Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Periprosthetic joint infection (PJI) is a challenging and increasingly common complication after total joint arthroplasty (TJA). In 2009, 2.18% of both total hip arthroplasty (THA) and total knee arthroplasty (TKA) patients were treated for PJI, with an associated annual cost of $566 million. The definition of acute has been controversial. The Musculoskeletal Infection Society (MSIS) defines acute postoperative PJI as cases with symptoms presenting within 3 months of the index procedure and acute hematogenous PJI as patients with less than 6 weeks of symptoms, with onset more than 3 months from the index arthroplasty. The International Consensus Meeting (ICM) on Musculoskeletal Infection similarly defines acute postoperative PJI as onset during the first 3 postoperative months provided that patients are symptomatic for less than 3 weeks. Others have proposed 4- and 6-week cutoffs for acute PJI. Acute postoperative PJI accounts for 17% of all PJI.
This chapter will discuss the diagnosis and operative treatment of acute postoperative PJI. Prevention and management of associated wound complications will also be reviewed.
Diagnosis of PJI in the early postoperative period can be particularly challenging due to normal postoperative pain, variable wound appearances, and normal elevations of commonly used serum markers. Several studies, however, have examined optimal cutoffs for traditional markers of infection, including the erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), synovial white blood cell (WBC) count, and synovial polymorphonuclear cell percentage (PMN%) in the acute postoperative period. Yi et al. examined 73 patients who underwent reoperation within the first 6 weeks following THA and had a consistent evaluation for PJI. Of the 73 patients, 36 met modified MSIS criteria for acute PJI. The authors found that the serum CRP was strikingly accurate for diagnosis (area under the curve [AUC] 93%) with a suggested cutoff value of 93 mg/L and associated sensitivity of 88% and specificity of 100%. Serum ESR was found to be less reliable, with an AUC of 73% and a suggested cutoff value of 44 mm/h (sensitivity 92%, specificity 53%). Of the markers studied, the synovial WBC count was found to be the most accurate (AUC 98%), with a suggested cutoff of 12,800 cells/μL having a sensitivity of 89% and specificity of 100%. The differential was likewise quite accurate (AUC 91%), with a suggested cutoff of 89% PMNs associated with a sensitivity of 81% and specificity of 90%.
Bedair et al. performed a similar study in which they reviewed 146 patients who required aspiration within the first 6 weeks following TKA. Of these, 19 (13%) were considered infected based on positive culture on solid media, two broth cultures growing the same organism, or intraoperative findings of gross purulence. Once again, synovial WBC count was the most accurate marker for acute postoperative PJI with an AUC of 97%. A threshold of 27,800 cells/μL was most specific (84% sensitivity, 99% specificity), and a threshold of 10,700 cells/μL was most sensitive (95% sensitivity, 91% specificity). Similarly, serum CRP was extremely accurate for diagnosis, with an AUC of 73% and a suggested cutoff of 95 mg/L (68% sensitivity, 66% specificity). The ESR in this study was not found to be a reliable diagnostic marker (AUC 51%). Interestingly, a synovial differential of 89% PMNs was once again chosen as the optimal cutoff value, with a sensitivity of 84% and specificity of 69% (AUC 81%).
Based on these two studies, we recommend a thorough history and physical examination combined with serum CRP as the first step whenever there is concern for acute postoperative PJI. If the clinical suspicion for infection is high or if the serum CRP is near or above 100 mg/L (100 is just an easier number to remember), the joint is aspirated and the fluid is sent for synovial fluid WBC count, differential, and culture. If the synovial fluid WBC count is greater than 10,000 cells/μL (again, 10,000 is easier to remember than the values described in the studies by Yi et al. and Bedair et al. ) and/or the differential is greater than 90%, infection is presumed. If uncertainty remains regarding infection status, the cultures may be followed to determine the appropriate treatment, but this is rarely needed if antibiotics have not been administered prior to evaluation.
Irrigation and debridement combined with a modular bearing surface exchange is the most commonly used treatment for an acute postoperative infection in North America. Reported results are extremely variable and depend partly on how success is defined. The MSIS proposed a four-tier outcome-reporting tool, also adopted by the 2018 ICM, to standardize the definition of successful PJI treatment in the literature ( Table 27.1 ). , Successful management of PJI includes tiers 1 and 2, in which the infection is controlled with or without chronic antibiotic therapy, respectively. An infection is considered controlled only in patients who no longer meet the MSIS criteria for PJI and those who do not require any additional surgery. Tiers 3 and 4, considered failed treatment, include patients requiring reoperation, unanticipated spacer retention, or those who die following treatment of PJI.
| Tier | Definition |
|---|---|
| Tier 1 | Infection control with no continued antibiotic therapy |
| Tier 2 | Infection control with patient on suppressive antibiotic therapy |
| Tier 3 | Need for reoperation and/or revision and/or spacer retention (assigned to subgroups of A, B, C, D, E, and F based on the type of reoperation) |
| A | Aseptic revision greater than 1 year from initiation of PJI treatment |
| B | Septic revision (including debridement, antibiotics, and implant retention [DAIR]) greater than 1 year from initiation of PJI treatment (excluding amputation, resection arthroplasty, and fusion) |
| C | Aseptic revision less than or equal to 1 year from initiation of PJI treatment |
| D | Septic revision (including DAIR) less than or equal to 1 year from initiation of PJI treatment (excluding amputation, resection arthroplasty, and fusion) |
| E | Amputation, resection arthroplasty, or fusion |
| F | Retained spacer |
| Tier 4 | Death (assigned to subgroups of A or B) |
| A | Death less than or equal to 1 year from initiation of PJI treatment |
| B | Death greater than 1 year from initiation of PJI treatment |
The primary contraindication to any type of a debridement procedure is a loose implant. , While the appropriate timing for a debridement is again controversial, we tend to perform them within the first 6 weeks postoperatively; at least in our practice, patients seem to present most commonly around the 4-week mark. While some surgeons have advocated for serial debridements, , we generally will perform only a single debridement, as failure of an irrigation and debridement usually suggests to us that the implants require removal. Finally, surgeons may want to consider delaying surgical intervention until the causative organism has been identified. While this is not typically done in our own practice, the knowledge that an organism is resistant to standard antibiotics, for example, may encourage the clinician to pursue more aggressive management, including implant removal.
Successful treatment relies on timely and aggressive debridement of infected tissue and removal of all modular components. For this reason, arthroscopic debridement is discouraged, as it significantly limits the surgeon’s exposure to all infected surfaces and precludes modular component exchange. , Removal of modular components is essential not only to eliminate a potential site of early biofilm formation and decrease the overall bioburden but also to improve visualization for debridement of the infected tissue. , Choi et al. reported a 53% success rate at 3 years after treatment of acute postoperative and acute hematogenous TKA PJI when the polyethylene component was exchanged compared with a 0% success rate with polyethylene retention ( P < .001). Similarly, in patients with acute postoperative and acute hematogenous THA PJI, Grammatopoulos et al. found 10-year implant survival rates of 86% with a modular bearing exchange and 68% without ( P = .02).
Arguably, the most critical portion of the procedure is the debridement. In either a hip or a knee, a generous incision and exposure is recommended to fully visualize the joint space and to complete a full synovectomy. Around the knee, it is typically possible to define a clear interval between the joint capsule and synovium ( Fig. 27.1 ). Exploiting this interval facilitates a near complete anterior synovectomy; unfortunately, obtaining access posteriorly is far more challenging without removal of the implants. Retained implants can also be scrubbed with a brush to remove any potential biofilm that may be present.
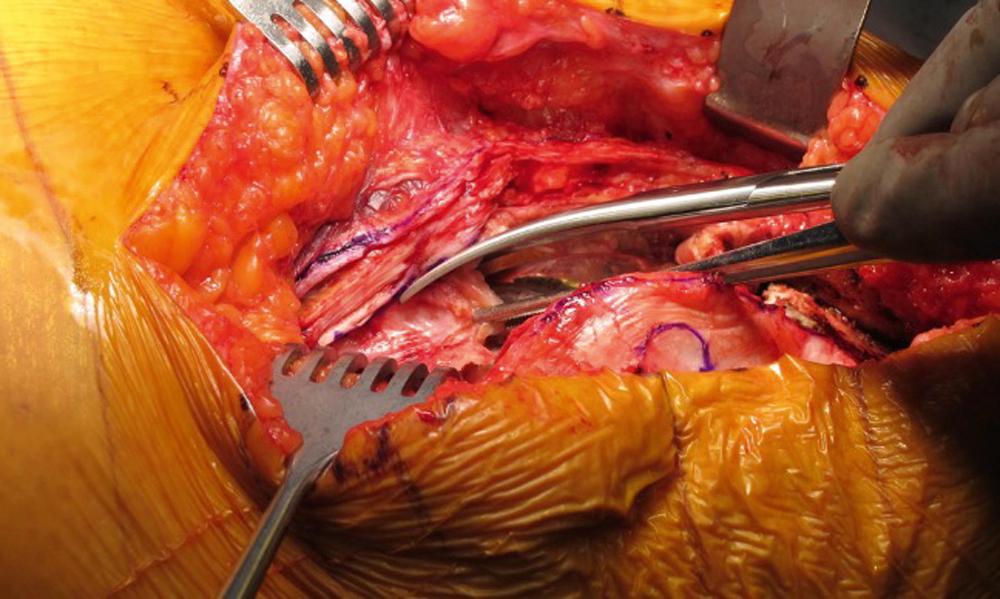
During the debridement, five or more tissue samples are typically sent for culture. We recommend taking these tissue samples from periprosthetic regions that clinically appear most infected, including the deep tissue as well as the modular interfaces. In addition to culture, periprosthetic tissue should be sent for histopathology. , Although perioperative antibiotics are frequently withheld until intraoperative cultures are obtained, there is no evidence that antibiotics administered at the time of anesthesia induction reduce intraoperative culture sensitivity. Thus, we do not routinely hold antibiotics preoperatively at our institution. In practice, patients are typically aspirated prior to returning to the operating room; thus, a fluid sample has already been sent for culture. We start antibiotics after the aspiration and prior to surgery and continue them postoperatively.
The type and volume of irrigation solution has also been extensively investigated. In general, 6 to 9 L should be used. , The volume of irrigation appears to be a more important predictor of success than the type of solution, as studies utilizing less than 6 L reported largely inferior results irrespective of irrigant. , Normal saline is the most common solution, but dilute povidone-iodine, Castile soap, sterile water, and antibiotic solutions have been used as well. , , After exposing Staphylococcus aureus and Escherichia coli to six common irrigation solutions in vitro, Goswami et al. found that only dilute povidone-iodine, chlorhexidine, and sodium hypochlorite were effective against both organisms. Of these, dilute povidone-iodine was the least cytotoxic to chondrocytes, osteoblasts, and fibroblasts. In patients undergoing primary THA and TKA, Brown et al. reported that a 3-minute dilute povidone-iodine wash was associated with an acute PJI rate of 0.15% compared with 0.97% in patients who received a normal saline wash ( P = .04). Calkins et al. similarly found that dilute povidone-iodine was more effective than normal saline irrigation in a randomized controlled trial with 457 patients undergoing aseptic TJA revisions, with acute PJI rates of 0.4% and 3.4%, respectively, in patients who did and did not get a dilute betadine lavage ( P = .038).
Recent literature has suggested that topical vancomycin inserted into the wound may reduce the incidence of acute postoperative PJI. Although controversial, we have recently included this in our own protocol for managing acute PJI. Riesgo et al. found that addition of 1 g vancomycin powder placed both sub- and suprafascially for acute PJI resulted in a failure rate of 16.7% at 27 months compared with a 37% failure rate at 43 months in a matched cohort that did not receive vancomycin powder ( P < .05). However, this study was confounded by significantly longer follow-up in the control group. Additionally, there is no clear support for intraarticular antibiotics or resorbable antibiotic-impregnated pellets at this time. Future randomized controlled trials are needed to investigate vancomycin powder further.
Our current regiment now includes a three-step protocol for irrigation following removal of the modular bearing surface and complete synovectomy. First, we irrigate the wound using pulse lavage with 3 L of normal saline. We then soak the wound for 2 minutes in a 50:50 solution containing 100 mL of 3% hydrogen peroxide and 100 mL of sterile water. Next, we pulse lavage the wound again with 3 L of normal saline and soak the wound for 3 minutes with dilute, sterile povidone-iodine solution (22.5 mL of 10% povidone-iodine solution mixed with 1 L of normal saline). Lastly, we again aggressively pulse lavage the wound with 3 L normal saline. It is important to avoid mixing different irrigation agents together, as certain combinations have been shown to create potentially harmful precipitates. For knees, we then place a laparotomy sponge soaked in dilute povidone-iodine into the wound between the retained tibial and femoral components and provisionally close the skin with a running #2 nylon suture. All of the drapes are removed, and the extremity is completely re-prepped and draped. A completely new set of instruments are also utilized. During this interval, all members of the surgical team change their scrubs and head/shoe covers. The wound is then reopened, the new bearing is trialed, and, after a final irrigation with dilute povidone-iodine and saline, the new bearing surface is inserted. Vancomycin powder 1 g is placed into the joint and a standard closure is performed. This protocol is based on our recent favorable experience with a similar one-stage exchange protocol used for the management of chronic PJI. For acute postoperative PJI in hips, we now rarely employ this strategy as the majority of our hips are cementless devices, which are treated more easily and more effectively with a one-stage exchange in our opinion.
The choice of antibiotic regimen utilized after surgical treatment for acute PJI depends on the offending organism and often relies on the expertise of an infectious disease specialist. While waiting for cultures to result, we typically start with dual antibiotic therapy, including intravenous (IV) cefazolin and vancomycin. We believe that empiric methicillin-resistant Staphylococcus aureus (MRSA) coverage is essential but prefer cefazolin for other Gram-positive bacteria due to its superior susceptibility profile. Cefazolin has been proven to prevent infection after primary and revision arthroplasty; additionally, it is both cost-effective and bactericidal, rather than bacteriostatic. The definitive choice of antibiotics should ultimately be culture directed and continued for a minimum of 6 weeks postoperatively.
While the majority of acute PJI is caused by Gram-positive bacteria, infections caused by Gram-negative bacteria are increasingly common and may respond especially well to fluoroquinolones. , , Fluoroquinolones have among the highest success rates in Gram-negative PJI and have been shown to protect against recurrence. , , Additional treatment with oral antibiotics is generally recommended. In our practice, oral antibiotics are typically prescribed for at least 1 year following IV treatment, although there is little data to support a specific duration of treatment. Chronic lifelong suppression with oral antibiotics can be considered for patients who are at high risk for failure or who have undergone complex reconstructions where additional surgical intervention may not be possible. However, it is important to recognize that extended oral antibiotic treatment is not without risk. These patients should be followed in conjunction with an infectious disease specialist to assist with monitoring them. Ensuring appropriate care for a patient with acute PJI requires a multidisciplinary effort consisting of surgeons, infectious disease specialists, nutritionists, and nursing and clinical staff to help coordinate care. ,
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here