Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Pathology, also referred to as histopathology, anatomical pathology or morbid anatomy, is the study of abnormal or diseased tissues and organs. This book is intended for the student approaching histopathology for the first time and aims to give a thorough but concise grounding, both in basic pathological processes and in specific diseases of the various organs and systems. Pathology is fundamental to the study, understanding and diagnosis of disease and therefore to successful treatment. We assume in this text that the student has already studied histology, the microscopic appearances of normal tissues, and has a basic understanding of the workings of the microscope (see Appendix 1 in Wheater’s Functional Histology, 6th edition ). Most student doctors who use this text will not become specialist pathologists, but almost all will have regular interactions with pathologists as part of their normal work and a basic understanding of pathology, even if only an ability to understand written pathology reports, is vital. For instance, what is the difference between adenocarcinoma, adenomatoid tumour and carcinoid tumour? These are three very different tumours with different behaviours requiring very different treatments and the non-pathologist receiving such a report needs to understand the difference.
As part of the diagnostic process, the pathologist uses a range of techniques. Many pathological changes can be identified by routine histological staining methods (see Appendix 1 ). If these do not allow specific identification of the disease then the pathologist has at their disposal a range of ancillary techniques, including immunohistochemistry , electron microscopy and various molecular tests. These allow precise diagnosis of many conditions that otherwise have similar clinical manifestations. For this reason, a biopsy is often carried out to obtain tissue for diagnostic purposes. This may be an incisional biopsy, i.e. removing a small sample of a lesion, or an excisional biopsy, which removes the entire lesion and may also be a curative procedure.
The pathologist’s role may also include autopsies or post-mortem examinations, some of which may be forensic, i.e. investigation of suspicious or unexplained deaths. Furthermore, molecular pathology has an increasing role in the diagnosis of diseases, guiding targeted therapies and providing prognostic information.
The other, equally important branches of pathology, such as haematology, clinical chemistry and microbiology are not covered here.
In this chapter we aim to give an overview of the basic techniques used in histopathology so that the reader will have a basis for understanding the subsequent chapters.
Histopathology works with tissue samples and cytopathology with cell samples. Both end up with the examination of biological material from a patient on a glass slide using a microscope. Before the specimen can be examined in this way a number of steps are usually needed. In the case of a histopathology specimen, after removal from a patient, the tissue undergoes the following steps:
Fixation (which may take up to 48 hours)
Macroscopic (gross) examination and selection of the parts to be submitted for microscopic examination (depending on the size of the specimen)
Processing – takes from 2 hours to overnight
Embedding in paraffin wax
Microtomy (cutting very thin slices) and placing sections onto a glass slide
Staining and cover-slipping
The finer details of how these procedures are carried out are available in technical texts and form part of the role of an essential profession allied to pathologists, known as biomedical scientists. The steps are briefly summarised in Fig. 1.1 . Following all of these processes, the final product is a slide or set of slides ready for examination by a pathologist. The number of paraffin blocks, and therefore slides, depends on the size of the specimen. A core biopsy of a breast lesion will generate one block whereas a wide local excision or lumpectomy from the breast may generate several blocks, including blocks containing the tumour, the surgical margins, any lymph nodes removed at the time of surgery and normal breast tissue adjacent to the lesion.

Sometimes several of these steps are bypassed to perform a frozen section examination. This generally happens during an operation whilst the patient is still anaesthetised. The aim of a frozen section is to guide how the operation should proceed, for example to establish whether the tumour has spread and is inoperable or to establish the resection margins of a known malignant tumour. For example, in a breast cancer resection a diagnosis of cancer might trigger the surgeon to remove lymph nodes draining the tumour site as well as the primary lesion.
Cytopathology is also a branch of pathology, but in this case the pathologist examines single cells from the patient rather than intact tissue. A specimen for cytopathology may be obtained by fine needle aspiration (FNA) when a needle is inserted into the lesion, such as a breast lump, and cells obtained by suction. Cells may also be obtained by scraping them from a surface (exfoliative cytology) , which is the technique used for cervical screening (the Pap smear). Fluids such as urine, sputum or a pleural effusion can also be obtained for cytological examination. The resulting specimen is smeared on a glass slide, fixed, stained and examined under the microscope. A major advantage of cytology examinations is that they require much less processing and preparation time than routine histopathology. However, great skill is required to interpret the appearances of individual cells without the advantage of seeing the tissue architecture. Several examples of FNA specimens are given throughout this book, for example bronchial brushings in the diagnosis of lung cancer (see Figs 12.17–12.21 ).
A list of different types of specimens that may be taken for diagnostic or treatment purposes is shown in Table 1.1 below.
| Branch of pathology | Type of biopsy | Procedure | Examples |
|---|---|---|---|
| Histopathology | Core biopsy | Sample of a lesion removed by insertion of a needle to take a core of tissue (often uses a ‘biopsy gun’) | Core biopsy of breast mass Core biopsy of kidney in glomerulonephritis |
| Incisional biopsy | Removes part of a lesion | Biopsy of skin rash | |
| Excision biopsy | Removes entire lesion | Benign breast lump | |
| Radical excision | Removes entire lesion plus additional tissues as required for cure or staging | Radical nephrectomy for malignant kidney tumour removes surrounding fat, adrenal gland and lymph nodes | |
| Cytopathology | Aspiration cytology | Aspiration of a collection of fluid through a needle – may be part of a drainage procedure | Aspiration of pleural fluid to determine if the pleural fluid contains malignant cells |
| Fine needle aspiration | Aspiration of a solid lesion using a very small diameter needle and suction | Aspiration of cells from a breast lump | |
| Exfoliative cytology | Removal of cells from the surface of tissue | Cervical screening |
Immunohistochemistry (also known as immunocytochemistry, immunostaining and immunoperoxidase) is a technique for identifying antigens in tissues or cells and uses the principles of the antigen–antibody reaction . In brief, antibodies that react with a particular antigen are linked to an enzyme that converts a colourless substrate to a coloured product. Thus, if an antibody that is specific for cytokeratin is applied to tissue containing epithelial cells, the antibody binds to those cells but not to other cell types (i.e. fibroblasts and smooth muscle cells do not contain cytokeratin). The non-bound antibody is washed off the tissue section and the non-coloured substrate is added. Under suitable conditions, the enzyme attached to bound antibody is converted to a coloured product that highlights the epithelial cells only. A summary of the processes involved in immunostaining are given in Fig. 1.2 .
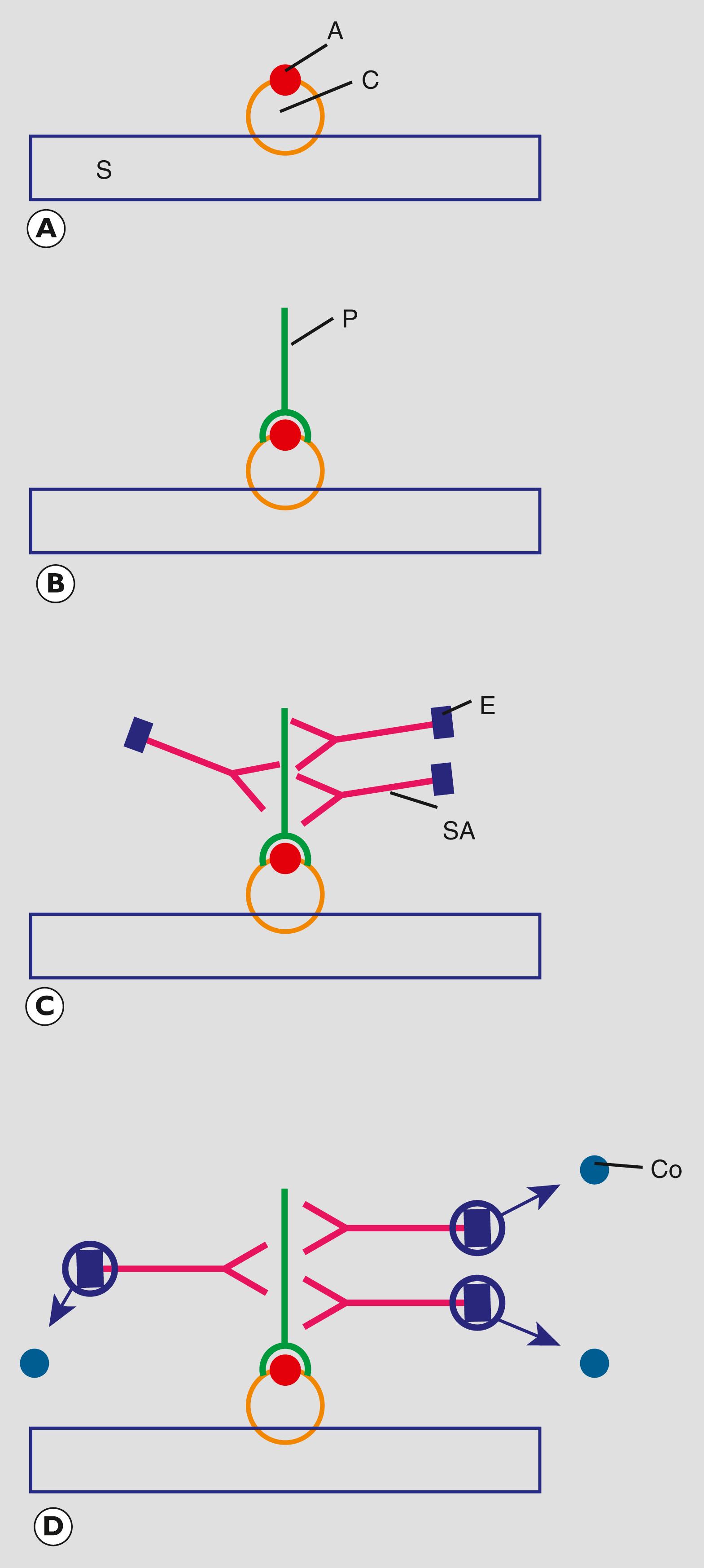
Pathologists use immunohistochemistry in everyday practice in a variety of situations including:
to identify the site of origin of a primary tumour (e.g. TTF-1 in lung cancer)
as prognostic markers (e.g. p16 positivity in oropharyngeal carcinoma)
to predict the clinical response to specific treatments (e.g. HER 2 in breast cancer)
to aid in the diagnosis of certain tumours (e.g. CD117 in gastrointestinal stromal tumours)
to demonstrate invasion (e.g. basal cell markers in prostate cancer)
as a screening test for specific mutations (e.g. mismatch repair markers for Lynch syndrome).
A antigen of interest C cell in tissue Co coloured compound E chromogen P primary antibody S slide SA secondary antibody
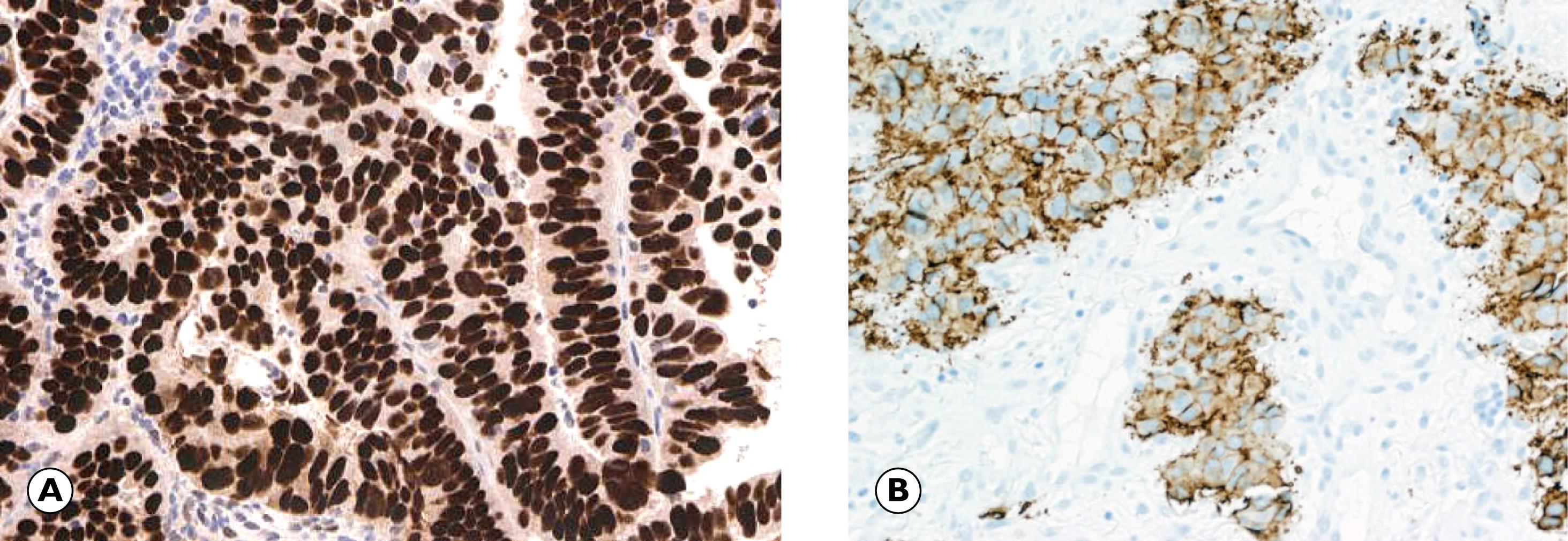
There are a vast number of antibodies used in pathology and some of these are referred to in the relevant sections of the book. To aid understanding, a summary of the common types of immunohistochemical (IHC) markers and the tissues they stain are given in Tables 1.2 and 1.3 . Interpretation of immunohistochemical stains can be challenging. A range of factors are assessed to decide whether or not the marker is positive, including which cells are staining, which part of the cell is stained, how many cells are staining and the intensity of staining (weak, moderate or strong). Patterns of staining within the cell include nuclear, membranous, cytoplasmic, perinuclear dot or a combination. Nuclear and membranous staining patterns are illustrated in Fig. 1.3 . Scoring systems exist for particular markers, such as the Allred score for the hormone receptors oestrogen receptor (ER) and progesterone receptor (PR) in breast cancer (see Fig. 18.12 ).
| Tissue type | Markers |
|---|---|
| Epithelial | CK7, CK20, CAM 5.2, EMA, AE1/3 |
| Myoepithelial | Smooth muscle actin (SMA), S100, Calponin |
| Mesenchymal | SMA, Desmin (smooth muscle), Vimentin |
| Neuroendocrine | CD56, Synaptophysin, Chromogranin |
| Melanocytic | S100, Melan A, HMB45 |
| Neural | S100, Neurofilament, GFAP |
| Germ cell | hCG, αFP, PLAP, OCT4 |
| Lymphoid | Vast number of Cluster Differentiation (CD) markers, e.g. CD20 (B cell), CD3 (T cell) |
| Marker | Organ |
|---|---|
| Thyroid transcription factor (TTF1) | Lung/ thyroid |
| Prostate specific antigen (PSA) | Prostate |
| Hepatocyte paraffin 1 (HepPar 1) | Liver (hepatocellular carcinoma) |
| CDX2 | Gastrointestinal tract |
| Wilms Tumour 1 (WT1) | Ovary (serous carcinoma), mesothelium |
| Gross Cystic Disease Fluid Protein (GCDP 15) | Breast |
| Introduction to pathology | Key points | Figure |
|---|---|---|
| Basic histology techniques | Study of tissues Variety of specimen types Processing of specimen important to create blocks and slides for pathologist review (fixation, macroscopic assessment, trimming, processing, embedding, microtomy, staining). |
Table 1.1 Fig. 1.1 |
| Cytology | Study of cells rather than tissues Includes fine needle aspirate, effusion fluid and cervical cytology |
Table 1.1 |
| Immunostaining | Identifying antigens in tissues using antigen–antibody reaction and visualising in the tissue or cell with a chromogen reaction | Fig. 1.2 |
| Immunohistochemistry interpretation | Variety of antibodies in clinical use to help diagnose cancers, identify the primary tumour site in metastatic tumours and provide prognostic and predictive information. Many factors assessed when using IHC, such as proportion and intensity of staining. | Tables 1.2, 1.3Fig. 1.3 |
| Electron microscopy | Assessment of the ultrastructure of a cell including the organelles and the nucleus. Use now limited largely to renal and muscle pathology (see Chapter 15 , Chapter 23 ) | |
| Molecular techniques PCR | Amplification of DNA targets for detection, e.g. EGFR mutation testing in lung cancer | 1.4 |
| FISH | Detection of target using DNA probe labelled with fluorescein marker and viewed under fluorescence microscope, e.g. ALK mutation in lung cancer | 1.5 |
| Next-generation sequencing (NGS) | Sequencing of large sections of DNA/RNA or even whole genome | |
| Bioinformatics | Using applied software/statistics to analyse and interpret biological data such as those generated from NGS. | |
| Digital Pathology | Slides digitally scanned and assessed on a computer screen instead of microscope. | 1.6 |
Since its invention in the 1930s, the electron microscope has provided great insights into the fine structure of cells and tissues, as well as the structure and related function of subcellular organelles such as mitochondria, the plasma membrane and the nucleus. Without electron microscopy, our knowledge of these structures would be negligible. In some cases, electron microscopy is also used for diagnostic purposes. However, many of its early applications have been replaced by immunohistochemistry, which has proven to be faster, cheaper and often more accurate. Despite this, electron microscopy remains vital for the diagnosis of many renal and skeletal muscle disorders and several examples are shown in Chapter 15, Chapter 23 respectively.
There has been an exponential growth in knowledge and understanding of molecular biology and the genetic basis of disease in the last decade. As such, the repertoire of molecular tests utilised in pathology is expanding. These tests can be used in a variety of clinical scenarios including:
diagnosis of cancer
diagnosis of inherited genetic syndromes
newborn screening
predicting response to specific therapies
guiding prognosis in certain cancers.
Molecular pathology is an essential component of personalised medicine which uses the biological features of the cancer to help decide which is the best treatment for that particular patient. Detailed discussion of the intricacies of the molecular biology of cancer are not possible here, however a summary of the main points are covered in the clinical boxes in this chapter.
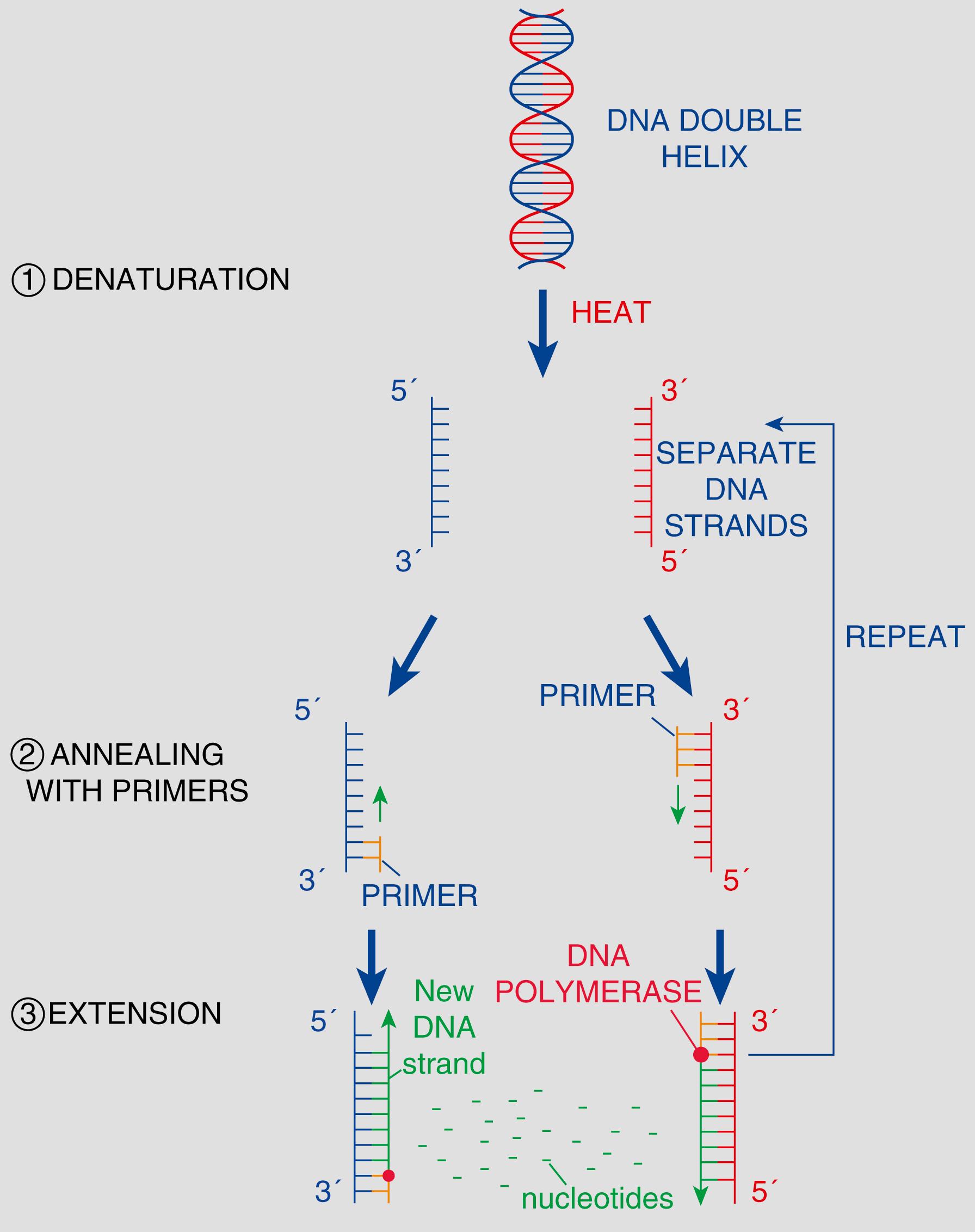
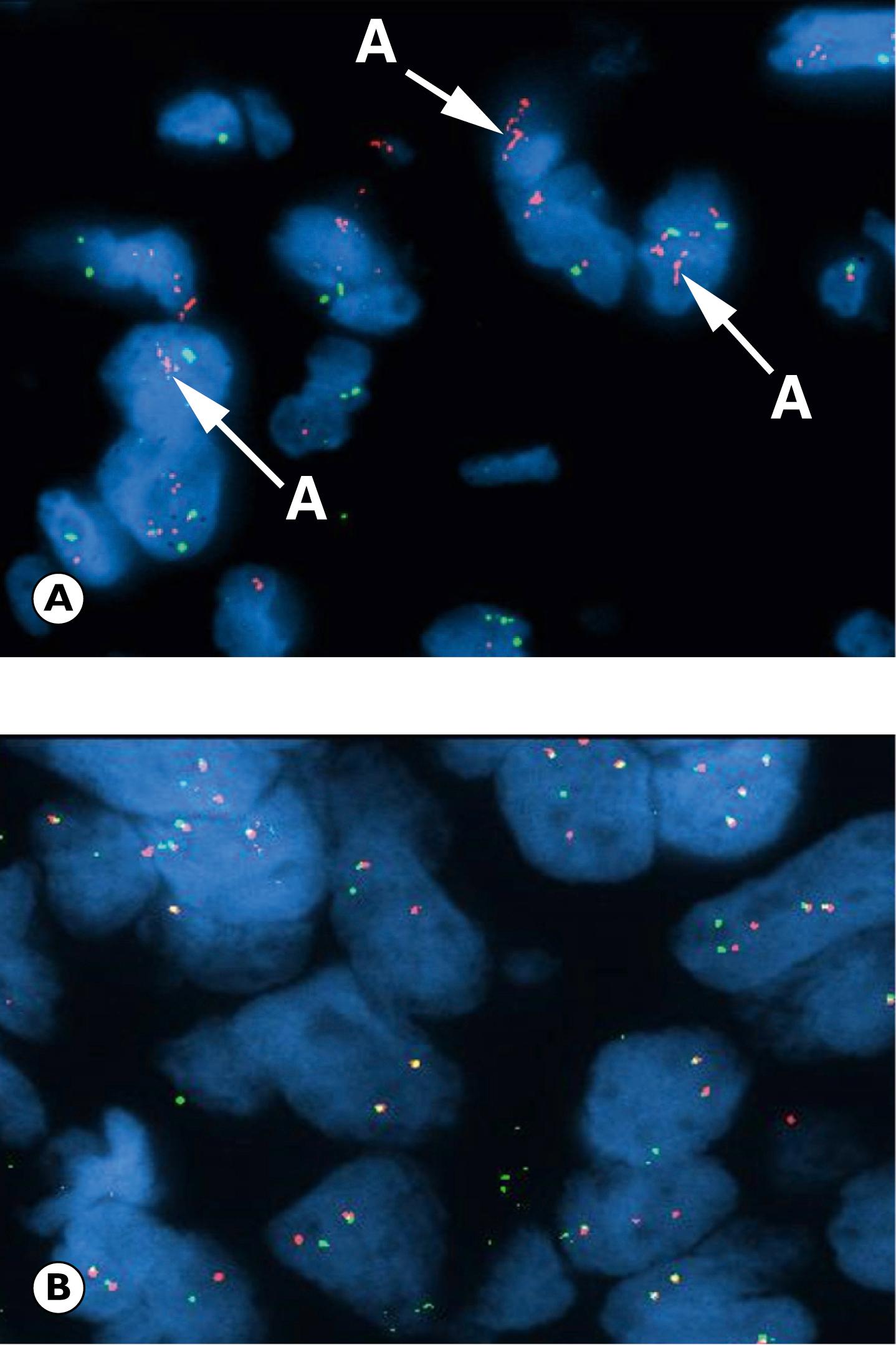
Currently, the commonest tests in use are polymerase chain reaction (PCR) and in situ hybridisation (ISH/FISH) and these are summarised in Figs 1.4 and 1.5 . Specific tests important to pathology practice are discussed in the relevant chapters.
Our knowledge of the molecular origins of cancer grows at an exponential rate, such that any account of the field is destined only to be a snapshot in time of our understanding. Nevertheless, the basic principles underlying the cellular events in the development of cancers remain. In essence, all cancers arise as a consequence of non-lethal damage to DNA (i.e. the cell survives the damage). Typically, the genes damaged are in one of three classes with roles regulating the cell cycle:
Proto-oncogenes , which code for proteins regulating cell growth and differentiation. Activation by either genetic damage (mutation) or increased expression results in the development of oncogenes.
Tumour suppressor genes , which code for proteins involved in regulating the cell cycle, repairing damaged DNA or, if the DNA is damaged beyond repair, inducing programmed cell death (apoptosis) .
MicroRNAs (miRNA) , which, unlike the other genes involved in the development of cancer, do not code for proteins but are single-stranded RNA molecules with a role in regulation of the cell cycle.
Such non-lethal damage frees the cell from the normal restraints and controls on growth and maturation leading to uncontrolled, clonal expansion of the abnormal cell.
There are four mismatch repair genes whose function is to repair DNA damage (MLH1, MSH2, MSH6, PMS2). Mutations result in inactivation of these genes and occur in two principle forms; inherited Lynch Syndrome as a result of germline mutation and sporadic (non-inherited) mutations usually as a result of epigenetic mechanisms, e.g. hypermethylation of the MLH1 gene.
Microsatellites are repetitive short sequences of DNA also known as short tandem repeats. When a mutation occurs in the tumour, these sequences of damaged DNA are not repaired and therefore accumulate. This is known as MSI-unstable or MSI-high . These sequences can be detected in tumour samples by multiplex PCR , using multiple primers at the same time to detect different sequences of DNA. There is also loss of expression of the protein product of the gene, which can be detected using immunohistochemistry.
These tests are routinely carried out in colorectal cancer specimens and endometrial cancers in many laboratories. The clinical aspects of MSI in colorectal cancer are discussed further in Ch. 13 .
A amplified DNA of ERBB2 gene target
As noted above, oncogenes encode proteins controlling proliferation, programmed cell death (apoptosis) or both. Activation of these genes, with resultant abnormal production of their protein products ( oncoproteins ), can take place in a variety of ways:
Point mutation : resulting in a permanently active gene, such as in the RAS oncogene, which encodes a signal transduction protein. Mutations in this gene are very common, occurring in around 20% of all human cancers.
Translocation : This is a common molecular abnormality in cancer cells. The result is that either the oncogene comes to lie adjacent to a gene that is constitutively expressed or relocation of the gene results in its fusion with a second gene, producing a new protein with increased activity. The former is a common event in haematological malignancies such as in Burkitt lymphoma , where the C-MYC oncogene on chromosome 8 becomes relocated adjacent to the immunoglobulin heavy chain gene on chromosome 14, which is constitutively expressed in mature B lymphocytes. In 95% of cases of chronic myeloid leukaemia , translocation results in the ABL gene on chromosome 9 fusing with the BCR gene on chromosome 22, the outcome being the BCR-ABL gene, also known as the Philadelphia chromosome . The protein product of this fused gene is constitutively active and has a role in activating proteins controlling the cell cycle.
Amplification : Here, multiple copies of a gene are formed, resulting in increased production of the oncoprotein. This is of particular note in tumour progression and hence correlates with poor prognosis. One such example is the ERBB2 ( HER2/neu ; human epidermal growth factor receptor 2) gene in breast carcinoma, where amplification of the gene correlates with poorer prognosis.
Next-generation sequencing (NGS) permits the amplification of large sections of DNA and RNA, including whole genomes, which can then be analysed using bioinformatics, a process combining computing and scientific study that puts results into clinical context. It is likely that NGS will be a routine test in molecular pathology in the coming years, providing a molecular microscope at the level of the cellular DNA and RNA within the cells.
Knowledge of the molecular origins of cancer, with an awareness of the genes and their protein products which drive cancerous cells, has provided researchers with targets for intervention in cancer treatment. This offers clinicians the opportunity to deliver treatment to patients based on the particular molecular signature of their specific tumour, so-called personalised or individualised therapy . Thus, molecular assessment of many cancers has become part of the routine pathology work-up in a growing number of cancers. Throughout this book, many examples of diagnostic molecular pathology providing insight into tumour behaviour and/or directing therapy are given. Here, we discuss the principles of these therapies using two common examples.
Those protein products which are expressed on the cell surface, in theory, provide a target for attack by appropriately designed monoclonal antibodies. This principle forms the basis of many targeted therapies in cancer. We have already mentioned the ERBB2 (HER2/neu; human epidermal growth factor receptor 2) gene in breast carcinoma, which, when amplified, is associated with a poorer prognosis. This protein has also provided an opportunity for development of targeted therapy through production of a monoclonal antibody to HER2/neu (trastuzamab; trade name Herceptin), which has proved successful in treatment of some patients with breast carcinoma, specifically those tumours shown to over-express the HER2/neu receptor. Thus, assessment of HER2 status is now commonplace in the evaluation of breast carcinoma.
In addition to monoclonal antibodies targeting cancer-associated proteins, a number of small molecules have been developed that can interfere with the function of oncoproteins. Tyrosine kinases are enzymes which phosphorylate proteins and have key roles in cell growth and differentiation (among other functions). One example where tyrosine kinase activity is increased in cancers is in relation to the BCR-ABL gene in chronic myeloid leukaemia (CML) where the protein product of the gene has tyrosine kinase activity. To counter this, inhibitors of tyrosine kinase have been developed and are now used in routine practice with considerable success, not only in treatment of CML, but also in other cancers where tyrosine kinase activity is implicated in the development of the tumour, such as gastrointestinal stromal tumours.
Until recently, most pathologists used traditional microscopy to analyse glass slides. With technological advances, pathologists are now able to examine virtual slides using a computer system without relying upon a traditional microscope. This is facilitated by digital scanning of glass slides. This has many advantages including the ability to share images with experts in other centres in real-time and in the interpretation of cell counts and immunocytochemistry. Many of these processes can be automated, thus improving turnaround time within the laboratory. A typical digital pathology set-up is shown in Fig. 1.6 .

It is well recognised that, in the same way that each individual’s appearance and personality are subtly different, their tumours also show subtle differences which can alter an individual’s prognosis. As pathologists, it is our responsibility to assess accurately each tumour at the macroscopic, microscopic and molecular level and to record all of these factors within the pathology report to permit a more personalised approach to treatment.
An example would be in the treatment of lung cancer. A tumour will now be described macroscopically, its histological subtype identified with microscopy and immunohistochemistry and then, subsequently, these tumours will be tested for EGFR and ALK mutations, as well as for the presence of PD-L1 receptors on the cell surface. This allows a patient-centred treatment approach, taking into account specific biological therapies and clinical trial opportunities.
Histological samples, following removal from a patient, undergo a number of preparation steps in the laboratory in order to produce a glass slide. Choose the correct order of steps from the list. Choose one CORRECT answer:
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here