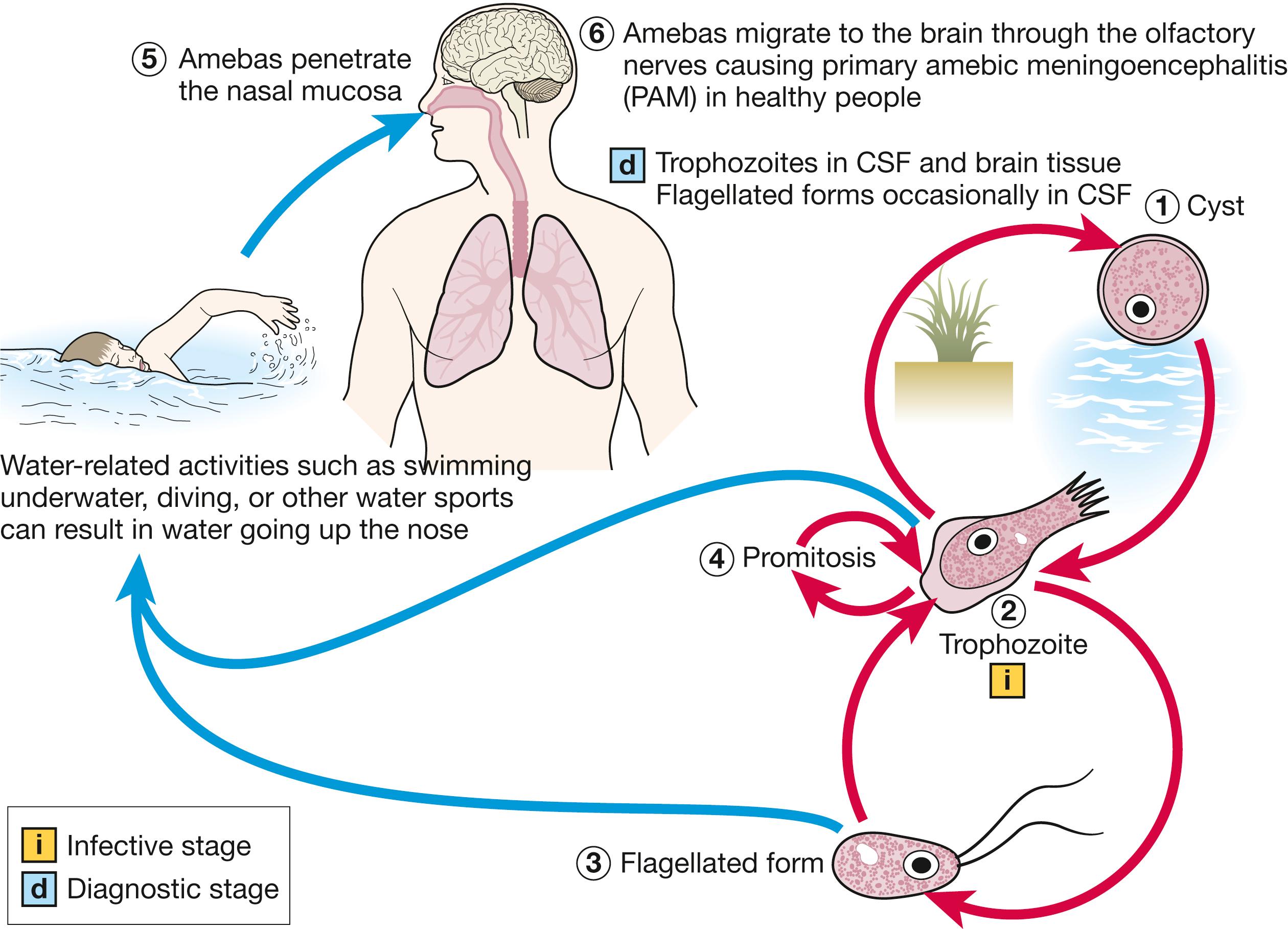
When causing human infection, amebas enter the nasal cavity, are phagocytosed by sustentacular cells of the olfactory neuroepithelium, and migrate along the unmyelinated axonal fibers of the olfactory nerve, through the cribriform plate, and into the subarachnoid space. The incubation period varies, with a median of 5 days, but has been reported to range from 1–9 days. The olfactory bulbs, the base of the frontal and temporal lobes, and the cerebellum primarily are involved in a hemorrhagic, necrotizing process.
In 1965 Fowler and Carter demonstrated that free-living amebas can invade the human brain and cause rapidly fatal meningitis, which was subsequently termed primary amebic encephalitis. , Worldwide prevalence cannot be estimated reliably; however, >16 countries have reported cases of PAM since the 1960s, and N. fowleri is distributed worldwide (except Antarctica). , As of 2018, 381 cases had been reported in the literature.
In the US, from 1937–2013, 142 cases were reported. The majority of cases have been otherwise healthy, young children with recreational freshwater exposure during the summer. , , The age of US patients has ranged from 8 months to 66 years, with a median age of 12 years, and the majority (>75%) have been male. Most cases have been reported from southern states. However, a case of PAM acquired in Minnesota was reported in 2012, raising concern that ongoing climate change could contribute to a wider geographic distribution and increased incidence of PAM. , Based on epidemiologic data, a Centers for Disease Control and Prevention (CDC) study estimated that undiagnosed PAM causes an annual average of 16 deaths in the US.
The predilection for young, healthy people may reflect behavior (e.g., more likely to remain in the water for longer periods of time, more active in the water, and more prone to diving and underwater swimming) that stirs bottom sediment that may contain amebas. In addition to recreational freshwater exposure, disease has been acquired through exposure to other recreational activities in hot springs, artificial whitewater, water parks, and improperly managed swimming pools. Reported clusters have also been related to nasal irrigation or other exposures with infected tap water. Similarly, examination of treated water drinking systems worldwide also has also shown the presence of N. fowleri. It is hypothesized that inhalation of cyst-containing dust could represent another portal of infection, although the overwhelming majority of cases have a direct epidemiologic link to water exposure.
Given the rapid progression and significant mortality rates associated with PAM, a high degree of clinical suspicion should be maintained in the setting of children and young adults presenting with acute neurologic symptoms and a history of recent freshwater exposure. The initial presentation can be indistinguishable from that seen in acute bacterial meningoencephalitis and may be nonspecific with symptoms of fever, headache, nausea, and vomiting. Consistent with olfactory nerve involvement, some patients may notice an altered sense of smell and taste. Additional symptoms and signs of central nervous system (CNS) involvement include altered mental status, meningismus, and seizures, with rapid progression to coma and death. PAM often progresses so rapidly that focal neurologic signs do not develop. Intracranial pressure is increased terminally and commonly causes herniation of the uncus and cerebellar tonsils. The mortality rate is >95%, and death usually occurs a median of 5 days (range 1–18) after the onset of symptoms. ,
Although the risk is not well defined, PAM is not an absolute contraindication to organ donation. One report noted no transmission of PAM to 21 transplant recipients from 5 organ donors who died of PAM.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here