Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
A pulmonary nodule is defined as a rounded or irregular opacity measuring up to 3 cm in diameter. There are many etiologies, both benign and malignant, of lung nodules ( Table 22.1 ). Some opacities can resemble a nodule but are truly nodule mimics and can be identified as such in cross-sectional imaging. Lung nodules are extremely common, with some studies indicating that more than 50% of smokers older than 50 years of age demonstrate nodules on computed tomography (CT). There is also variation in incidence according to geography. People living in regions with endemic fungal infections such as histoplasmosis and coccidioidomycosis have a higher incidence of nodules. The prevalence of malignancy in incidentally detected nodules ranges widely but is estimated to be anywhere from 1% to 5%.
| Benign Causes (Nodule Mimics) | Malignant Causes |
|---|---|
Neoplastic
Infectious
Noninfectious inflammatory
Vascular
Congenital
Other
|
|
Assessment of various features of nodules is helpful in determining the significance and may help in identifying potential malignant nodules. The major features include attenuation, shape and contour, size and evolution, location, and enhancement.
Pulmonary nodules are primarily classified by their attenuation into solid and subsolid nodules. Subsolid pulmonary nodules are further divided into pure ground-glass and part solid nodules. Pure ground-glass nodules demonstrate hazy increased attenuation without obscuring the bronchi and vessels within the nodule. Part-solid nodules contain components of both ground-glass and soft tissue (solid) attenuation ( Fig. 22.1 ).
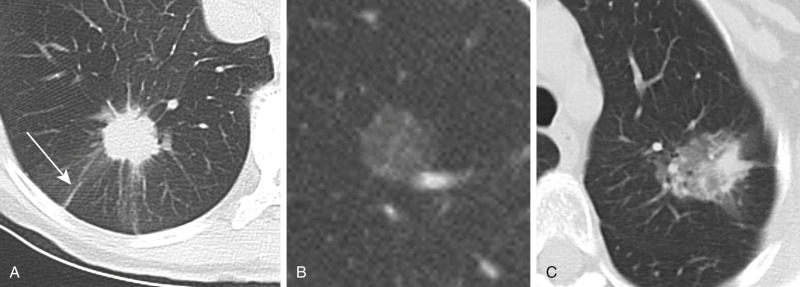
The presence of fat within a nodule can indicate a benign etiology such as a hamartoma or lipoid pneumonia. Fat appears as an area of low attenuation measuring between −50 to −150 HU ( Fig. 22.2 ). If fat is suspected but the Hounsfield units are not at least −30 HU, a reliable diagnosis of hamartoma cannot be made on CT. Magnetic resonance imaging evaluation, however, may be considered for assessment of microscopic fat in large nodules.
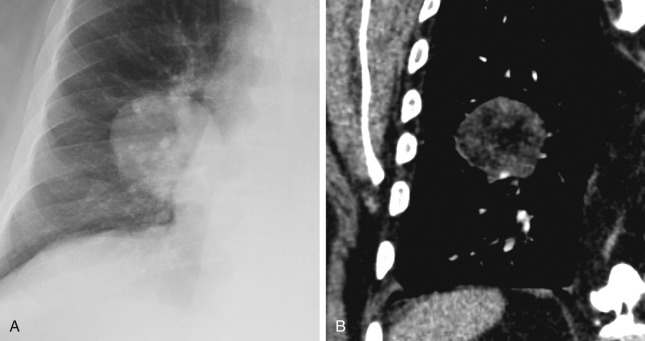
Hounsfield values greater than +200 HU indicate the presence of calcification on CT. The pattern of calcification plays an important role in evaluating whether a nodule is benign or malignant ( Fig. 22.3 ). Benign patterns include diffuse, central, laminar, and popcorn. Diffuse central and laminated calcifications are typical with granulomatous infection. Popcorn calcifications are seen in cartilaginous tumors and hamartomas. All other patterns of calcification, including punctate, eccentric, and amorphous, are considered nonspecific and are seen in up to 10% to 15% of primary lung cancers ( Fig. 22.4 ). Lung metastases from chondrosarcoma or osteosarcoma may also manifest with calcifications but should demonstrate interval growth unlike granulomas.

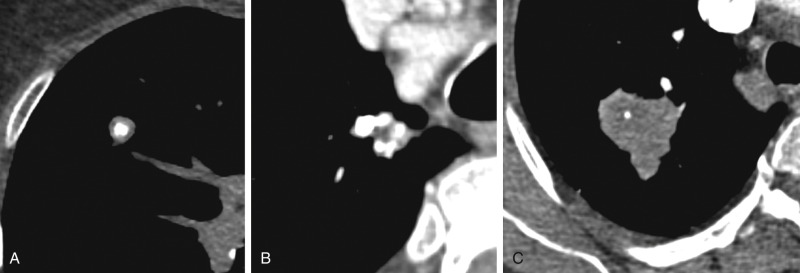
Cavitation is frequently found in infectious and inflammatory conditions (e.g., vasculitis). However, it can also be seen in malignancy, often of squamous cell histology. Cavitation is the result of necrosis, which may be related to ischemia or tissue destruction from coagulation or caseous necrosis. Cavitary nodules with smooth walls are more typical in benign lesions ( Fig. 22.5 ). Thick, nodular, and irregular walls are more common with primary and metastatic tumors, but these are not specific for benign or malignant lesions.
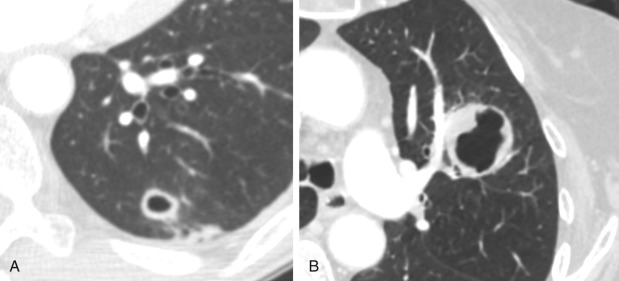
Cysts should be distinguished from cavities. In contrast to cavities, the presence of a cyst does not imply necrosis. Cysts are smooth, thin-walled structures that are likely related to dilated airspaces from obstruction of distal bronchioles. Irregular nodular thickening of a cyst wall or interval development of a solid component is worrisome for malignancy ( Fig. 22.6 ).
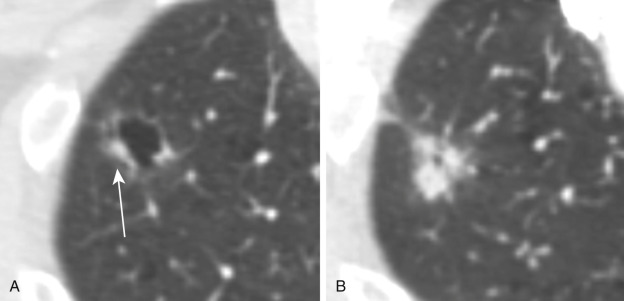
Some nodules demonstrate air bronchograms within them. This can be seen in association with certain infections and pulmonary lymphoma, as well as in lung adenocarcinoma ( Fig. 22.7 ). Other nodules demonstrate what is known as a “reverse halo sign,” which can be seen in organizing pneumonia as well as in certain infections, after radiation treatment, or in pulmonary neoplasms ( Fig. 22.8 ).
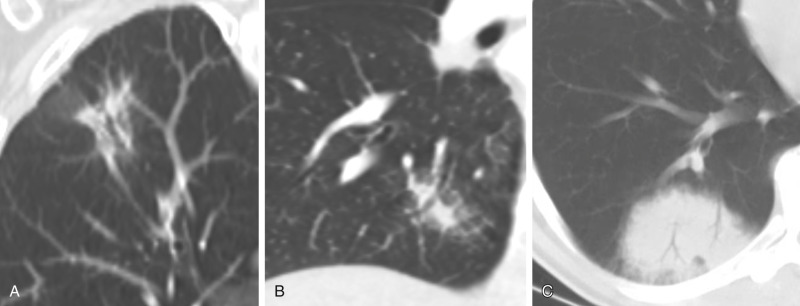
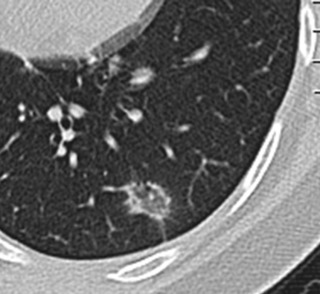
Nodules can be categorized broadly into various shapes, including triangular, linear, oval, and spherical. Triangular and linear or oval shapes are more commonly associated with benign nodules. A triangular nodule along the pleural surface is virtually pathognomonic for an intrapulmonary lymph node ( Fig. 22.9 ). Intraparenchymal nodes also have ovoid and reniform or kidney-bean shapes. A spherical shape, however, is less reassuring and needs careful analysis.
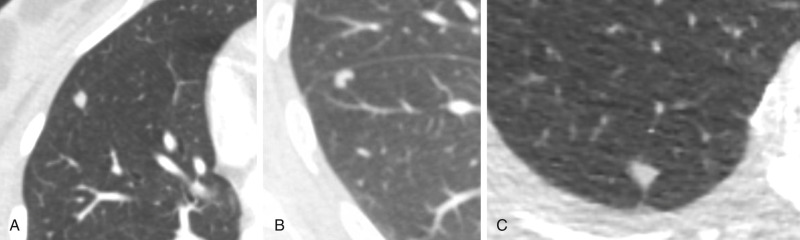
Nodule contour or margin may be described as smooth, lobulated, irregular, or spiculated ( Fig. 22.10 ). Although both benign and malignant etiologies can take any of those forms, a spiculated contour or margin has a positive predictive value for malignancy of 90% and is thought to be related to growth of malignant cells along the pulmonary interstitium. Lobulated margins may appear to be caused by differential growth rates within nodules and are seen in both malignant and benign etiologies. Although in general benign nodules tend to have a smooth contour, up to 20% of primary lung malignancies also have smooth margins.
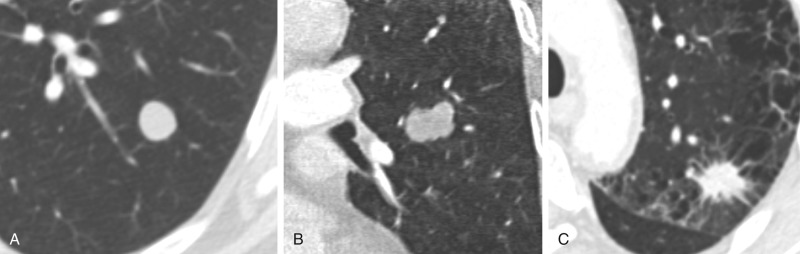
Enhancement after administration of intravenous contrast in a nodule can be helpful in differentiating benign and malignant nodules. An enhancement value of less than 15 HU is associated with 99% positive predictive value of benign lesion. Because increased blood flow is seen in many inflammatory conditions as well as in malignancy, an enhancement value greater than 15 HU is indeterminate (≈58% of cases are malignant). Decreased enhancement can also be seen in malignant nodules because of the presence of mucin and necrosis. Cavitary lesions and nodules less than 10 mm in size should not be evaluated for enhancement by CT. Patterns of enhancement such as rapid peak to enhancement and washout may also be used to predict malignant potential. Because it is not a standard practice to obtain unenhanced and contrast-enhanced chest CT, it is often not possible to determinate the degree of enhancement. Dual-energy CT can generate virtual noncontrast images and can potentially help to better characterize pulmonary nodules (see Chapter 1 ).
The location of a pulmonary nodule may also help in stratification of risk. Lung cancers tend to occur most frequently in the right upper lobe, but metastases tend to be lower lobe predominant and multiple and show greater variability in size. Perifissural and subpleural solid nodules often represent intrapulmonary lymph nodes. A central versus peripheral location may serve as a hint to type of pathology because certain cancers such as adenocarcinomas and metastases tend to be peripheral, but squamous cancers are more often found near the hila.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here