Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
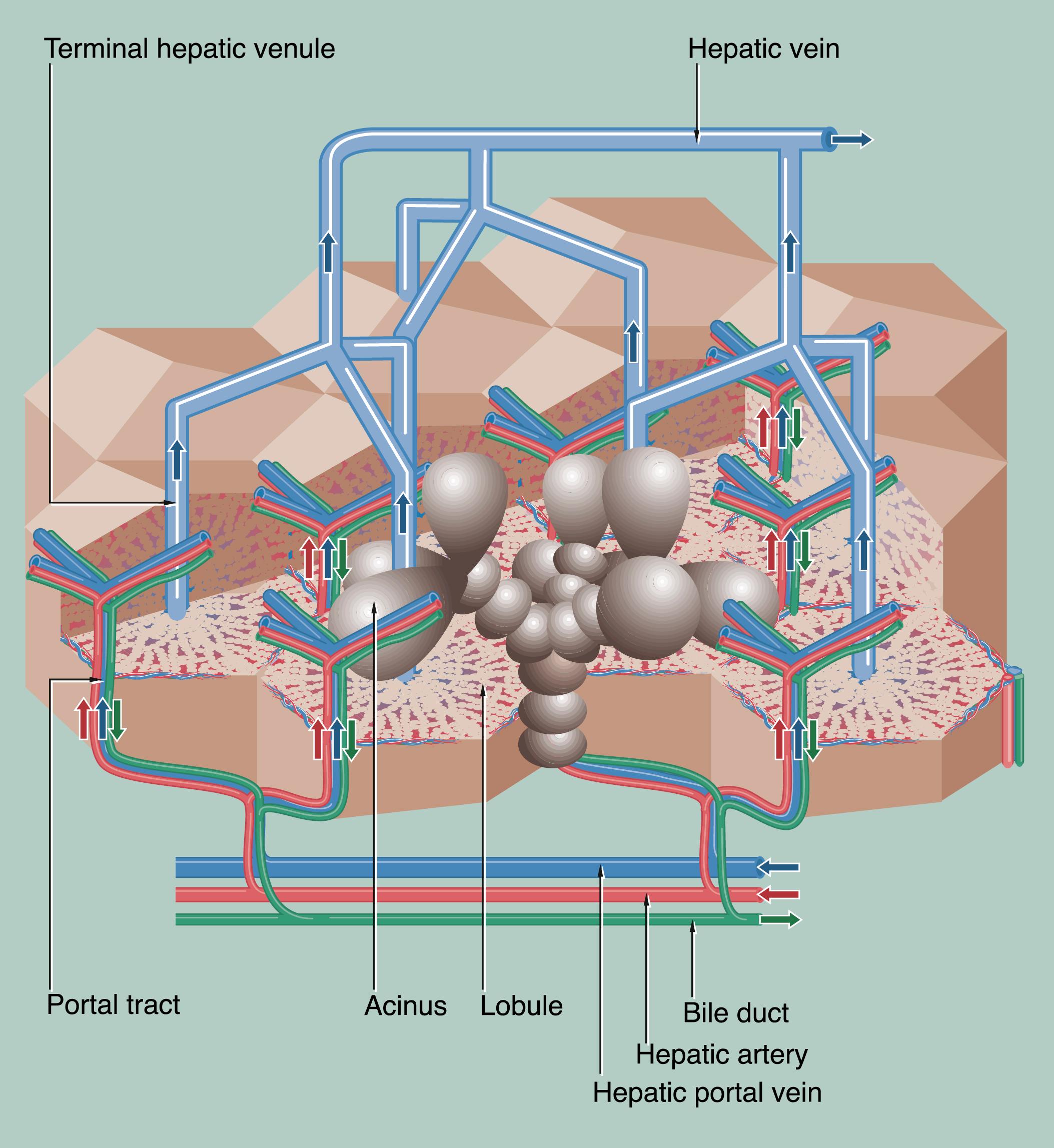
The liver is affected by a wide range of disorders, the most important of which are listed below. The liver performs many different metabolic functions and liver disease produces diverse clinical symptoms and signs. The pathophysiology of the more common symptoms and signs is described in Table 14.1 .
Acute hepatic necrosis has been described in Ch. 2 . This usually results from exposure to certain drugs or poisons or occurs as part of a fulminant viral infection.
Acute hepatitis has many causes. It may resolve or become persistent (chronic hepatitis) .
Chronic hepatitis is diagnosed when biochemical manifestations of liver cell damage have persisted for more than 6 months. It may eventually progress to cirrhosis.
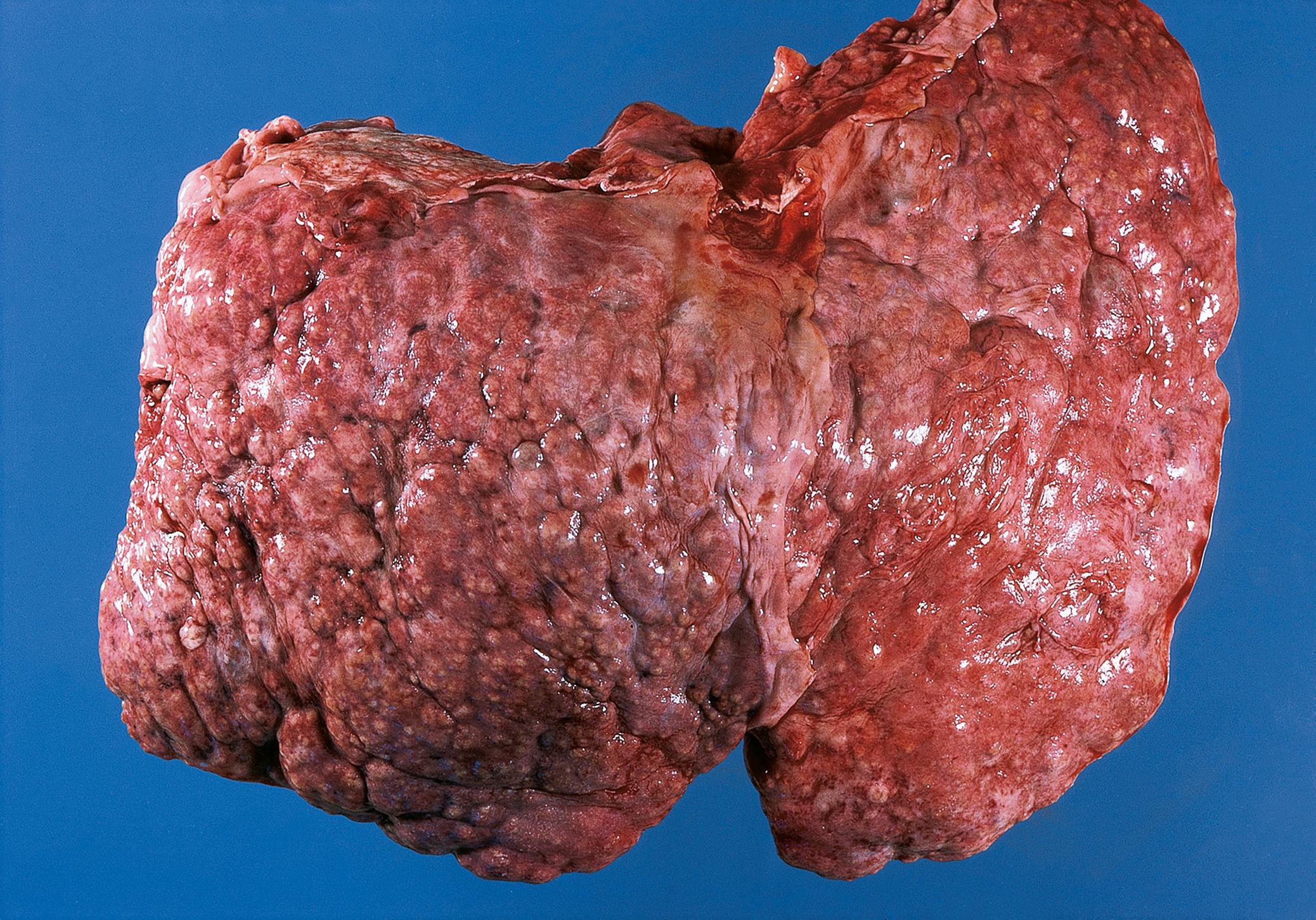
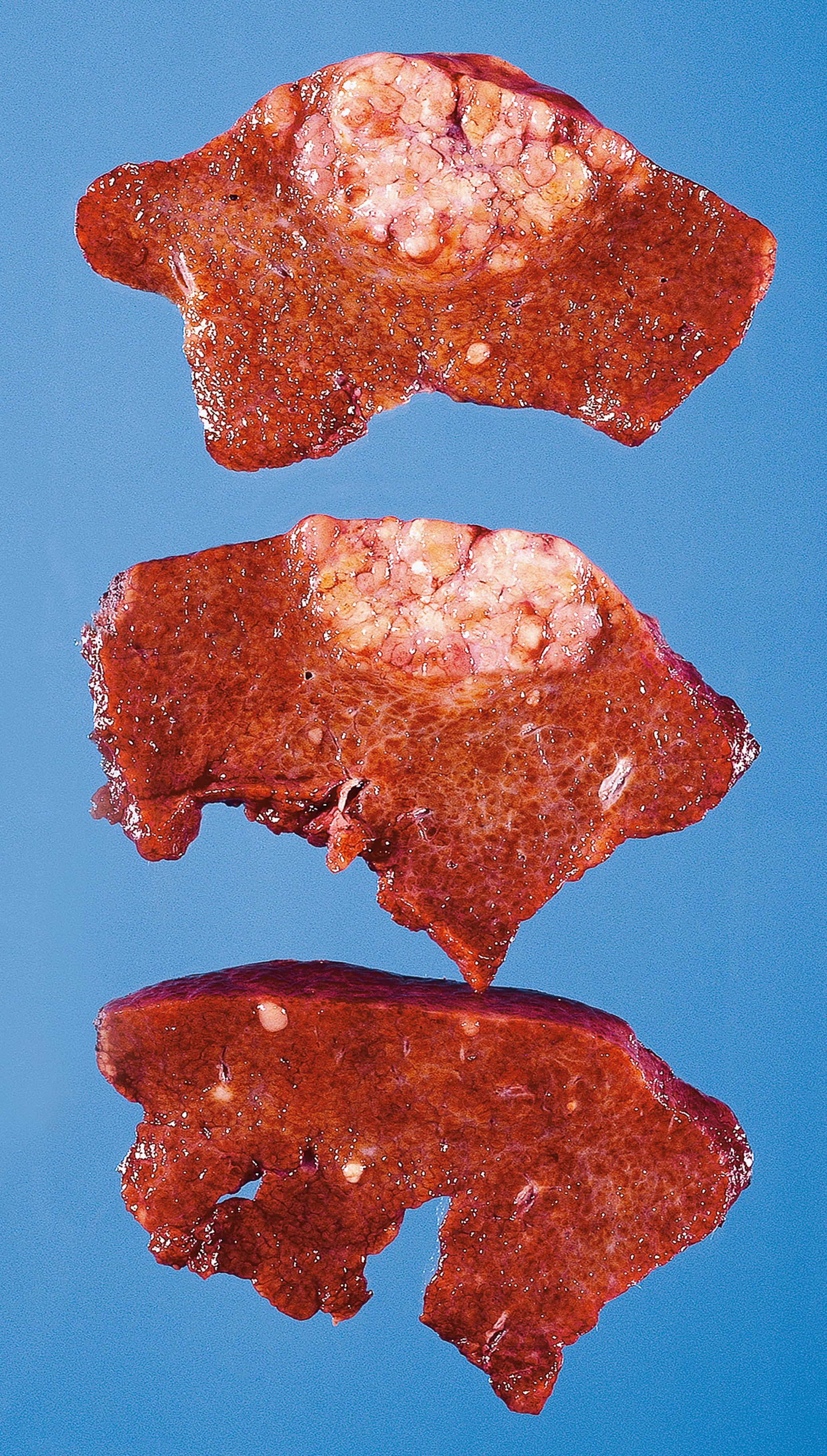
Cirrhosis is the end stage of various types of liver disease in which there is liver cell loss and architectural distortion over a long period. It is characterised by destruction of normal liver architecture, which is replaced by regenerative nodules of hepatocytes separated by bands of fibrous tissue. There may be evidence of continuing active damage to liver cells. Eventually, liver function is impaired or fails totally (chronic liver failure) . The distortion of vascular architecture leads to portal hypertension . Various different types of cirrhosis are illustrated and discussed in Fig. 14.5 .
Malignant disease frequently involves the liver, most commonly as metastatic spread, especially from primary lesions in the gut, breast and lung (see Fig. 7.16). Less frequently, the liver becomes diffusely infiltrated in haematolymphoid malignancies such as Hodgkin and non-Hodgkin lymphomas.
Primary malignancy of the liver, hepatocellular carcinoma ( Fig. 14.7 ), is uncommon and most often arises where there is pre-existing cirrhosis.
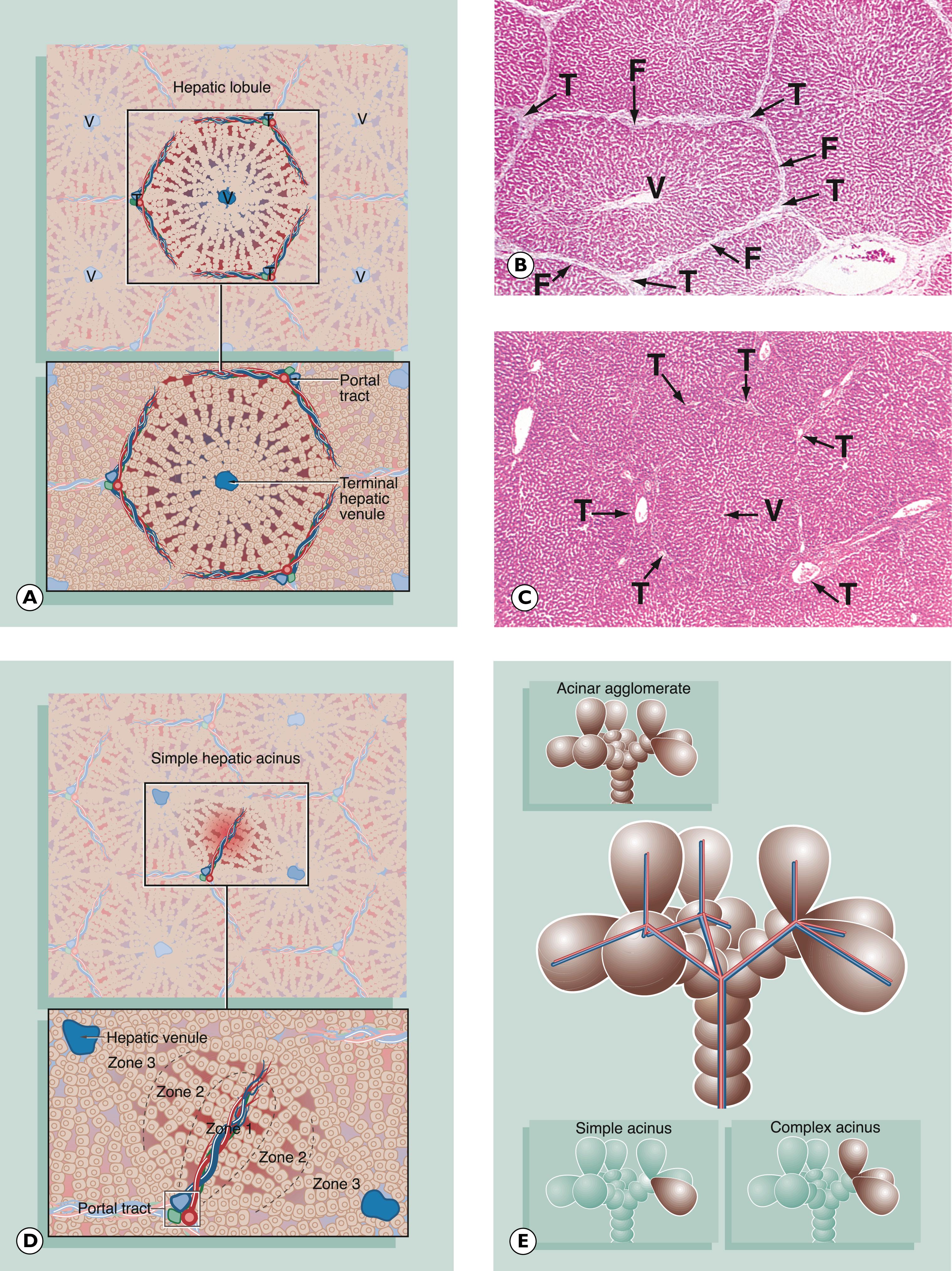
Right-sided cardiac failure involves the liver when raised venous pressure is transmitted to the central hepatic venules, resulting in congestion of the sinusoids with blood. Hepatocytes in the centrilobular area (zone 3 of the liver acinus) may then undergo atrophy or even frank necrosis.
Inborn errors of metabolism , often reflecting single gene defects, can result in the abnormal accumulation of various metabolites within hepatocytes. These are known as storage diseases and include glycogen storage diseases , mucopolysaccharidoses , lipidoses , haemochromatosis ( Fig. 14.6 ) and Wilson’s disease . Liver biopsy may be useful in the diagnosis of such disorders.
C Councilman bodies H hydropic degeneration In inflammatory cells
| Sign/Symptom | Clinical feature | Mechanism |
|---|---|---|
| Jaundice | Yellow discolouration of tissues owing to bile pigments | Failure of metabolism or excretion of bile |
| Bleeding | Easy bruising and prolonged clotting time of blood | Failure of hepatic synthesis of clotting factors |
| Oedema | Swelling of dependent parts owing to extracellular accumulation of water | Failure of hepatic synthesis of albumin resulting in reduced plasma oncotic pressure |
| Ascites | Fluid in peritoneal cavity | Low serum albumin and portal hypertension |
| Gynaecomastia | Enlarged male breasts | Failure to detoxify endogenous oestrogens |
| Encephalopathy | Altered consciousness, lack of coordination, may lead to coma | Failure to detoxify ammonia and excitatory amino acids which result from protein breakdown |
| Haematemesis and/or melaena | Vomiting blood and passing blood | Bleeding from oesophageal varices or per rectum owing to portal hypertension |
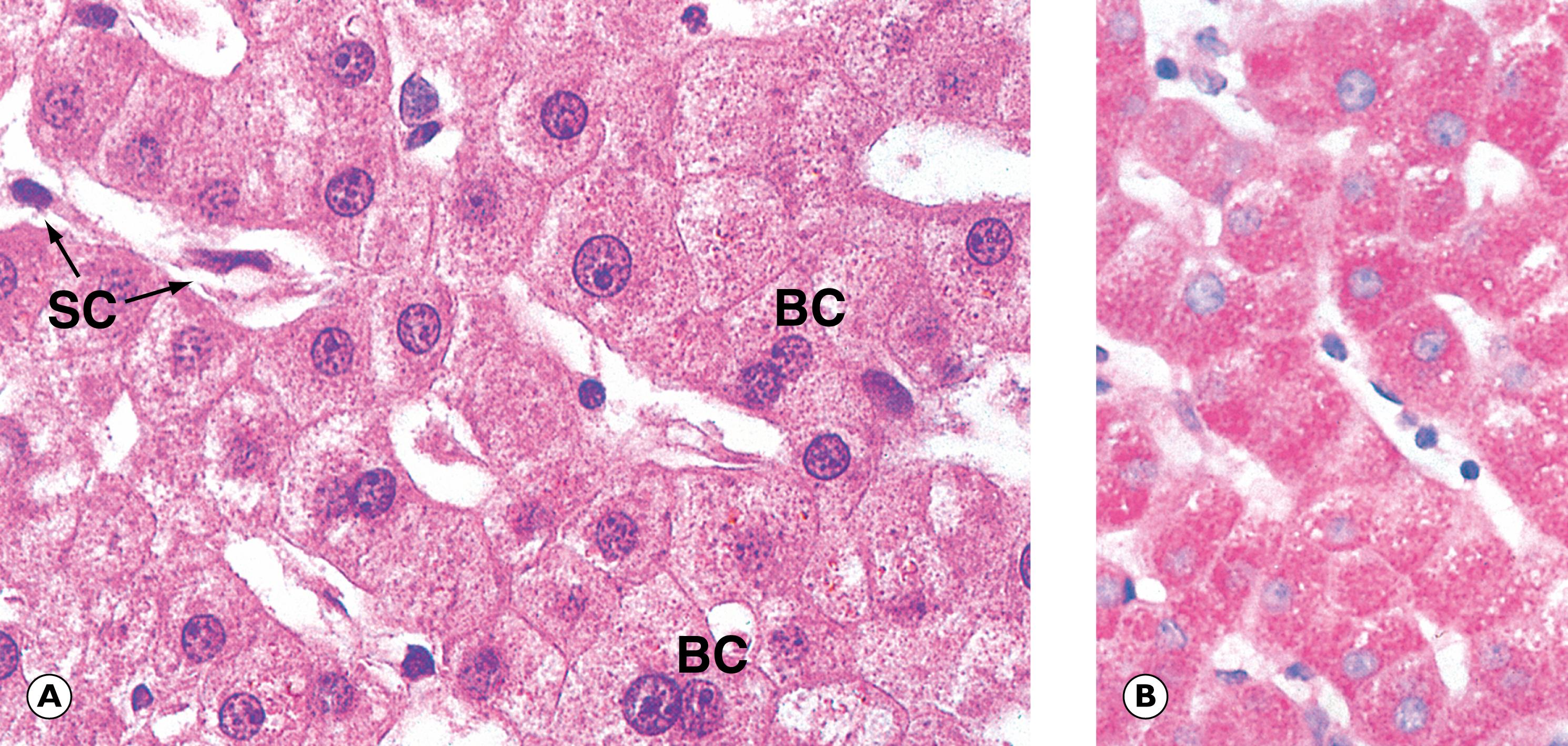
Hepatocytes ( E-Fig. 14.1 H ), with their high degree of metabolic activity, are readily disturbed by toxins and demonstrate the histological cellular responses known as ballooning, fatty change and necrosis as described in Ch. 2 . Acute inflammation of the liver parenchyma is usually marked by focal accumulation of inflammatory cells, usually in relation to necrotic hepatocytes. The exception to this is in the formation of hepatic abscesses , which usually develop either as a result of bacterial infections from the biliary tract or from a septic focus in the abdomen drained by the portal venous system to the liver.
Acute hepatitis is a general term for inflammation of the liver parenchyma, which can then be further classified according to aetiology. The four most important groups of conditions causing acute hepatitis are:
Viral hepatitis: This is illustrated in Fig. 14.1 and its various subtypes are outlined in Table 14.2 .
| Hepatitis | Mode of transmission | Acute hepatitis | Chronic Hepatitis | Notes |
|---|---|---|---|---|
| A | Faecal oral | Y | N | Mild self-limiting illness common |
| B | Blood borne, Oro-mucosal | Y | Y | Transmission from blood derived fluids. Prevention via vaccination strategies. |
| C | Blood borne | Y | Y | Transmission – blood products, tattoos, sharing needles. Curable with new oral antiviral agents |
| D | Blood borne | Y | Y | Only in association with hepatitis B |
| E | Faecal oral | Y | Y | Can cause chronic infection in immunocompromised patients |
Toxins: Alcohol is the most common hepatic toxin ( Fig. 14.2 ).
Drugs: Hepatitis may be caused by the anaesthetic gas halothane, particularly after repeated exposure. Isoniazid, a drug commonly used in the treatment of tuberculosis, results in acute hepatitis in a small proportion of cases. Many other drugs occasionally cause hepatitis.
Systemic infections: Infections caused by Leptospira and Toxoplasma usually involve the liver as part of disseminated disease. Other systemic infections may cause multiple minute infective lesions as in bacterial septicaemia and miliary tuberculosis (see Fig. 5.7).


This is a relatively common disorder that closely resembles alcoholic liver disease, but which occurs in the absence of excessive alcohol intake. It is often diagnosed incidentally due to the discovery of deranged liver function tests upon routine blood testing. Patients may have no symptoms or clinical signs of liver disease at the time of diagnosis. It is thought that this process may account for a large proportion of patients who present with cirrhosis or end-stage liver disease without any preceding history of overt liver pathology (a pattern of disease often referred to as cryptogenic cirrhosis ).
Of interest, epidemiological studies suggest that non-alcoholic fatty liver disease is very prevalent in developed country populations and it is strongly associated with other common metabolic disorders such as obesity, dyslipidaemia, impaired glucose tolerance and non-insulin dependent diabetes mellitus. Like alcoholic liver disease, there is a spectrum of pathological changes from simple steatosis through non-alcoholic steatohepatitis (known as NASH ) to eventual hepatic fibrosis and cirrhosis. Clearly, given the morphological similarity to alcohol-related liver disease, this diagnosis can only be made after correlation of the clinical and pathological findings.
B ballooning degeneration F fatty change H Mallory’s hyaline N necrosis
When inflammation of the liver continues without improvement for 6 months or more, the condition is described as chronic hepatitis . Conventionally, this term excludes chronic inflammation of the liver caused by alcohol, bacterial agents and biliary obstruction. The main causes of chronic hepatitis are summarised below:
Viral infection
Hepatitis B : chronic hepatitis occurs in ∼ 5%–10% of cases (but ∼80% if hepatitis D superinfection)
Hepatitis C : chronic hepatitis occurs in ∼85% of cases
Other hepatitis viruses
Autoimmune disease
Autoimmune hepatitis
Usually occurs in middle-aged women
Positive serum auto-antibodies (anti-nuclear antibodies, anti-smooth muscle antibodies)
Often associated with other auto-immune diseases, e.g. rheumatoid arthritis, thyroid disease.
Primary biliary cirrhosis
Usually occurs in middle aged women
Positive anti-mitochondrial antibodies in most patients
Autoimmune destruction of intrahepatic bile ducts
Despite name, cirrhosis occurs only with end-stage disease
Primary sclerosing cholangitis
Usually young or middle-aged, more common in men
Strongly associated with inflammatory bowel disease
Fibrous obliteration of intrahepatic and extrahepatic bile ducts
Toxic/metabolic
Wilson’s disease : disorder of copper metabolism with CNS effects (hepatolenticular degeneration)
α1-Antitrypsin deficiency : protease inhibitor deficiency causing chronic lung and liver damage
Drug-induced hepatitis : many causes and various histological patterns
Histologically, chronic hepatitis is characterised by:
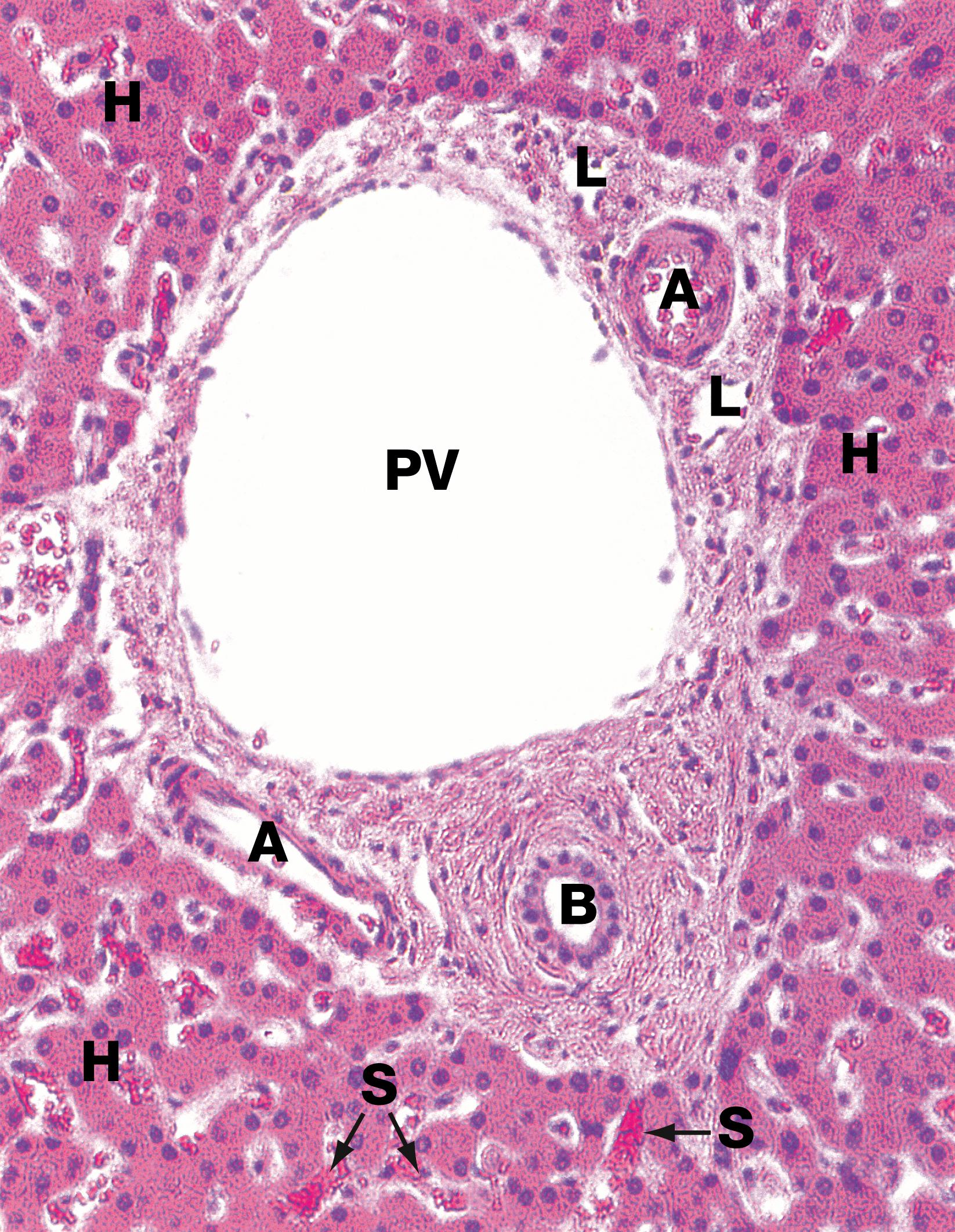
The presence of inflammatory cells (mainly lymphocytes) in portal tracts ( E-Fig. 14.2 H ) or scattered within the liver lobules, or both.
Necrosis of hepatocytes, either concentrated upon the limiting plate of hepatocytes around the portal tracts ( interface hepatitis , previously called piecemeal necrosis ) or scattered within the liver lobules ( lobular hepatitis , previously called spotty necrosis ), or both.
The presence of fibrosis, either in the portal tracts, or extending from the portal areas into the adjacent parenchyma as short septa, or forming ‘bridges’ across the lobular structure, causing architectural distortion.
Assessment of these features can be used to determine how active the disease is ( grade of chronic hepatitis) and how far the disease has progressed ( stage of disease). The general principles of grading and staging are outlined below.
Various scoring systems have been devised for grading and staging hepatitis. These aim to give information about appropriate treatment and likely disease prognosis. In this context, grade is used to reflect the severity of any ongoing inflammation or necrosis. The extent of such damage can be assessed within portal areas and within the liver lobule. When there is significant inflammation, drug treatment may be initiated to reduce continuing liver damage and to prevent the development of progressive disease with fibrosis.
Stage is a means of assessing the degree of damage to the connective tissue framework of the liver. Fibrosis tends to begin within the portal areas and fine septa then extend out into the parenchyma. Eventually, if disease continues to progress, bands of collagen form between adjacent portal tracts, dividing the liver parenchyma into distorted nodules and culminating in cirrhosis .
On a global scale, chronic viral hepatitis is an important public health problem. Both hepatitis B and hepatitis C may give rise to symptomatic chronic hepatitis or to an asymptomatic carrier state. Normally, when hepatitis B is acquired in adulthood, the risk of chronic viral carriage is around 1%–10%. In the developing world, transmission of hepatitis is commonly from mother to child (vertical transmission) . This is important because when infection occurs at the time of childbirth, chronic carriage will occur in 90%–95% of cases. These patients may develop chronic liver fibrosis with eventual cirrhosis. Cirrhosis is a major risk factor for the development of hepatocellular carcinoma and in some areas where hepatitis B infection is highly prevalent, hepatocellular carcinoma (a rare tumour in most developed countries) is the most common form of malignancy.
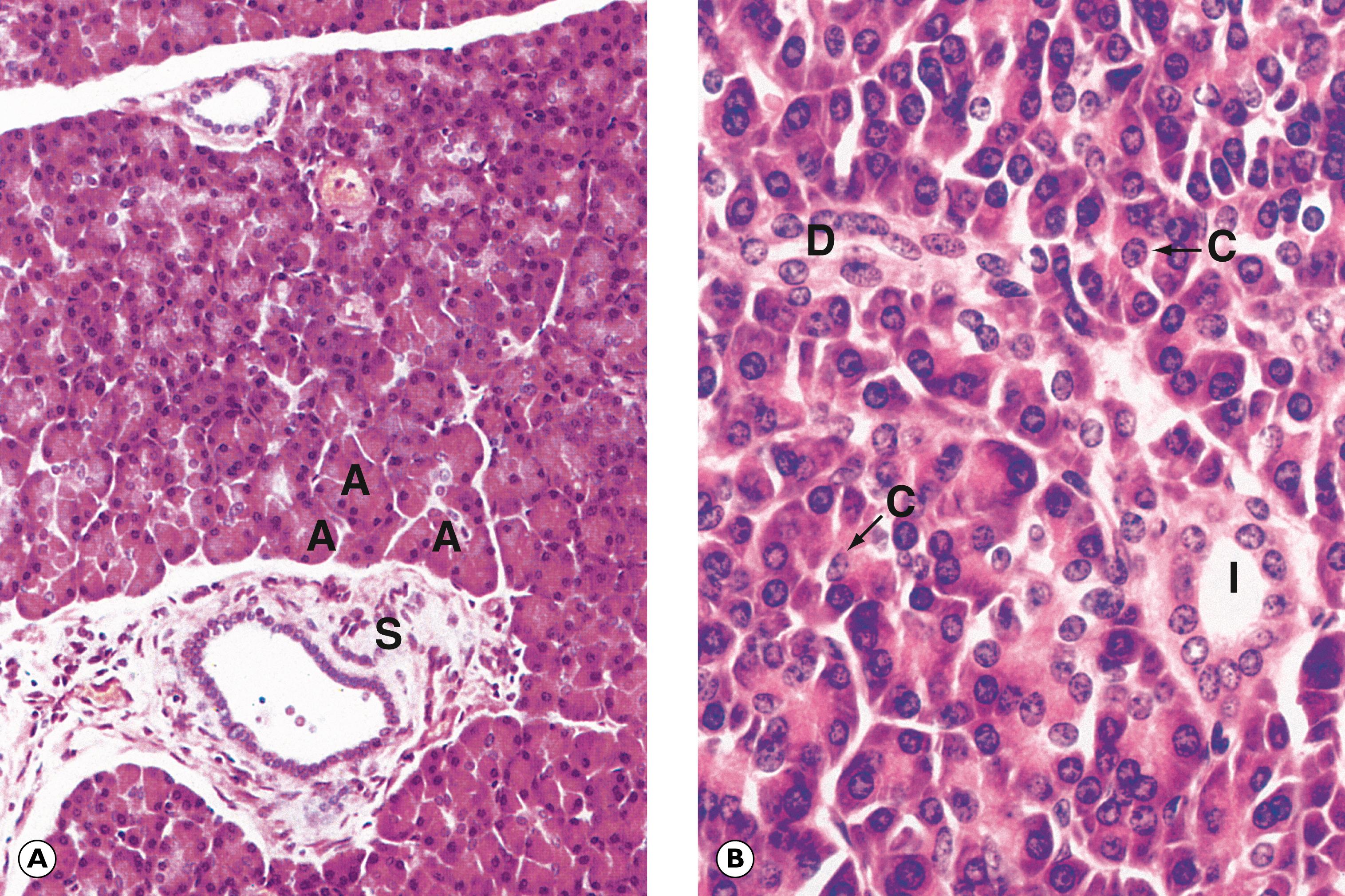
A artery B bile duct Bn binucleate cell C chronic inflammation D bile ductules F fibrosis G ground glass hepatocytes Gr granuloma L limiting plate LN lobular necrosis N necrosis P periportal hepatocytes V vein
F fibrous tissue In inflammation N nodule P portal area
Genetic haemochromatosis is an autosomal recessive disorder that occurs due to a single amino acid substitution within the protein encoded by the HFE gene. Clinical presentation tends to be earlier in men than in women because normal menstrual blood loss in premenopausal females reduces the extent of iron deposition within the tissues and so exerts a degree of physiological control. The diagnosis may be first suspected due to deranged liver function tests, but other clinical signs include skin pigmentation and the development of diabetes mellitus due to chronic pancreatic damage. These features gave rise to the old clinical description of haemochromatosis as bronzed diabetes , a term that serves as a useful aide memoir.
The liver is commonly affected by metastatic malignant tumours, particularly metastatic carcinomas. For this reason, staging of most newly diagnosed malignant tumours involves biochemical testing of liver function, as well as careful radiological assessment to identify any evidence of tumour deposits. When such tests do reveal mass lesions, percutaneous liver biopsy may sometimes be undertaken to confirm the diagnosis histologically. More often, liver biopsy is used when a patient is found to have multiple tumour deposits in the liver but without any known primary site of tumour. In this setting, the pathologist plays a vital role in confirming the diagnosis of malignancy and in investigating the likely site of origin of the tumour.
The pathological examination of a liver biopsy that is obtained from a mass lesion begins with assessment of a routine H&E stained section to confirm whether the biopsy has successfully targeted the tumour mass. If a tumour is present, it is then necessary to decide whether it could be a primary liver tumour, either a hepatocellular carcinoma or possibly a benign liver lesion such as a haemangioma (see Fig. 11.8). If it is a metastatic malignant tumour, it is necessary to determine the type of tumour, e.g. carcinoma, melanoma, lymphoma, etc. Immunohistochemical staining is useful but is always guided by the histological appearance on H&E staining.
Metastatic adenocarcinoma is very common and immunostaining can help to distinguish likely sites of origin such as colon, lung, breast, etc. This allows the pathologist to direct further investigation of the patient. Sometimes, confirming a diagnosis in this way is sufficient to guide further management, allowing immediate treatment of the tumour with appropriate chemotherapy and avoiding the need for further biopsy.
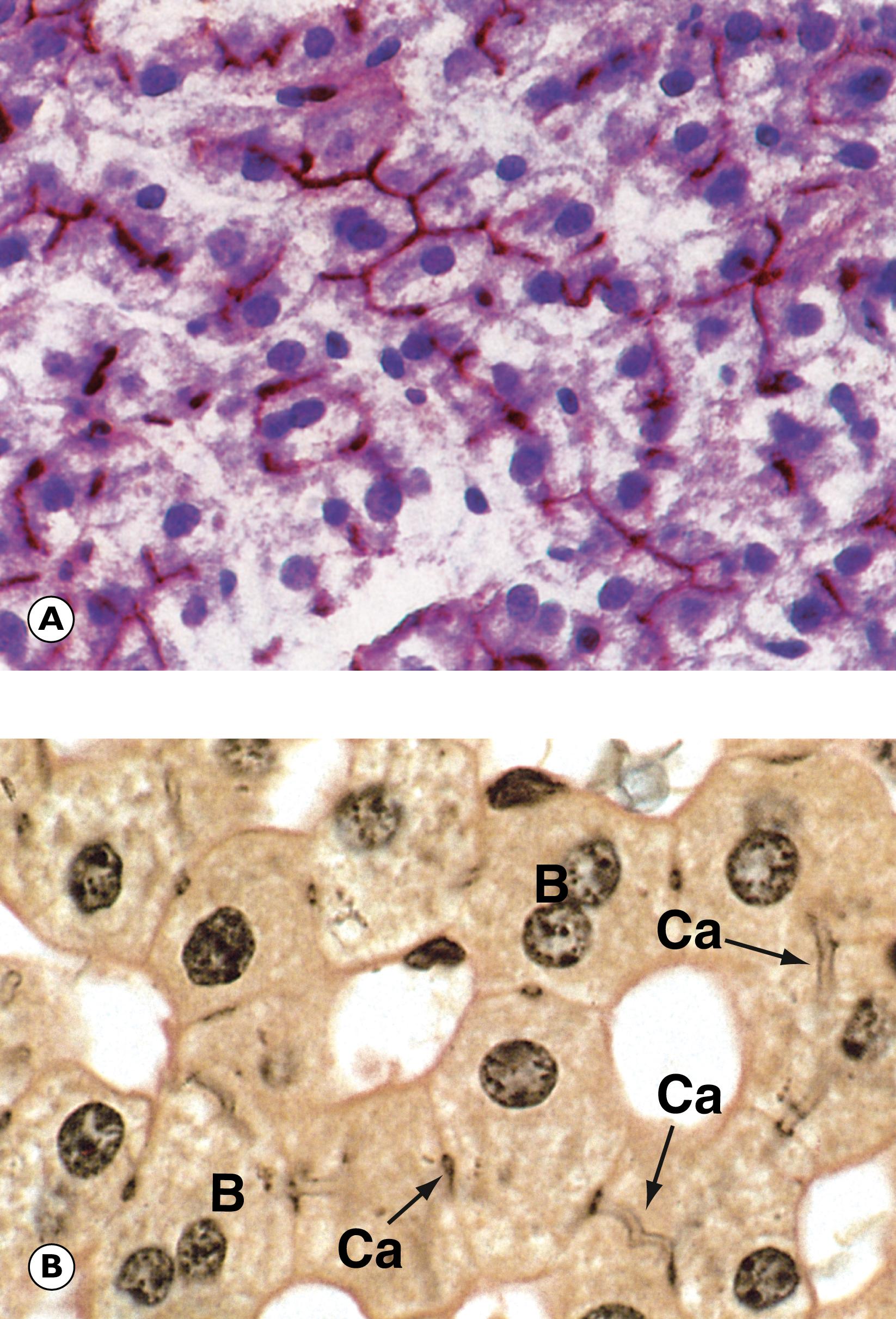
Cholestasis is the term applied to accumulation of bile within the liver. This is illustrated in Fig. 14.8 . Gallbladder disease is a common surgical problem in developed countries and is often associated with stone formation and chronic obstruction of the cystic duct leading to chronic cholecystitis ( Fig. 14.9 ). Cholesterolosis describes the deposition of cholesterol and other lipids within the lamina propria of the gallbladder. This is commonly seen in cholecystectomy specimens and is illustrated in Fig. 14.10 . Primary carcinoma of the gallbladder is very rare ( Fig. 14.11 ). Tumours of the biliary system are relatively rare and usually take the form of highly malignant adenocarcinomas known as cholangiocarcinomas ( Fig. 14.12 ).
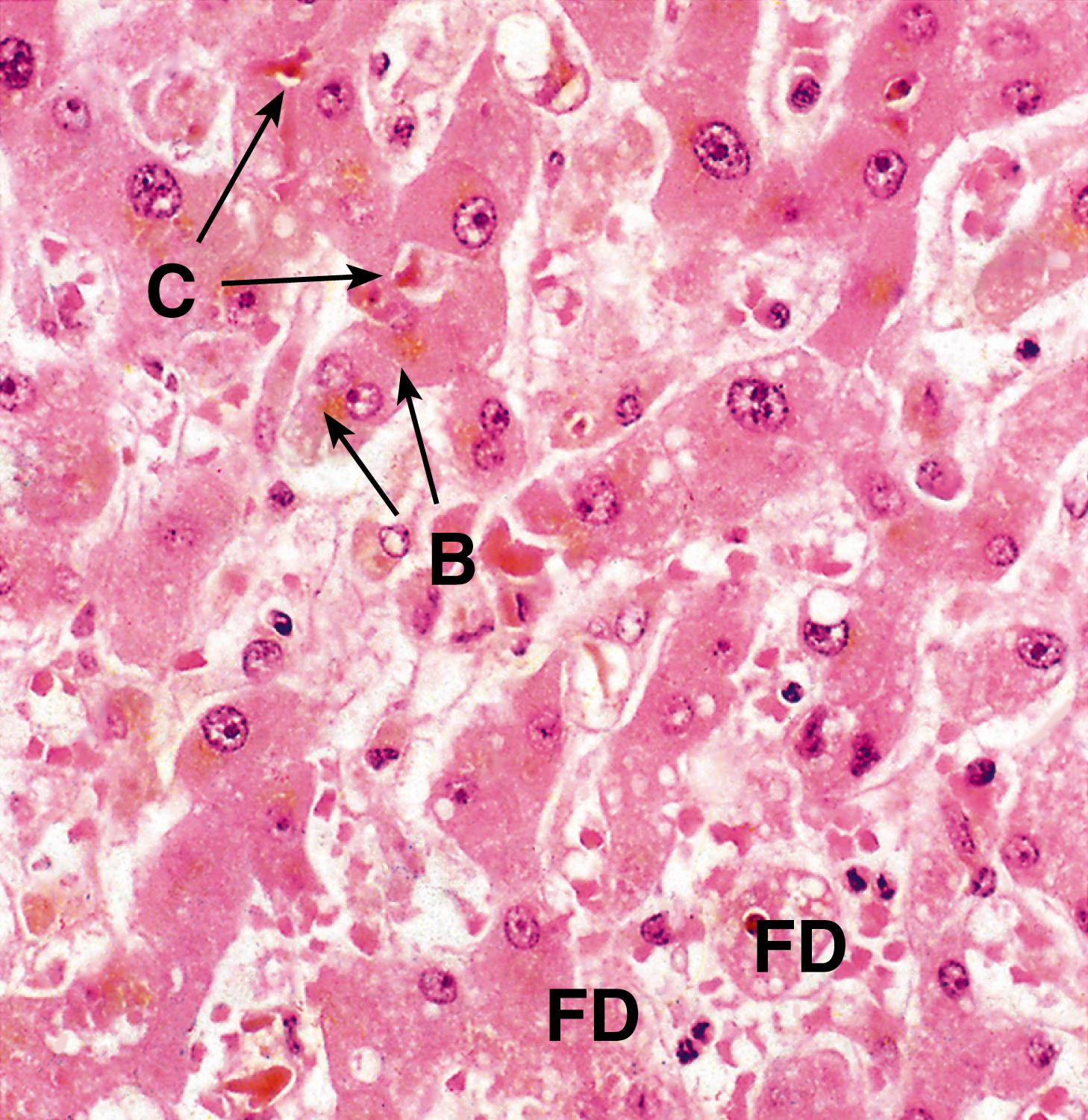
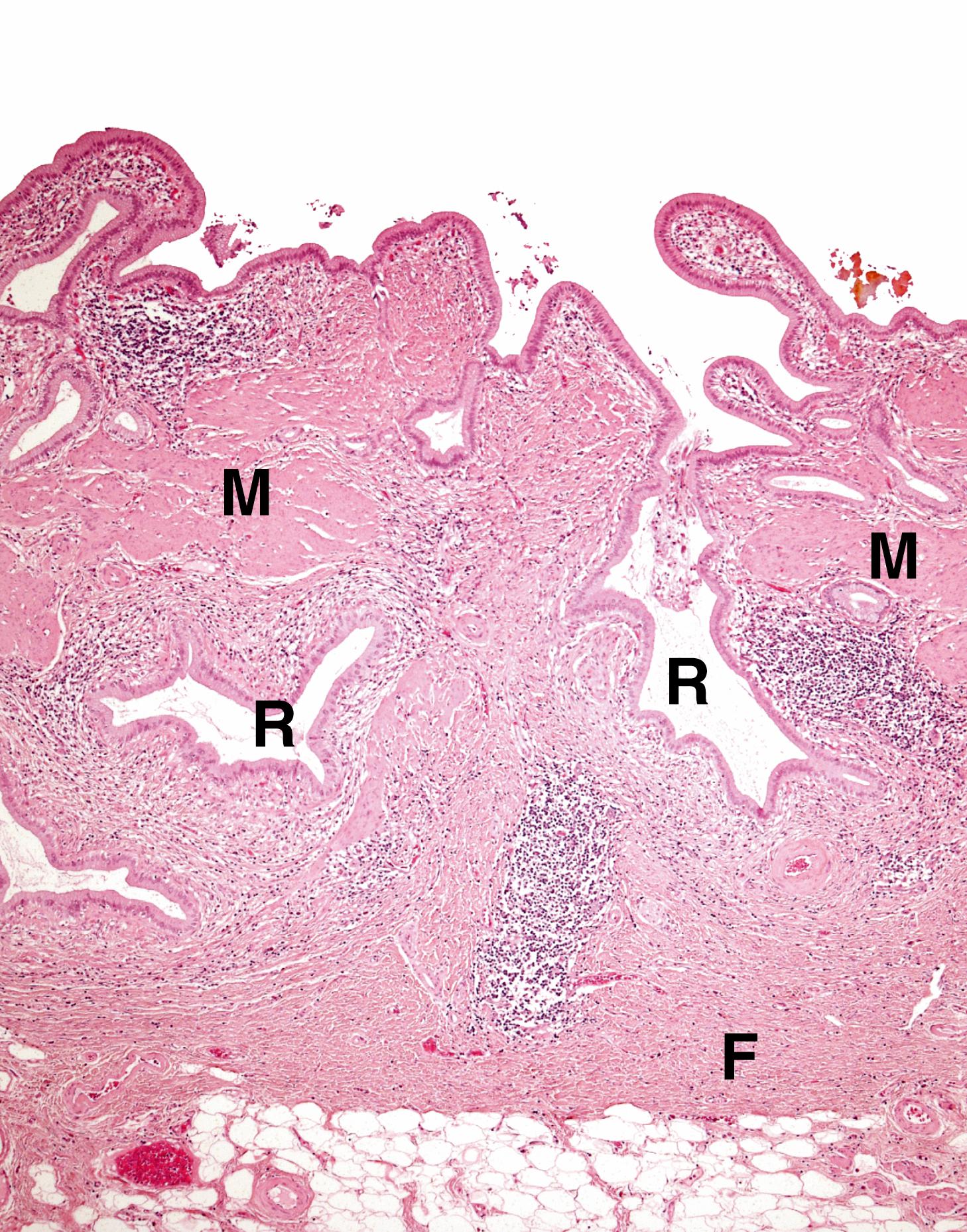
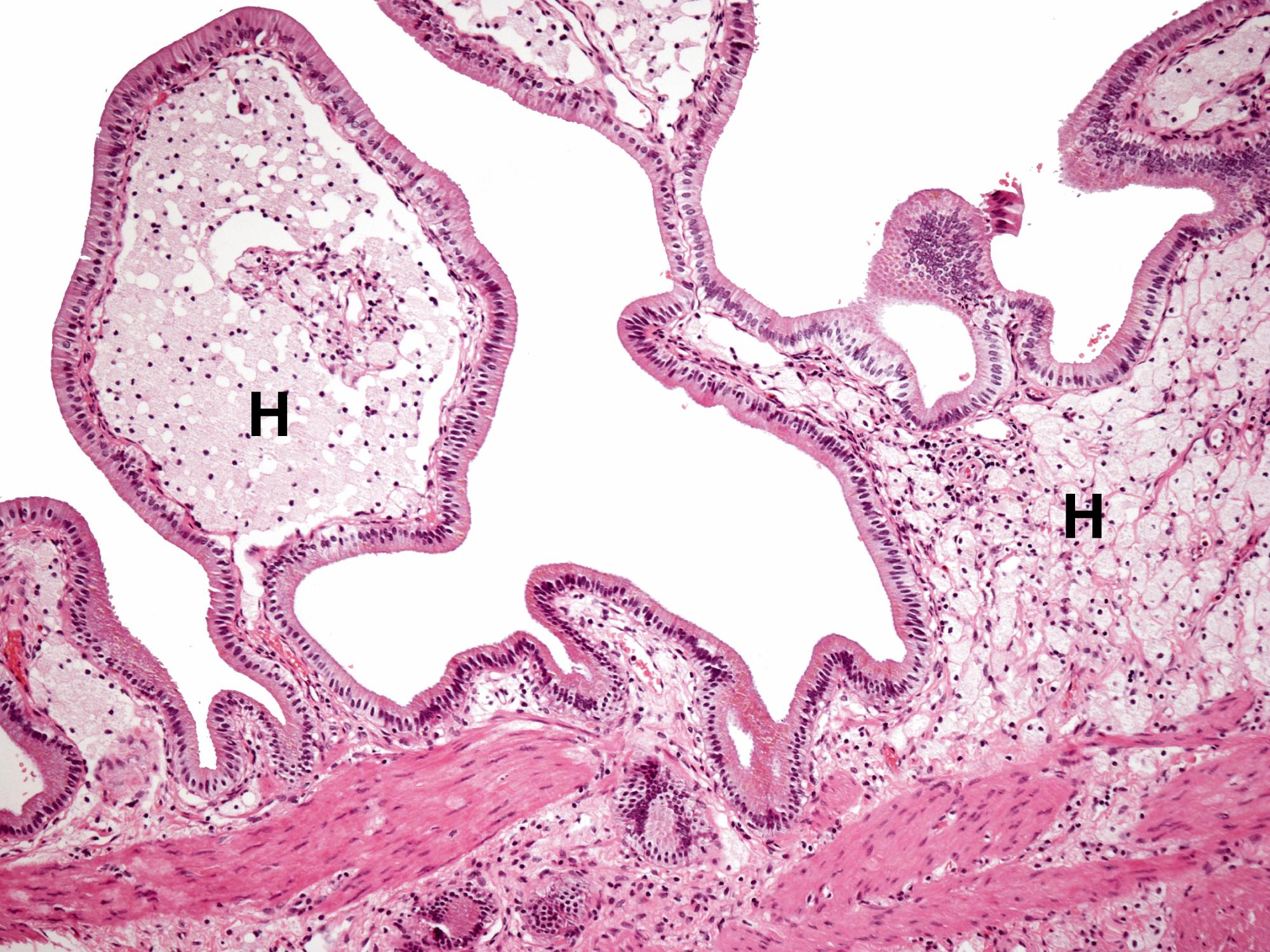
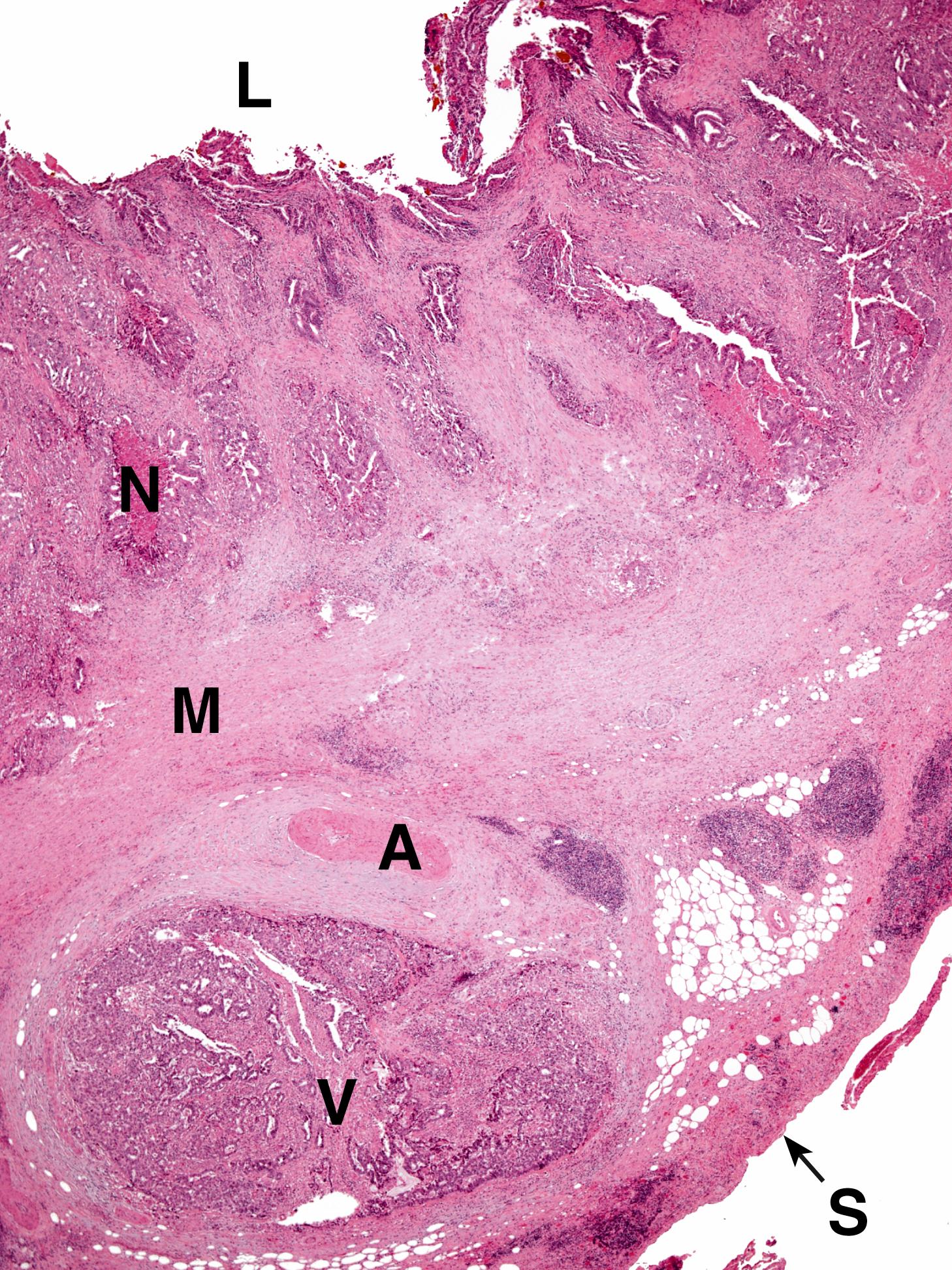
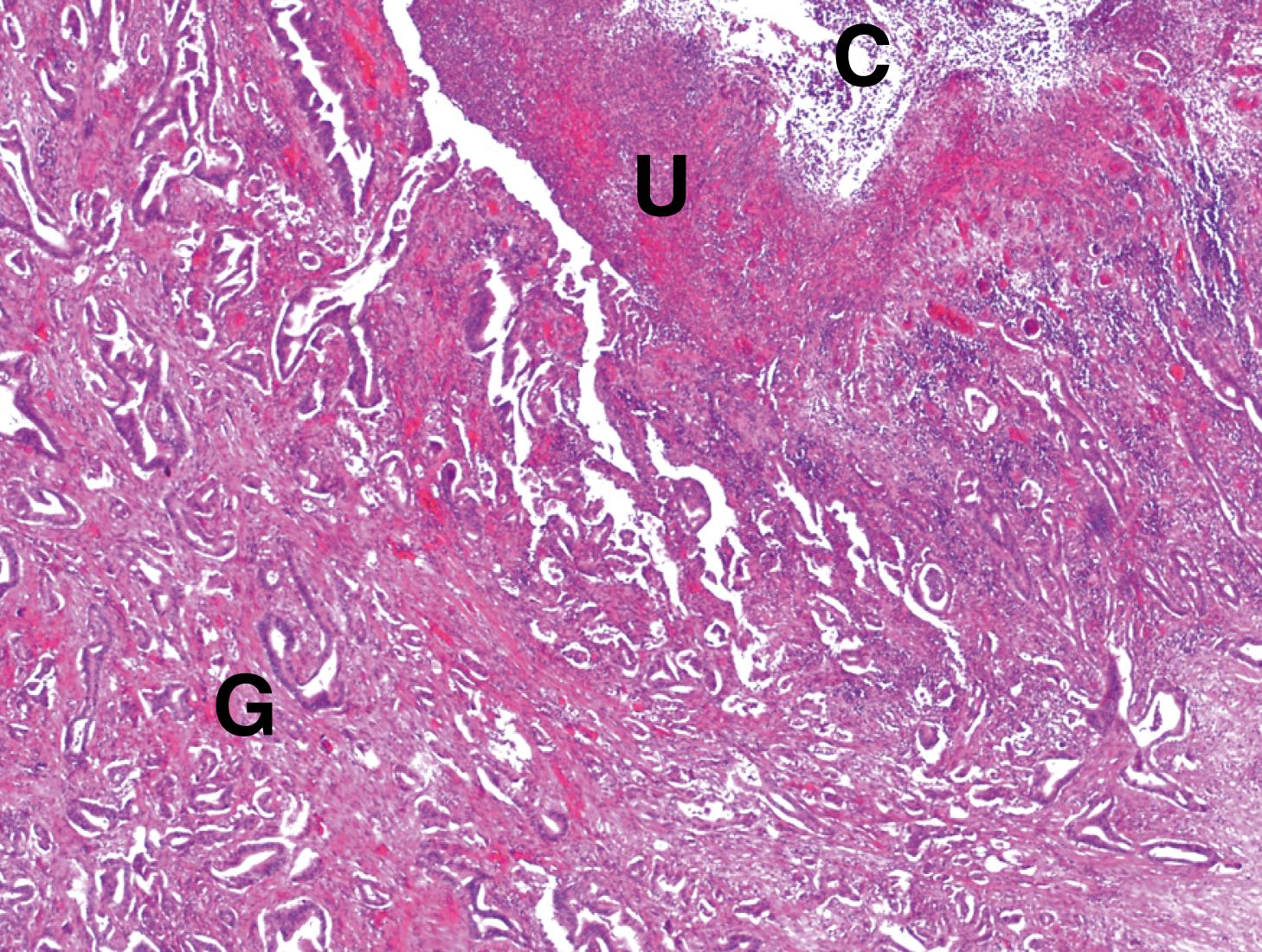
B bile C canalicular bile plugs D duct-like structure F fibrosis FD feathery degeneration H normal hepatocytes M muscle hypertrophy R Rokitansky-Aschoff sinuses T malignant tumour cells
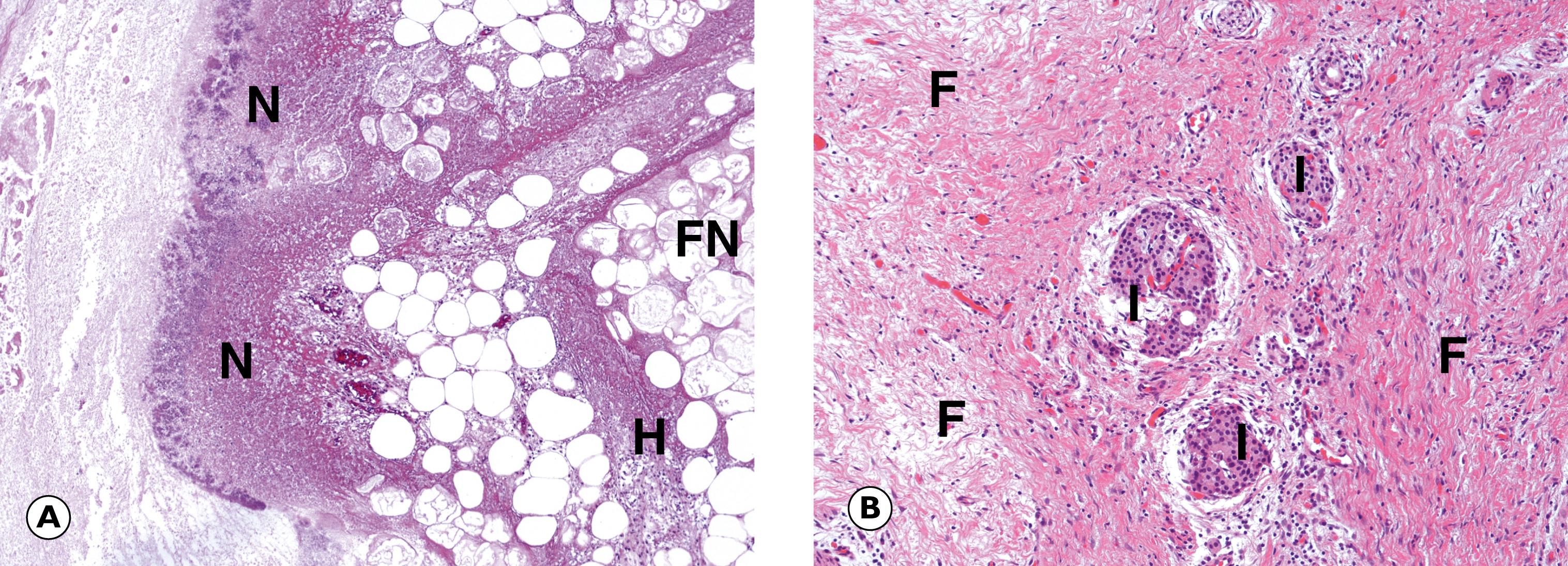
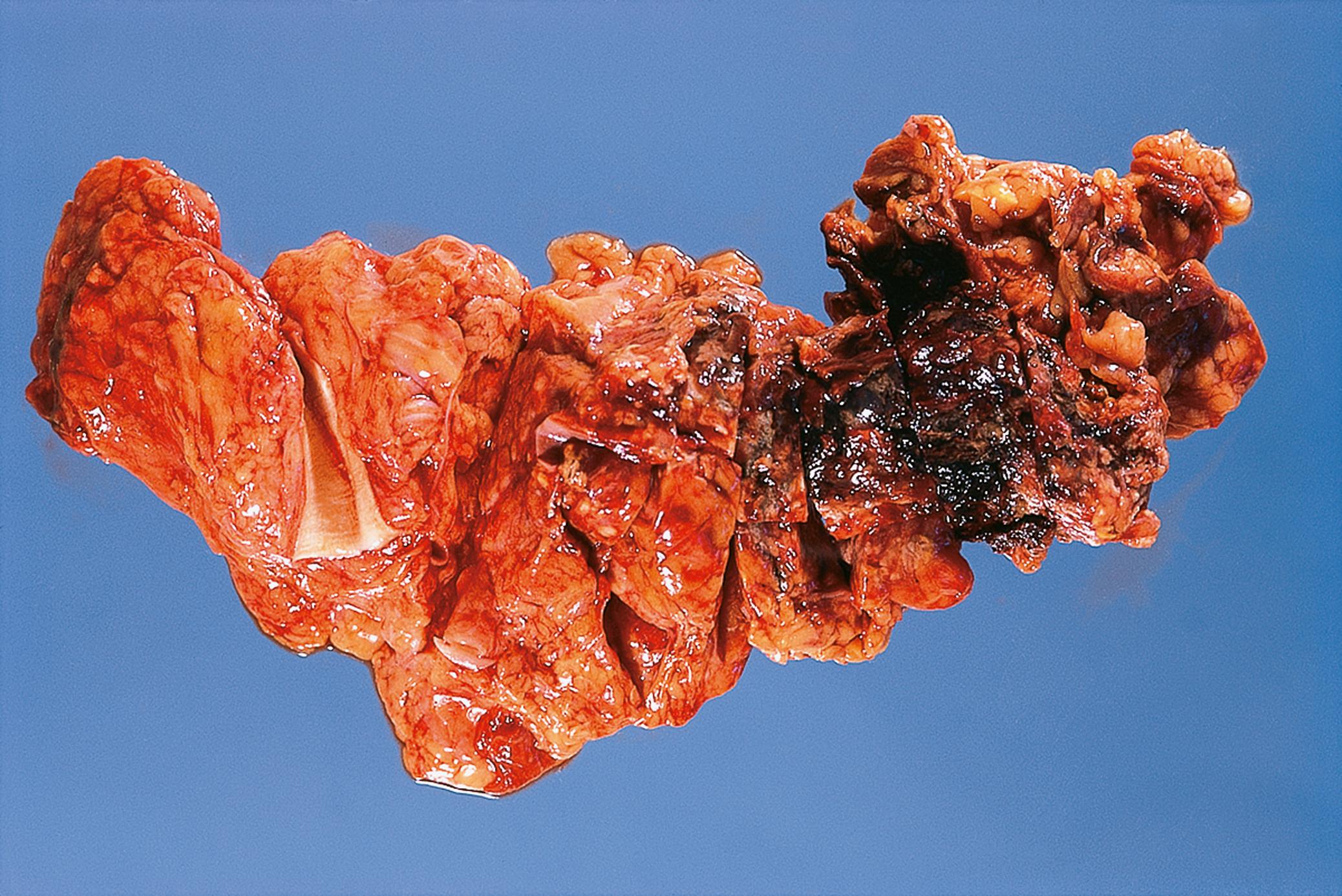
Inflammation of the pancreas ( E-Fig. 14.9 H ) (pancreatitis) may present in acute or chronic form. Acute pancreatitis is thought to occur as a result of inappropriate activation of pancreatic enzymes, which causes autodigestion of the pancreas and triggers an inflammatory cascade, which can result in necrosis both within the pancreas itself and in extra-pancreatic adipose tissues. Alcohol excess and gallstones are by far the two most common causes in western countries ( Table 14.3 ). Acute pancreatitis can range from a mild, self-limiting illness to severe pancreatitis with shock and multi-organ failure due to extensive necrosis and haemorrhage. Immediate complications of acute pancreatitis include infection, haemorrhage and shock. Long-term complications include pseudocyst formation as a result of resolution of large areas of necrosis and chronic pancreatitis. Chronic pancreatitis is characterised by fibrotic replacement of the pancreas. Alcohol excess is the commonest cause of chronic pancreatitis, which is frequently associated with intractable epigastric pain and pancreatic insufficiency. Chronic pancreatitis is also a risk factor for pancreatic cancer ( Fig. 14.14 ).
|
|
|
|
|
|
|
|
A artery C common bile duct F fibrosis FN necrotic fat G malignant glands H lipid-laden histiocytes I islets of Langerhans L lumen M muscularis propria N necrosis S serosa U ulcer V vein with tumour
Pancreatic ductal adenocarcinomas (PDAC) arise from the pancreatic ductal epithelium and are of great importance because of their insidious manner of growth, often remaining undetected until an advanced stage ( Fig. 14.14 ). Other primary malignant tumours of the pancreas can arise from the acinar structures (acinic cell carcinoma) or the islets of Langerhans (neuroendocrine tumours) . Neuroendocrine tumours of the pancreas show similar histological features to their counterparts in the gastrointestinal (GI) tract (see Fig 13.13) and are assessed in a similar fashion. Like GI neuroendocrine tumours, most pancreatic tumours are non-functioning; however, some produce excess hormone products including insulin (insulinoma) and glucagon (glucagonoma) . Other uncommon tumours that can involve the pancreas include metastases and lymphoma.
Most PDACs arise in the head of the gland where they tend to obstruct the common bile duct, thus presenting with obstructive jaundice. Tumours arising in the body and tail of the pancreas are very difficult to detect clinically and are often at an advanced stage at diagnosis. Histologically, PDACs are composed of glandular elements resembling the ductal structures in the gland, usually with marked surrounding desmoplasia. Often there is associated obstructive chronic pancreatitis. Precursor lesions include mucin-producing cystic neoplasms ( Fig. 14.15 ) and Pancreatic Intraepithelial Neoplasia (PanIN ) , which is a form of dysplasia in the pancreatic ducts ( Fig. 14.14 ).
Pancreatic tumours are usually diagnosed by a combination of radiology (CT scan and/or MRI), endoscopic ultrasound and cytological assessment (FNA of pancreas or common bile duct brushings). PDAC is usually treated with a combination of chemotherapy and surgery. A Whipple’s procedure or pancreato-duodenectomy is usually required to remove tumours in the head of pancreas along with the gallbladder, a portion of duodenum, common bile duct and associated lymph nodes. Many centres now perform pylorus-preserving surgery to preserve the outflow function of the stomach. Unfortunately, only a small proportion (10%–15%) of patients with PDAC are suitable for surgical intervention due to advanced tumour stage at diagnosis or poor medical fitness to withstand the stresses of such a major operation.
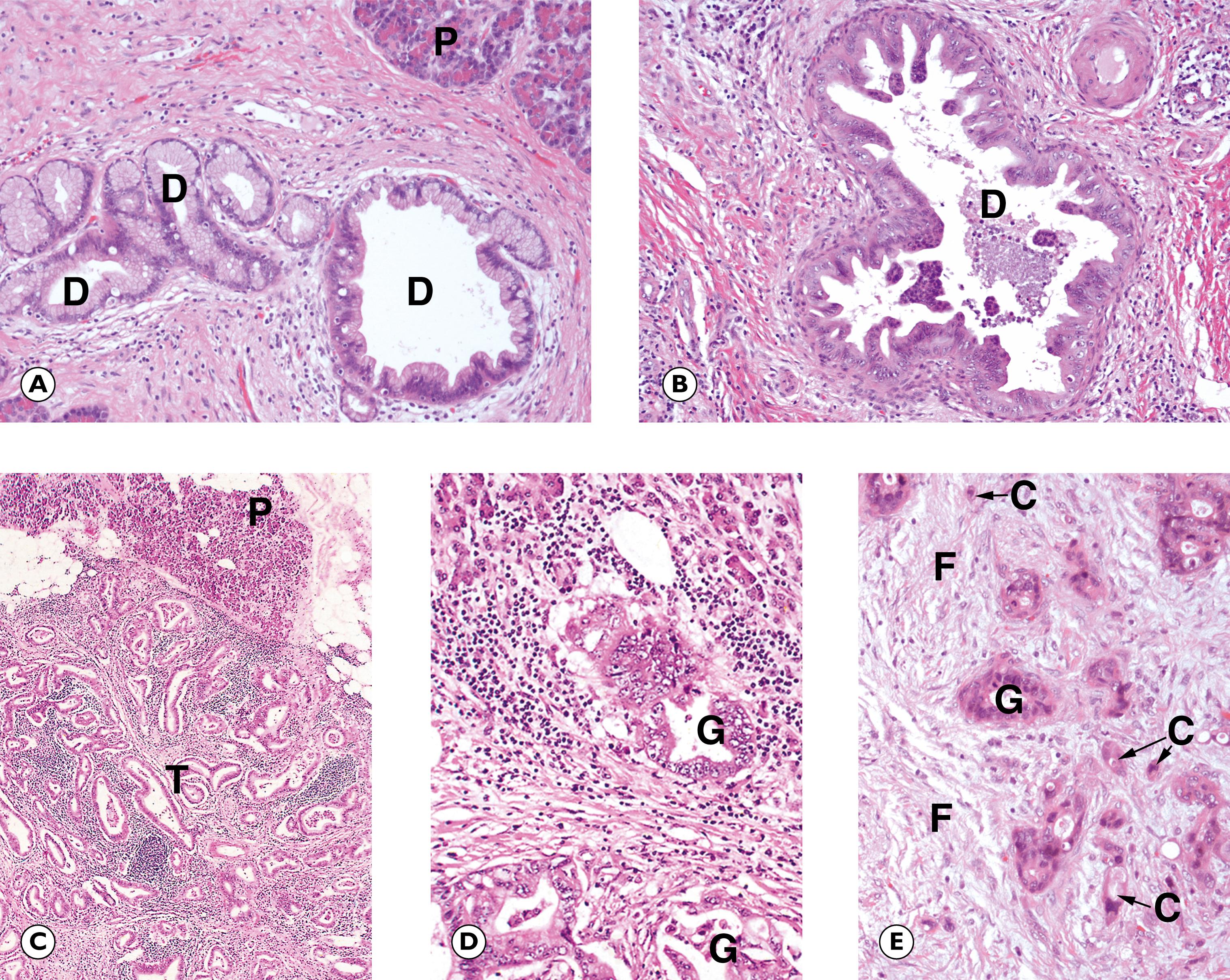
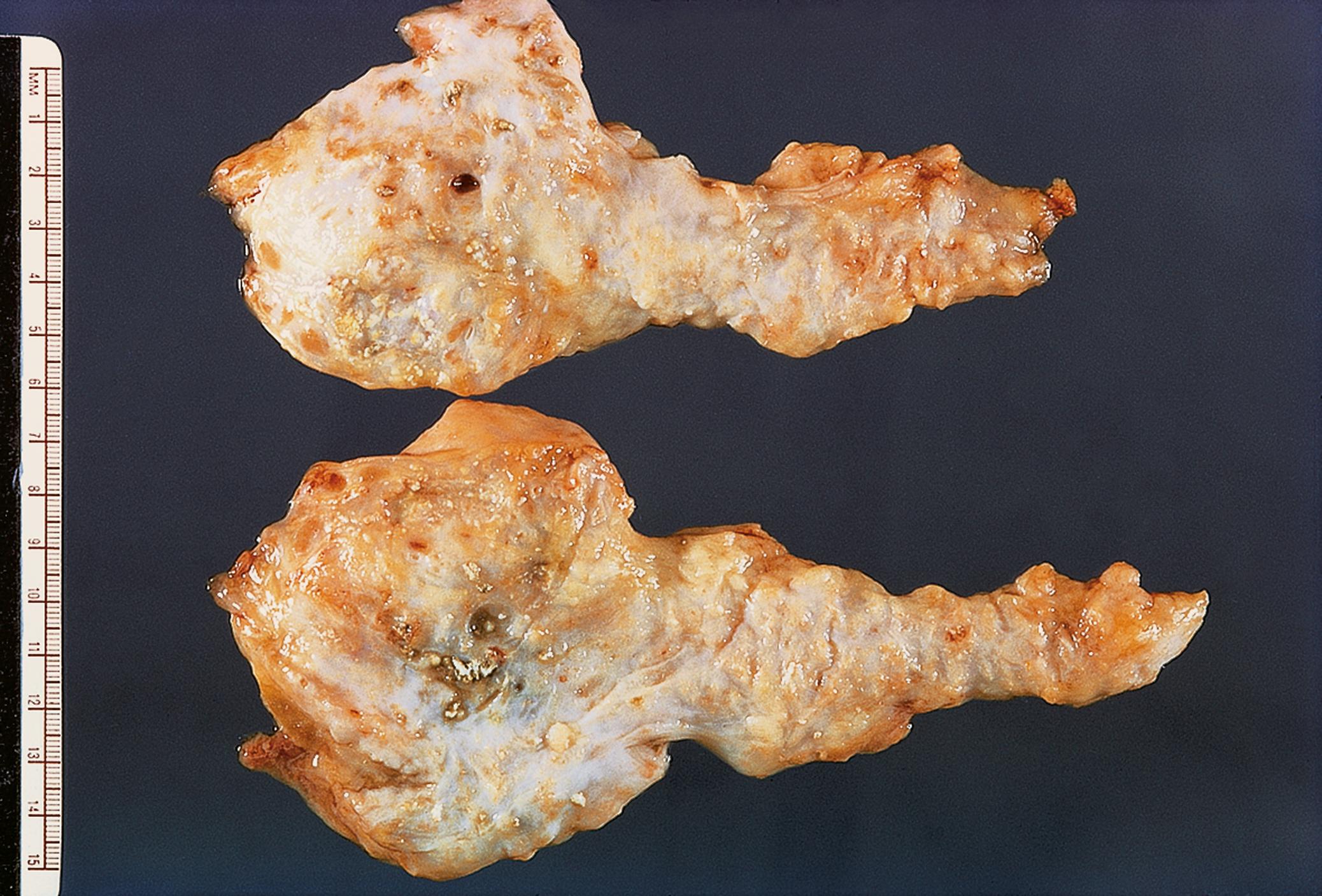
Cystic lesions are often detected incidentally following imaging for another condition. There are a variety of different types of cystic lesion that can involve the pancreas, listed in Table 14.4 . The commonest cyst in the pancreas is the pseudocyst , so-called because it is devoid of an epithelial lining. Pseudocysts arise as a complication of pancreatitis. True cysts of the pancreas contain an epithelial lining. There are two types of mucinous cyst associated with a risk of invasive PDAC and therefore are usually followed up and treated surgically when any worrying features arise. These are intraductal papillary mucinous neoplasms (IPMN) and mucinous cystic neoplasms (MCN) and are described in Fig. 14.15 .
| True cysts: Neoplastic | Mucinous: mucinous cystic neoplasm intraductal papillary mucinous neoplasm Non-mucinous: serous cystadenoma |
| True cysts: Non neoplastic | Lymphoepithelial cyst Epidermoid cyst Congenital cyst |
| Other | Cystic degeneration of pancreatic ductal adenocarcinoma, neuroendocrine tumour, metastasis or acinar cell carcinoma Pseudocyst |
| Disorder | Main features | Figure |
|---|---|---|
| Inflammatory liver diseases | ||
| Acute hepatitis | Cloudy swelling, fatty change, liver cell necrosis- various causes May resolve or persist as chronic hepatitis |
14.1 |
| Viral hepatitis | Hepatotropic viruses (A,B,C,D,E, etc.) or others (CMV, EBV, HSV, etc.)Hep A and E faecal-oral transmission, no chronicity or carrier state Hep B (D) and C blood borne, chronic hepatitis and carrier states |
14.1 |
| Alcoholic liver disease | Steatosis: fat droplets in hepatocytes Steatohepatitis: ballooning degeneration, neutrophils, Mallory’s hyaline.Fibrosis/cirrhosis- initially perivenular and pericellular collagen |
14.2 |
| Non-alcoholic fatty liver disease | Same morphological spectrum as alcoholic liver disease Association with obesity, dyslipidaemia, NIDDM |
Like 14.2 |
| Chronic hepatitis | Persisting more than 6 months, various causes Portal/interface/lobular inflammation (grade)Extent of fibrosis (stage) |
14.3 |
| Primary biliary cirrhosis | Early autoimmune damage to bile ducts with granuloma formation Later interface hepatitis, ductular proliferation and progressive fibrosis |
14.4 |
| Cirrhosis | Diffuse process, end stage of chronic liver damage Formation of regenerative nodules divided by fibrous bands |
14.5 |
| Haemochromatosis | Defect of HFE gene, iron overload with deposition in liver, heart, endocrine organs, joints and skinProgressive cirrhosis | 14.6 |
| Liver tumours | ||
| Haemangioma | Benign proliferation of blood vessels in the liver Common incidental finding on imaging |
11.8 |
| Hepatic metastases | Usually carcinoma, especially GI tract (portal drainage), breast, lung. Tendency to be multiple. | 7.16 |
| Hepatocellular carcinoma | Primary malignant tumour arising from hepatocytes- may secrete bile. Serum alpha-fetoprotein useful serum marker. Often single nodule. | 14.7 |
| Biliary disease | ||
| Cholestasis | Extrahepatic or intrahepatic obstruction to bile flow. Plugs of bile in liver canaliculi. Feathery degeneration of hepatocytes. | 14.8 |
| Chronic cholecystitis | Usually due to gallstones- cholesterol, pigment or mixed types. Chronic scarring, muscle hypertrophy, Rokitansky-Aschoff sinuses. | 14.9 |
| Cholesterolosis | Deposition of lipid-laden histiocytes in lamina propria. Gross appearance of ‘strawberry gallbladder’. | 14.10 |
| Gallbladder carcinoma | Uncommon, usually elderly patients. Origin from surface intestinal metaplasia and dysplasia. Presents late, often liver infiltration. | 14.11 |
| Cholangiocarcinoma | Adenocarcinoma arising in intrahepatic or extrahepatic bile ducts. Typically causes intense fibrous stromal reaction. | 14.12 |
| Pancreatic disease | ||
| Acute pancreatitis | Gallstones and alcohol main risk factors. Pancreatic enzymes cause tissue damage and necrosis. High risk of multi-organ failure. | 14.13A |
| Chronic pancreatitis | May follow repeated episodes of acute pancreatitis. Commonly alcohol related. Scarring and loss of gland tissue. Back pain, risk of diabetes. | 14.13B |
| Pancreatic adenocarcinoma | Usually head of pancreas, insidious growth. Presents late with obstructive jaundice. Poor prognosis. | 14.14 |
| Pancreatic endocrine tumours | Origin from islet cells. Resemble carcinoid tumours seen at other sites. Uncommon. Named for secretory product, e.g. insulinoma, glucagonoma. | 13.13 |
| Pancreatic cystic lesions | Often incidental finding. Broadly divided into non-neoplastic and neoplastic. MCN and IPMN associated with pancreatic adenocarcinoma. | 14.15 |
C single infiltrating malignant cells D ducts F desmoplastic stroma G malignant gland I intestinal type epithelium O ovarian type stroma P pancreas T tumour
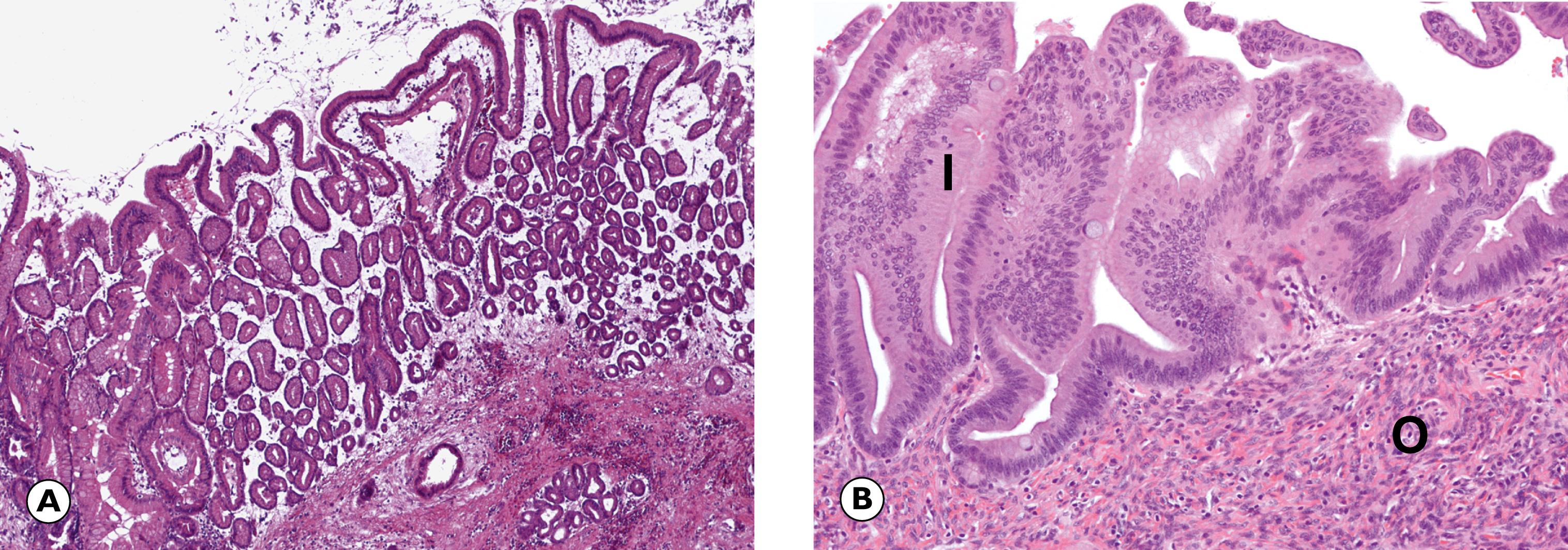
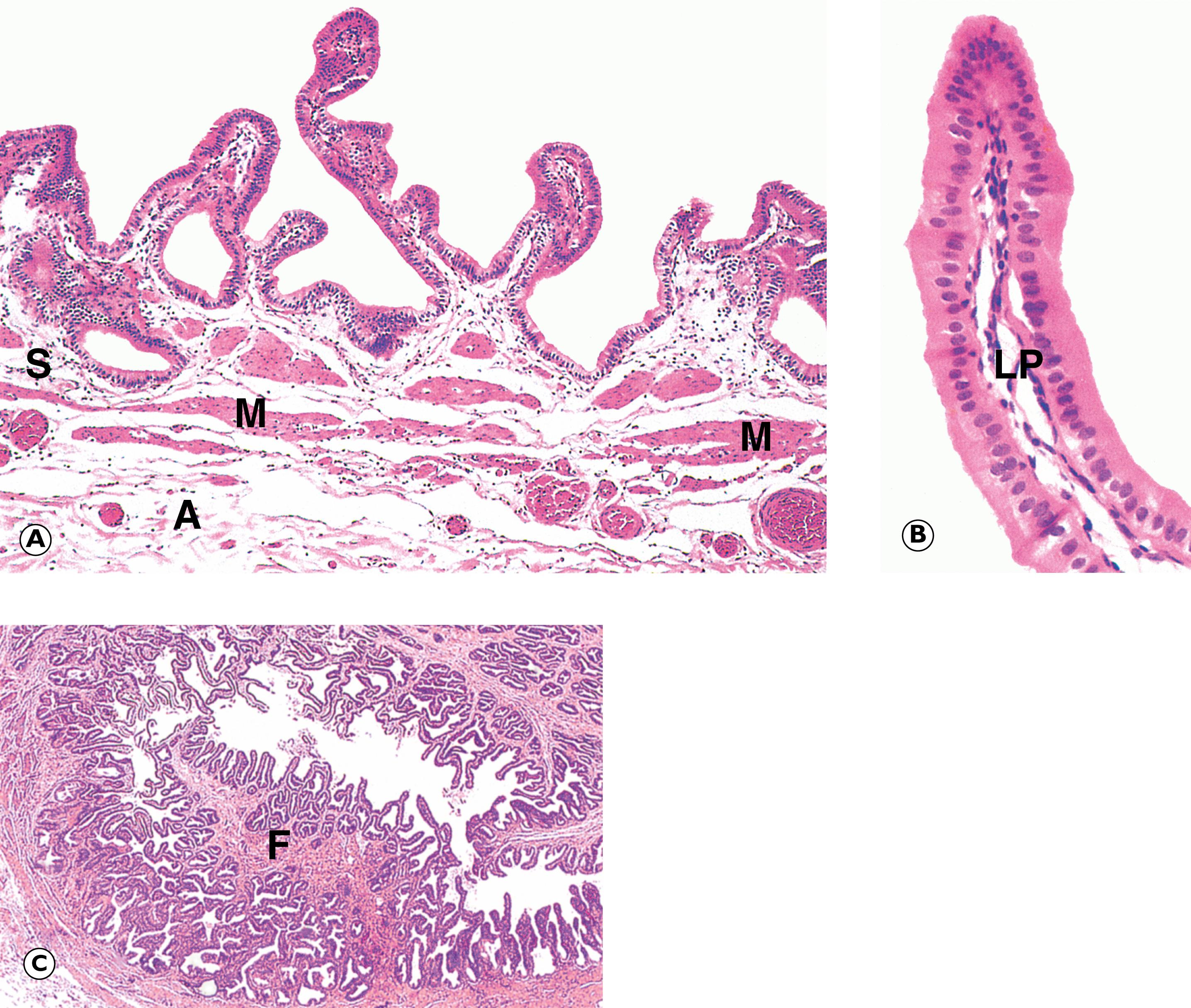
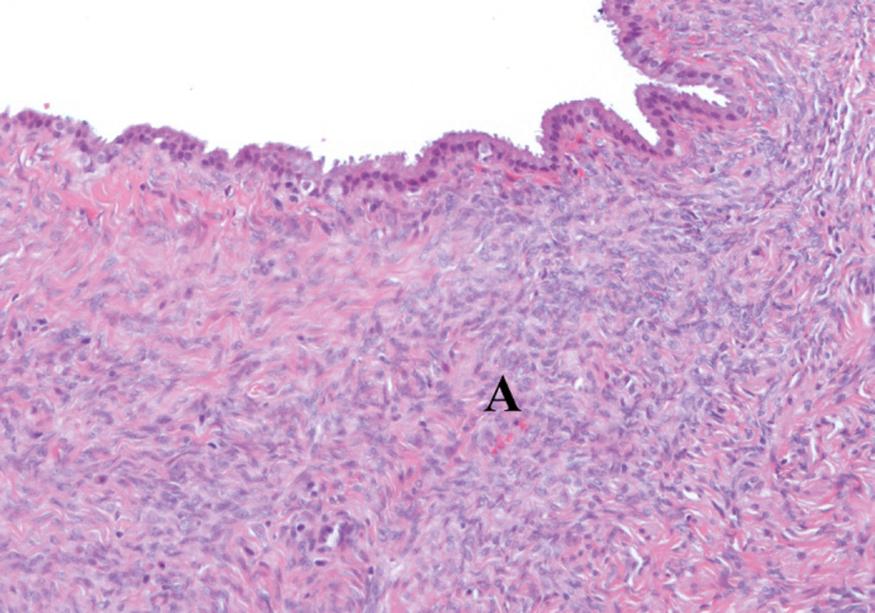
A 52-year-old woman presents with abdominal pain and a subsequent CT scan shows an incidental multiloculated cyst in the tail of the pancreas. This is surgically removed and a section is illustrated above. What is the characteristic feature shown in A? Select ONE answer.
Fibrosis
Mucinous epithelium
Inflammatory cells
Dysplasia
Ovarian type stroma
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here