Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Malignant mitral valve prolapse (MVP) syndrome has been recently described in a subset of patients who may be at greater risk for sudden cardiac death (SCD), made up of young women with mostly bileaflet MVP, midsystolic click, biphasic or inverted T waves in the inferior leads, and polymorphic/right bundle branch block (RBBB) morphology complex ventricular arrhythmias, in the absence of significant regurgitation. Subsequent clinic-pathologic data, matching autopsy and cardiac magnetic resonance (CMR) findings in arrhythmic MVP patients provided evidence of fibrosis in the papillary muscles and inferobasal left ventricular (LV) wall and showed mitral annulus disjunction (MAD) and systolic curling. At the same time, other groups demonstrated that MVP patients with previous cardiac arrest are characterized by having premature ventricular beats (PVBs) arising from Purkinje tissue that are triggers for ventricular fibrillation.
The following cascade of events has been put forward to explain electrical instability in MVP patients, where MAD and systolic curling represent the “primum movens.” They account for the paradoxical increase in the MV annulus diameter during systole, the myxomatous degeneration of MV leaflets, and the localized myocardial stretch in the LV inferobasal wall and papillary muscles (PMs), with segmental hypertrophy and fibrosis. Malignant arrhythmias in MVP probably result from the combination of the substrate (regional myocardial hypertrophy and fibrosis, Purkinje fibers) and the trigger (mechanical stretch) eliciting PVBs. Prospective studies are now needed that are able to match myocardial imaging scar and electrophysiologic substrates and assess the therapeutic role of implantable cardioverter-defibrillators (ICDs), targeted catheter ablation, and surgical repair in selected MVP patients with a high arrhythmia burden and risk.
Since the original description by Barlow in the 1960s, ,2 the existence of an arrhythmic variant of MVP has been recognized. With the recent introduction of the term arrhythmic malignant MVP , , its definition and implications in terms of diagnosis, risk stratification, and therapy are a matter for discussion among clinical cardiologists. At the same time, there has been increasing attention on MVP as a cause of cardiac arrest. Although SCD is most commonly the result of ischemic heart disease or cardiomyopathies, there is a distinct subset of patients who experience cardiac arrest/SCD but lack these common substrates. , In the group of patients with otherwise unexplained cardiac arrest, the significance of MVP remains controversial. Overall, the prevalence of MVP in pathology series of SCD is low, and often MVP is not even mentioned among the cardiovascular causes of SCD in the young. The absence of a uniform diagnostic criteria of MVP in the general, forensic pathology practice and the frequent consideration of MVP as a finding of uncertain significance are major obstacles to obtaining data on the contribution of MVP to SCD. Recently MVP was identified in a small proportion (2.3%) of SCDs in the general population of the Oregon Sudden Unexpected Death Study.
At the same time, the estimated occurrence of SCD in patients with MVP is low, at 16 to 41 per 10,000 per year (0.2%–0.4% per year). Although SCD is usually ascribed to ventricular fibrillation, the relationship between MVP and ventricular fibrillation has not been clearly elucidated. Furthermore, studies in which MVP has been associated with SCD hypothesize a role for coexisting risk factors of electrical instability, making it difficult to define MVP as causative.
Recently a systematic review and meta-analysis focused on the prevalence of MVP in the community, the prevalence of MVP in SCD and unexplained SCD patients, and the incidence of SCD in the general MVP population. The far higher prevalence of MVP in unexplained SCD cohorts than in the general population indirectly provides supports for an association of MVP with sudden death. Although the population of MVP at risk of SCD seems small, the potential number of “at-risk” individuals is significant. The “arrhythmic” MVP patient is usually female, with nonsyndromic bileaflet or single leaflet myxomatous degeneration of the mitral valve (MV), electrocardiographic (ECG) repolarization abnormalities, and polymorphic/RBBB morphology complex ventricular arrhythmias. ,
The echocardiographic features of the MV are those of single-leaflet or bileaflet displacement greater than 2 mm beyond the long-axis annular plane and greater than 5 mm leaflet thickening.
Moreover, the pathologic features of the MV are consistent with the MVP resulting from myxomatous degeneration of the MV (i.e., accumulation of proteoglycans leading to leaflet thickening and redundancy, chordae elongation, interchordal hooding, and annular dilation, classically ascribed to dysregulation of the transforming growth factor (TGF) β1 pathways. This entity should be kept distinct from the rarer forms of MVP, such as that caused by fibroelastic deficiency and the X-linked nonsyndromic variant related to Filamin-A gene mutation. ,
It should be clear that the 2% to 3% prevalence of MVP in the general population simply refers to the echocardiographic definition of at least 2 mm displacement, whereas the actual prevalence of “arrhythmic” MVP patients remains to be established.
A variable prevalence of ventricular arrhythmias has been reported in MVP series, reflecting the different definitions of MVP, the populations studied, the complexity of ventricular arrhythmias, and the fact that ECG recording has not been systematically performed. ,
It is well known that severity of regurgitation in MVP is an important determinant of the incidence of SCD and that LV volume overload is associated with a high recurrence rate of ventricular arrhythmias. , Kligfield et al. first emphasized that significant mitral regurgitation is an important predisposing factor for SCD in MVP. Their data showed that the prevalence of frequent PVBs is 40% in patients with hemodynamically relevant mitral regurgitation compared with 3% in those without.
The 10-year mortality of asymptomatic MVP patients is reported to be 19%, and severe mitral regurgitation and impaired LV function are the most important risk factors for mortality. Nevertheless, the detection of MVP in SCD/aborted SCD suggests that an association between hemodynamically uncomplicated MVP and life-threatening arrhythmias also exists. It is noteworthy that 40% of patients with SCD reported by Grigioni et al. were categorized as New York Heart Association (NYHA) class I.
Overall, the reported incidence of PVBs in MVP, as evaluated by 24-hour ECG Holter monitoring, varies from 49% to 92% in the adult population. , Both localized reentry and triggered automaticity have been implicated, with triggering from either a remote PVB focus or a sinus beat.
Early studies looked for features of PVBs predictive of malignant ventricular arrhythmias and a poor prognosis. In 1980 Lichstein examined the vectorcardiogram of PVBs in MVP and found that the most common site of origin was the posterobasal portion of the LV, a feature consistent with the hypothesis that mechanical irritation produced by the billowing valves dumping blood into the LV could trigger the arrhythmias.
More recently, Sriram et al. investigated the morphology of ventricular arrhythmias in a cohort of MVP patients with otherwise “unexplained” out-of-hospital cardiac arrest. They noted that patients with bileaflet MVP had a significantly higher burden of PVBs and more frequent ventricular bigeminy and nonsustained or sustained ventricular tachycardia (VT) compared with patients without bileaflet MVP. In nine patients, mostly with bileaflet MVP, they also noted PVBs with outflow tract origin morphologies alternating with PM or fascicular origin ECG morphologies. The outflow tract PVBs always originated from the LV and in some cases from both the LV and right ventricular (RV) outflow tracts. Cardiac arrest survivors with bileaflet MVP more commonly showed such alternating PVB configurations compared with those without MVP. In our autopsy series of 43 young SCD cases (more than half female) with MVP caused by myxomatous degeneration, ventricular arrhythmias, when documented, were almost always of RBBB morphology, rather than left bundle branch block (LBBB) morphology.
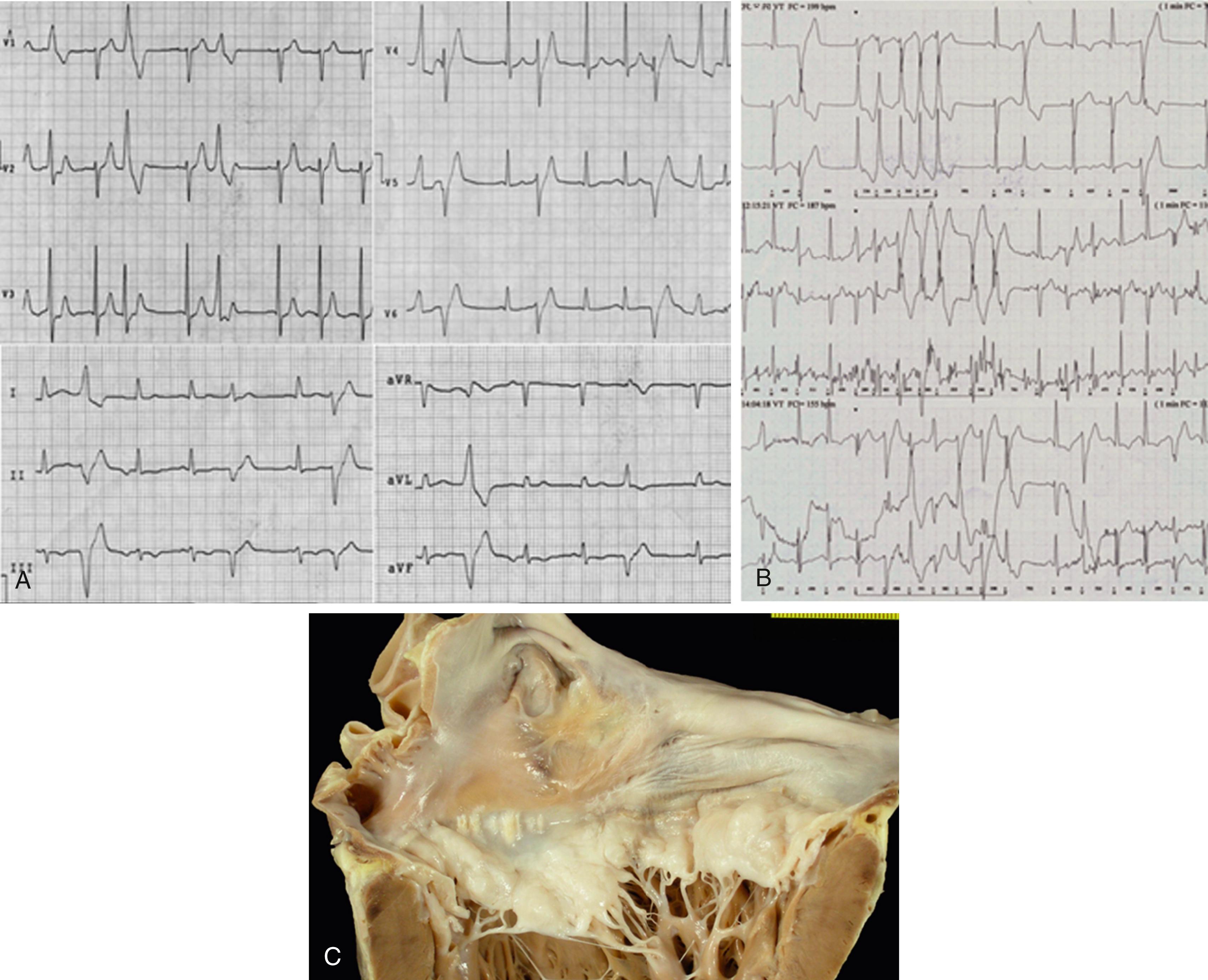
Thus there is an agreement that the so-called arrhythmic MVP syndrome is characterized from an electrocardiographic viewpoint by complex PVBs arising from one or both PMs, fascicular tissue, and outflow tract, as well as by T-wave inversion in the inferolateral leads , ( Fig. 94.1 ). Moreover, in electrophysiologic studies, PVB sites of origin have been mapped to the PMs, the LV outflow tract, and the mitral annulus, suggesting that PVBs arising close to the prolapsing leaflet and adjacent structures are the arrhythmic triggers.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here