Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
C.R.E. discloses significant research funding (to Vanderbilt University) from Medtronic Inc., Boston Scientific Inc., and Boehringer Ingelheim Inc. He is on the advisory board/consultant for Medtronic Inc., Boston Scientific Inc., AtriCure, Inc., and Abbott Medical Inc. A.N.K. is a speaker for Janssen Pharmaceuticals and Zoll.
The left atrial appendage (LAA) is formed from the left wall of the primary atrium during the fourth week of embryo development. The mature LAA moves freely in the pericardial sac overlapping portions of the left ventricle, left atrium, and pulmonary artery. It uniquely develops into a tubular, hooked, or crenellated structure with an orifice that smoothly contours into the left atrium. The LAA is trabeculated with muscle bridges or bars running in a parallel web (pectinate muscles) forming pits and saccules where blood stasis can lead to thrombus formation.
The first detailed assessment of LAA morphology using resin casts in 198 cases at necropsy was published in 1995, demonstrating a wide variation in LAA volume (0.7–19.2 mL), orifice width (5–40 mm), and length (16–51 mm). We described that the principal LAA axis was markedly bent or spiral in 70% of cases. The LAA volume and orifice width was larger in the subjects known to be in permanent atrial fibrillation (AF). The shape and size of the LAA are highly variable; in a 1997 human autopsy study, the LAA was found to have many different anatomic cavity configurations including 54% bilobed, 23% trilobed, 3% four-lobed, and 20% single lobed.
The endocardial ostium of the LAA is typically elliptical with its long axis obliquely oriented in relation to the mitral valve annulus. It is common to see small pits in the left atrial wall extending toward the mitral annulus or along the LAA–pulmonary vein (PV) ridge. The LA wall at these sites can become exceedingly thin (0.5 mm) and their association with AF-related stroke is not known. , The dynamic nature of the LAA also factors into the ostial contour and in highly mobile (contractile) variants; it can appear to nearly collapse completely shut with atrial systole. The opposite scenario is observed with a dilated LAA in permanent AF with minimal contractility and a seemingly immobile orifice.
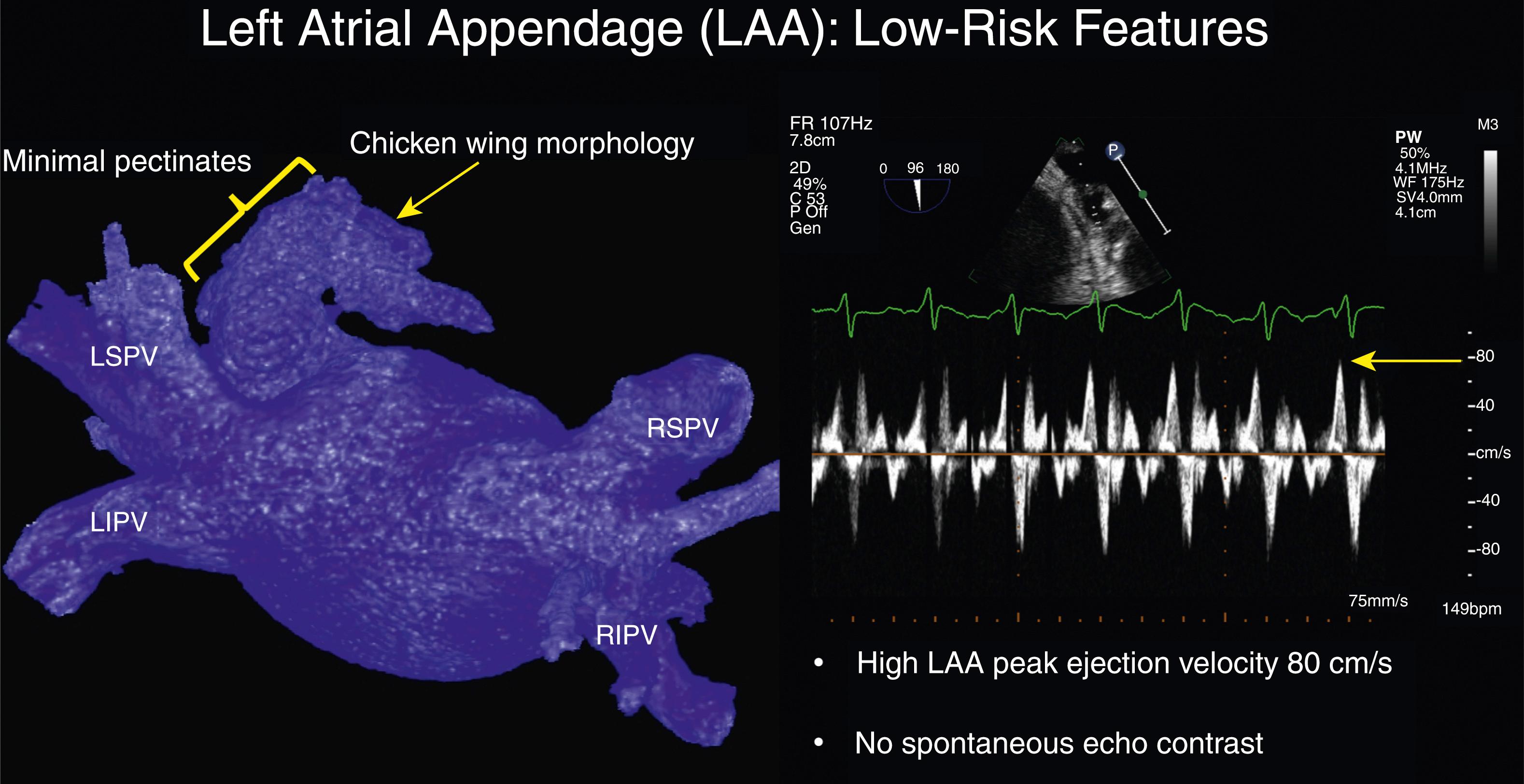
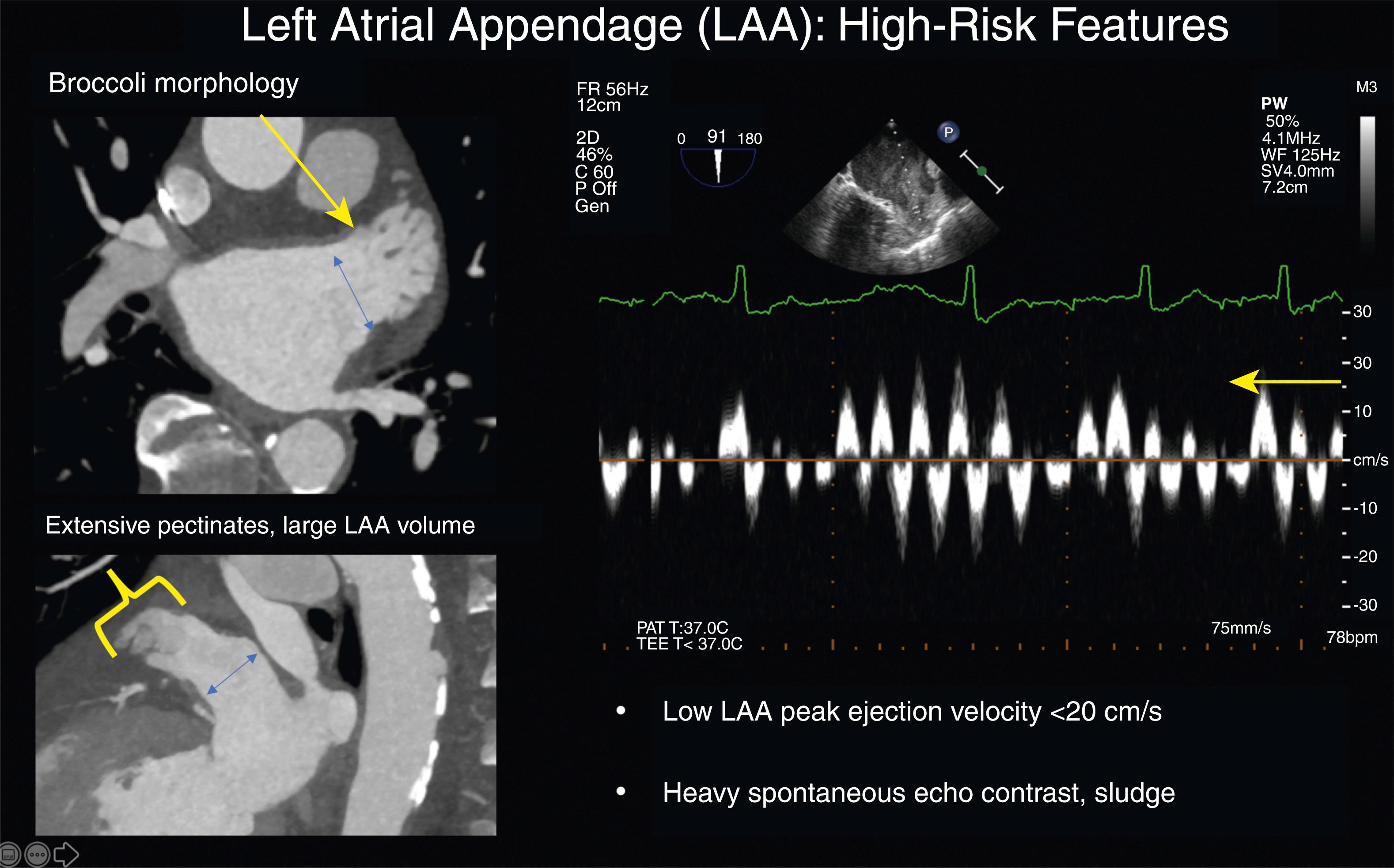
A relationship between LAA morphology and AF-related stroke risk appears linked to the gross description by computed tomography angiography (CTA) or magnetic resonance imaging (MRI) of the LAA as “chicken wing” (CW) ( Fig. 77.1 ) or “non–chicken wing” (NCW) appendage. In one series 48% of studied LAA anatomies were defined as CW, with a lower observed rate of embolic events in low CHADS (congestive heart failure [CHF], hypertension history, age ≥75 years, diabetes mellitus history, stroke or transient ischemic attack symptoms previously) score patients presenting for AF ablation. , The extent of pectinate muscles (typically 1–2 mm thick) creating webbed bundles and pits in the LAA wall may account for this observation. A study of silent cerebral ischemia demonstrated reduced trabeculations or pectinates in the more common CW LAA shape with extensive trabeculations in the less common broccoli or cauliflower LAA. Patients with heavy pectinates were observed to have a higher rate of cerebral ischemia. It is unknown when the LAA takes its final mature shape and whether the morphology of the LAA can change with alterations in LAA volume.
Congenital absence of the LAA has been reported in 17 patients to date with confirmatory CT or cardiac MRI and transesophageal echocardiography (TEE) imaging, in some cases, combined with direct left atrial angiography. The majority of cases were found incidentally during evaluation for cardioversion, AF ablation or LAA occlusion (LAAO). None of these cases have been confirmed at autopsy and the resultant effect of absence of the LAA on left atrial thrombus formation and AF-related embolic stroke risk is not known.
The gold standard for noninvasive evaluation of LAA anatomy and the presence of LAA thrombus likely remains TEE, although use of gated CTA with late phase imaging (60 seconds after initial LA imaging sequence) and cardiac MRI with gadolinium enhancement have shown comparable sensitivity to exclude clot. Detailed LAA anatomy for LAAO devices or AF ablation may favor CTA over TEE; however, TEE can provide functional assessment of LAA contractility and emptying velocity (LAA-EV). TEE also provides excellent views of the interatrial septum for transseptal access and can identify “valvular AF” in cases of rheumatic aortic and mitral valve disease. Examples of a low-risk and high-risk LAA are shown in Figs. 77.1 and 77.2 .
Markers of systemic thrombosis and fibrinolysis appear additive to excluding patients who do not have LAA thrombus. A recent study incorporating the use of a D-dimer level less than 270 ng/mL in an atrial thrombus exclusion (ATE) score was tested to rule out LAA thrombus by TEE in 2494 patients. ATE score was given for factors associated with the presence of atrial thrombus (hypertension, history of stroke, heart failure, D-dimer level >270 ng/mL). An ATE score of 0, or low risk, had 100% sensitivity for absence of LAA clot, whereas a CHADS 2 score of 0 failed to predict five patients with LAA clot on TEE.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here