Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Malignant tumors arising from or displaying differentiation along the lines of the various elements of the nerve sheath (e.g., Schwann cell, perineurial cell, fibroblast) are collectively referred to as malignant peripheral nerve sheath tumors (MPNSTs). Because MPNSTs recapitulate the appearance of various cells of the nerve sheath, they range in appearance from tumors that resemble a neurofibroma to those resembling an adult-type fibrosarcoma. Rarely, tumors arising from nerves or neurofibromas display aberrant lines of differentiation. For example, angiosarcomas may arise in nerves or in preexisting neurofibromas. By convention, these tumors are not considered MPNSTs but are classified according to their aberrant line of differentiation. Although primitive neuroectodermal tumors may also rarely arise from peripheral nerves, they are considered along with Ewing sarcoma (see Chapter 33 ).
The diagnosis of MPNST has traditionally been one of the most difficult and elusive lesions among soft tissue tumors in the past because of a lack of standardized diagnostic criteria. Even now, no specific biomarkers are widely available to establish the diagnosis with certainty. Although there is general agreement that a sarcoma can usually be considered an MPNST if it arises from a peripheral nerve or a neurofibroma, there is less agreement about the diagnostic criteria for tumors occurring outside these settings. As a result, the incidence of this sarcoma, as reported in the literature, has varied depending on the stringency of the diagnostic criteria.
A sarcoma is assumed to be an MPNST if one of three criteria can be met: (1) the tumor arises from a peripheral nerve and shows no aberrant or heterologous line of differentiation; (2) it arises from a preexisting benign nerve sheath tumor, usually a neurofibroma; or (3) the tumor displays a constellation of histologic features that are seen in tumors arising in the foregoing situations and are considered typical of malignant Schwann cell tumors. These features include the (1) dense and hypodense fascicles alternating in a marble-like pattern consisting of (2) asymmetrically tapered spindled cells with irregular buckled nuclei, or (3) immunohistochemical or electron microscopic evidence of Schwann cell differentiation in the context of a fibrosarcomatous-appearing tumor. In addition, there are features that are less specific but frequently occur in Schwann cell tumors, including nuclear palisading, whorled structures that vaguely suggest large tactoid structures, peculiar hyperplastic perivascular change, and occasionally, heterologous elements (e.g., cartilage, bone, skeletal muscle). Clearly, these criteria allow inclusion of only MPNSTs that are reasonably well differentiated or have not yet obscured their site of origin. Nonetheless, this approach offers an acceptable degree of diagnostic reproducibility and eliminates the potpourri of spindle cell tumors that have been diagnosed as MPNSTs in the past.
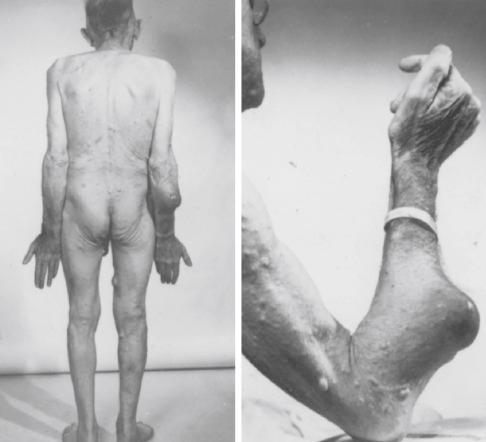
MPNSTs account for approximately 5% to 10% of all soft tissue sarcomas, and about one-fourth to one-half occur in the setting of neurofibromatosis 1 (NF1). This range probably reflects differences in diagnostic criteria and patient referral patterns. In most large studies the percentage of patients with MPNST with and without NF1 is about equal. Although the lifetime risk of developing an MPNST in the setting of NF1 is about 10%, the risk approaches 30% in patients with symptomatic plexiform neurofibromas . In addition, patients with large rearrangements of the NF1 gene, so-called microdeletions, have a lifetime risk of MPNST of 16% to 26%. Patients with germline NF1 microdeletions also often have facial, skeletal, and cardiac abnormalities and large numbers of neurofibromas. This is postulated to be the result of inactivation of additional tumor suppressor genes, including SUZ12 , ADAP2, and RNF135 , adjacent to the microdeletion. Patients with NF1 microdeletions require especially close clinical surveillance.
Patients with NF1 develop sarcomas usually after a relatively long latency (10-20 years), and in some cases the MPNSTs are multiple. MPNSTs in childhood are recognized but infrequent. The exact mechanism of malignant transformation or tumor progression in NF1 is not fully understood but involves a multistep process in which genes other than NF1 also participate (see later). Aside from the foregoing genetic predilection to develop MPNST, little is known about the pathogenesis of sporadic tumors in humans. About 10% to 20% of cases result from therapeutic or occupational irradiation after a latent period of more than 15 years. These tumors do not differ significantly from other MPNSTs.
MPNST is typically a disease of adult life because most tumors occur in patients 20 to 50 years old (median age: 35). Patients with NF1 usually present at a slightly earlier age and have larger tumors than those with sporadic lesions. Men predominate in studies that report a high percentage of patients with NF1, because of the slight bias for men in this disease. In sporadic cases of MPNST, the gender ratio is approximately equal.
As with other sarcomas, these lesions present as enlarging masses that are usually noted several months before diagnosis. Pain is variable but is more prevalent in those patients with NF1. In fact, pain or a sudden enlargement of a preexisting mass in this setting should lead to immediate biopsy to exclude the possibility of malignant transformation of a neurofibroma. Fluorodeoxyglucose positron emission tomography, which allows visualization of glucose metabolism by cells, has been reasonably successful in identifying malignant change in plexiform neurofibromas and may give some indication of the grade of the lesion. MPNSTs that arise from major nerves typically result in a striking constellation of sensory and motor symptoms, including projected pain, paresthesias, and weakness. The symptoms rarely antedate the detection of a mass.
Most MPNSTs arise in association with major nerve trunks, including the sciatic nerve, brachial plexus, and sacral plexus. Consequently, the most common anatomic sites include the proximal portions of the upper and lower extremities and the trunk. Comparatively few arise in the head and neck, a feature that contrasts with the distribution of the schwannoma .
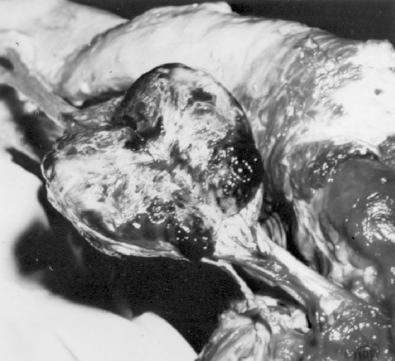
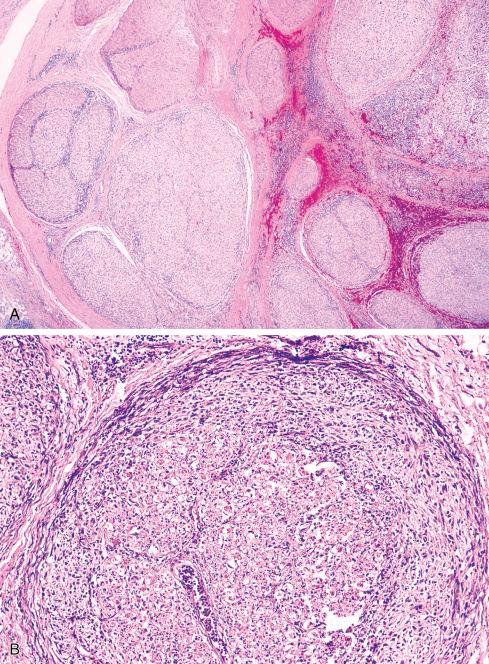
In its classic form, an MPNST arises as a large fusiform or eccentric mass in a major nerve ( Fig. 27.1 ). Thickening of the nerve proximally and distally to the main mass usually indicates spread of the neoplasm along the epineurium and perineurium. In NF1 patients, MPNST may develop in a preexisting neurofibroma ( Fig. 27.2 ). Most of these lesions are deeply situated; only rare ones arise from superficial neurofibromas. Regardless of the clinical setting, the gross appearance of the MPNST is essentially similar to that of other soft tissue sarcomas. It is usually large, averaging more than 5 cm in diameter, and has a fleshy, opaque, white-tan surface marked by areas of secondary hemorrhage and necrosis. This appearance contrasts with the white mucoid appearance of the typical neurofibroma.
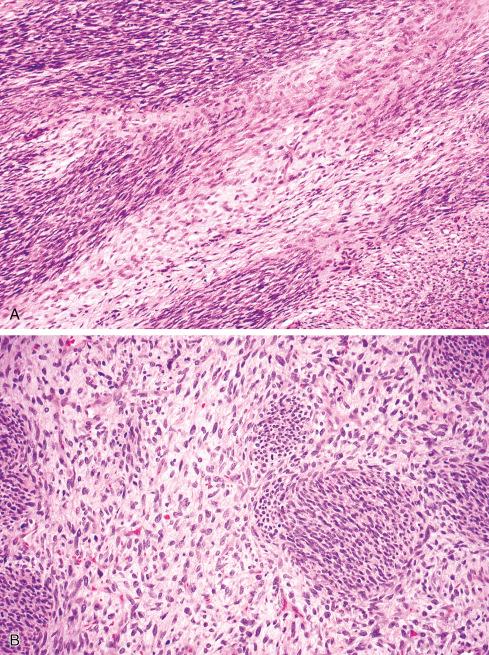
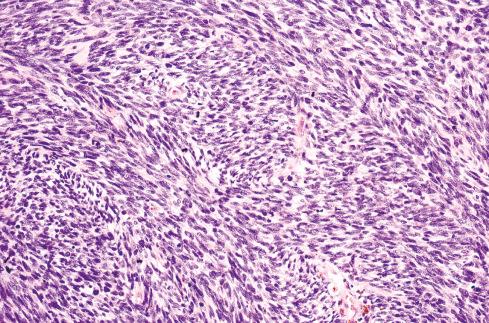
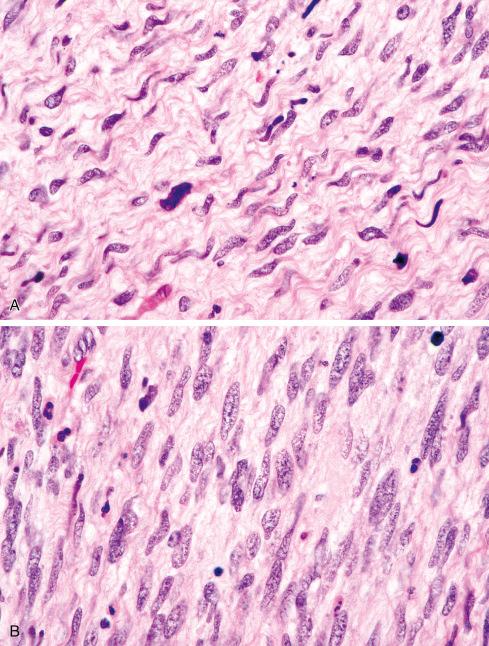
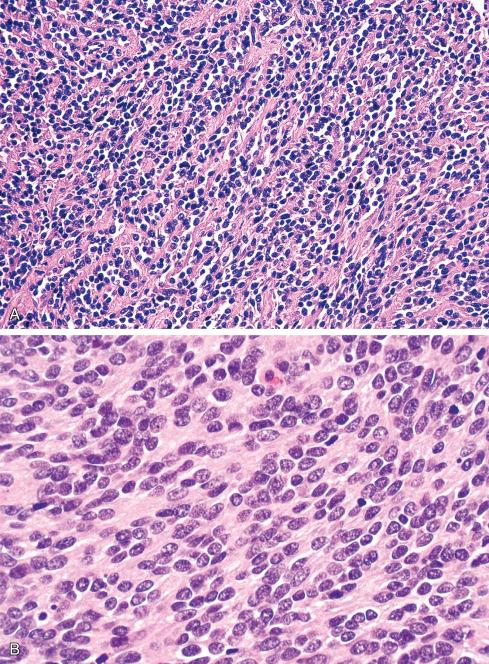
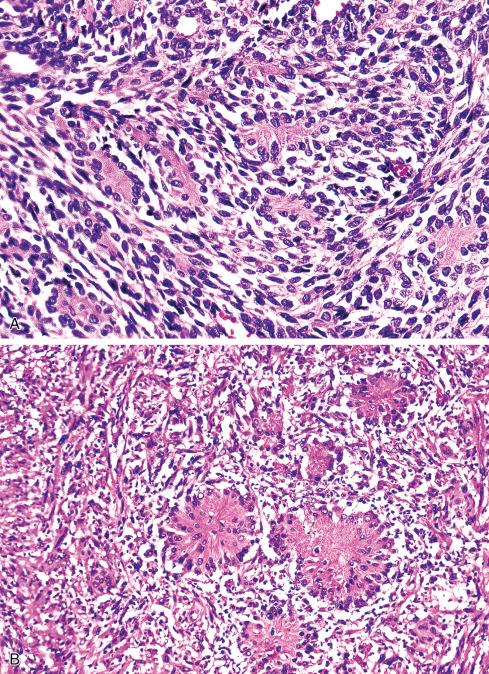
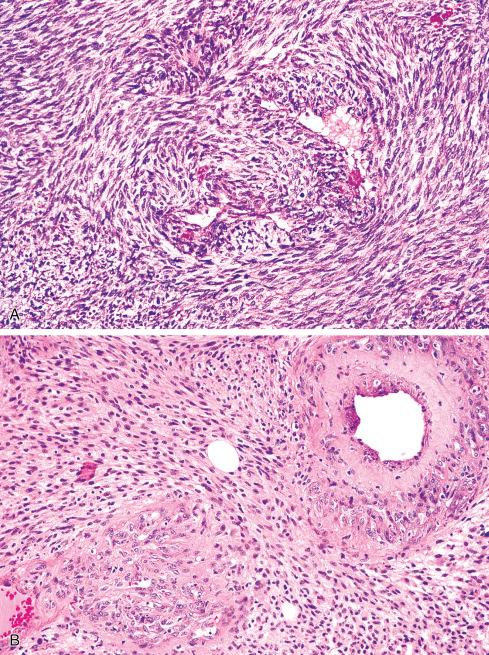
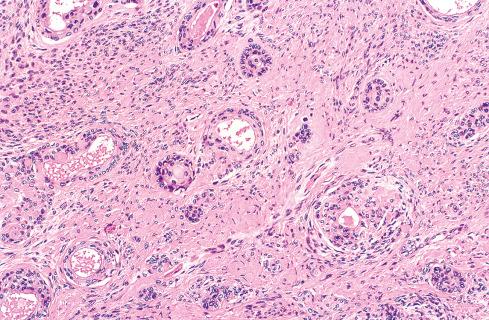
Most MPNSTs resemble adult-type fibrosarcomas in their overall organization, with certain modifications ( Figs. 27.3 to 27.12 ). Classically, the cells recapitulate the features of the normal Schwann cell. Unlike the symmetrically spindled cells of adult-type fibrosarcoma, they have irregular contours. In profile, the nuclei are wavy, buckled, or comma shaped, whereas when viewed en face, they are asymmetrically oval ( Fig. 27.7 ). The cytoplasm is lightly stained and usually indistinct. The cells can range from spindled in shape to fusiform or even rounded, and thus the lesion can mimic other spindle cell sarcomas or even a round cell sarcoma ( Fig. 27.9 ). The cells are arranged in sweeping fascicles, but there is greater variation in organization than in adult-type fibrosarcoma or monophasic synovial sarcoma. Densely cellular fascicles alternate with hypocellular, myxoid zones ( Figs. 27.3 to 27.6 ), which swirl and interdigitate with one another, creating a marble-like effect ( Fig. 27.3 ). Others display a peculiar nodular, curlicue, or whorled arrangement of spindled cells ( Fig. 27.10 ), crudely suggesting tactoid differentiation. Nuclear palisading may be present in a minority of cases and is usually a focal finding ( Fig. 27.12 ).
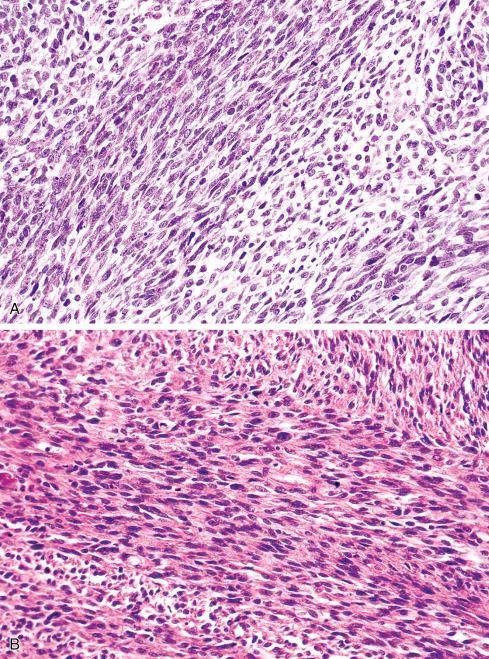
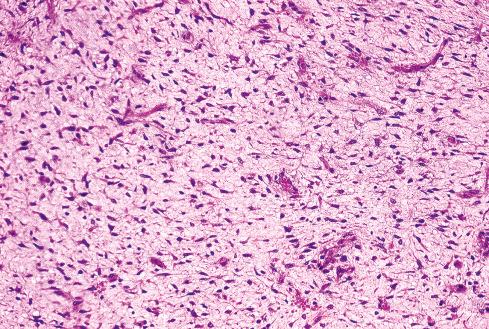
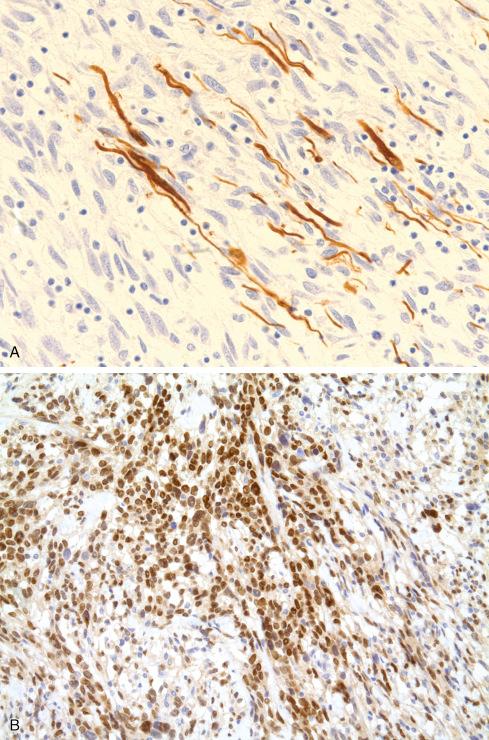
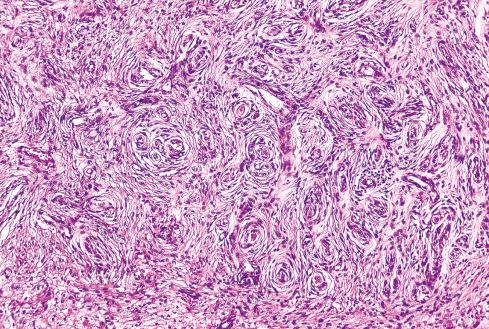
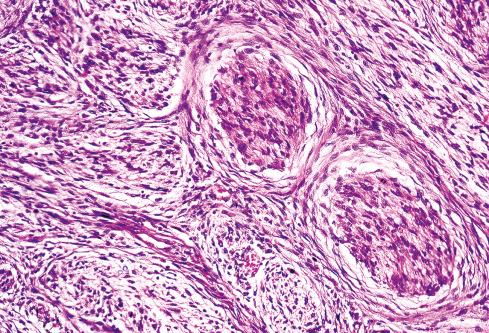
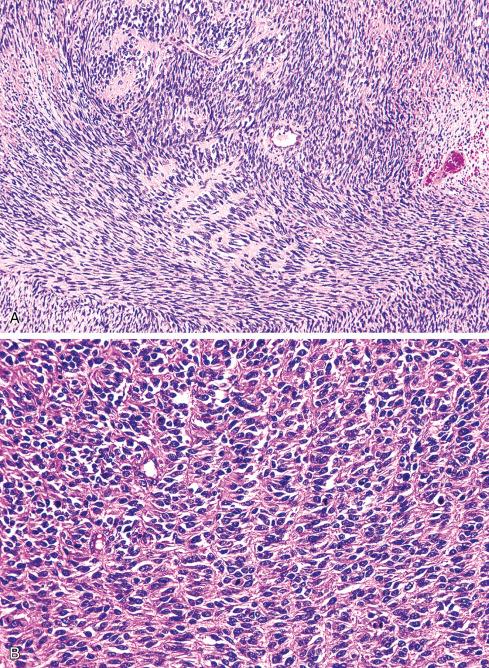
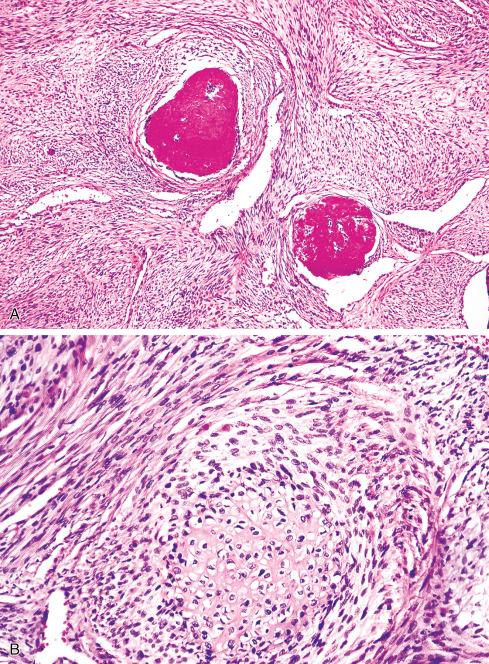
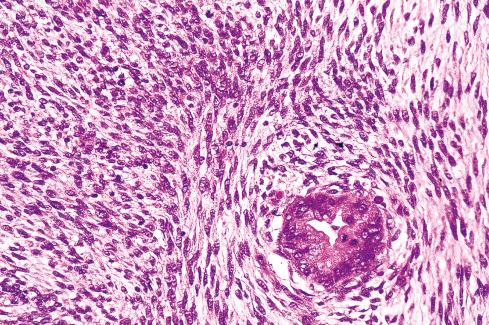
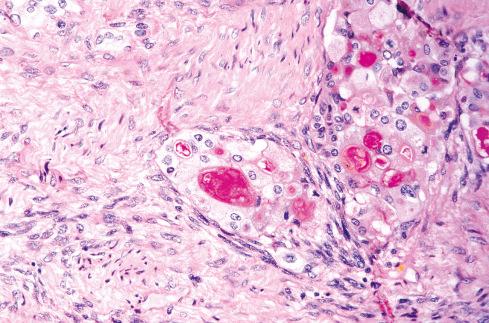
Several other, subtle features are quite characteristic of MPNST but are not completely specific, so they must be evaluated in the context of the previous discussion before a given tumor is labeled MPNST. These features include hyaline bands and nodules, which in cross section can be likened to giant rosettes ( Fig. 27.13 ); extensive perineural and intraneural spread of tumor ( Fig. 27.14 ); and a peculiar proliferation of tumor in the subendothelial zones of vessels so that the neoplastic cells appear to herniate into the lumen ( Fig. 27.15 ). Small vessels may also proliferate in the walls of or around large vessels. It must be emphasized that none of these features is specific for MPNST and may also be seen, for example, in synovial sarcoma. Heterologous elements, present in about 10% to 15% of MPNST, seem to be more common in MPNSTs than in other sarcomas. Mature islands of cartilage and bone are the most common elements ( Fig. 27.16 ), whereas skeletal muscle, mucin-secreting glands, and squamous islands are rare. The finding of heterologous elements in a monomorphic spindle cell sarcoma should suggest the diagnosis of MPNST ( Figs. 27.17 to 27.22 ).
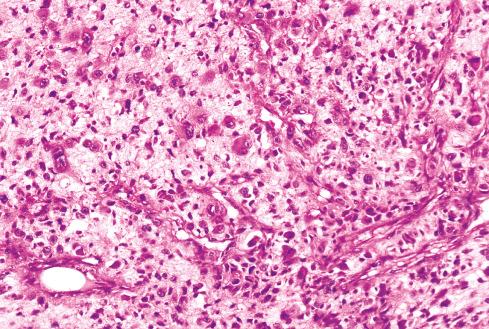
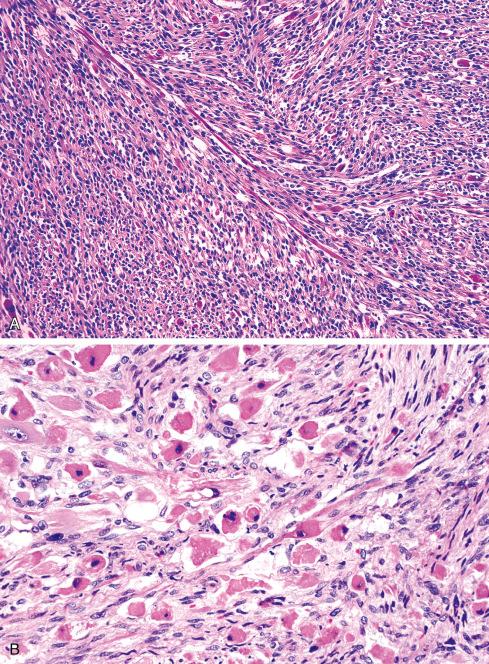
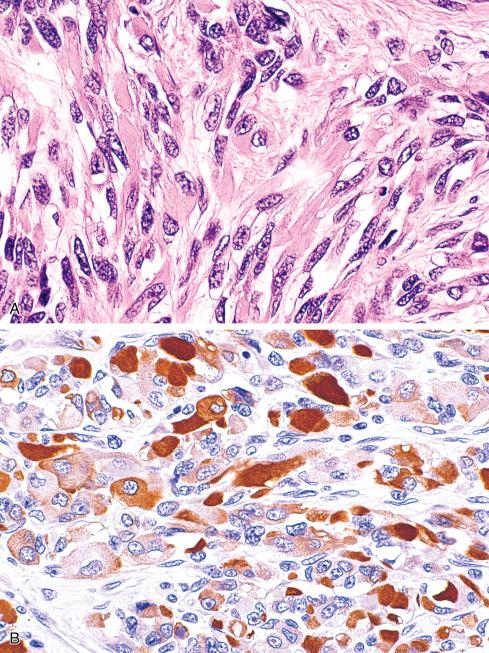
Although most MPNSTs conform to this description, a small percentage appear quite different. Some of these tumors closely resemble neurofibromas, except they manifest a greater degree of cellularity, pleomorphism, and mitotic activity. These lesions are typically found in the setting of NF1 and may represent rare examples of low-grade MPNST. Criteria have been proposed for the distinction of low-grade MPNST from “atypical neurofibromatous neoplasm of uncertain biologic potential” (ANNUBP) (see Chapter 26 ). At the opposite end of the spectrum are anaplastic MPNSTs, which may be difficult to distinguish from other pleomorphic sarcomas. They are composed of plump, spindled, and giant cells intermixed with hemorrhage and necrosis, similar to that seen in glioblastoma multiforme . Although these pleomorphic tumors have been documented more often in the setting of NF1, this may reflect the general reluctance to diagnose an anaplastic MPNST outside of NF1. Diagnosis of these tumors depends on identifying areas of typical MPNST. Infrequently, MPNSTs contain areas of primitive neuroepithelial differentiation consisting of cords or nests of small round cells and, in extraordinary cases, even rosettes. Primitive neuroepithelial differentiation appears to be a more common feature of MPNST in children.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here