Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Mites, including chigger and scabies mites, are among the smallest arthropods, with most barely visible without magnification. Only about 20 species of the more than 3000 species of chigger, animal, plant, and scabies mites are of any medical importance, and most of these are simply biting nuisances and do not transmit infectious diseases. Mites are closely related to ticks but not as prodigious at blood-feeding. They also do not transmit as broad a range of infectious microbial diseases as ticks. The most serious diseases transmitted by mites are scrub typhus and rickettsialpox.
Only biting larvae of Asian scrub typhus chiggers ( Leptotrombidium spp.) can transmit scrub typhus caused by Orientia tsutsugamushi (formerly Rickettsia tsutsugamushi ), and only biting house mouse mites (Liponyssoides sanguineus) can transmit rickettsialpox caused by Rickettsia akari. Both scrub typhus mites and house mouse mites are, like ticks, capable of inheriting bacterial infections by transovarial transmission and maintaining infections in several mite generations as bacteria are passed from adult to juvenile stages (nymphs and larvae) by transstadial transmission. Originally considered vectors of a rodent zoonosis, scrub typhus chiggers are the main environmental reservoirs of O. tsutsugamushi in endemic regions, with much smaller secondary reservoirs in wild rodents. Common house mice are the zoonotic reservoirs of R. akari, not only in crowded urban apartment buildings in the United States but also in mice-infested buildings, such as sheds and barns, in more rural locations worldwide.
Mites may be commonly classified as scabies mites (see Chapter 293 ), trombiculid or chigger mites (also called chiggers, red bugs, and itch mites), human follicle mites, dust mites, and a variety of animal, plant, and wood mites ( Table 295.1 ). All mite species develop close generational associations with their ecosystems and zoonotic reservoirs, often referred to as mite islands. Mite islands usually border cleared land and scrub bush and have several habitat requirements, including grassy vegetation with warm soil temperatures and high humidity, frequently visiting rodent hosts to feed larvae, and sufficient small insect fauna to feed nymphs and reproducing adults. Humans stumbling onto mite islands are at significantly higher risk for multiple larval chigger bites or trombidiosis worldwide or scrub typhus in the endemic regions of Eurasia and Asia.
| FAMILY, GENUS, SPECIES | COMMON NAMES (PLANT OR ANIMAL MITE) | GEOGRAPHIC DISTRIBUTION | MAINTENANCE IN NATURE | CLINICAL MANIFESTATIONS | INFECTIOUS DISEASE TRANSMISSION |
|---|---|---|---|---|---|
| Sarcoptidae | |||||
| Sarcoptes scabiei var. hominis | Scabies (itch) mite (human mite) | Worldwide | Obligate ectoparasite of humans, human reservoir | Classic scabies Atypical scabies |
None |
| Trombiculidae | |||||
| Neotrombicula autumnalis | European harvest mite (animal mite) | Europe | Free-living ectoparasites of small mammals and birds | Scrub itch (trombidiosis) | None |
| Eutrombicula alfreddugesi | American chigger mite (animal mite) | Western Hemisphere | Free-living ectoparasites of small mammals and birds | Scrub itch (trombidiosis) | None |
| Eutrombicula sarcina | Asian chigger mite (animal mite) | Asia, Australia | Free-living ectoparasites of small mammals and birds | Scrub itch (trombidiosis) | None |
| Leptotrombidium deliense | Asian rodent chigger (animal mite) | Southeast Asia, Japan, Philippines, South Pacific, Australia | Free-living ectoparasites of rodents and insectivores, transovarial/transstadial passage of infectious disease agent | Scrub typhus (tsutsugamushi disease) | Orientia tsutsugamushi (formerly Rickettsia tsutsugamushi), causative agent of scrub typhus |
| Leptotrombidium akamushi, Leptotrombidium pallidum, and Leptotrombidium scutellaris | Japanese rodent chiggers (animal mites) | Japan | Same | Same | Same |
| Leptotrombidium arenicola and Leptotrombidium fletcheri | Malaysian rodent chiggers (animal mites) | Malaysia | Same | Same | Same |
| Leptotrombidium pavlovskyi | Russian rodent chigger (animal mite) | Far east of former Soviet Union | Same | Same | Same |
| Demodicidae | |||||
| Demodex folliculorum | Hair follicle mite | Worldwide | Obligate ectoparasite of man, human host reservoir in hair follicles | Benign follicular (scaling) dermatitis Chronic blepharitis (demodicidosis) |
None |
| Demodex brevis | Sebaceous gland mite | Worldwide | Obligate ectoparasite of man, human host reservoir in sebaceous glands | May potentiate granulomatous acne | None |
| Pyroglyphidae | |||||
| Dermatophagoides pteronyssinus | European house dust mite (human mite) | Worldwide | Free-living ectoparasites of man; live in human bedrooms, especially in mattresses; feed on human skin detritus | House dust mite allergies and asthma | None |
| Dermatophagoides farinae | American house dust mite (human mite) | Worldwide | Same | Same | None |
| Dermanyssidae | |||||
| Liponyssoides sanguineus (formerly Allodermanyssus sanguineus ) | House mouse mite | North America, Northern Europe, Asia, and Africa | Free-living ectoparasites of field mice, transovarial/transstadial passage of infectious disease agent | Rickettsialpox | Yes (Rickettsia akari) |
| Dermanyssus gallinae | Red poultry (chicken) mite | Worldwide | Free-living ectoparasites of domestic and wild birds | Poultry workers' dermatitis of hands | None |
| Macronyssidae | |||||
| Ornithonyssus bacoti | Tropical rat mite | Temperate and tropical regions worldwide | Free-living ectoparasites of large rodents: Rattus rattus, Rattus norvegicus | Urticarial papulovesicular to pustular dermatitis | None |
| Ornithonyssus bursa | Tropical fowl mite | Same | Free-living ectoparasites of domestic and wild birds | Pruritic papules in a scabietic distribution: finger webs, axillae, groin, buttocks | None |
| Laelapidae | |||||
| Laelaps echidnina | Spiny rat mite | Worldwide, the most prevalent rodent mite species in the United States | Free-living ectoparasites of large rodents: Rattus rattus, Rattus norvegicus | Nonspecific mite-bite dermatitis | None |
| Pyemotidae | |||||
| Pyemotes tritici ventricosus | Grain (hay) itch mite | Worldwide | Free-living ectoparasites of straw-, hay-, grain-, and rice-eating moths, beetles, weevils | Grain workers' pruritic vesicular eruption | None |
| Pediculoides ventricosus Newport | European wood beetle itch mite | Worldwide | Free-living ectoparasites of straw-, hay-, grain-, and rice-eating moths, beetles, weevils | Solitary to multiple highly erythematous pruritic macules, some of which have attached macular tracts resembling comet tails | None |
| Pyemotes herfsi | Oak leaf gall mite | Europe, introduced into the United States | Free-living ectoparasites of gall-making larvae of oak trees | Pruritic, erythematous, vesicular eruptions of limbs, face, and neck | None |
| Acaridae a | |||||
| Carpoglyphus lactis | Cheese and dried fruit mites | Worldwide | Free-living ectoparasites of cheeses and dried fruits | Cheese and fruit workers' dermatitis | None |
| Tyrophagus putrescentiae | Copra (dried coconut meat or kernel) mite | Copra (dried coconut meat or kernel) growing areas | Free-living ectoparasites of coconut copra | Copra itch | None |
| Glycyphagidae a | |||||
| Glycyphagus domesticus | Grocer's mite | Worldwide | Free-living ectoparasites of fruits and vegetables | Grocer's itch | None |
| Glycyphagus destructor | Hay mite | Worldwide | Free-living ectoparasites of cut hay | Hay workers' and hay wagon riders' allergy, asthma, rhinitis, conjunctivitis | None |
a The acarid and glycyphagid plant mites may rarely enter the gastrointestinal tract if swallowed with food and cause intestinal distress (gastrointestinal acariasis). They may also be inhaled in aerosols and cause bronchial irritation and respiratory distress (respiratory acariasis). They may crawl into the urethra to cause dysuria (urinary acariasis). The mites can be recovered from feces, sputum, and urine. Treatment is supportive. Although they may rarely infest humans, plant mites are all free living and do not reproduce in human dead-end hosts.
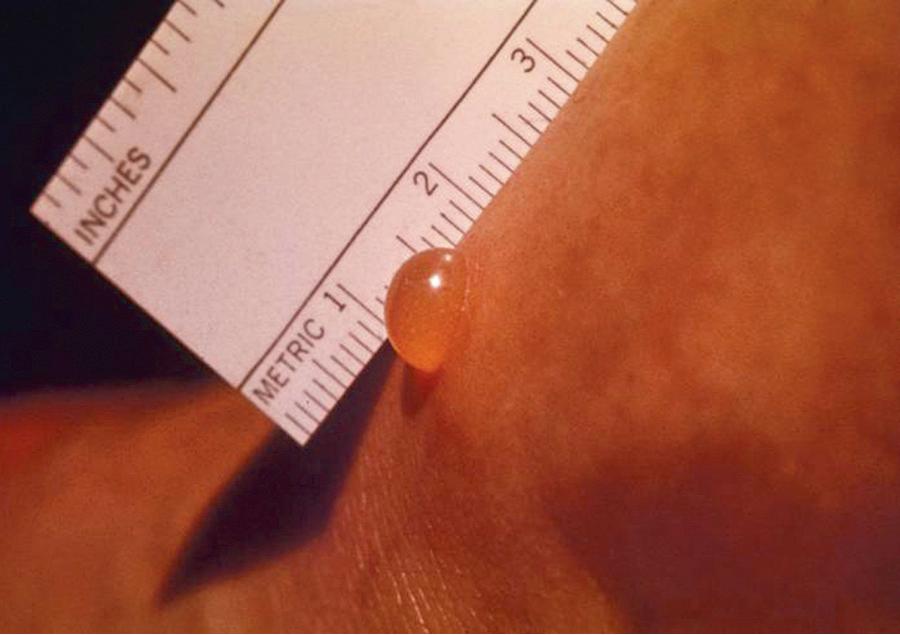
Among the trombiculid chiggers (the Trombiculidae), including the scrub typhus–transmitting Leptotrombidium spp., only the larvae are human and animal ectoparasites. The larger chigger nymphs and adults are free living and feed on small insects and their eggs. All trombiculid larvae exhibit a unique method of feeding on their human hosts and transmitting salivary secretions, which may contain O. tsutsugamushi in endemic regions. When larval mites have selected a human host, they will congregate where the skin is soft, warm, and moist, particularly where clothing is tight against the skin, such as under waistbands, undergarment elastic bands, and socks. Initially painless, chigger bites cluster in these regions on the genitalia, perineum, thighs, buttocks, waist, and ankles and become symptomatic in 3 to 6 hours ( Fig. 295.1 ). Larvae pierce the skin with sharp mouthparts and inject tissue-dissolving saliva to create a pool of lymph, other body fluids, and dissolved epithelial cells to drink from (see Fig. 295.1 ). Unlike ticks, mites are not blood feeders, but tissue juice feeders. The repeated injection of saliva into the bite wound induces a host reaction that forms a strawlike hollow tube, known as a hypostome or stylostome, which extends downward into the host's skin, anchoring the mite firmly. Some trombiculid larvae remain attached to and feeding on human hosts for up to a month, but the larval vectors of scrub typhus feed only for 2 to 10 days before dropping to the ground engorged and ready to mature into free-ranging nymphs.
All of the noninfectious chigger larvae can cause trombidiosis or trombiculiasis (trombiculidiasis), with the American chigger mite (Eutrombicula alfreddugesi) being the most common culprit in the United States; the European harvest mite, Neotrombicula autumnalis, the most common culprit in Europe; and the Asian chigger, Eutrombicula sarcina, the most common culprit in Asia. In the United States, clusters of trombiculid larval bites are often referred to as “chiggers,” a colloquial term for the intensely pruritic groupings of erythematous welts inflicted by American chigger mites. In Asia and Australia, similar clusters of larval mite bites may be referred to as “scrub itch,” especially in Australia, where scrub itch mites, Trombicula hirsti, commonly cause trombidiosis. Among the scrub typhus–carrying Leptotrombidium larval chigger mites, Leptotrombidium deliense, the Asian rodent chigger, is a principal vector throughout eastern Asia and Eurasia.
Unlike North American “chiggers,” scrub typhus is a serious and potentially fatal zoonotic infectious disease caused by O. tsutsugamushi organisms transmitted by bites from infected Leptotrombidium species chigger larvae. As a result of experiences with scrub typhus among US troops in the Pacific during World War II and later during wars in Korea, Vietnam, and Afghanistan, scrub typhus was originally considered a regional zoonosis confined to the “tsutsugamushi triangle,” which extended from Pakistan in the west to the Pacific coast of Russia in the east to northern Australia in the south. In 2006, additional cases were detected in the Middle East caused by a new species, Orientia chuto , and in Chile caused by O. tsutsugamushi. In 2016, three additional cases of scrub typhus caused by O. tsutsugamushi were confirmed serologically (with enzyme-linked immunosorbent assay [ELISA] and indirect immunofluorescent antibody assay [IFA]) and molecularly (with polymerase chain reaction [PCR] detection of DNA in the 47-kD and 56-kD genes) in Chile, which is 12,000 km away from the tsutsugamushi triangle. Although the zoonotic reservoirs and larval mite vectors of scrub typhus outside of the tsutsugamushi triangle remain unidentified, there is now a wider global distribution of the disease than initially assumed.
After O. tsutsugamushi –infected Leptotrombidium chigger bites, an 8- to 10-day incubation period precedes the onset of classic clinical manifestations of scrub typhus with bite eschar, regional lymphadenopathy, conjunctival injection, central nervous system (CNS) manifestations (e.g., headache, confusion, delirium, transient hearing loss), and centrifugal rash ( Fig. 295.2 ). Many cases are nonclassic with nonspecific clinical manifestations and go undiagnosed, especially when serologic tests are unavailable. In the temperate regions of Eurasia, there is a definite scrub typhus seasonal transmission cycle determined by peaking temperatures and humidity during weeks of marked seasonal change in late spring to early summer and again in late fall to early winter. In the tropics, scrub typhus transmission occurs year-round. Fatal complications may include adult respiratory distress syndrome (especially in older patients), hypotensive shock, acute renal failure, encephalomyelitis, and disseminated intravascular coagulation.
Although the most common CNS manifestations of scrub typhus are meningitis or meningoencephalitis, Yun and coauthors recently reported a case of acute transverse myelitis in a 67-year-old man with dysuria and lower-leg weakness associated with serologically confirmed scrub typhus in Korea and successfully treated with doxycycline and corticosteroids.
In 2016, Thipmontree and coauthors reported another very rare complication of scrub typhus in a 74-year-old Thai woman with acute abdominal symptoms requiring surgery, due to a ruptured spleen. In this case, scrub typhus was confirmed by a fourfold increase in immunoglobulin M (IgM) titer by ELISA and positive PCR targeting the 47- and 56-kD genes of O. tsutsugamushi . The patient underwent splenectomy and was treated successfully with intravenous chloramphenicol and oral doxycycline over 10 days.
The house mouse mite, Liponyssoides sanguineus, maintains a rickettsial zoonosis in its preferred house mouse (Mus musculus) reservoir and can transmit rickettsialpox caused by R. akari through its bites. Although initially described in clusters in crowded apartment complexes in large US cities, including New York, Boston, Cleveland, Philadelphia, and Pittsburgh, rickettsialpox has now been reported in rural areas of the eastern United States and eastern Europe Many experts believe that rickettsialpox is underreported and more widely distributed in silent sylvan cycles worldwide. The incubation period and initial clinical manifestations of rickettsialpox mirror those of scrub typhus, with eschar formation at the bite site within 10 to 12 days followed by fever, chills, severe headache, conjunctival injection, and truncal maculopapular then vesicular rash. Illness is typically mild, and regional lymphadenopathy is uncommon ( Table 295.2 ). Unlike with scrub typhus, complications are rare, but they may include thrombocytopenia and interstitial pneumonia.
| SCRUB TYPHUS | RICKETTSIALPOX | |
|---|---|---|
| Bacterial agent | Orientia (formerly Rickettsia ) tsutsugamushi a | Rickettsia akari |
| Mite vector | Larvae of Leptotrombidium species of Asian rodent chigger mites | Common house mouse mites, Liponyssoides sanguineus |
| Incubation period | 8–10 days (range, 8–20 days) | 10–12 days |
| Mite-bite site | Painless initial bite, eschar at bite site (50%) | Painless initial bite, eschar at bite site |
| Presenting constitutional symptoms | Fever, chills, headaches, myalgia, pathognomonic hearing loss (30%) | Fever, chills, severe headache, myalgia |
| Conjunctival injection | Present | May be present |
| Regional lymphadenopathy | Present regionally and tender | Usually absent |
| Associated rash exanthema | Delayed truncal onset, erythematous macules, then maculopapules that spread peripherally | Abrupt truncal onset, erythematous macules that develop central vesicles in crops |
| Pulmonary findings | Cough, tachypnea, dyspnea, bibasilar rales | Asymptomatic bibasilar rales |
| Radiographic findings | Infiltrates common | Usually normal |
| Potential complications | Adult respiratory distress syndromes, acute renal failure, disseminated intravascular coagulation, encephalomyelitis | Thrombocytopenia |
| Differential diagnosis | Infectious mononucleosis, leptospirosis, tularemia, anthrax, spotted fever rickettsioses | Chickenpox, tick-bite eschar |
| Diagnostic methods | Screening: rapid dipstick recombinant 56-kDa protein antigen test Serodiagnostic: indirect immunofluorescent antibody tests, immunoperoxidase assays |
Serodiagnostic: immunofluorescent antibody assay for IgG to both R. akari and R. rickettsii, with follow-up cross-adsorption testing for predominant antibodies |
| Confirmatory: microscopic isolation of agent from blood or tissues, polymerase chain reaction for agent DNA (or RNA) | Confirmatory: isolation from skin biopsy specimen | |
| Recommended treatment | Tetracycline 500 mg orally 4 × daily × 1 wk, or doxycycline 100 mg twice daily × 1 wk, or IV chloramphenicol, 50–75 mg/kg/day × 1 wk (only for complicated cases) | |
| In childhood and pregnancy, consider the macrolides: azithromycin, clarithromycin, or roxithromycin | Doxycycline 100 mg orally twice daily × 7–10 days | |
| Outcomes/case-fatality rates (%) | 1%–15% | <1% |
a Although initially classified among the Rickettsia, the causative agent of scrub typhus, Orientia tsutsugamushi (originally Rickettsia tsutsugamushi ) has now been reclassified into a separate genus, Orientia, based on molecular evidence that its cell wall differs significantly from that of Rickettsia both ultrastructurally and in its component proteins.
Dermatophagoides spp. dust mites have highly allergenic exoskeletons, body fragments, and feces, all of which can be easily aerosolized during bed making and pillow fluffing. Allergens from living and dead dust mites frequently cause allergic rhinitis and asthmatic bronchitis in predisposed, atopic persons. The American house dust mite, Dermatophagoides farinae, is now distributed worldwide, as is the European house dust mite, Dermatophagoides pteronyssinus. House dust mites prefer to live in bedrooms year-round, especially in mattresses and carpets in warm, humid homes. They exhibit maximum growth and reproduction during seasonal warming cycles at ambient temperatures at or above 25°C and relative humidity at or above 75%.
Scabies and follicle mites are the only exclusively human ectoparasitic mites and cannot transmit infectious diseases. Less serious but more common than scabies is infection with the human follicle mites: Demodex folliculorum inhabits hair follicles, and Demodex brevis inhabits sebaceous glands. These diminutive (0.1–0.4 mm) human mites feed on sebum and exfoliated skin while lodged in human hair follicles or sebaceous gland pores.
Demodex mites or face mites are commensal ectoparasites that cluster in hair follicles and sebaceous glands on the nose, eyelids, and nasolabial folds and have even been found living in earwax (D. folliculorum). All of the developmental stages of Demodex mites occur over an egg-to-egg cycle of 13 to 15 days entirely within hair follicles or sebaceous glands, especially in females overusing cream-based facial cosmetics and adolescents with increased sebaceous gland activity. Other than causing comedones or “blackheads,” Demodex infections cause few adverse symptoms and rarely need treatment, unless infections are associated with acne, blepharitis, impetigo, rosacea, or seborrheic dermatitis. In some cases, infection or demodicosis may be precipitated by immunosuppression from stress, advancing age, or coexisting illness.
Although of limited clinical significance, a number of animal, plant, and wood mite species can cause bothersome erythematous papulovesicular eruptions if encountered. Bites from the red chicken or poultry mite, Dermanyssus gallinae, can cause a pruritic dermatitis, usually on the backs of the hands and forearms in poultry workers ( Fig. 295.3 ). Both St. Louis encephalitis virus and western equine encephalitis virus have been isolated from naturally infected red chicken mites, but they are not preferred vectors for these mosquito-borne arboviruses (see Fig. 295.3 ). Bites from the rat mite Ornithonyssus bacoti, which is ubiquitous in the temperate areas of Europe and the Americas, can cause a papulovesicular dermatitis in stockyard and warehouse workers. The rat mite can also transmit Rickettsia typhi, the agent of murine typhus from rat to rat, maintaining the rodent zoonosis, but is incapable of human transmission. The bird mite, Ornithonyssus bursa, is a common ectoparasite of pigeons worldwide and a frequent cause of mite infections with maculopapular dermatitis of the finger webs and axillae in pigeon breeders and fanciers.

Like chigger mites, the plant and wood mites do not transmit infectious diseases but are common causes of annoying infections with pruritic, erythematous maculopapular rashes on the limbs and face of arborists, landscapers, and campers.
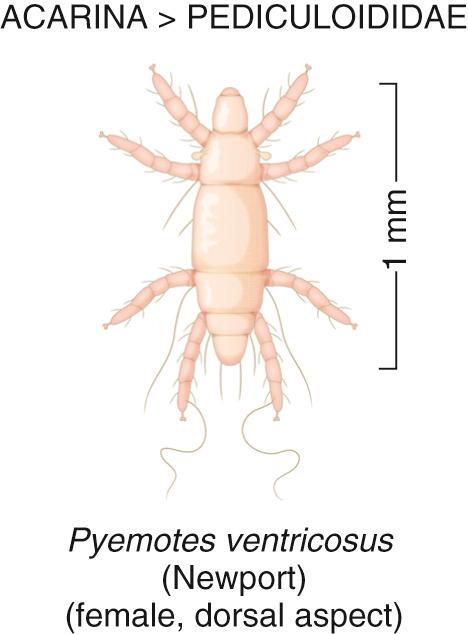
The North American hay itch mite, Pyemotes tritici ventricosus, feeds preferentially on the larvae of insects that infest cane, hay, straw, and some grains, especially rice ( Fig. 295.4 ). In 1965, Fine and Scott were the first investigators to describe P. tritici ventricosus dermatitis in the southern United States, where hayrides, cane furniture, and straw rugs are popular. Such exposures frequently place patients in skin contact with infested hay, straw, or cane furniture during peak mite feeding and breeding seasons in the spring and summer. Hay itch mite dermatitis is characterized by pruritic, maculopapulovesicular eruptions on the limbs and trunk, which resolve rapidly with topical corticosteroid therapy.
A close relative of the North American hay itch mite, P. tritici ventricosus, the oak leaf gall mite (Pyemotes herfsi), which preferentially feeds on insect larvae in oak trees, caused an outbreak of plant insect mite dermatitis in the United States in 2004. Over 300 residents of Pittsburg, Kansas, sought immediate medical attention for an intensely pruritic, erythematous maculopapular rash clustering on the face, neck, and limbs ( Fig. 295.5 ). All lesions healed within days after topical treatment with antihistamines and corticosteroids.
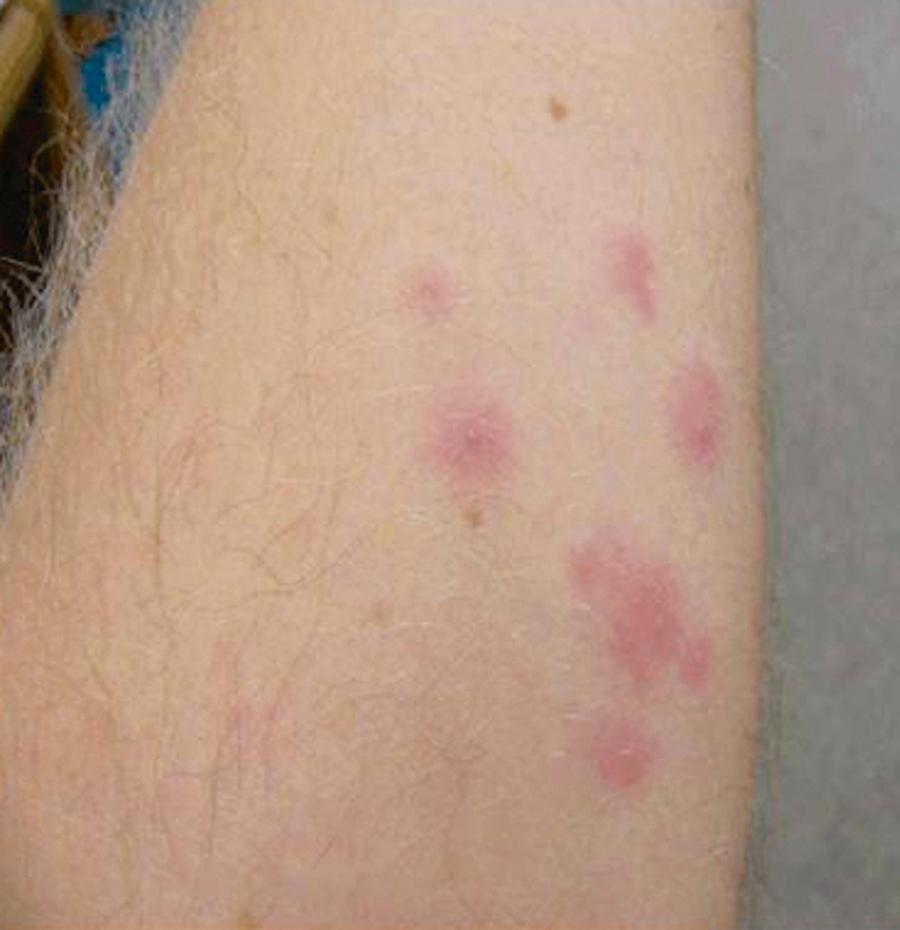
Three separate outbreaks of dermatitis affecting over 100 patients caused by the European hay itch mite (Pyemotes ventricosus) were reported from Spain in 2000. A similar outbreak, also suggestive of mite bite–induced dermatitis, was reported in southeastern France in 2006. The dermatitis was characterized by solitary to multiple, highly erythematous pruritic macules; some were accompanied by contiguous, linear erythematous macular tracts that resembled comet tails ( Fig. 295.6 ). In a 2007 outbreak investigation of an additional 42 cases of dermatitis with comet tail signs in the same region, Del Giudice and coworkers identified P. ventricosus mites as causative agents and described the epidemiology and outcomes of P. ventricosus infections in homes and humans.
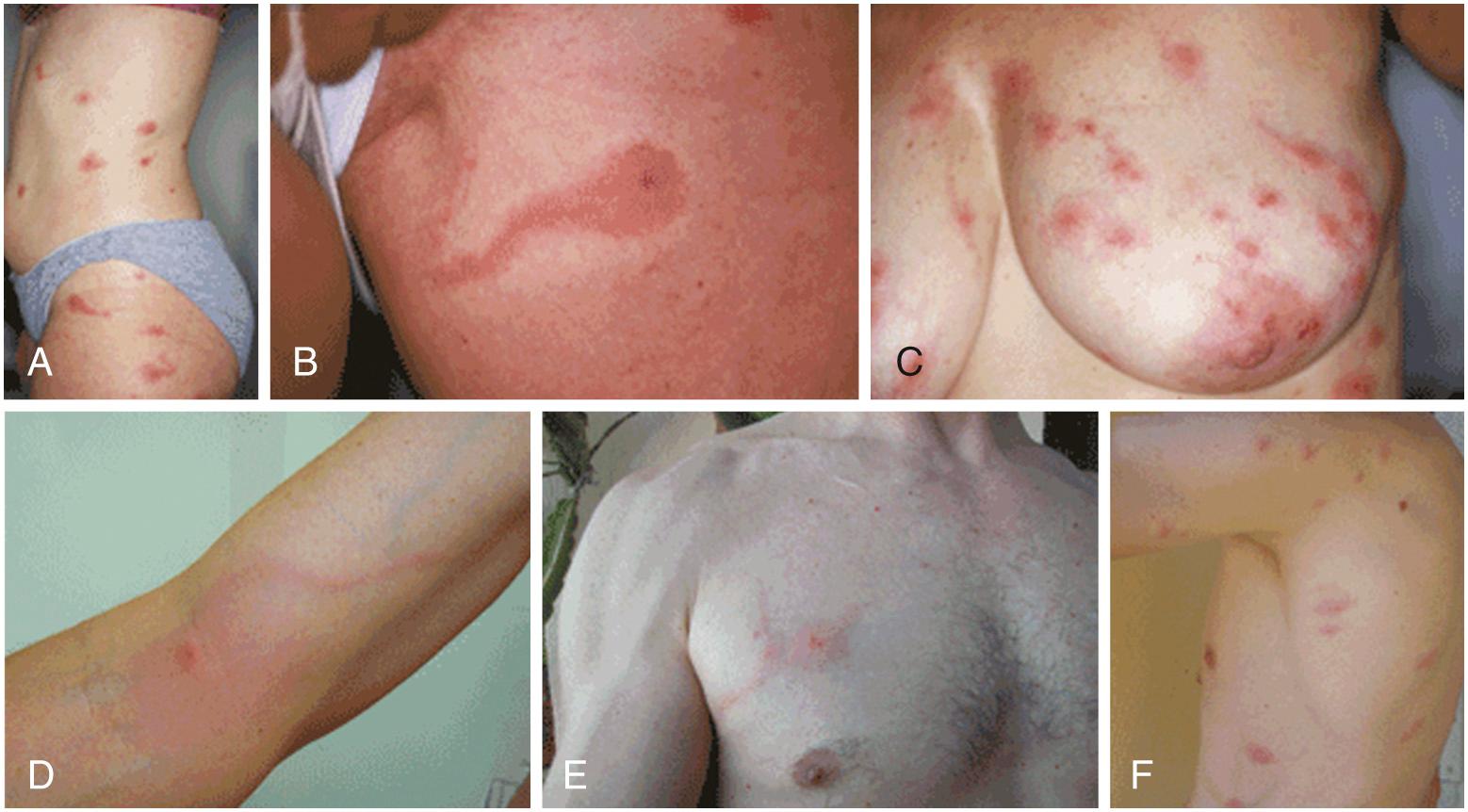
Most residences of index case patients with P. ventricosus dermatitis were infested with live furniture beetles, Anobium punctatum, which do not bite or infest humans. However, adult P. ventricosus mites, common ectoparasites of furniture beetles, were present in stereomicroscopic examination of wood dust beneath beetle-infested furniture. Confocal laser scanning microscopy (CLSM) of a central microvesicle in a maculopapular lesion on an experimentally infested coinvestigator demonstrated an ovoid foreign body consistent with a P. ventricosus mite. Both naturally occurring and experimental infections caused the characteristic maculopapular rash of P. ventricosus dermatitis, again associated with comet signs (see Fig. 295.6 ). Although oral prednisone (0.5 mg/kg) rapidly relieved pruritus, P. ventricosus dermatitis would persist or recur in index case patients until beetle-infested furniture was removed from households or patients permanently vacated their infested residences, which were often in resort regions.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here