Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Meningiomas are among the most common of central nervous system (CNS) neoplasms and are by far the most common of the extra-axial tumors. Our understanding of meningioma classification, grading, and molecular genetics has evolved greatly over time and continues to be refined. Both environmental and genetic factors have been implicated in meningioma formation, although most examples are idiopathic. Meningioma genetics is covered later in this chapter, whereas the meningioma-associated syndrome NF2 is discussed further in Chapter 22 . Proposed environmental factors include irradiation (discussed later), hormone exposures (discussed later), and head trauma, although only the first of these has been unequivocally established. The latter is occasionally the basis for medicolegal cases, though it is hardly a proven cause. In Cushing and Eisenhardt's 1938 monograph, a surprising 32% of intracranial meningiomas had a prior history of head injury, and a modern epidemiologic study estimated a 4.33 odds ratio for developing meningiomas 10 to 19 years after head trauma. Rare case reports feature meningiomas arising at sites of prior skull fracture, cranioplasty, or lodged foreign material, such as shrapnel, implicating wound healing and arachnoidal proliferation as a possible mechanism. However, most epidemiologic studies have not found a significant correlation. Similarly, evidence to date argues against the common notion that cell phones cause meningiomas and other brain tumors, although admittedly this is a relatively new technology and more follow-up may still be needed.
In contrast, meningiomas caused by irradiation are well documented, and this is an irrefutable risk factor. As with other radiation-associated neoplasms (see Chapter 21 ), these tumors must arise in the prior radiation field, develop after long latency (typically years), differ histologically from the originally irradiated disease process, and arise more frequently in irradiated than in control patients. Radiation-induced meningiomas are sometimes divided into low-dose (<10 Gy), medium-dose and miscellaneous (10 to 20 Gy), and high-dose (>20 Gy) categories. The low-dose group has been most extensively studied, mostly in children receiving scalp irradiation for tinea capitis in Israel during the 1960s, although recent studies have similarly implicated prior exposures to computed tomography (CT) imaging and dental x-rays (mostly the older and somewhat higher dose Panorex films in young children). The miscellaneous group includes direct application of radium, thorotrast ventriculography, and myelography, whereas high-dose examples mostly follow therapeutic irradiation for cancer. Radiation-induced meningiomas typically afflict younger patients, with median onset being 45, 32, and 34 years for the high-, medium-, and low-dose groups, respectively. The average latencies are 35, 26, and 19.5 years, respectively. Children appear to be particularly sensitive (“window of vulnerability”), with the latency often being shorter than in adults. Providing further evidence, atomic bomb survivors are also at increased risk of developing meningiomas, the distance from the epicenter of the explosion being inversely associated with overall risk.
Lastly, it is of interest that allergic diseases, such as asthma and eczema, have been confirmed in epidemiologic studies as protective (i.e., lower risk) for subsequent meningioma development.
Meningiomas are a clinicopathologically diverse group of neoplasms with morphologic, immunohistochemical, and ultrastructural features resembling normal meningothelial cells. More specifically, the majority resemble the arachnoidal cap cell, although rare variants such as the microcystic meningioma resemble other cell types (arachnoidal trabecular cell). Like their normal meningothelial counterparts, meningiomas can display a wide range of epithelial- and mesenchymal-like properties. Mesenchymal features include a prominent spindled morphology in the fibroblastic and sarcoma-like meningiomas, an ability to produce collagen and other extracellular matrix proteins, and the finding of mesenchymal “metaplasia” in a subset of otherwise classic meningiomas (discussed further under Histologic Variants and Grading ). Epithelial features include epithelioid morphology in meningothelial (and other variant) meningiomas, the presence of desmosomes and other intercellular junctions, epithelial membrane antigen (EMA) positivity, the capacity for papillary growth, the formation of gland-like lumens in the secretory variant, and occasional carcinoma-like histology in anaplastic meningiomas.
As an appropriate candidate cell of origin, arachnoidal cap cells are essentially found in the same locations as meningiomas (see Chapter 2 ). These include
the external layer of the leptomeninges and arachnoid granulations attaching loosely to the inner surface of the dura (e.g., dural-based and, less often, leptomeningeal-based meningiomas );
extensions along perivascular Virchow-Robin spaces into the underlying brain (rare intracerebral meningiomas );
the tela choroidea (meningeal invagination) at the stromal base of the choroid plexus (intraventricular meningiomas); and
rare meningothelial rests/heterotopias, traumatic displacements, metaplasias/aberrant differentiation, and perineural extensions (calvarial/intraosseous, scalp, sinonasal, pulmonary, and other extradural meningiomas).
The light- and electron-microscopic similarities include frequent intranuclear inclusions, interdigitating cell membranes, intercellular junctions, and cytoplasmic filaments. Arachnoidal cap cells are unique cell types that are also functionally similar to meningiomas. Their known functions include:
ensheathing or wrapping around each other, the surface of the CNS, and proximal portions of nerve roots, forming part of the CNS–cerebrospinal fluid (CSF) and blood-CSF barriers (recapitulated in the whorls of meningiomas);
helping to maintain CSF homeostasis, both in terms of protein secretion (recapitulated in the pseudopsammoma bodies of secretory meningiomas ) and the regulation of intracranial pressure;
providing trophic support for migrating neuroglial cells during development; and
taking on macrophage-like functions rarely, including some limited capacity for phagocytosis and rheumatoid nodule formation; the most macrophage-like meningioma is the metaplastic-xanthomatous subtype.
The wide-ranging biologic and histologic continuum of meningiomas has fascinated physicians since the beginning of the 20th century. One of the greatest single advances in our understanding of these tumors came in 1938 when Drs. Harvey Cushing and Louise Eisenhardt published their detailed monograph on 313 meningiomas accumulated over nearly 30 years. Over the next few decades, variants became better characterized and new techniques such as karyotyping, electron microscopy, and immunohistochemistry were subsequently applied. In 1982, a second major monograph by John Kepes summarized the many advances of that time, along with personal observations obtained from a review of approximately 1300 cases. Since then, a few more aggressive variants have been described or clarified, hemangiopericytomas (now termed solitary fibrous tumor/hemangiopericytoma) have been removed as a biologically distinct tumor type (see Chapter 14 ), grading criteria have been extensively revised, our understanding of the molecular pathology has greatly increased, and therapeutic modalities have evolved. The classification scheme utilized by most pathologists today is the World Health Organization (WHO) 2016 edition.
As with other neoplasms, the issue of meningioma histogenesis has long been debated. The earliest terms attempted to lump these tumors in with better known extracranial categories, resulting in clumsy and inaccurate diagnoses, such as fungus of the dura mater, sarcoma, carcinoma, mesothelioma, endothelioma, meningoexothelioma, and arachnoidal/meningeal fibroblastoma. In 1922, recognizing that these tumors were distinct from systemic tumor types, Cushing proposed the term meningioma, a clinically practical alternative based primarily on anatomy, rather than unsubstantiated histogenetic notions. Various hypotheses on the cell of origin were posited nonetheless, including dural, endothelial, fibroblastic, and epithelial cell types. In 1831, Dr. Richard Bright raised the possibility that they might originate from the inner arachnoidal lining of the dura, rather than the dense fibrous tissue of the dura proper. Similarly, Cleland and, later on, Schmidt emphasized the similar histology of most meningiomas with the cell clusters that line arachnoid villi. Since then, these arachnoidal cap cells have been considered the most likely, though still unproven, cell of origin for meningiomas.
Meningioma accounts for about 36% of over 356,000 primary brain tumors reported to the Central Brain Tumor Registry of the United States in the 2008 to 2012 census; it is the single most common tumor subtype overall and also accounts for over half of all nonmalignant CNS tumors. The incidence is roughly 7.86 per 100,000 person-years (4.68 in males and 10.62 in females), with a mean diagnostic age of 65 years. Meningioma incidence figures have progressively risen over time, possibly reflecting ever-increasing radiologic detection of incidental tumors. In fact, incidental meningiomas are quite common, especially in the elderly where they are reported in up to 2.3% of autopsies. The prevalence of meningioma is much higher in families with NF2 and rarely in those with Gorlin, Cowden, Li-Fraumeni, Turcot, or von Hippel-Lindau syndromes as well (see Chapter 22 ). It also occasionally runs in families without any well-established tumor predisposition syndromes.
Meningiomas are most common in middle-age to elderly adults and vary in incidence with race in the United States, affecting Blacks, Whites, and Asians/Pacific Islanders in decreasing order, respectively. However, they are considerably more common in women than men, with a roughly 2 to 1 female–male ratio. This sex predilection is even higher in thoracic spinal meningiomas, where the ratio is nearly 9 to 1. Pediatric meningiomas are distinctly rare, but are more likely than adult counterparts to present with large tumor size, cyst formation, lack of dural attachment, high-grade histology, aggressive behavior, and/or aggressive variant histology, particularly the clear cell or papillary subtypes. They lack any female predominance and are more likely to present in unusual locations, such as lateral ventricles, posterior fossa, and spinal epidural regions. Predisposing factors often include NF2 or a history of prior radiation, although over half are still sporadic. In comparison to adult cases, the clinical behavior is more difficult to predict.
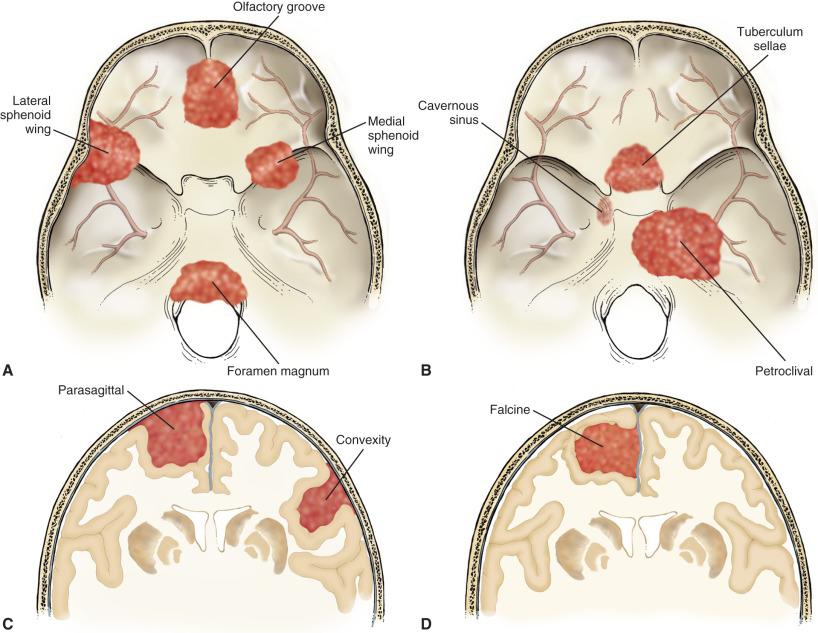
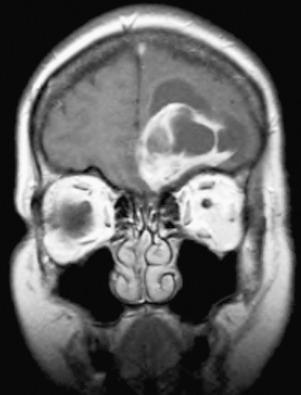
Meningiomas occur all along the craniospinal axis ( Fig. 13.1 ). Intracranially, they are most common over the convexity and parasagittal regions, with approximately half extending to the superior sagittal sinus and half affecting more lateral areas ( Fig. 13.2A ). Falcine meningiomas attach to the dense fibrous tissue of the falx rather than the sinus per se, while parasagittal meningiomas are located slightly more laterally ( Fig. 13.1C and D ). “Skull base meningiomas” are often technically challenging to resect and variably involve olfactory groove, parasellar region, lateral or medial sphenoid wing ( Fig. 13.2B ), foramen magnum, cavernous sinus, or tuberculum sella ( Fig. 13.1A and B ). Orbital (optic pathway) meningiomas or those that involve the anterior visual pathway in general are often highly invasive and frequently recur, despite a benign histologic appearance. Recent studies suggest some heterogeneity in this location, with those involving the spheno-orbital region being most clinically aggressive, especially those with 1p and 6q chromosomal losses. Nevertheless, advances in radiation therapy have generally improved local control for these tumors. In the posterior fossa, they arise in regions such as the petrous bone, tentorium cerebelli, and foramen magnum. Tentorial meningiomas may be supratentorial, infratentorial, or both.
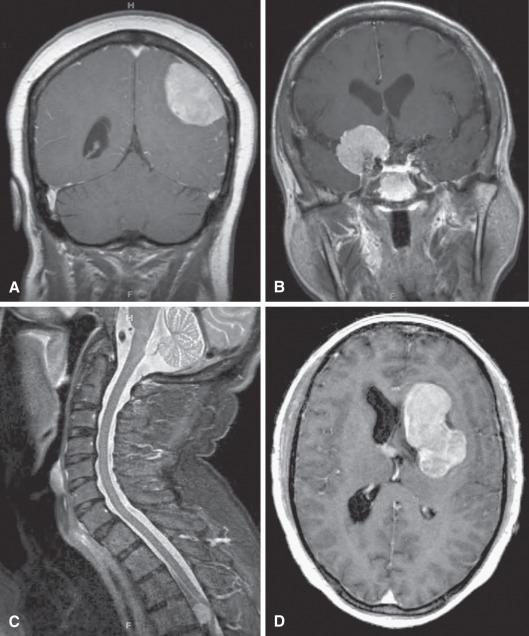
Spinal meningiomas occur mostly in the thoracic region ( Fig. 13.2C ), although males are less likely to show this regional predilection. Cervical and thoracic examples mostly present anterior and posterior to the cord, respectively. Most cover multiple segments and present as intradural extramedullary tumors similar to schwannomas. Less commonly, however, they present as leptomeningeal or epidural masses.
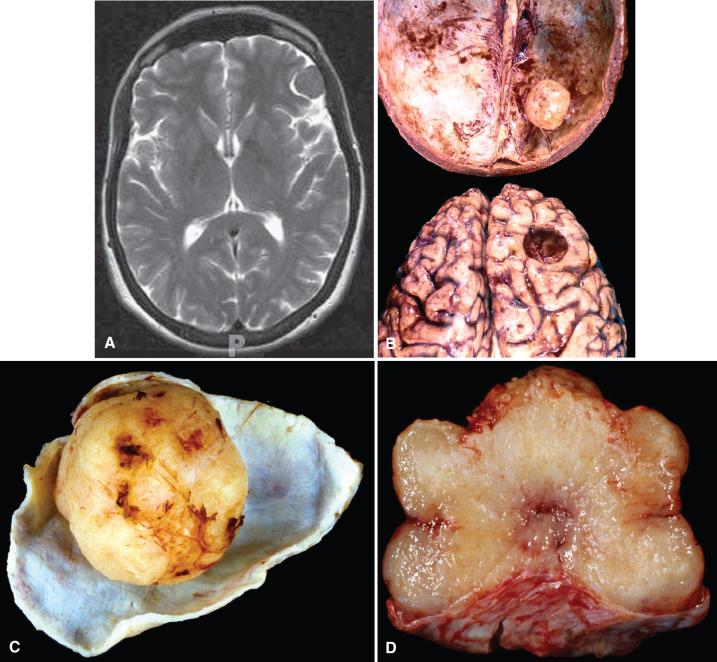
Intraventricular meningiomas are relatively rare (0.5% to 3%) and most likely originate from the tela choroidea of the choroid plexus ( Fig. 13.2D ). Lateral ventricles are most often involved, and there is an unexplained left-sided predilection. Most reach a large size by the time they present, making complete resection difficult.
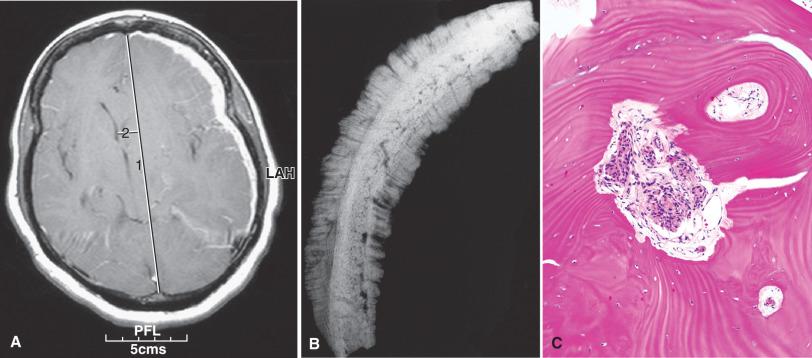
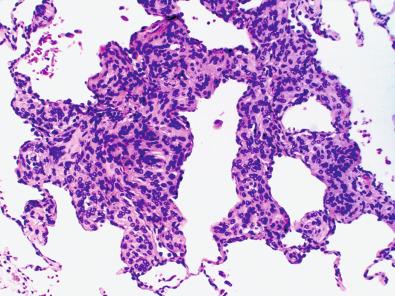
Extracranial or extradural meningiomas have been reported in the orbit (see previous discussion), scalp, paranasal sinuses, nasal cavities, and the skull (“intraosseous meningioma”). In the latter, one must first rule out the possibility of bone invasion from a thin en plaque or carpet-like dural-based meningioma ( Fig. 13.3 ). The origin of other extracranial meningiomas is controversial. In cases where extension of intracranial meningioma has been ruled out, meningothelial heterotopias or rests seem most plausible. Peripheral nerve, lung, salivary gland, and the soft tissue of the little finger are even rarer sites of supposed involvement. In the lung, minute meningothelial pulmonary nodules are thought to arise from reactive or metaplastic changes and provide one viable histogenetic explanation ( Fig. 13.4 ). The function of these unusual structures is unclear, but may involve drainage of pulmonary edema fluid. Some of the older case reports could also include misdiagnosed perineuriomas, given the extensive morphologic and immunohistochemical overlap of this rare entity (see Chapter 15 ).
Clinical manifestations of meningiomas vary with location and may also include nonspecific symptoms of mass effect. Headaches, seizures, personality changes, and localizing signs such as hemiparesis, sensory deficits, and ataxia are most common. Parasagittal meningiomas may cause leg weakness combined with urinary incontinence. Parasellar and orbital meningiomas often produce visual symptoms that occasionally lead to blindness. Similarly, hearing loss is common in the cerebellopontine angle meningiomas. Spinal examples compress the cord, leading to motor and sensory deficits. Those in locations with greater room for growth (e.g., lateral ventricles) can be asymptomatic for years, finally reaching a critical mass that brings it to clinical attention. It is also well known that patient morbidity and mortality increase with peritumoral cerebral edema, as does the likelihood of psychiatric manifestations.
The WHO grade II meningiomas (atypical, clear cell, chordoid) are characterized by considerably increased risks of recurrence when compared with the benign (WHO grade I) counterparts, even after a gross total resection. They also have a modest but statistically significant increased mortality rate. Their accurate identification is therefore of critical importance. The estimated 5-year recurrence rate for atypical meningiomas is 40% even after gross total resection, as compared with 5% for similarly treated benign meningiomas. After subtotal resection, the vast majority of atypical meningiomas recur. At a minimum, these tumors require careful radiologic follow-up; whether they should be treated with ancillary radiation therapy remains controversial and often depends on other variables as well, such as patient age and extent of resection.
In addition to increased recurrence rates, WHO grade III tumors (anaplastic, papillary, rhabdoid) are associated with significant risks of highly aggressive local disease, widespread dissemination, and tumor-related patient death. As such, they are considered malignant meningiomas. They lack the usual female predominance of benign meningiomas and present either as anaplastic from the outset (de novo) or via progression from atypical or benign meningiomas. Either way, the prognosis is poor with median survival of 2 to 5 years. Roughly a quarter of patients live considerably longer, and most of their tumors lack 9p21 deletions (see Molecular Genetics section).
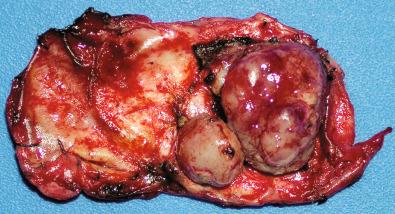
Multiple meningiomas are typical of NF2, but are also seen in sporadic adult patients, representing roughly 3% of surgical and up to 8% of postmortem cases. In some patients, there is one predominant meningioma with one or more satellite masses ( Fig. 13.5 ), either in close proximity or at a distance; they often show the same variant type morphology, many being transitional or psammomatous. The number of multiple meningiomas varies, with as many as 47 histologically similar tumors reported in one non-NF2 patient. Often, the same NF2 gene mutation is found in multiple meningioma patients in all the tumors removed from a single patient, consistent with a monoclonal origin despite the multicentricity. This suggests intradural spread as a mechanistic explanation (see Fig. 13.10C and D ), which is further supported by the common finding of microscopic tumor foci in otherwise grossly unremarkable dura samples from meningioma patients. In the rarer case of a child with multiple meningiomas, this is nearly pathognomonic for NF2.
Neuroimaging plays a critical role in the diagnosis of meningiomas and their therapeutic planning. Meningiomas are commonly isodense to gray matter on noncontrast CT and T1-weighted magnetic resonance imaging (MRI) studies, making it difficult to appreciate their borders. However, they are avidly contrast enhancing, such that meningiomas as small as 3 mm are often detectable. Radiologists use the “dural tail” sign as a helpful feature to diagnose meningioma or to confirm that the lesion is indeed extra-axial, rather than parenchymal in origin (see Fig. 13.2A ). This refers to the tail-like extension of contrast enhancement into adjacent dura. In fact, this is a nonspecific finding commonly encountered in nearly all dural-based masses, but given that meningioma is so much more common, it often leads to the correct diagnosis. Microscopically, this dural tail often consists merely of reactive hypervascular dura, although small foci of tumor can also be seen occasionally. T2-weighted images vary with tumoral consistency, such that firm, fibroblastic meningiomas often appear hypointense, whereas hyperintensity is often associated with soft, vascular, and/or meningothelial meningiomas. Calcifications appear dark on T1 MRI and bright on CT. Necrosis is typically also dark on T1, but bright on T2.
Most meningiomas display a smooth, rounded edge that appears to compress rather than invade the adjacent brain ( Fig. 13.6 ). An irregular tumor–brain interface has been associated with increased risk of finding brain invasion and other high-grade features on histology ( Fig. 13.7 ). Other features that have been associated with more aggressive meningiomas include increased signals on both T1- and T2-weighted MRI, “mushrooming” into the adjacent brain (i.e., irregular contour), extensive edema, lack of calcifications, central necrosis, and low apparent diffusion coefficient (ADC) values on diffusion-weighted imaging. However, these radiopathologic correlations are far from perfect and there are many exceptions.
Extent of radiologically detected peritumoral cerebral edema (dark on T1, bright on T2 MRI) is highly variable, and, not surprisingly, it is often more prominent in symptomatic versus incidentally detected meningiomas. Extensive edema has also been associated with large tumor size, recruitment of pial vascular supply, location in the convexity or middle fossa, an irregular (often “mushroom-like”) brain–tumor interface, T2 bright signal intensity, microscopic brain invasion, histologic malignancy, and variant histology that includes secretory, microcystic angiomatous, and/or lymphoplasmacyte-rich features (see Histologic Variants and Grading section).
Meningiomas show a diverse range of gross appearances, depending mostly on their locations, growth patterns, and histologic subtypes (see Histologic Variants and Grading section). Most are rubbery, well circumscribed, spherical, and firmly attached to the inner surface of the dura (see Fig. 13.6 ). The tumor surface is usually smooth or bosselated, like cauliflower. The cut surface is white, yellow, or tan, often with intersecting white streaky bands of fibrous tissue. Xanthomatous or lipomatous “metaplasia” imparts a yellow color, whereas foci of metaplastic cartilage and bone appear white. Psammoma bodies (“sand-like” from the Greek word psammos ) impart a gritty cut surface. Decalcification is often needed for the psammomatous variant, ossified tumors, and those with bone invasion. The microcystic variant has a moist glistening cut surface, consistent with Masson's original name of “meningioma humide.” Secretory meningiomas have a tan-red gland-like appearance. Hemorrhage and necrosis are relatively rare findings in benign meningiomas, but are seen more commonly after therapeutic embolization. More cellular tumors such as atypical and anaplastic meningiomas tend to be softer and more fleshy, the latter often being extensively necrotic on cut surface (see Figs. 13.5, 13.7C and D ).
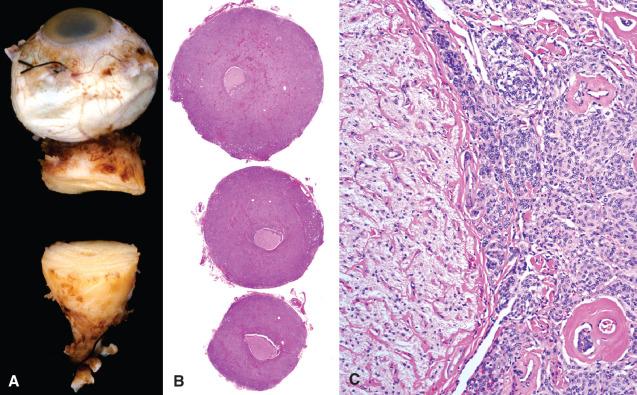
The cerebral cortex adjacent to meningiomas often appears compressed and distorted, sometimes forming a cavity that molds perfectly around the protuberant surface of the meningioma (see Fig. 13.6B ). Nevertheless, the brain–tumor interface is typically distinct, with most meningiomas easily peeling away. Those that don't are either brain invasive or merely adherent to the leptomeninges. In contrast to true brain invasion, infiltration of adjacent dura, venous sinuses, bones, and soft tissue is relatively frequent, even in benign examples. For example, orbital meningiomas are often highly invasive of adjacent bone and soft tissue, typically surrounding and compressing the optic nerve with resulting loss of vision ( Fig. 13.8 ). Other cranial and spinal nerves/ganglia are occasionally invaded by meningioma as well.
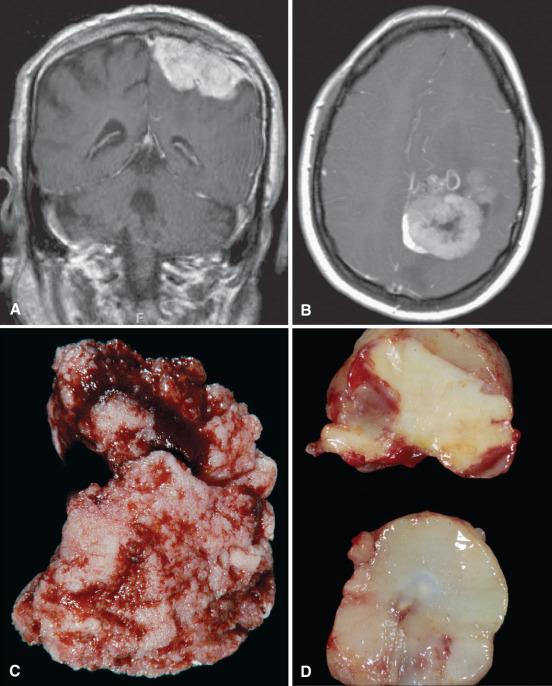
Some meningiomas are dumbbell shaped with tumor masses growing out on both sides of a dural reflection, such as the falx or tentorium. Intraventricular meningiomas are often large, potentially expanding and distorting the ventricular cavity, often with resultant obstructive hydrocephalus (see Fig. 13.2D ). The en plaque meningiomas are defined by a carpet-like growth pattern, resulting in long stretches of thickened, sometimes rough or shaggy dura (see Fig. 13.3A ). They are most commonly encountered in the skull base, especially the sphenoid wing, where they tend to invade the adjacent bone, with associated hyperostosis ( Fig. 13.3B and C ).
Cystic meningiomas represent 2% to 4% of all cases ( Fig. 13.9 ). They are mostly benign and are seen more commonly in children. Any meningioma can develop grossly evident cysts, although the microcystic variant is most frequent, with macrocysts likely forming from coalescence of microscopic ones. In some of these cases, the dural attachment is subtle and there may even be ring enhancement, mimicking glioblastoma. Cystic elements are intratumoral and/or peritumoral with suspected etiologies including secretory, reactive, and degenerative changes. In rare cases, a cystic meningioma may form within a preexisting malformation, such as an arachnoid cyst.
The histopathologic spectrum of meningiomas is extremely wide (see next section), although there are several characteristic features, seen in most tumors regardless of subtype or grade. Key features include whorl formation, psammoma bodies, and characteristic cytologic features such as nuclear clearings (“holes”) and pseudoinclusions. As stated earlier, one of the common characteristics of meningothelial cells is to wrap around various structures including themselves, resulting in cellular whorls ( Fig. 13.10A ). Over time, the cells within these whorls deposit intercellular collagen, progressively leading to hyalinized whorls, typically with concentric laminations. As these structures mineralize, one sees concentric calcifications known as psammoma bodies. Cytologically, most meningioma cells are moderately sized with ample eosinophilic cytoplasm and oval nuclei with delicate chromatin, often displaying numerous clear vacuoles (“holes”) and pseudoinclusions ( Fig. 13.10B ). The latter represent cytoplasmic invaginations into the nuclei, such that on cross section, they appear membrane bound with identical tinctorial qualities to that of the cytoplasm (i.e., eosinophilic, rather than clear). These cytologic features are by no means specific to meningiomas, but are helpful for the differential diagnosis nonetheless, particularly in variants that do not otherwise show classic architectural features, such as whorling. The vast majority of meningiomas, even the most benign, invade the dura, and this likely represents a common mode of spread beyond the point of dural attachment ( Fig. 13.10C and D ). Other histologic features such as cytoplasmic characteristics, degree of collagenization, stromal background, architectural growth patterns, vascular changes, and invasiveness into bone, soft tissue, and brain are highly variable and discussed in greater detail in the next section.
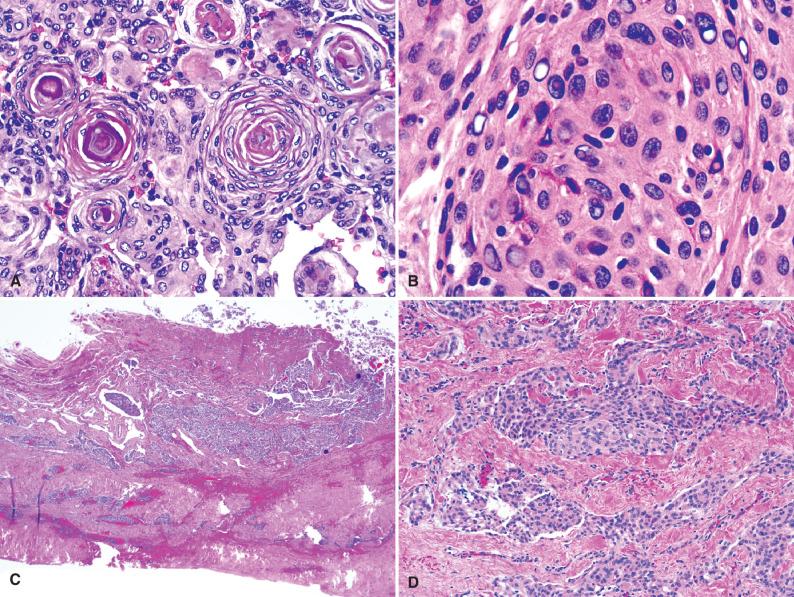
The vast histopathologic spectrum encountered in meningiomas is astonishing and has led to several complex classification schemes with numerous variants described over the years. In 1938, Cushing and Eisenhardt stratified meningiomas into 9 main types and 20 subtypes. Since that time, several variants have been reclassified as distinct entities. For example, the meningeal solitary fibrous tumor/hemangiopericytoma is now considered a distinct form of sarcoma (see Chapter 14 ). However, in the WHO 2016 classification scheme, there remain nine mostly benign (WHO grade I), three intermediate category (WHO grade II), and three malignant (WHO grade III) subtypes ( Table 13.1 ). For the more aggressive subtypes, it remains unclear what percentage of the variant histology must be present before the higher grade is automatically assigned. For the four subtypes in this category (clear cell, chordoid, papillary, rhabdoid), it has been suggested that the WHO grade II or III designation only be assigned if the majority of the tumor (>50%) shows the appropriate morphology ; nevertheless, a study of rhabdoid meningiomas lacking overt features of malignancy was associated with a mostly favorable prognosis even when more than 50% of cells appeared rhabdoid. In contrast to these rare variants, any of the more common meningioma subtypes may qualify as either atypical (WHO grade II) or anaplastic (WHO grade III) if diagnostic criteria are met even focally ( Table 13.2 ), either at initial presentation or in subsequent recurrences. These subtypes are discussed in greater detail in this chapter.
| Meningioma Variants That Are Mostly Benign (WHO Grade I) | WHO Grade II Meningiomas |
|---|---|
| Meningothelial | Atypical (any variant plus criteria) |
| Fibrous (fibroblastic) | Clear cell |
| Transitional (mixed) | Chordoid |
| Psammomatous | |
| Angiomatous (vascular) | |
| Microcystic | WHO Grade III Meningiomas |
| Secretory | Anaplastic (any variant plus criteria) |
| Lymphoplasmacyte rich | Papillary |
| Metaplastic | Rhabdoid |
| Benign Meningioma (WHO Grade I) |
|
| Atypical Meningioma (WHO Grade II) (Any of Three Major Criteria) |
|
| Anaplastic (Malignant) Meningioma (WHO Grade III) (Either of Two Criteria) |
|
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here