Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This involves the selective injection of contrast medium into the right and left coronary arteries and the left ventricle (while recording the resultant moving images)
Being replaced by non-invasive CCT and CMR
A percutaneous femoral arterial catheterization is the usual approach (an alternative percutaneous route is the radial artery) ▸ at least 3 shaped catheters are required – one for each coronary artery and one for the left ventricle) ▸ low osmolality contrast media is used with rapid filming at 25+ frames/s ▸ images are acquired in multiple orientations with very short exposures (5–10 ms) to freeze cardiac motion
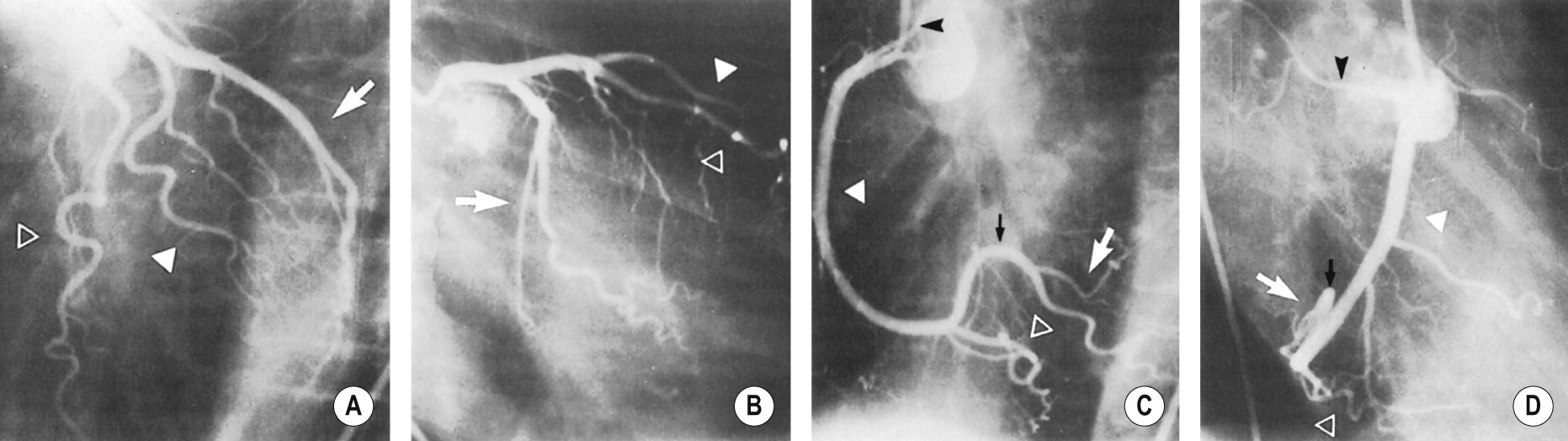
The severity and length of a stenosis ▸ the presence of a complete occlusion (± collateral vessels) ▸ the number of affected vessels ▸ the vessel diameter and stenosis configuration
Significant (>50% diameter reduction) accessible stenoses in 1 or 2 vessels (with a diameter > 2 mm) are treated by angioplasty or stenting
Left main coronary artery disease or significant disease affecting all 3 coronary arteries is usually best treated with a CABG
Poor ventricular function is associated with an increased risk, but a greater potential benefit (the prognosis is related to the degree of ventricular dysfunction)
High-risk patients with unstable angina or non-ST elevation myocardial infarction may benefit from early angioplasty or stenting
May miss mild or non-stenotic disease
A normal coronary angiogram does not exclude disease, and a stenotic plaque may be the tip of the iceberg
This allows imaging of the heart and great vessels through a limited chest wall acoustic window ▸ transoesophageal echocardiography can also image the heart
This can estimate blood flow and pressure gradients
This uses IV microbubble contrast ▸ it improves the definition of the margins of the ventricular cavity and enhances the accuracy of echocardiography for assessing ventricular function in poor echocardiography patients ▸ it can also assess myocardial perfusion
Demonstration of the effects of IHD on ventricular function
A large infarct will appear hypoechoic, whereas fibrotic scar tissue is stiffer than normal myocardium and appears hyperechoic
Detecting structural complications such as a VSD, papillary muscle dysfunction (causing mitral regurgitation) and ventricular thrombus
Demonstrating the origins of the main coronary arteries (especially with transoesophageal echocardiography) ▸ demonstrating any anomalous origins and coronary artery aneurysms (e.g. Kawasaki's disease)
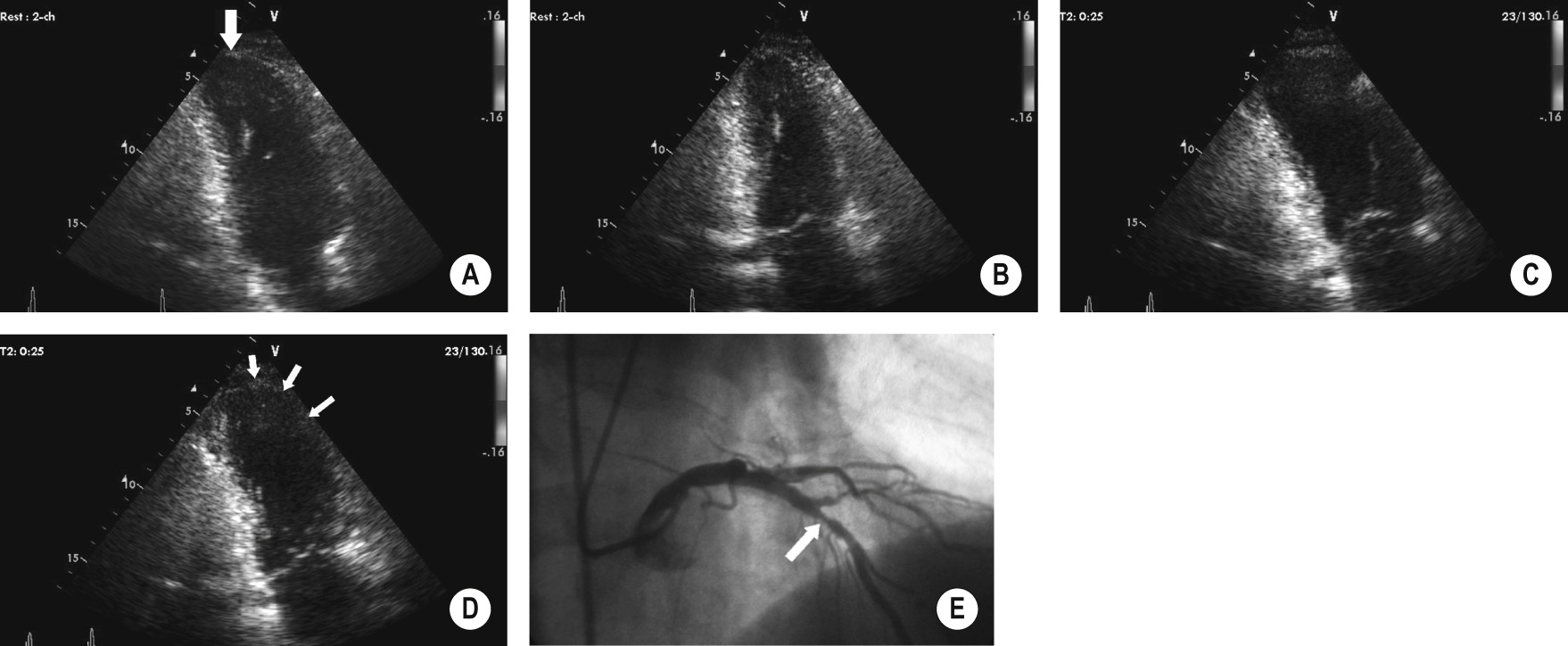
This detects reversible wall motion abnormalities (indicating reversible ischaemia) using the same stimulants as for CT or MR stress imaging
It enables risk stratification for patients with known or suspected IHD (a normal study indicates a very good prognosis)
It enables preoperative risk assessment (this determines myocardial viability)
It is more sensitive for detecting ischaemia than a conventional ECG-based exercise test ▸ the endpoint of the test is the appearance of new wall motion abnormalities (chronic ischaemia causes diffuse dysfunction, an acute myocardial infarction causes more localized changes)
Factors affecting the accuracy of the technique: the threshold for defining a significant stenosis ▸ whether there is single or multivessel disease ▸ whether an adequate stress is achieved (especially for exercise-based protocols) ▸ the presence of other disease processes which affect myocardial function (such as cardiomyopathy, microvascular disease, or hypertrophy)
| Vascular | Haematoma False aneurysm Arteriovenous fistula Mycotic aneurysm Retroperitoneal haematoma Acute occlusion Arterial dissection |
| Cardiac | Arrhythmias (catheter manipulation) Myocardial infarction Coronary dissection Systemic embolus (including stroke) Myocardial perforation |
| Contrast medium | Heart failure (reduced with low-osmolality contrast agent) Arrhythmias (sinus bradycardia, sinus arrest, ventricular fibrillation in 0.1-1.0%) ECG changes (wide QRS, long QT, ST changes, change in QRS axis) Hypotension (reduced with low-osmolality contrast agent) Allergic/idiosyncratic (reduced with non-ionic contrast agent) Renal impairment (possibly reduced with low-osmolality contrast agent) |
|
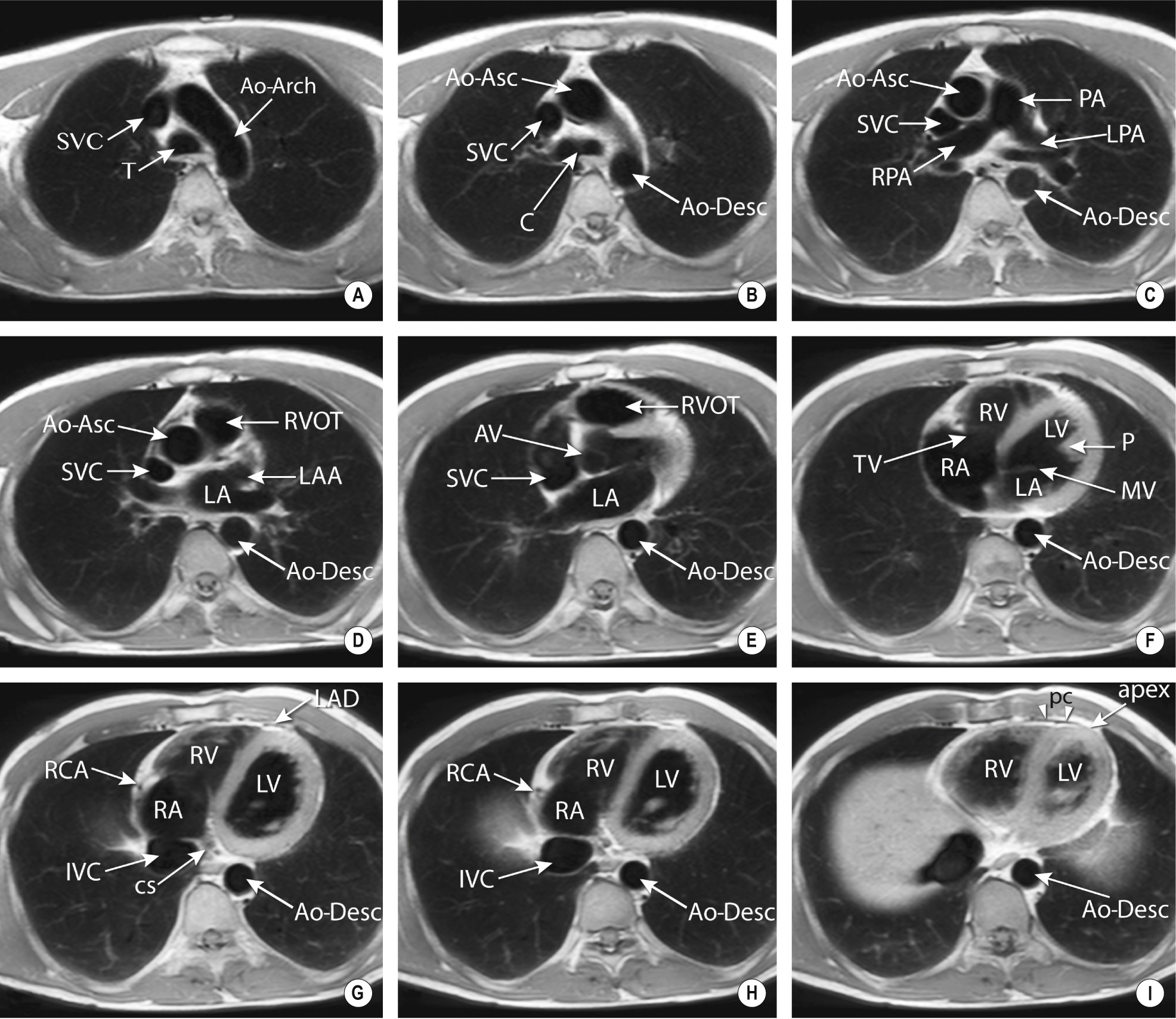
3D imaging technique – any imaging plane can be reconstructed with a slice thickness down to 0.5 mm
Single heart beat acquisition with wide volume detectors (up to 16 cm) or high pitch helical scanning (flash scanning) ▸ multiple (3-4) heart beat acquisition with narrow detector coverage (4 cm) and step-and-shoot ‘sequential’ acquisition
ECG-gating and heart rate control (oral or IV β-blocker, target heart rate <65 bpm) are used to minimize cardiac motion artefact ▸ sublingual nitroglycerin immediately prior to the scan is used for coronary vasodilatation
Prospective ECG-gating: image acquisition at a certain time point of the cardiac cycle (R-R interval), either diastole (70-80%) or systole (30-40%) or systole and diastole (30-80%) ▸ it is used for coronary artery assessment depending on the heart rate
Higher or unstable heart rates require imaging in systole or both systole and diastole
Retrospective ECG-Gating: data acquisition throughout the cardiac cycle (0-100% of R-R interval) only for functional assessment
Higher radiation dose due to longer exposure ▸ dose modulation reduces radiation doses
Temporal resolution: 75-175 msec depending on the scanner type ▸ best temporal resolution of 75 msec with dual source cardiac CT scanner
2 X-ray tubes and 2 sets of detectors ▸ only ![]() rotation for data acquisition
rotation for data acquisition
Coronary CTA: dual-head injection pumps allow contrast injection to be followed by saline injection (or a mixture of contrast and saline) using biphasic or triphasic injection protocols (cf. single-head pumps) ▸ volume of contrast ranges depending on the scanner type from 60-100 ml with an injection rate of 4-6 ml/s
Radiation doses range significantly (0.5-8 mSv) depending on the protocol, scanner type, body habitus and heart rate
Multiplanar reconstruction (MPR): this enables the viewing of any 2D section from a 3D dataset (the spatial resolution is the same for any orientation with near isometric voxels) ▸ a curved MPR can follow any tortuous anatomy
Maximum intensity projection (MIP): the highest attenuation voxels (CTA) or highest signal voxel (MRA) along any ray (line) are identified and projected onto a 3D image
Surface shaded display (SSD): all voxels with an attenuation or signal above a certain threshold are represented as a 3D image (which can be rotated in any direction) ▸ an imaginary source of illumination is used to provide 3D perspective
Volume-rendered techniques: volumes with a certain range of attenuations or SI are assigned a colour or grey scale density ▸ 3D reconstructions can benefit from the removal of areas of a certain attenuation or SI (e.g. bone for CTA or fat for MRA)
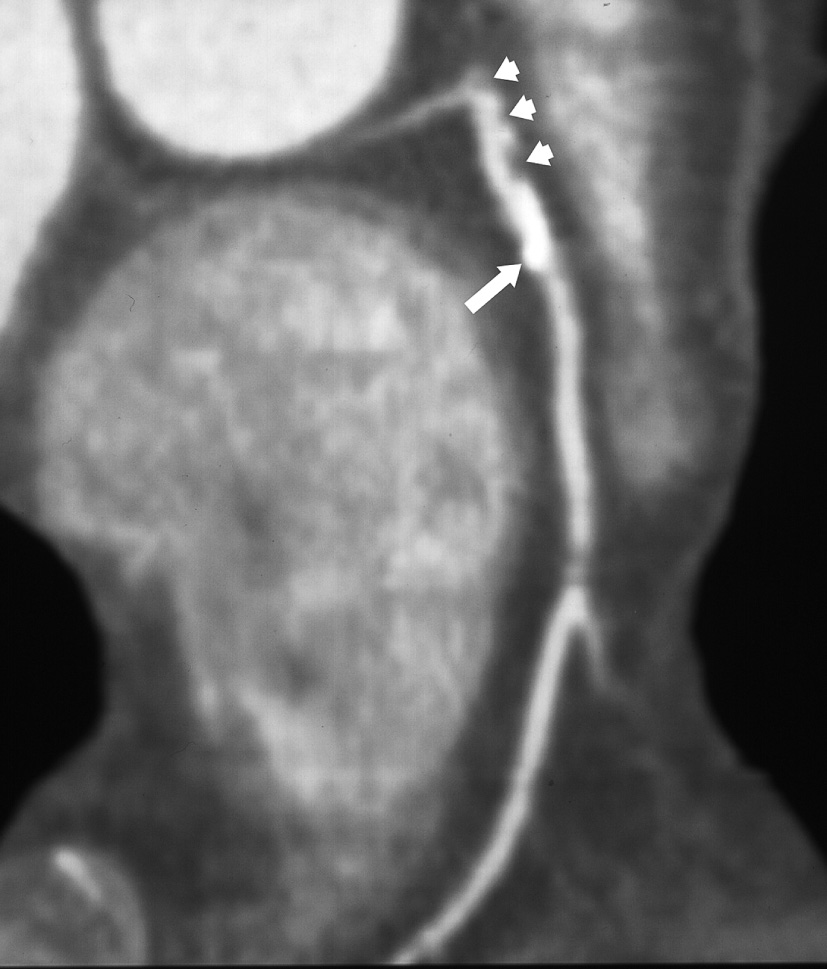
Agatston calcium score : this quantifies the presence of high attenuation material within the coronary arteries ▸ the amount of coronary calcium roughly correlates to the plaque extent, but is not related to plaque stability (weakly related to the degree of luminal stenosis) ▸ tissue densities ≥ 130 HU are used as an attenuation level for calcified plaque ▸ alternatively the actual volume of plaque is quantified (CVS: total calcium volume score) ▸ some studies suggest that a high calcium score predicts an increased risk of an adverse coronary event
It can detect plaque of the coronary artery wall and assess the degree of luminal narrowing ▸ narrowing may be severe (>70%), moderate (50-69%), mild (25-49%) or minimal (<25%)
CT fractional flow reserve (FFR) and CT perfusion are emerging applications for assessment of haemodynamic significance of stenosis, increasing the accuracy of CTA (a purely anatomical test)
It has a very high negative predictive value for coronary artery disease (95-99 %) when images are diagnostic
Non-diagnostic images mainly due to cardiac motion artefact (caused by high heart rate), blooming artefact (caused by heavy calcification, stents, metallic clips)
In addition (in up to 10% of cases) MDCT can detect significant non-cardiac diagnoses (e.g. pneumonia, lung cancer, PE)
Coronary artery dominance: the artery that gives rise to the posterior descending artery
Right dominant (85%) ▸ left dominant (7%) ▸ co-dominant (8%)
Assessment of anatomy and patency of grafts post CABG
Assessment of suspected coronary artery anomalies
Evaluation of aortic pathology
Evaluation of congenital heart disease and pericardial disease pathology
Evaluation pre and post procedures:
TAVI (trans-catheter aortic valve implantation) ▸ TMVI (trans-catheter mitral valve implantation) ▸ AF ablation
Assessment of valvular heart disease in selected cases
Assessment of myocardial scarring and ventricular function are feasible but of limited use in clinical practice
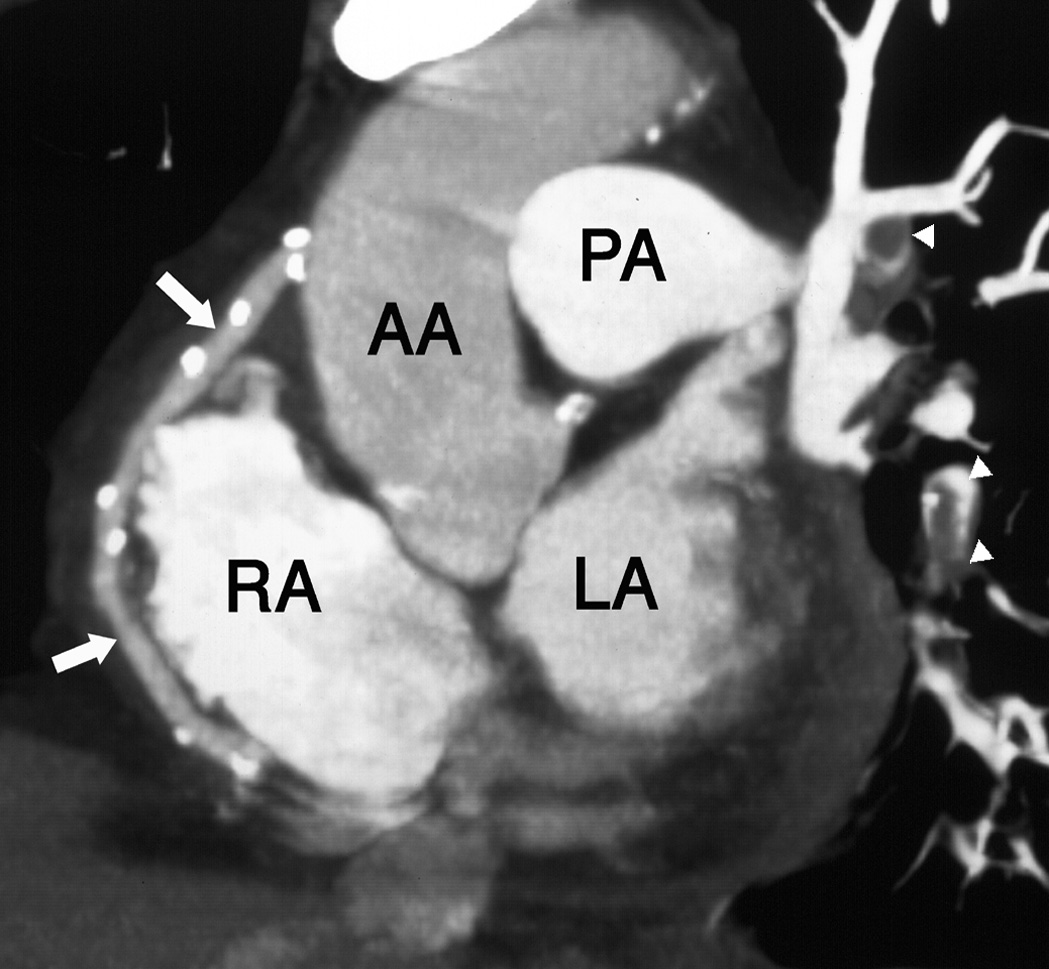
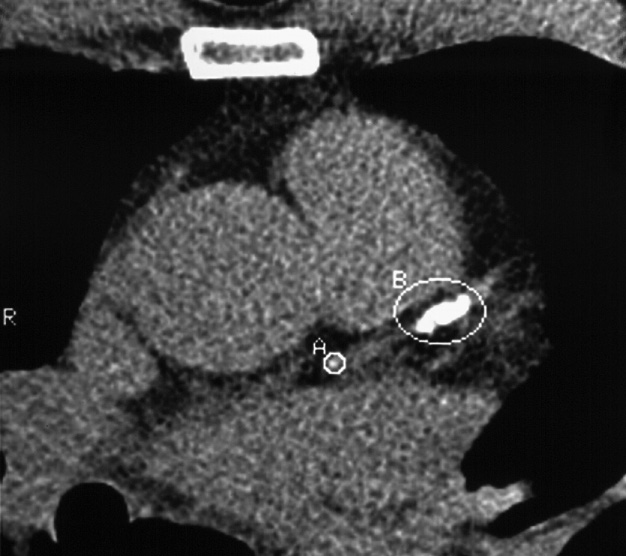
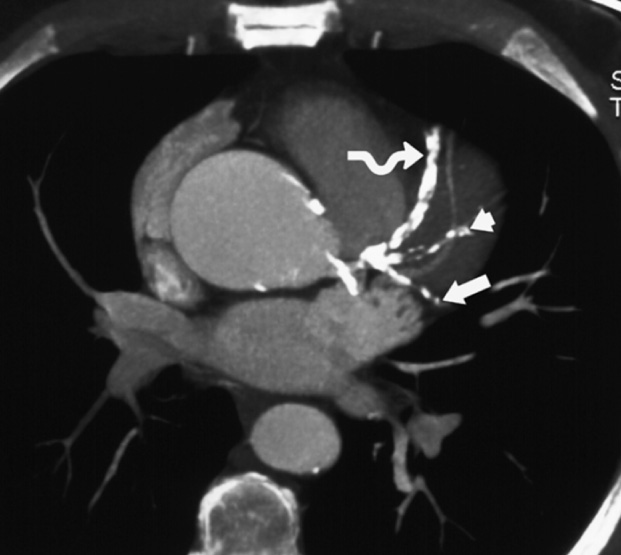
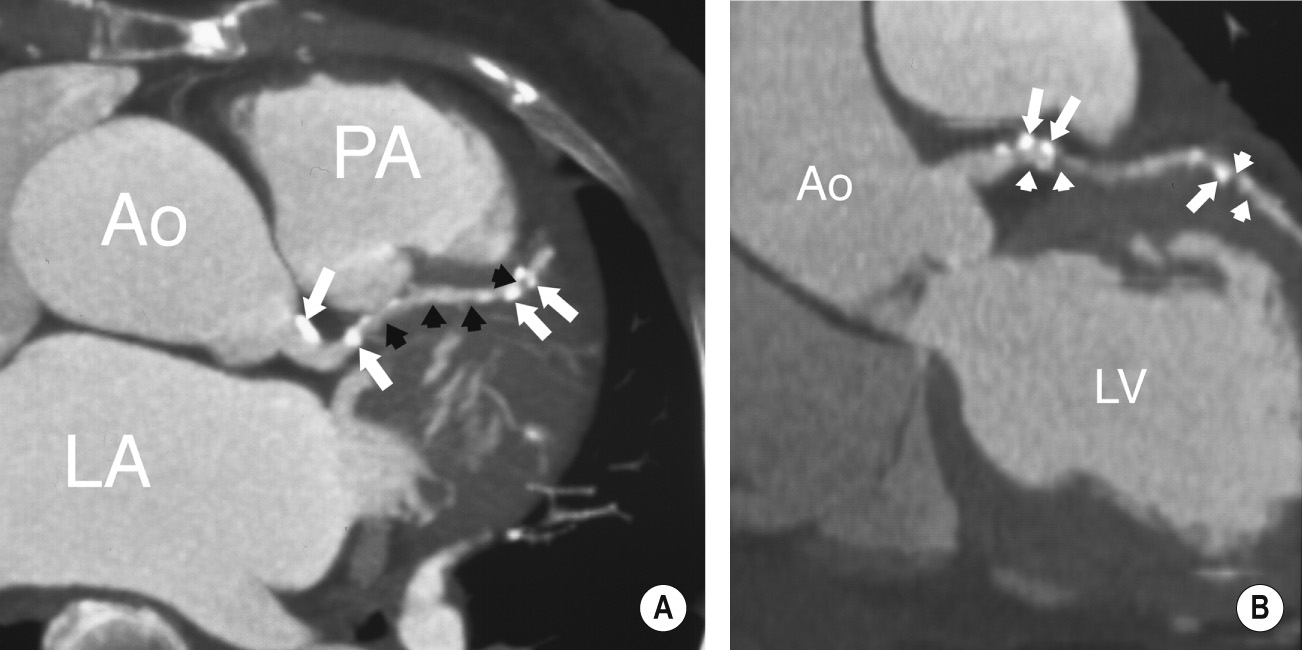
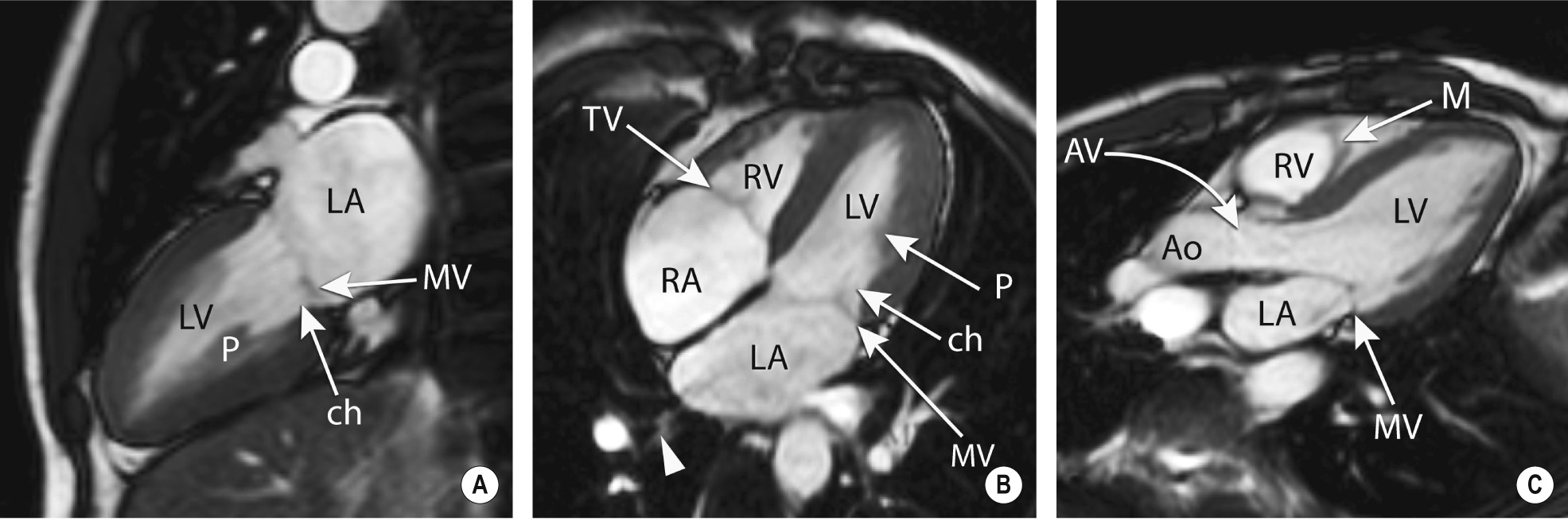
‘Black blood’ imaging: the myocardium and vascular wall appear bright and the blood dark ▸ useful for high spatial resolution static anatomical imaging (but rather slow to perform)
180° inverting pulse applied to the imaging volume followed by a slice selective 180° ‘de-inversion’ pulse: net result is inverted spins outside the imaged slice but spins within the imaged slice are unchanged (they have experienced both inversion and de-inversion) ▸ the time at which this is applied is the time at which the longitudinal magnetization of blood has reached zero from the initial inversion – therefore blood flowing into the slice will generate no signal (‘black blood’) ▸ variable signal from slow or in-plane blood flow (producing artefacts)
‘Bright blood’ imaging: blood produces a higher SI than that seen with spin-echo sequences
This is because only one radiofrequency pulse is used and there is less time for the blood to move out of the imaging plane between slice selection and image acquisition (turbulent blood flow will produce areas of signal loss)
The gradient-echo sequence can be repeated more rapidly with a reduced RF flip angle, allowing the acquisition of cine loops ▸ this also allows quantification of ventricular stroke volumes (comparing end-systolic and end-diastolic volumes)
This allows quantification of flow velocities
Positive velocity encoding: if a gradient is applied in the direction of blood flow for a finite time and then turned off, the relationship of phase will change in relation to the two ends of the gradient (the protons at the stronger end of the gradient precess at a faster rate during its application than those at the weaker end)
Negative velocity encoding: when the gradient is turned off, the rate of precession becomes constant again in the slice, but the phase relationship has changed and the phase signature remains ▸ when the gradient is reversed and applied for the same period of time as the original gradient, the phase signature of still material is cancelled, but flowing blood moving during the gradients to a different phase territory retains a phase change proportional to its velocity
Simple subtraction of the images obtained with positive and negative velocity encoding will generate a phase image whose pixel value is directly proportional to the blood flow velocity
T1WI: the heart is imaged during the bolus administration of a gadolinium contrast agent
Ischaemic areas will demonstrate delayed enhancement
T1WI: accurately determines CABG patency-possibly that of the infarct-related artery after coronary thrombolysis
It has an equivalent accuracy to MDCTA (however MDCTA is quicker and easier to use)
Prior to a GE cine loop a special selective excitation pulse produces a 2D grid structure within the myocardium (narrow planes of very low SI appearing as a latticework)
This moves and deforms throughout the cardiac cycle allowing assessment of movement
Prospective gating: the QRS complex of the ECG triggers the imaging such that the image is formed from a specific part of the cardiac cycle
Retrospective gating: the imaging sequence is repeated continuously with a constant repeat time while the ECG is monitored ▸ the data are sorted at the completion of the sequence and adjusted to give images at specific parts of the cardiac cycle
Prevention of respiratory motion (breath holding) is important
Usually obtained in orientation to the axes of the heart (as for echocardiography)
Myocardial function at rest: end-diastolic and end-systolic volumes can be calculated from the endocardial outlines using fast cine GE imaging
Myocardial function during stress: this allows stress assessment for inducible ischaemia and reversible dysfunction using dobutamine or adenosine infusions
Myocardial perfusion and viability: it is possible to measure the myocardial perfusion from the peak myocardial enhancement ▸ delayed hyperenhancement identifies infarcted or non-viable myocardium
Coronary arteries and bypass grafts: useful for assessing anomalous coronary arteries and graft patencies
Valvular heart disease: velocity mapping allows measurements of peak velocities across stenoses ▸ it provides quantification of regurgitation (GE sequences show turbulent blood flow as areas of signal loss) ▸ phase contrast imaging allows assessment of the stenosis severity
Acute myocarditis: focal wall motion with abnormal high signal on T2WI and contrast enhancement on T1WI
Cardiac infiltration:
Sarcoidosis: T2WI: high SI ▸ T1WI + Gad: enhancement ▸ delayed contrast-enhanced MRI: enhancement
Amyloidosis: thickening of the myocardium, interatrial septum, valves, leaflets and papillary muscles ▸ atrial dilatation ▸ pleural and pericardial effusions ▸ T2WI: high SI ▸ T1WI + Gad: enhancement
Myocardial iron overload: a diffuse reduction in contraction ▸ reduced myocardial SI
CMR can also assess: the pericardium ▸ cardiac thrombus and tumours ▸ cardiomyopathies ▸ congenital heart disease
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here