Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Yeasts and yeastlike fungi are commonly encountered in human tissue specimens from patients with fungal infections. Alterations in normal anatomy, due to the presence of indwelling catheters, and alterations in normal microbiota, which occur with antibiotic use, provide opportunities for fungi to cause disease. It is not surprising then that yeasts from our own microbiota, such as Candida albicans, have become one of the most common causes of nosocomial infections, particularly for patients in intensive care units. Alterations in the normal human immune response, due to viral, pharmacologic, or genetic causes, also increase the risk for fungal infections. Examples of these include HIV-infected patients with thrush or disseminated histoplasmosis and patients receiving corticosteroids or other immunosuppressive agents who develop cryptococcal meningitis or pneumocystosis. Alternatively, some fungal infections are accidental pathogens, which is the case for the endemic dimorphic fungal pathogens. The asexual reproductive cells (e.g., conidia) of many of these fungi are small enough to remain aloft when aerosolized. When inhaled, these travel to the distal airways, where they germinate and can cause infection. Human disease caused by these fungi usually occurs after an environmental exposure. Examples include histoplasmosis after exposure to bat guano or coccidioidomycosis after exposure to aerosolized arthroconidia in the desert. Finally, some fungi enter the human body by traumatic inoculation, such as members of the Sporothrix schenckii complex, the so-called rose gardener’s disease.
The definitive identification of these organisms on histopathology is possible in some instances using morphologic criteria alone (e.g., the presence of a demonstrable capsule indicative of Cryptococcus ), whereas in other instances it is not possible to achieve this level of differentiation (e.g., non- C. glabrata species wherein only budding yeasts and pseudohyphae are seen). Regardless, a detailed description of the features present usually allows for a clinically relevant categorization, which may aid in the selection of therapy.
The morphologic features that are most important for the categorization of these microorganisms are size, shape, the uniformity versus variability in size, the type of budding (e.g., narrow-neck versus broad-based budding), the presence or absence of pseudo- or true hyphae, the presence or absence of cell-wall pigment, and other special features, such as the presence of a capsule (e.g., Cryptococcus ) or internal septations (e.g., Prototheca ). These features, when used in conjunction with the inflammatory response, afford the separation of these fungi into clinically important groups. This chapter concentrates predominantly on the most commonly encountered pathogens in this group and only briefly mentions the less commonly encountered organisms.
Yeasts and yeastlike fungi are commonly separated into three groups based on size. These are small (2 to 5 μ), medium sized (6 to 10 μ), or large (> 10 μ), and, of course, there is some overlap. The organisms in the small category that will be covered include Histoplasma capsulatum, Sporothrix schenckii, Candida glabrata, Penicillium marneffei, Malassezia spp., and Pneumocystis jiroveci . It should be noted that the endospores from a ruptured spherule of Coccidioides spp., the microforms of Blastomyces, and the misnamed “acapsular” variants of Cryptococcus neoformans also appear as small yeastlike forms but will be discussed in another section. The organisms in the medium-sized category that will be discussed include Candida species other than C. glabrata and Cryptococcus spp. There will be a brief mention of less commonly encountered fungi in this category, such as Trichosporon spp. and Blastoschizomyces capitatus. It should be noted that as endospores grow into immature spherules of Coccidioides, they, too, are in this category. The organisms in the large category that are considered here include Blastomyces dermatitidis, the mature spherules of Coccidioides spp., and Paracoccidioides brasiliensis.
The inflammatory response present in any infection depends on the antigenic characteristics of the invading microorganism and the host immune system. For patients with intact immune systems, the typical cellular immune responses include neutrophilic infiltrates in response to Candida species, necrotizing and non-necrotizing granulomas with Histoplasma, necrotizing granulomas with Coccidioides, and pyogranulomatous inflammation in response to Blastomyces, Sporothrix, and chromoblastomycosis, the latter disease being covered elsewhere. Atypical cellular immune responses may be present for patients with immunodeficiencies. The type of inflammatory response in affected patients depends on the type and severity of the immunosuppression. In patients with mild immunosuppression, the type of inflammation is usually classic, but the intensity of the response may be attenuated. In patients with severe immunosuppression, such as in patients with advanced HIV infection (i.e., AIDS), the only response may be the phagocytosis of the yeasts by macrophages and tissue necrosis, an example of which is seen in such patients with disseminated histoplasmosis.
There are a number of histochemical stains used in the assessment of tissues that contain fungal elements. Many fungi are visible in routine hematoxylin and eosin (H&E)-stained histologic sections, such as Candida species. In contrast, other fungi may be difficult to detect in H&E-stained sections, such as Histoplasma, Sporothrix, and sometimes the dermatophytes. If a fungus is suspected based on the inflammatory response or clinical history, then histochemical stains specifically designed for the detection of fungal elements should be performed.
The periodic acid-Schiff (PAS) and Grocott-Gomori methenamine silver (GMS) stains are useful for the identification of fungi in histologic section. These are nonspecific stains that highlight the cell walls of these organisms. Predigestion of the tissue with diastase or amylase is useful in conjunction with the PAS stain, especially in glycogen-rich tissues that stain intensely with PAS, because the enzyme activity will clear some of the background staining and allow for better identification of diastase/amylase-resistant fungal elements. The GMS is a nonspecific silver precipitation stain, which not only stains fungi but also stains common bacteria, mycobacteria, and Nocardia . Unfortunately, it may also precipitate on neutrophils and other tissue elements, particularly in heavy preparations, which may be misleading. Stains useful for the identification of Cryptococcus neoformans include mucicarmine, Alcian blue, and Fontana-Masson stains. The two former stains highlight the polysaccharide capsule of C. neoformans/C. gattii, whereas the latter stains melanin in the Cryptococcus cell wall. Fontana-Masson is also useful to confirm the dematiaceous nature of fungi, if not readily obvious in the H&E-stained sections.
The sensitivity of histochemical stains depends on the number and type of organism present, the choice of stain, the ability of the observer to recognize the pathogen (e.g., H. capsulatum is small and easily overlooked), the magnification used, and the time dedicated to the search. The specificity of the assay is influenced by the stain quality and the familiarity of the observer with organism morphology.
The organisms covered in the small size category include Histoplasma capsulatum, Sporothrix schenckii, Candida glabrata, Malassezia spp., Pneumocystis jiroveci, and Penicillium marneffei.
HISTOPLASMOSIS
Histoplasma capsulatum var. capsulatum is the most widely distributed of the endemic mycoses, being present in many parts of the world. Histoplasma capsulatum var. duboisii is a variant that exists in Africa, is larger, and may be confused with Blastomyces dermatitidis, but because of the paucity of infections it is not covered further. In North America, H. capsulatum is endemic to the Mississippi and Ohio River valleys but is also present in other areas. The fungus is associated with bat and bird guano, nitrogen-rich substrates that support fungal growth. This association increases the risk of disease in cave explorers. Immunosuppressed individuals, particularly those with advanced HIV disease and those receiving tumor necrosis factor inhibitors, are at risk for disseminated histoplasmosis.
Histoplasma capsulatum var. capsulatum.
There has been significant research in the genetics and physiology of H. capsulatum, as well as the pathophysiology of histoplasmosis. The detection and confirmation of genetic elements that contain unique nucleic acid sequences (i.e., signature sequences) afford the molecular detection of H. capsulatum by in situ hybridization, polymerase chain reaction (PCR), and nucleic acid sequencing. More recently, characterizations of the protein profile of the fungus are allowing for the identification of these and other fungi by mass spectrometry.
Histoplasmosis can range from mild and self-limited disease, to nodular-to-cavitary lung disease, to disseminated and possibly fatal infection. There is a predilection for these fungi to involve the adrenal glands. Meningitis, which is often basilar, may also be caused by H. capsulatum. Mild cases of histoplasmosis are often subclinical and consist of a transient influenza-like illness. Patients with progressive pulmonary infections have fever, shortness of breath, a cough that is often nonproductive, and chest pain. Other respiratory signs and symptoms, such as hemoptysis, may also occur, depending on the severity of disease. Those with disseminated disease have high fever and may develop oral ulcers, skin lesions, and constitutional symptoms. Patients with meningitis have the typical signs and symptoms of meningitis and may also develop focal neurologic deficits including those involving cranial nerves. Histoplasma capsulatum has also been associated with fibrosing mediastinitis, but definitive causation has not been proven. This disease is thought to possibly be secondary to a hyperimmune response rather than direct damage caused by the fungus. These patients may present with superior vena cava syndrome and nonspecific respiratory symptoms due to compression of airway structures.
The diagnosis of active infections is commonly achieved using a combination of culture, cyto- and histopathology, serology, and antigen testing. A urinary antigen test is available that is most sensitive in active and disseminated disease. This test is known to have some cross-reactivity in patients with blastomycosis. Serology and biopsy, in conjunction with radiologic findings, are used to diagnose fibrosing mediastinitis, as the infection with Histoplasma is largely thought to be remote.
The radiologic features depend on the type of disease. Imaging studies from patients with remote disease show calcification of hilar lymph nodes. These patients may also have numerous, small calcified nodules in the liver and spleen. Bilateral, diffuse, ill-defined nodular infiltrates may be seen in patients with disease limited to the lungs or with disseminated disease. In contrast, solid or cavitary
Endemic mycosis caused by Histoplasma capsulatum.
Endemic to the Mississippi and Ohio River valleys.
Associated with bird and bat guano.
Immunosuppressed patients are at risk for disseminated disease.
Histoplasmosis can range from a mild, self-limited disease to a nodular or cavitary lung disease to disseminated, possibly fatal disease.
Symptoms are dependent on disease presentation.
Histoplasma capsulatum can also cause a basilar meningitis.
A hyperimmune response to H. capsulatum leads to fibrosing mediastinitis, which can present with superior vena cava syndrome.
Features depend on the type of disease.
Calcification of hilar lymph nodes and calcified nodules in the liver and spleen can be seen.
In pulmonary disease, findings can range from diffuse, ill-defined, nodular infiltrates to solitary or cavitary nodules; old, fibrotic histoplasmomas may appear as “coin lesions” on chest radiographs and may be excised to exclude malignancy.
Basilar meningeal enhancement is seen in meningitis.
Immunocompetent patients with noncomplicated disease recover after an influenza-like illness.
Progressive pulmonary disease is treated with systemic antifungal therapy and supportive care.
Basilar meningitis has a more guarded prognosis, and death can occur secondary to herniation.
Endocarditis often requires surgical valve replacement in addition to antifungal therapy.
nodular disease is seen in patients with progressive pulmonary disease. When focal pulmonary disease is contained by the immunologic response, then a “coin lesion” will be seen on chest x-ray due to the histoplasmoma. Meningeal enhancement, particularly in the basilar aspect of the brain, is demonstrable in patients with meningitis. Constriction of mediastinal contents and calcifications are seen in patients with fibrosing mediastinitis.
Multiple small calcified nodules in the spleen and liver are evidence of remote infection. These are usually too hard to cut with a scalpel or microtome blade, and decalcification is necessary for histologic sectioning. These old, inactive fibrocaseous nodules that contain H. capsulatum yeast forms are common findings at postmortem examination in patients who have lived in endemic areas.
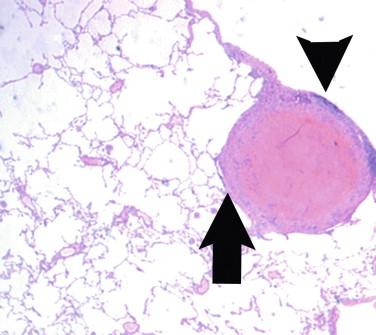
The gross anatomic findings of a larger, pulmonary histoplasmoma are similar to the other non-neoplastic “-omas” caused by infectious agents. These findings consist of a circular structure on a cut surface that often contains friable material in the center that is easily disrupted. This necrotic center is surrounded by a firmer rim of tissue. The rim may become very firm (i.e., fibrous) when longstanding disease is present, as the granulomatous inflammation is replaced by fibrosis.
Active pulmonary disease appears as a firm pneumonic process that is tan and raised compared with the surrounding pulmonary parenchyma. The nodules and cavitary lesions that may be formed are similar to those formed by tuberculosis and cannot be reliably differentiated by gross examination alone. These are firmer than the areas of pneumonia, also raised, but not so hard as to preclude sectioning. If cavitation is present, the central material is brown to tan, friable, and easily dislodged. Large vegetations are seen in patients with endocarditis. There may be enlargement of the spleen in patients with disseminated disease. The meninges are clouded to whitened, and foci of hemorrhage may be present.
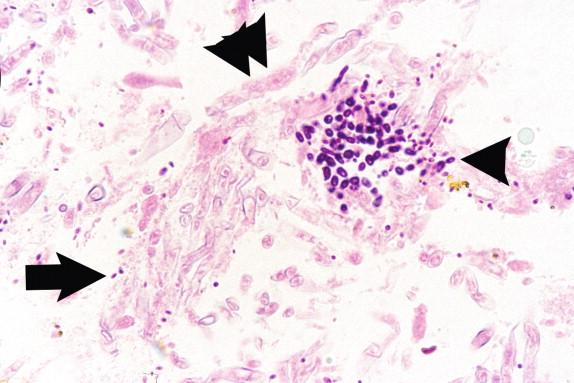
The inflammatory response to H. capsulatum in the immunocompetent patient is usually non-necrotizing or necrotizing granulomas ( Figure 26-1 ). The profoundly immunocompromised patient may only be able to respond to the infection through phagocytosis of the yeasts by macrophages and monocytes. Patients with an attenuated immune response have an inflammatory response between these two extremes, which is largely dependent on the degree and type of immunosuppression.
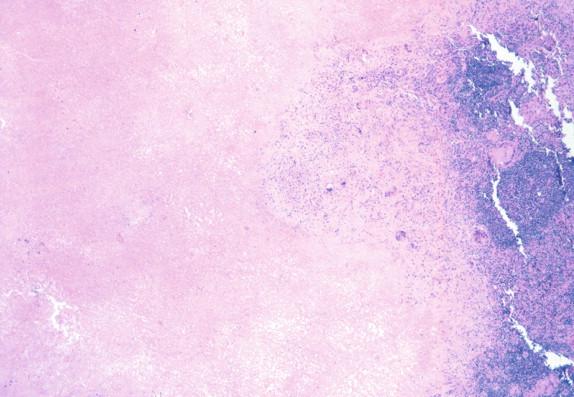
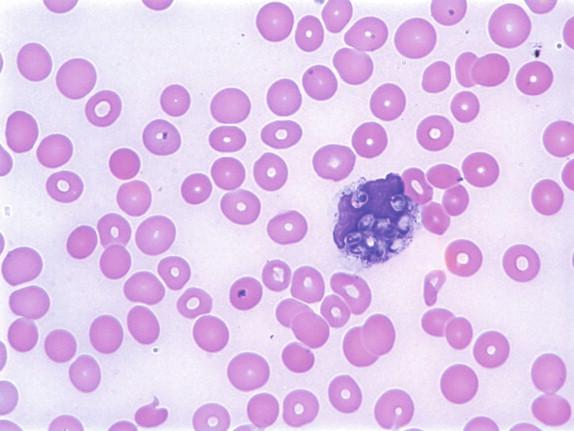
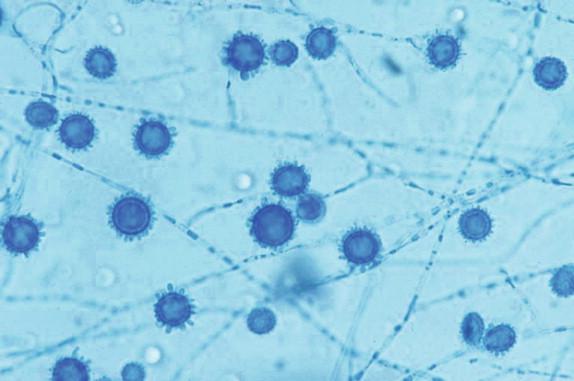
Histoplasma capsulatum is a small yeast (2 to 5 μ) that is oval to round and relatively uniform in size ( Figure 26-2 ). These yeasts may be difficult to see by H&E staining alone and are best highlighted by fungal stains. The yeasts reproduce by single, narrow-based buds. In old histoplasmomas (i.e., the residual lesion following the successful containment of the infecting Histoplasma by the immunologic response), wherein the fungus is likely nonviable as evidenced largely by negative cultures, the active granulomatous inflammation is largely replaced by fibrosis with a remaining cuff of lymphocytes ( Figure 26-3 ). The yeasts in old, inactive histoplasmomas may appear clustered in areas of necrosis, which is evidence of former phagocytosis by histiocytes, or may be more dispersed; in some instances, there is a mixture of clusters of yeasts and more dispersed yeasts.
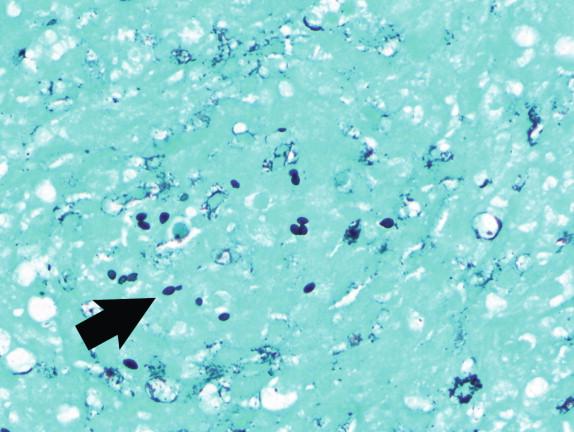
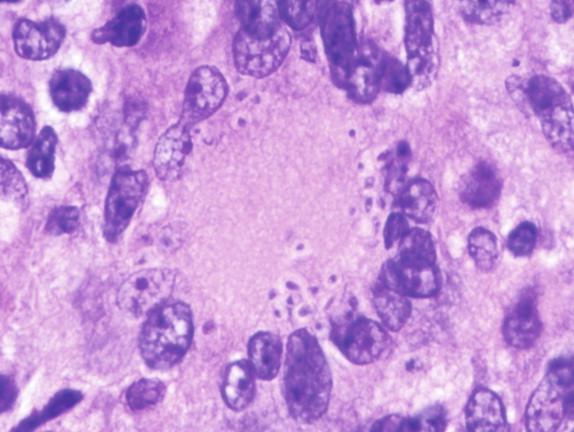
In active granulomas, the yeasts may be present in giant cells, in the areas of necrosis, or elsewhere within the granuloma. A clearing around the yeast cells may be seen in H&E-stained sections, which may give the appearance of a capsule ( Figure 26-4 ). This clearing is actually due to the poorly staining yeast wall and retraction of its cytoplasm during fixation; it is how this fungus received its species designation “ capsulatum .” In the profoundly immunocompromised host the yeasts are found distributed throughout the reticuloendothelial system and may also be seen in circulating phagocytes ( Figure 26-5 ). Atypical yeast forms may be seen in cardiac valves from patients with endocarditis and when there are overwhelming numbers of organisms in the immunocompromised host. These include pseudohyphae-like elements and enlarged or giant yeast cells ( Figure 26-6 ). Rarely, even hyphal forms have been reported in cases of endocarditis (the form typically seen in the environment), thus presenting a confounding histologic picture.
Necrotizing or non-necrotizing granulomas are seen in immunocompetent patients.
Yeast forms are 2 to 5 μ, oval to round and uniform in size, and are seen in macrophages and areas of necrosis.
Yeasts may be difficult to see on H&E and are best highlighted by fungal stains.
Not readily available.
Species-specific laboratory-developed PCR assays are available through reference laboratories. In situ hybridization assays have been developed and verified but are not commonly used. The internal transcribed spacer (ITS) and D2 regions have been identified that are useful for identification following broad-range PCR and DNA sequencing.
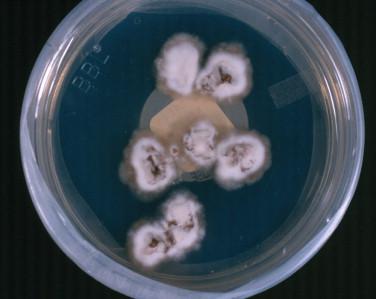
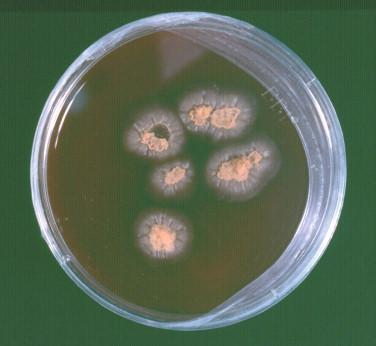
In the laboratory, H. capsulatum is a slow-growing mold that produces a white to off-white cottony colony at 25° C ( Figure 26-7 ). The organism may have a more waxy, yeastlike appearance on blood-containing media ( Figure 26-8 ). The fungus grows on cycloheximide-containing media. Microscopically, the fungus produces tuberculate macroconidia ( Figure 26-9 ) and microconidia. Confirmatory testing of molds with this type of conidiation is necessary, as saprophytic environmental mimics exist. Morphologic mimics, however, are extremely rare in clinical practice. The traditional confirmatory tests performed on the cultured isolate such as mycelial-to-yeast conversion and exoantigen testing have largely been replaced by genetic probe testing or molecular confirmation by other methods. Fungal serology, Histoplasma urinary antigen testing, and more recently laboratory-developed real-time PCR assays are other tests that may be used for the diagnosis of histoplasmosis.
The clinical differential diagnosis for pulmonary histoplasmosis includes neoplasia, tuberculosis, and other infections, particularly those caused by other fungal pathogens (e.g., cryptococcosis and coccidioidomycosis). The differential diagnosis for disseminated disease includes Hodgkin disease and other hematolymphoid malignancies, tuberculosis, enteric (i.e., typhoid) fever, and other systemic infectious diseases. The differential diagnosis for meningitis includes predominantly those agents that produce a more indolent manifestation. These include tuberculous meningitis, chronic granulomatous encephalitis, and other causes of fungal meningitis. Neoplastic disease and other fibroproliferative processes are in the differential diagnosis of fibrosing mediastinitis.
By histopathology, the yeasts of Histoplasma must be differentiated from other small yeasts, particularly Sporothrix and Candida glabrata, which also have narrow-neck budding. The clinical presentation, associated immune response, and visibility on H&E stain ( C. labrata yeasts are usually basophilic on H&E, whereas Histoplasma and Sporothrix may be difficult to detect) are all helpful features that usually allow for a morphologic diagnosis.
The prognosis for patients with histoplasmosis depends on the particular type of disease present, as well as on the presence of comorbid conditions, particularly those that compromise the immune system. Immunocompetent hosts with noncomplicated disease most commonly fully recover after an influenza-like illness and may never know that they have had histoplasmosis. Patients with more significant and progressive pulmonary disease require systemic antifungal therapy, as well as supportive care. These patients usually recover with appropriate medical attention. Treatment for basilar meningitis also requires antifungal therapy and supportive care, but the prognosis is more guarded, as death may occur due to cerebral or cerebellar herniation. Patients with endocarditis caused by this pathogen require surgical valve replacement in conjunction with antifungal therapy. Antifungal therapy is not useful in patients with fibrosing mediastinitis, and surgical intervention may be needed to release entrapped vital structures.
Amphotericin B remains an important antifungal agent for patients with severe and disseminated disease, with lipid formulations being preferred because of lower toxicity. Itraconazole is effective and commonly used in less severe disease.
SPOROTRICHOSIS
Sporotrichosis, also known as “rose gardener’s disease,” most commonly occurs after the traumatic inoculation of this organism into the skin in association with some type of plant material, such as a thorn, sphagnum moss, and hay. Disease is, therefore, sporadic. The organism spreads proximally from the site of inoculation through the lymphatics to cause local lymphadenopathy and may produce draining sinuses. Rarely, the inhalation of conidia may result in pulmonary sporotrichosis. Disseminated disease is also rare but may occur in immunocompromised patients.
Sporothrix schenckii and related Sporothrix species.
Comparative genetic studies have demonstrated that more than a single species within the Sporothrix genus can cause sporotrichosis. Assessments of the genetics of Sporothrix species have resulted in the identification of genus-level and species-specific sequences that allow for the detection of the genera and the differentiation of the species within this complex.
The majority of patients with sporotrichosis have disease that is limited to the extremities. Evidence of the primary inoculation site may or may not be present at the time of physical examination. The patient develops distal-to-proximal lymphangitic spread of the infectious agent from the affected extremity. There is often enlargement of the epitrochlear and associated lymph nodes in patients with infection of the upper limb. Draining sinuses may also form. The few patients who contract pulmonary sporotrichosis may develop serious pulmonary disease. These patients have nonspecific signs and symptoms of pneumonia, including fever, chills, and cough. Localizing signs of pulmonary consolidation may be present on physical examination, particularly in patients with progressive disease that is localized to a single focus in the lung.
The diagnosis is commonly achieved by culturing the causative agent or demonstrating the presence of yeasts in tissue with the appropriate morphology in association with a pyogranulomatous inflammatory response.
Radiologic studies are not usually needed to assess patients with sporotrichosis. In the rare instances of pulmonary sporotrichosis, pulmonary infiltrates may be seen but are nonspecific. Nodular and cavitary disease in the upper lobe of the lung that mimics tuberculosis has also been described.
Disease of the extremities, also known as rose gardener’s disease, which occurs after traumatic inoculation of Sporothrix schenckii and related Sporothrix species that is characterized by lymphatic spread, local lymphadenopathy, and draining sinuses.
Distributed worldwide but more common in the tropics.
Disease is sporadic.
Occurs in individuals that have exposure to thorny plants, sphagnum moss, or hay.
Disease is most often limited to the extremities.
Patients present with distal-to-proximal lymphangitis with associated lymphadenopathy.
Draining sinuses and evidence of the inoculation site may or may not be present.
Not usually performed for disease of the extremities.
Prognosis of sporotrichosis of the extremities is extremely good with therapy.
Itraconazole is the treatment of choice.
Large specimens are not usually submitted for a gross examination. The specimens are usually biopsies of the skin and subcutaneous tissues. These often disclose a disruption of the epidermis and redness with crusted drainage. The in situ gross features were described previously.
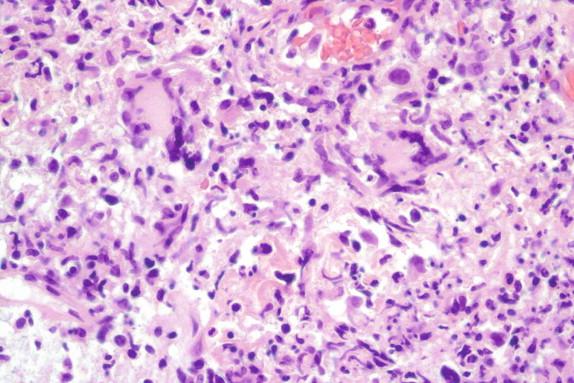
The inflammatory response in sporotrichosis is pyogranulomatous ( Figure 26-10 ). Splendore-Hoeppli material, which is an eosinophilic aggregate of proteinaceous material deposited around the fungus, may be present in histologic sections. When pyogranulomatous inflammation is seen in the skin, the foremost diagnostic possibilities include Sporothrix schenckii, chromoblastomycosis, and disseminated blastomycosis.
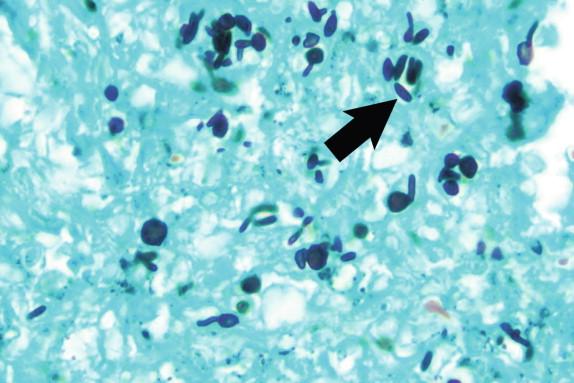
Sporothrix schenckii in tissue appears as small yeasts, which are variable in size and shape ( Figure 26-11 ). These may be round, oval, or elongated and fusiform in shape. The round and oval forms are indistinguishable from other small yeast forms. The fusiform yeasts are the classic “cigar bodies” described in patients with sporotrichosis (see Figure 26-11 , arrow). These are frequently found closer to the epidermis and may represent the earliest phase of yeast-to-mycelial conversion, but this remains speculative. When budding is found, a single, narrow-based bud is usually present.
Not available.
Laboratory-developed nucleic acid amplification assays have been described that detect S. schenckii, but these are not commonly used for diagnosis.
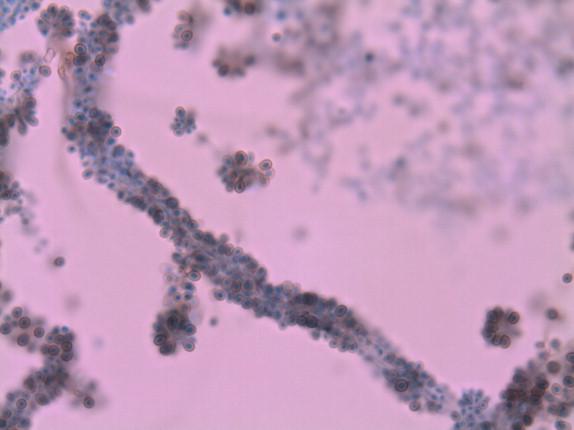
This organism grows at 25° C, initially as an off-white moist, wrinkled colony that turns darker and more filamentous with time. The yeastlike colony that is present early in the recovery of this fungus consists of elongated yeasts that may be mistaken for Ustilago. When yeast-to-mycelial conversion is complete, the microscopic morphology consists of narrow-diameter hyphae. Simple, single-celled conidia are produced that are connected to the site of conidiation by a threadlike attachment (i.e., a denticle). These conidia may be formed singly along both sides of the hyphae (i.e., a sleeve arrangement) ( Figure 26-12 ) or in a flower petal-like arrangement (i.e., a floret). In addition to the microscopic morphology, the identification of Sporothrix schenckii is accomplished by demonstrating mycelial-to-yeast conversion through incubation at 37° C.
There is a limited differential diagnosis of microorganisms that cause lymphangitic spread in the extremities. The most commonly encountered pathogens in this group are Bartonella henselae in cat scratch disease, Mycobacterium marinum, Prototheca wickerhamii, and Nocardia brasiliensis. Other nontuberculous mycobacteria, such as M. haemophilum and M. abscessus, also cause infections of the extremities. Distal-to-proximal lymphangitic spread may also occur when more common organisms, such as S. aureus, cause infections in the distal extremities. Use of histochemical stains such as Gram, GMS, and Ziehl-Neelsen allow for differentiation of these organisms.
The morphologic differential diagnosis includes other small yeasts, such as H. capsulatum and C. glabrata species. The former commonly has a more uniform appearance and, as it is likely present in the skin of a patient with disseminated disease, would appear in macrophages or an attenuated granulomatous response. Candida glabrata in the skin would likely be the result of fungemia; it, too, would be more uniform that Sporothrix, basophilic on H&E staining, and associated with a neutrophilic inflammatory response. The “cigar bodies” are absent in both histoplasmosis and candidiasis due to C. glabrata. The pyogranulomatous response generated in most patients with sporotrichosis is also a strong clue to the etiologic agent.
The prognosis for patients with noncomplicated disease of an extremity is good with the appropriate antifungal therapy. Although potassium iodine has been the traditional therapeutic agent and may still rarely be used in refractory infections, it has largely been replaced by itraconazole as the drug of choice for these infections. The prognosis is more guarded for patients with pulmonary sporotrichosis, but cures may be achieved once the diagnosis is made and the appropriate therapy is given. Lung function is permanently lost in the areas of the lung in which a well-formed nodule or cavity is produced. The prognosis of the rarely encountered disseminated sporotrichosis is poor for the immunocompromised patient. Recovery depends largely on the reversal of the immunocompromised state. If this can be achieved and the appropriate antifungal therapy is given, then the patient may survive. Amphotericin B has been used in the treatment of disseminated sporotrichosis.
INFECTIONS CAUSED BY CANDIDA GLABRATA
Candida glabrata is an opportunistic yeast that is part of the human microbiota. Like C. albicans, it may cause mucocutaneous disease that is associated with disruption of the bacterial flora by antibiotics. It is commonly found in patients with other comorbid conditions. Candida glabrata is an important cause of nosocomial fungemia, second only to C. albicans in many institutions. Fungemia is often secondary to an infection associated with an indwelling intravascular catheter but may also be due to endocarditis.
Candida glabrata.
The ITS and D2 regions have been thoroughly examined for all Candida species. Regions have been identified that allow for the molecular species-level identification of this fungus.
Apart from hepatosplenic candidiasis, C. glabrata causes the same types of infections as other Candida species. It is among the most common cause of fungemia and is often associated with indwelling intravascular catheter infections and mucocutaneous infections. Bloodstream infections are associated with fever and chills, whereas the mucocutaneous infections are associated with the same types of white plaques and white discharge that occur with infections by C. albicans and other Candida species. Other infections include urinary tract infections and wound infections. The former, if a true infection, demonstrates the same type of signs and symptoms caused by other lower urinary tract pathogens. The differentiation of true infection versus contamination may be difficult and is often based on the clinical presentation, the location of the yeasts within the tissue, and the associated host response.
Candida glabrata is an opportunistic yeast that can cause mucocutaneous disease and fungemia.
It is the second most common Candida species causing infections in most institutions.
It causes of infections in patients with altered microbiota secondary to antibiotic use or intravascular catheters.
Mucocutaneous disease presents with white plaques identical to those seen due to C. albicans.
Fungemia presents with fever and chills usually in association with an intravascular catheter.
Usually not performed.
Prognosis depends largely on the appropriateness of the antifungal therapy and comorbid conditions.
Fungemia is associated with significant mortality.
Susceptibility to antifungals is unpredictable, so susceptibility testing should be performed.
The diagnosis is most commonly achieved by culturing the microorganism from the site of infection. The identification of C. glabrata in histologic sections is highly suggested by denoting the very small size of the yeasts, the absence of pseudohyphae, the presence of a neutrophilic response, and the anatomic location. These are often seen in biopsies from the gastrointestinal tract and occasionally wounds. Although these features are useful in excluding other pathogenic fungi in the differential diagnosis, the pathologist is not usually as confident with this diagnosis as compared to that of other fungi. If the pathologist does not feel confident to make the definitive diagnosis, then the possibility of this pathogen should be mentioned in a comment, because the presence of this organism has antimicrobial implications as described later.
Radiologic studies are not usually needed to assess patients with infections caused by C. glabrata.
There are no gross anatomic findings that are distinctive for C. glabrata. Metastatic lesions from a bloodstream infection appear red due to associated hemorrhage. The vegetations in infective endocarditis caused by C. glabrata are large, similar to other causes of fungal endocarditis.
Candida glabrata is morphologically distinct from the other Candida species. This organism is spherical-to-oval and produces single, narrow-based buds ( Figure 26-13 ). They are relatively uniform in size, much smaller (2 to 5 μ) than other Candida species, and are unable to produce either true or pseudohyphae. This is the only Candida species that does not produce pseudohyphae. The site of infection, as well as the inflammatory response, is useful in differentiating C. glabrata from Histoplasma. Candida glabrata is more likely to be found in a mucosal location, whereas H. capsulatum is more likely to be found in the lungs, lymph nodes, or bone marrow. As previously noted, C. glabrata elicits a neutrophilic response, whereas H. capsulatum generates a necrotizing and non-necrotizing granulomatous response in the immunocompetent patient. Finally, C. glabrata yeasts are often basophilic on H&E, whereas Histoplasma yeasts are often eosinophilic and difficult to identify.
Small (2 to 5 μ), spherical to oval yeast forms with no true or pseudohyphae.
Associated with a neutrophilic immune response.
Not available.
An in situ hybridization assay is commercially available for the identification of this yeast from positive blood cultures. Experimentally, this assay also performs well on histologic sections, but full validation is needed for routine use. The presence of species-specific unique sequences in the D2 and ITS regions affords a molecular target for identification of this yeast. This, however, is not usually performed in routine practice.
Candida glabrata grows readily in the laboratory, producing typical glabrous white to off-white, creamy colonies. This organism grows slower than other Candida species, particularly on the traditional sheep blood agar used in bacteriology, which is a clue to the identification. The growth of C. glabrata as yeasts at 25° C readily differentiates it from H. capsulatum, which grows as a mold. In practice, the culture that contains H. capsulatum usually appears negative by visual inspection during the first few days of incubation due to the slow growth of this organism, whereas visible growth due to C. glabrata should be present in this time frame. Candida glabrata is germ tube negative and identified by its assimilation pattern, small size, and morphology on cornmeal agar. The rapid assimilation of trehalose is another method that has been described for the prompt identification of this yeast.
Other Candida species, H. capsulatum, and other small yeasts and yeastlike fungi are included in the morphologic differential diagnosis. The differential diagnosis of mucocutaneous lesions includes other Candida species, but these are larger. There are various causes of endocarditis, bloodstream infection, and urinary tract infection, which are too numerous to describe in detail. Differentiation of these other causes from C. glabrata is made predominantly by culture or by morphologic assessment of histologic sections.
The prognosis of patients with infections caused by C. glabrata is largely determined by the appropriateness of antifungal therapy provided, in conjunction with the severity of disease and comorbid conditions. Fungemia, due to this and other Candida species, is associated with a significant mortality. Removing the nidus of infection, such as an associated intravascular access catheter, is important for eliminating the fungemia. The antifungal susceptibility profile of C. glabrata is not predictable. Approximately half of the isolates have minimum inhibitory concentrations (MICs) that are characterized as susceptible, dose dependent, one fourth are susceptible, and one fourth are resistant to fluconazole. The MICs of the fluconazole-resistant organisms, although susceptible to the newer triazoles, are elevated compared to the MICs of C. albicans. Antifungal susceptibility testing, therefore, is important to guide therapy. Prior to the development of the newer triazoles, Sobel and colleagues described the benefit of topical boric acid and flucytosine for the treatment of refractory vulvovaginitis caused by C. glabrata.
INFECTIONS CAUSED BY MALASSEZIA SPECIES
Malassezia species are commensal microbiota of the skin and skin appendages. The oils on the skin provide the fungus with the requisite long-chain fatty acids needed for the survival of some Malassezia species. The common skin disease caused by the proliferation of these fungi is tinea versicolor. Rarely, this organism also causes fungemia, which is inevitably associated with an intravascular access catheter and often associated with lipid-containing hyperalimentation. Rarely, Malassezia species have been associated with a more invasive lesion, such as granulomatous dermatitis.
Malassezia furfur is the most common cause of infection. There are, however, a variety of other lipid-dependent and lipid-independent Malassezia species that may also cause infection.
Prior to the widespread use of DNA sequencing for comparative genetic analysis, the organisms in this group were usually designated as M. furfur if they were lipid dependent and M. pachydermatis if they were lipid independent. It is now known that there are a variety of species within each of these categories that are not readily differentiated by traditional means. The term complex is, therefore, a practical way to denote these groupings of physiologically and morphologically similar fungi.
Tinea versicolor is a painless discoloration of the skin. It appears lighter than the normal pigmentation in dark-skinned individuals and darker than the normal pigmentation in light-skinned individuals. Although there is discoloration, no physical discomfort is associated with this lesion. The fungemia associated with the intravascular catheter-associated infections produces fever and other symptoms of bloodstream infections, but they are considerably milder than infections caused by more virulent microorganisms.
Malassezia furfur is a lipophilic commensal of the skin.
Other lipid-dependent and lipid-independent Malassezia species can cause infections also.
Tinea versicolor caused by Malassezia species is a common skin disease.
Fungemia is associated with intravascular catheters and lipid-containing hyperalimentation.
Tinea versicolor is a painless discoloration in the skin that varies from hypopigmentation in dark-skinned individuals to hyperpigmentation in light-skinned individuals.
Fungemia presents with fever and chills, but symptoms are milder than bloodstream infections caused by other, more virulent organisms.
Usually not performed.
Tinea versicolor is easily treated with topical or oral antifungal medications, but recurrences are common.
Fungemia carries a good prognosis with removal of the intravascular catheter and antifungal therapy.
The diagnosis of tinea versicolor is usually based on the clinical appearance of the lesion alone. Occasionally, a skin scraping for direct examination for fungal elements will be performed. It is rare that a skin biopsy is required. The diagnosis of fungemia caused by M. furfur complex is achieved using standard blood culture methods. Subcultures will require long-chain fatty acid supplementation for the cultivation of lipid-dependent species.
None.
Gross specimens are usually not submitted for diseases caused by Malassezia species. If they are submitted, they consist of small, nondescript biopsies.
The morphology of this fungus on direct examination of skin scrapings or from a biopsy from a patient with tinea versicolor would consist of the so-called “spaghetti and meatballs” appearance. This is a mixture of round yeasts with hyphal elements. The yeasts are very small, in the 2- to 3-μ range, and the hyphal elements tend to be short and blunt ended. This is peculiar, because this yeast does not make hyphal elements in culture or when infections occur in other anatomic sites. The skin biopsy from this lesion would also demonstrate the basket weave–like splaying of the stratum corneum.
The morphology of the yeasts when seen as commensals in skin biopsies obtained for another reason or in the bloodstream of a patient with fungemia is distinctly different from those present in tinea versicolor. In these instances, the yeasts have a characteristic “bowling pin” appearance ( Figure 26-14 ). There is a distinctive collaret seen at the base of the bud, which is the remnant from previous budding. This collaret, although highly characteristic, is not obvious on all yeasts and must be sought. A close inspection of the bud morphology demonstrates a broad base to the bud.
Skin biopsies reveal “basket weave” splaying of the stratum corneum and a mixture of round yeasts and hyphal elements resembling spaghetti and meatballs.
Yeasts seen in fungemia or colonizing the skin will have a “bowling pin” morphology, and a collaret at the base of the bud may be observed.
Not available.
DNA sequencing is needed to differentiate the numerous Malassezia species that have been described. This, however, is rarely performed, because it is not necessary for patient management decisions.
Lipid-dependent Malassezia species, such as M. furfur, will not grow on routinely used artificial media unless they are supplemented with long-chain fatty acids. This is commonly accomplished through the addition of sterile olive oil. Tween may also be used as a supplement. When a blood culture bottle signals positive, it is not uncommon for the initial subculture to fail to grow if the subtleties of the yeast morphology are not appreciated and the medium is not supplemented with olive oil. The failure of yeasts to grow on subculture from a blood culture bottle should raise the possibility of M. furfur complex, and another subculture attempt should be made with olive oil supplementation.
Dermatophytosis as well as noninfectious disorders that affect pigmentation, such as vitiligo and pityriasis alba, are in the clinical differential diagnosis of tinea versicolor. Other causes of bloodstream infections are in the differential diagnosis of fungemia caused by M. furfur complex.
By histopathology, Malassezia is differentiated from other small yeasts by its location in tissue (e.g., superficial epidermis in commensal infections), presence of yeasts and hyphal forms in tinea versicolor, and distinctive bud scar, which is seen best under oil immersion.
The prognosis is good for tinea versicolor, and it is readily treated with repeated topical application of over-the-counter medications, such as selenium sulfide (e.g., Selsun Blue) or miconazole (i.e., Lotrimin). Alternatively, oral antifungal agents such as ketoconazole are popular with patients, given that only a single oral dose is required. Although this treatment is effective, reinfection is common, especially in warm, humid weather. The prognosis is also good for patients with fungemia caused by M. furfur complex. It does, however, often necessitate the removal of the associated catheter and use of antifungal therapy. Itraconazole and amphotericin B are antifungal agents that are effective against Malassezia species.
PNEUMOCYSTOSIS
Pneumocystis jiroveci (pronounced “yee-row-vet-zee”) is an atypical fungus that was long thought to be a parasite. This organism primarily causes pneumonia in immunocompromised patients, particularly patients with defects in the T cell–mediated immunity. The prevalence changed from a rarely encountered pathogen to a readily recognized cause of pneumonia during the AIDS epidemic, especially prior to the development of highly active antiretroviral therapy (HAART). It is now a recognized pulmonary pathogen in a variety of immunocompromised hosts, including transplant recipients and patients receiving immunosuppressive medications.
Pneumocystis jiroveci.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here