Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Sedative hypnotic agents are an essential element in the care of pediatric patients. These drugs are used as premedicants, induction agents, and adjuvants for the maintenance of anesthesia or sedation. They are administered either as sole agents or in combination with other drugs. The multiple indications and routes of administration of these agents require an understanding of the effect of age on the pharmacokinetics and pharmacodynamics for each of these drugs.
The sedative–hypnotic effect of barbiturates is thought to occur mainly through interaction with central nervous system (CNS) gamma-aminobutyric acid (GABA) receptors, which causes a decreased rate of dissociation of the inhibitory neurotransmitter GABA. These agents are administered intravenously for induction and maintenance of anesthesia, but oral, sublingual, intranasal, rectal, and (less commonly) intramuscular routes are also used.
Barbiturates have historically been classified as long, intermediate, short, and ultrashort acting. Short-acting barbiturates (e.g., thiopental, methohexital, and pentobarbital) can be used to induce anesthesia in infants and children. These agents produce rapid induction of hypnosis with minimal relaxation or analgesia. The pharmacokinetics of short-acting barbiturates in infants, children, and adults were studied extensively by , , , , and . The rapid recovery from a single intravenous dose of thiopental or methohexital reflects redistribution from the effect site of the brain to inactive tissues ( ).
Neonates have a decreased ability to metabolize barbiturates ( ). The longer-acting barbiturates, which are in part excreted unmetabolized in the urine, would be expected to have prolonged or elevated blood levels ( ; ). Glucuronic acid conjugation of barbiturates develops rapidly and increases 30-fold during the first 3 weeks of life ( ). These agents are used less commonly now because other short-acting sedative–hypnotic agents have supplanted the short-acting barbiturates.
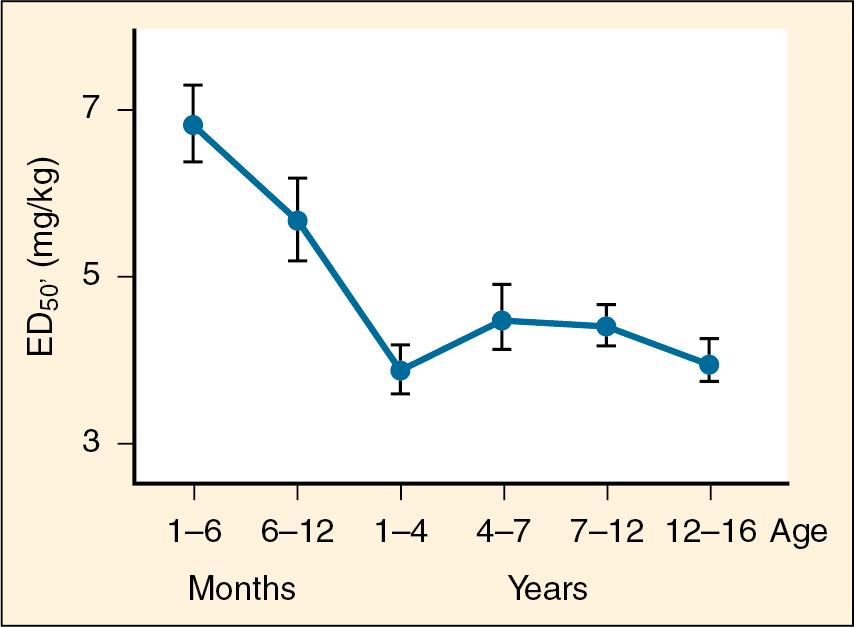
Thiopental is an ultra-short-acting barbiturate used as an intravenous induction agent in anesthesia. It is also used in critical care settings as a continuous infusion for the treatment of intracranial hypertension and status epilepticus. Hiccups, sneezing, and other respiratory irregularities are rarely seen on induction, and there is no excitement or extrapyramidal activity associated with its use. It decreases cerebrospinal fluid pressure and intraocular pressure. Awakening is quiet, occasionally interrupted by shivering, and is associated with a low incidence of nausea ( ). Porphyria, seldom encountered in the United States, is a specific contraindication to barbiturates ( ). Thiopental requirements for induction of anesthesia reveal an inverse relation with age. reported that the ED 50 of thiopental in infants is significantly greater (7 mg/kg) than that in adults (4 mg/kg) ( Fig. 9.e1 ) ( Table 9.1 ). studied the dose of thiopental to achieve satisfactory induction of ten healthy, unpremedicated neonates who were zero to 14 days old and 20 infants who were between 1 and 6 months old. In this study, the ED 50 for thiopental induction was 3.4 ± 0.2 mg/kg in neonates and 6.3 ± 0.7 mg/kg in infants aged 1 to 6 months. In an in vitro study in which the free fraction of thiopental was measured in the serum of neonates and adult volunteers, noted that neonates had a free drug fraction that was 1.5 to 2 times greater than that of adults. After a single intravenous dose, clinical effect is mainly terminated by redistribution. When repetitive or larger doses are used, redistribution becomes less effective and metabolism plays a more important role, explaining the shorter recovery times in children compared with adults.
| Indication | Route | Dose |
|---|---|---|
| Induction (neonate) | IV | 3–4 mg/kg |
| Induction (infants) | IV | 5–8 mg/kg |
| Induction (age 1–12 years) | IV | 5–6 mg/kg |
| Induction (age >12 years) | IV | 3–5 mg/kg |
| Sedation (age <6 months) | PR | 50 mg/kg |
| Sedation (age 6–12 months) | PR | 35 mg/kg |
| Sedation (age 1–5 years) | PR | 25 mg/kg |
| Seizures | IV | 2–3 mg/kg per dose (repeat prn) |
| Increased ICP | IV | 1.5–5 mg/kg per dose (repeat prn) |
Methohexital, a methylated oxybarbiturate, is more potent than thiopental by a ratio of about 3:1, is more rapidly eliminated, and produces more undesirable side effects ( ). Greater speed of recovery provides its principal indication, especially for outpatient care or for when a very brief effect is wanted, such as cardioversion or electroconvulsive therapy. compared intravenous induction of methohexital (3 mg/kg) and thiopental (7.3 mg/kg) in 41 infants aged 1 month to 1 year. In this study of short surgical procedures, recovery as measured by spontaneous eye opening after methohexital was significantly shorter than it was for thiopental. The incidence of involuntary muscular movement, hiccups, and respiratory irregularity during induction is definitely greater with methohexital than with thiopental. Methohexital can be administered via the intravenous, intramuscular, and rectal routes; however, some studies suggest that the high concentration of rectal methohexital can cause mucosal damage ( ).
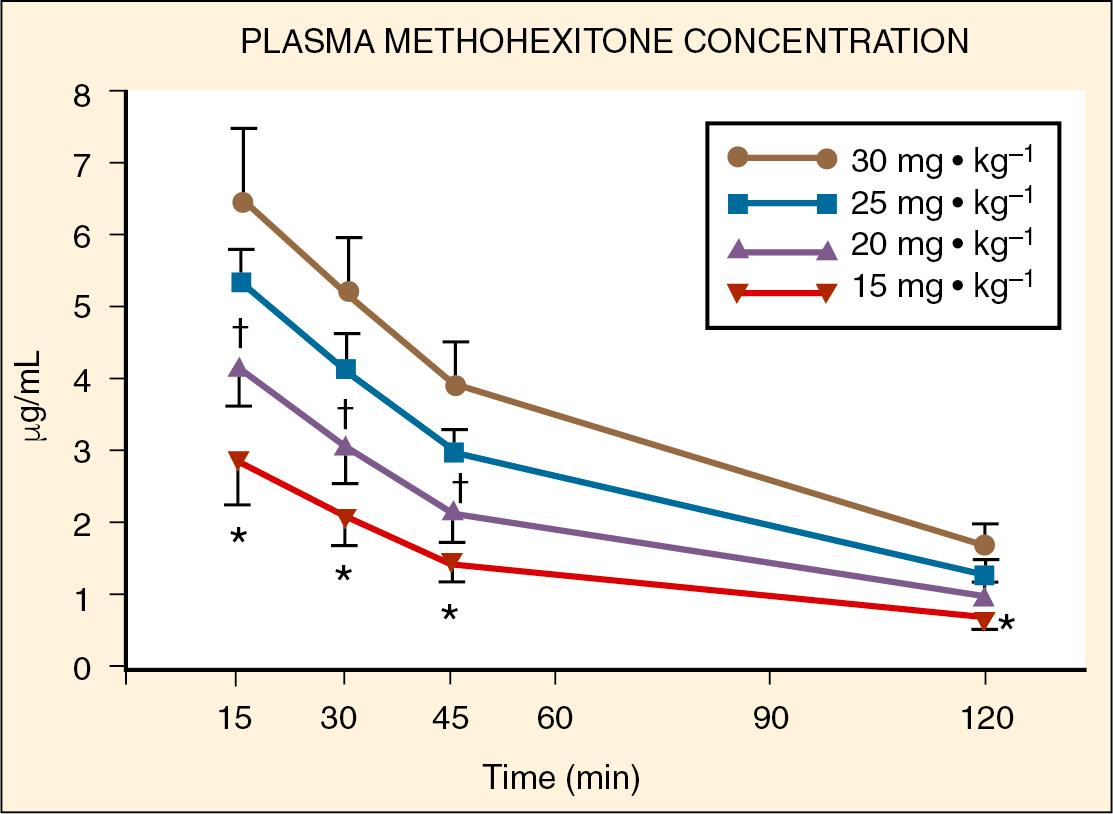
reported on the plasma concentrations of 60 children after doses of 15, 20, 25, or 30 mg/kg of rectal methohexital ( Fig. 9.e2 ). The dose of 30 mg/kg resulted in significantly higher plasma concentrations for up to 20 minutes. In a separate study of 12 patients premedicated with 25 mg/kg of 2% rectal methohexital, noted a significant increase in heart rate but no change in cardiac index, stroke volume, ejection fraction, or blood pressure using pulsed Doppler and two-dimensional echocardiography.
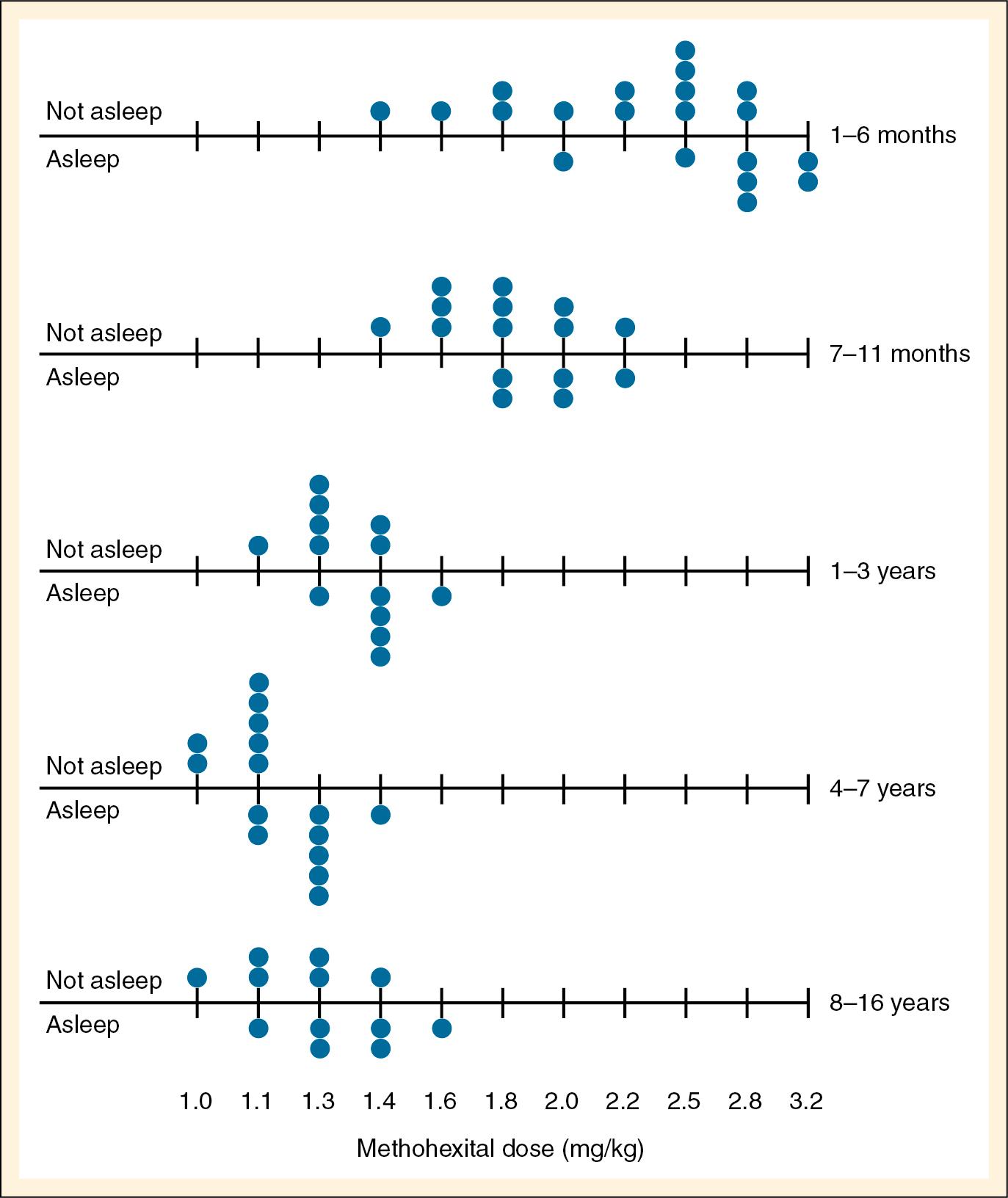
The intravenous dose for induction using methohexital dissolved in a lipid emulsion was determined by , who noted that the dose (adjusted by body weight) needed for induction in infants younger than 5 months was almost twice that for older children ( Fig. 9.e3 ). The recommended dose for intravenous use is 1 to 2 mg/kg; in children younger than 5 years, 25 to 30 mg/kg can be administered rectally ( Table 9.2 ).
| Indication | Route | Dose |
|---|---|---|
| Induction (>1 month and children) | IV | 1–2 mg/kg (1% solution) |
| Induction (>1 month and children) | IM | 6.6–10 mg/kg (5% solution) |
| Induction (>1 month and children) | PR | 25 mg/kg (1% or 10% solution) |
Pentobarbital has been used in pediatrics as a sedative drug for diagnostic imaging, with its effectiveness and relative safety demonstrated in a number of studies ( ; ; ; ; Table 9.3 ). The use of pentobarbital for sedation is waning due to increased adverse effects of vomiting, allergy, and prolonged recovery compared with the more favorable profile of propofol ( ). Pentobarbital as a single agent was associated with a longer time to arousal and time to discharge compared with propofol when the two drugs were used in a prospective trial for sedation in MRI ( ). found that propofol alone for MRI sedation resulted in faster onset and recovery time compared with a pentobarbital-based regimen consisting of midazolam and fentanyl, although efficacy was comparable. Failed or inadequate sedation using pentobarbital ranges from 1% to 16% ( ; ; ; ). Pentobarbital has also been used to control increased intracranial pressure or for treatment of refractory status epilepticus ( ). Use of pentobarbital as a continuous infusion as a backup for failed sedation has been described by ; however, subsequent studies describe a high incidence of complications related to continuous infusions ( ).
| Indication | Route | Dose |
|---|---|---|
| Sedation | IV | 1–3 mg/kg (repeat q 5–10 min prn) |
| Sedation | IM | 2–6 mg/kg (max 100 mg/dose) |
| Barbiturate coma (children and adults) | IV | 10–15 mg/kg load over 1–2 hours 1–3 mg/kg per hour maintenance for EEG burst suppression |
Pentobarbital is an oxy-analog of thiopental and is primarily metabolized in the liver with renal excretion of the metabolites, although a minority of the metabolic products are excreted in feces. The pharmacokinetics of pentobarbital has been well studied in adults, but the literature is limited in pediatrics. studied the pharmacokinetics of intravenous pentobarbital in neonates, infants, and young children (ages 3 days to 4.4 years) with congenital heart disease after open heart surgery. In an allometrically scaled model, they found total systemic clearance of 0.12 L/hr per kg, central compartment of 0.45 L/kg, and peripheral compartment of 0.98 L/kg, with an age effect on clearance for subjects younger than 12 months of age ( Table 9.4 ). The recommended dose for intravenous use is 1 to 3 mg/kg (to a maximum of 100 mg/dose) repeated every 5 to 10 minutes until the patient is asleep.
| Age (Months) | Percent Effect on Typical CL |
|---|---|
| 0.1 3 6 9 12 |
33% 73% 85% 94% 100% |
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here