Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
There has been a dramatic shift in the treatment of advanced cutaneous melanoma within the last 5 years. Until 2011, the only FDA-approved therapies for metastatic melanoma were dacarbazine chemotherapy or high-dose interleukin-2 (IL-2) immunotherapy, which each have limited efficacy and can carry significant toxicity. While response rates may range as high as 20%, dacarbazine generally fails to prolong overall survival (OS) ( ) and has typical chemotherapy-associated toxicities including bone marrow suppression, alopecia, nausea, vomiting, and fatigue that worsen patient quality of life. IL-2 has a 16% response rate, with 6% of patients achieving a complete response. Although IL-2 induces durable responses in a minority of patients, the trade-off is the toxic adverse events, which can be severe, including a septic shock-like presentation with hypotension, fevers, oliguria, nausea, and vomiting with a drug-related death rate of 2% ( ). IL-2 administration requires thorough monitoring in an intensive care unit in the event that vasoactive pressors are required. Elderly patients or those with significant medical comorbidities or poor performance status are not IL-2 candidates.
Newly available advanced melanoma therapies have enhanced efficacy over dacarbazine and IL-2 since FDA approval of the immunotherapies ipilimumab in 2011, followed by pembrolizumab and nivolumab in 2014, as well as the BRAF inhibitors vemurafenib in 2011 and dabrafenib in 2013. With these new therapies comes a unique set of adverse events that are manageable when promptly recognized and addressed.
Ipilimumab is an anticytotoxic T-lymphocyte antigen-4 (CTLA-4) monoclonal antibody that blocks the immune-inhibitory action of CTLA-4 and thereby promotes an immune response. Ipilimumab was initially studied in a phase II clinical trial that randomized previously treated advanced melanoma patients to three doses of ipilimumab: 10, 3, or 0.3 mg/kg every 3 weeks. Overall response rates (ORR) were 11.1%, 4.2%, and 0%, respectively, with drug-related adverse events becoming more prominent with increasing dose ( ). The phase III trial randomized patients to three treatment arms: ipilimumab 3 mg/kg plus a gp100 peptide vaccine, ipilimumab 3 mg/kg plus a placebo vaccine, or gp100 vaccine alone. Ipilimumab with or without vaccine demonstrated prolonged OS (10 and 10.1 months, respectively), compared to vaccine alone (6.4 months) ( ). In these trials and in a retrospective analysis of 14 completed phase I-III trials that enrolled melanoma patients to receive ipilimumab, drug-related adverse events occurred in up to 80% of patients. At least 60% of the toxicities were immune-mediated, most commonly manifesting as dermatitis (49%) including pruritis or rash, gastrointestinal effects (31%) including diarrhea and hepatitis, fatigue, and endocrinopathies ( ). Up to 25% of patients experienced grade 3 or 4 events ( ). Less than 1% of patients had rare immune-related adverse events (irAEs) such as uveitis, pneumonitis, pancreatitis, and nephritis ( ).
Programmed death-1 (PD-1) receptor monoclonal antibodies pembrolizumab and nivolumab came on the market after ipilimumab, for both single agent use or as combination therapy in the case of ipilimumab and nivolumab. Pembrolizumab, compared to ipilimumab, demonstrated improved response rates (33% vs 12%), progression-free survival (PFS) and OS in advanced melanoma patients with a decreased rate of grade 3–5 drug-related adverse events ( ). For pembrolizumab treated patients, adverse events had later onset times when compared to ipilimumab and there was a decreased rate of drug discontinuation. The most common adverse events for pembrolizumab are fatigue (20%), diarrhea (15%), rash (14%), and pruritis (14%) ( ). Similarly, single-agent nivolumab has also demonstrated efficacy in advanced melanoma with a 25% ORR ( ) and 1 year OS rates ranging from 62% to 73% ( ; ). Nivolumab is similarly well tolerated, with the most common adverse events being fatigue (20%), pruritis (17%), and nausea (17%). Only 7% of patients needed to discontinue nivolumab due to adverse events ( ).
Currently, the combination of ipilimumab and nivolumab has demonstrated a robust ORR of 40–61% which is higher than for either agent alone, with impressive 2 year OS rates of 88% ( ). While the combination therapy is extremely promising in terms of potential for durable responses, it carries significantly increased toxicity, with 93% of patients experiencing treatment-related adverse events, and thus candidates for combination immunotherapy must be carefully selected. Grade 3–4 treatment-related adverse events occurred in 53% of patients in the phase I combination therapy trial, most commonly reported as elevated serum lipase (13%), aspartate aminotransferase (AST) (13%), and alanine aminotransferase (ALT) (11%). The combination group also demonstrated higher rates of rash (55%), pruritis (47%), fatigue (38%), and diarrhea (34%) compared to single-agent immunotherapy alone ( ). Furthermore, a significant percentage of patients (21–36%) on combination ipilimumab/nivolumab require discontinuation of therapy due to adverse events ( ).
Despite some of the varying safety profiles between anti-CTLA-4 antibodies, anti-PD-1 antibodies, and combination immunotherapy, irAEs should be recognized and managed similarly for each agent. The time to onset of irAEs are generally similar among the immunotherapies, although irAEs associated with anti-PD-1 agents may be later in time of onset and slower to resolve than irAEs associated with anti-CTLA-4 agents. Chronologically, dermatologic manifestations are usually the first to arise, followed by diarrhea or colitis, which typically present after the first 1–3 cycles of immunotherapy. Liver toxicity usually occurs later, at least after the third cycle of therapy, and endocrinopathies present as late irAEs, even when the patient may have been off immunotherapy for months or even years ( ). Grade 3 or 4 treatment-related adverse events occur with highest incidence in patients treated with ipilimumab/nivolumab combination therapy (55%), and less frequently for single agent ipilimumab (27%), nivolumab (16%), or pembrolizumab (10%) ( ).
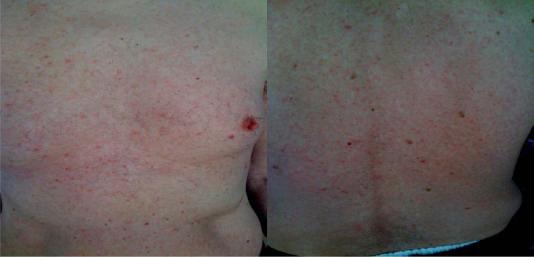
Dermatologic toxicity of immunotherapy usually arises early after the first or second cycle of therapy, tends to be mild, and presents as a maculopapular erythematous rash or as pruritis ( ). Pruritis and rash occur in up to 24% and 19% of ipilimumab-treated patients, respectively ( ), while skin disorders of any grade arise in 70% of patients treated with combination ipilimumab/nivolumab, although the majority are grade 1–2 ( ). Pruritis may occur to a slightly lesser degree with pembrolizumab compared to ipilimumab ( ).
Grade 1–2 pruritis and rash do not require dose reductions and usually resolve with administration of oral antipruritics, such as diphenhydramine or hydroxyzine, taken as needed and/or topical corticosteroids, such as betamethasone 0.1% cream or urea-containing creams, applied to the affected area ( ). If the rash does not improve within 1 week with these conservative therapies, oral corticosteroids should be considered. Vitiligo may also appear as a dermatologic irAE and is permanent, however is thought to represent a surrogate of response to treatment ( ). Rarely, severe or life threatening grade 3–5 immune-mediated dermatitis can occur, as represented by Stevens-Johnson syndrome, toxic epidermal necrolysis, or appearance of a bullous, ulcerated, or hemorrhagic rash which occurs in less than 1% of patients ( ). Grade 3 dermatologic irAEs are treated by holding the next immunotherapy dose and initiating oral corticosteroids dosed as prednisone 1 mg/kg daily, to be tapered over 4 weeks ( ). Grade 4 dermatologic irAEs mandate permanent discontinuation of immunotherapy and again, oral corticosteroid administration with 1–2 mg/kg of prednisone daily with a prolonged taper of 4 weeks or more is indicated ( ) ( Figs. 20.1 and 20.2 ).

Diarrhea is a common irAE, occurring in 32% of ipilimumab-treated patients enrolled on the phase III trial, with 5% of patients experiencing grade 3 diarrhea ( ). The degree of diarrhea appears to be dose-dependent as shown in the phase II ipilimumab trial in which grade 3–4 diarrhea occurred in 14% of patients on ipilimumab 10 mg/kg but in only 1% of patients on 3 mg/kg ( ). Diarrhea also occurs with the anti-PD-1 antibodies, but to a lesser extent and decreased severity, with an incidence of 15% for both nivolumab and pembrolizumab ( ). Patients on combination ipilimumab/nivolumab do experience higher rates of diarrhea of any grade (34–44%) ( ).
Onset of diarrhea usually occurs after the second cycle of immunotherapy, at a median time of 7.4 weeks after treatment initiation for ipilimumab. Grade 1 diarrhea consisting of less than 4 diarrheal bowel movements over baseline in 24 h should be managed with the antimotility agent loperamide. Patients should follow the American Dietary Association colitis diet, increase fluid hydration, and receive electrolyte repletion as needed ( ). Grade 2 diarrhea defined as 4–6 diarrheal stools per day over baseline should prompt an infectious work-up consisting of stool culture and examination for fecal leukocytes and Clostridium difficile and oral atropine/diphenoxylate four times daily can be added. If symptoms persist longer than 1 week, systemic corticosteroids at a dose of 0.5 mg/kg/day should be started and endoscopic exam should be considered to look for colitis, inflammatory bowel disease, or infection ( ).
For grade 3–4 diarrhea defined as at least 7 diarrheal bowel movements over baseline in 24 h, immunotherapy should be permanently discontinued. Intravenous fluid hydration and methylprednisolone 125 mg IV should be initiated. Oral prednisone 1–2 mg/kg daily or dexamethasone 4 mg every 4 h should follow as a slow taper over at least 4 weeks ( ). Average time to resolution of grade 2 or higher diarrhea treated with a median dose of prednisone 80 mg daily was 2.3 weeks for ipilimumab treated patients ( ). For patients who do not demonstrate symptomatic improvement within 48–72 h of high dose steroid initiation, the tumor necrosis factor alpha monoclonal antibody infliximab should be administered at a dose of 5 mg/kg ( ). Infliximab can be administered once every 2 weeks as necessary for persistent symptoms, but is contraindicated in patients with bowel perforation or sepsis. In the phase III ipilimumab study, four patients required infliximab for treatment of grade 3 or higher diarrhea and colitis ( ). If possible, tuberculosis testing is recommended prior to infliximab use ( ). If diarrheal symptoms worsen again while on decreasing doses of a steroid taper, the steroids should be increased back to a dose of 80–100 mg per day and then more slowly tapered and inflimximab can be reinitiated if necessary ( ). Although diarrheal prophylaxis has been investigated in patients receiving ipilimumab, a phase II trial demonstrated that prophylactic budesonide does not prevent grade 2 or higher ipilimumab-induced diarrhea and so prophylactic measures have not been recommended ( ).
Abdominal pain and presence of stool blood or mucous can be a sign of colitis, which can progress to bowel obstruction and rarely perforation (<1%) if not promptly addressed ( ). Surgical consult should be requested if the patient develops an ileus with severe diarrhea ( ). Anti-PD-1 agents have a lower rate of grade 3–5 colitis (1–3%) compared to ipilimumab (7%) ( ).
Immune-mediated hepatic impairment is uncommon and occurs in approximately 4% of ipilimumab treated patients ( ) and 2% of pembrolizumab treated patients ( ) usually occurring after the second cycle of immunotherapy ( ). Incidence of hepatic toxicity is 15–23% in patients treated with the ipilimumab/nivolumab combination, and 15% of cases are grade 3–4 ( ). The hepatitis usually presents as an asymptomatic rise in serum liver transaminases or bilirubin or can involve fatigue and fever ( ) and while infrequent, hepatitis can be severe and must be urgently managed ( ). Baseline liver function tests should be performed prior to each cycle of immunotherapy and clinicians should consider screening for hepatitis B or C prior to immunotherapy initiation, particularly given that immunotherapy trials typically exclude patients with active hepatitis B or C ( ). Clinicians should counsel patients against active alcohol use and ingestion of hepatotoxic drugs while on immunotherapy ( ).
Upon any noted rise in liver function tests, clinicians should suspect disease progression, medication toxicity, or viral hepatitis, all of which should be ruled out before diagnosing immunotherapy-mediated hepatitis. Once serum levels rise to greater than or equal to 2 times the upper limit of normal, repeat testing is recommended every 1–3 days until stabilization or a decrease is noted, and autoimmune hepatitis evaluation including antinuclear, antismooth muscle, antimitochondrial, and antiliver-kidney microsomal antibodies should be checked ( ). Immunotherapy should be held for grade 2 hepatotoxicity, defined as elevation of AST or ALT to greater than 2.5–5 times the upper limit of normal or elevation of total bilirubin to 1.5–3 times the upper limit of normal, and should be permanently discontinued for hepatotoxicity grade 3 or above ( ). Grade 3–5 hepatotoxicity consisting of elevated AST or ALT above 5 times the upper limit of normal or elevated total bilirubin above 3 times the upper limit of normal should be managed with 24–48 h of high-dose intravenous glucocorticoids followed by oral prednisone 1–2 mg/kg tapered over at least 4 weeks. Liver function tests should be monitored at least weekly, and daily if the levels are greater than 8 times the normal. Although immunotherapy-induced hepatitis is usually responsive to steroids, mycophenolate mofetil 500 mg orally every 12 h can be administered if liver function tests do not begin to improve 48 h after steroid initiation and consultation with a hepatologist at that time is recommended ( ). In a rare cases refractory to these therapies, antithymocyte globulin has been effectively used ( ).
Endocrinopathies including hypothyroidism, hypopituitarism, hypophysitis, adrenal insufficiency, increase in serum thyrotropin level, and decrease in serum corticotropin level were all reported in total in 8% of patients on the ipilimumab arm of the phase III trial. Grade 3 events included one case of hypopituitarism and two cases of hypophysitis. Grade 4 events consisted of hypopituitarism and decrease in serum corticotropin level ( ). In patients on combination ipilimumab/nivolumab in the phase I trial, 13% developed immune-mediated endocrinopathies ( ).
Immunotherapy-induced hypophysitis typically involves the anterior pituitary, which secretes thyroid stimulating hormone (TSH), adrenocorticotropic hormone (ACTH), leutinizing hormone (LH), follicle stimulating hormone (FSH), prolactin, and growth hormone. The most common immunotherapy-related endocrinopathy is hypophysitis and usually presents after the third cycle of ipilimumab ( ). In a retrospective analysis from Memorial Sloan Kettering Cancer Center (MSKCC), the incidence of ipilimumab-treated patients developing hypophysitis was 8% with a median time to onset of 4 months after therapy initiation, however distant cases can occur, as hypophysitis developed in two patients at 8 and 19 months postipilimumab initiation ( ). Symptoms of hypophysitis can often be vague and include headaches, visual changes, fatigue, nausea, abdominal pain, mental status changes, and altered bowel habits. Of the 19 patients in the MSKCC analysis who developed hypophysitis, 16 had secondary adrenal insufficiency which presents as hypotension and electrolyte abnormalities (hyponatremia, hyperkalemia), 11 had abnormal thyroid function tests, and 3 had loss of ACTH secretion. Brain MRI in these patients will demonstrate pituitary enlargement and/or abnormal enhancement ( ).
Treatment of hypophysitis first involves recognition, which can often be difficult as the symptoms are nonspecific and may mimic other common melanoma issues such as brain metastases, which are important to rule out. TSH, free T4, and electrolytes should be checked prior to each immunotherapy cycle. For symptomatic patients in which an endocrinopathy is suspected, blood should be drawn prior to steroid initiation for cortisol, ACTH, free T3, free T4, TSH, as well as testosterone in males and FSH, LH, and prolactin in females ( ). Immunotherapy should be held for any grade 3–4 endocrinopathy or in patients with symptomatic pan-hypopituitarism ( ). Intravenous high dose steroids with mineralocorticoid activity such as methylprednisolone 1–2 mg/kg should be initiated, followed by oral prednisone 1–2 mg/kg daily with at least a 4 week taper. Importantly, evidence of severe dehydration or hypotension should make the clinician suspect an adrenal crisis, which is also managed with rapid initiation of high dose methylprednisolone. Infectious causes should be ruled out and an endocrinology consult is recommended. Immunotherapy should be permanently discontinued in the event of adrenal crisis. Patients with secondary adrenal insufficiency require physiologic hydrocortisone replacement and given that pituitary dysfunction can be long-lasting or permanent, patients should be informed that steroid maintenance may be life-long ( ). In the MSKCC analysis, 9/19 patients did not recover pituitary functioning, while three regained secretion of ACTH and two recovered LH and FSH secretion ( ).
Hypothyroidism occurs at a rate of 6–10% in patients on ipilimumab, pembrolizumab, or nivolumab ( ). Time onset is variable, ranging from months to years after the initiation of immunotherapy ( ). Pembrolizumab has a slightly increased rate of both hypothyroidism (9% vs 2%) and hyperthyroidism (3% vs 2%) compared to ipilimumab ( ). Most patients are either asymptomatic or present with fatigue and this usually resolves with initiation of hormone supplementation ( ).
Neurologic irAEs are rare complications of immunotherapy, usually related to anti-CTLA-4 therapy, and are mostly documented as case reports but should be suspected in patients treated with immunotherapy who present with new sensory or motor deficits ( ). Reported immune-mediated neurologic deficits include neuropathy, chronic inflammatory demyelinating polyneuropathy, transverse myelitis, myasthenia gravis, and Guillane-Barre syndrome ( ). Peripheral neuropathies may be only slightly symptomatic and can resolve spontaneously, although persistent grade 2 neuropathy is treated by withholding the next dose of therapy and administering a prednisone or dexamethasone taper over 4 weeks ( ). For more serious neuropathies and other neurologic irAEs, treatment involves discontinuation of immunotherapy and high dose steroid initiation such as prednisone 1–2 mg/kg daily. Some patients have required infliximab or plasmapheresis ( ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here