Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The recent publications of the 4th edition of the World Health Organization (WHO) Classification of Tumors of the Haematopoietic and Lymphoid Tissues and the 5th Edition of the WHO Classification of Thoracic Tumors provide the most up to date diagnostic criteria for the diagnosis of thoracic lymphoid lesions and lymphoproliferative processes. The practice of hematopathology continues to lead the way in demonstrating the links between genotypic entities, phenotypic expressions, and predictive and prognostic markers. New insights into the diagnosis and subclassification of thymic neoplasms, follicular lymphomas, the family of human herpesvirus (HHV)-8-related lymphomas, and the previously somewhat neglected category of T cell lymphomas are well established. Immunoglobulin (Ig)G4 disease has become firmly established as an entity, and lung involvement is now well characterized in the literature. At the same time, molecular profiling has expanded beyond large B cell lymphoma to develop predictive and prognostic perspective on multiple types of hematologic disease.
A constant in this area is that morphologic, immunophenotypic, and immune-architectural features still drive the essential diagnosis of reactive and neoplastic proliferations, with genotypic data providing further resolving power when overlapping results cloud the distinction between specific entities. What has changed, though, is that the clinical team is increasingly taking an initial minimally invasive approach that yields quantitatively limited biopsy samples. To not squander material on useless tests, the pathologist managing the case must develop an accurate and tight differential diagnosis and be able to effectively use a broad array of immunohistochemistry (IHC), cytogenetic, and molecular genetic assays. Additionally, as regulatory control tightens, payers may no longer reimburse if there is the perception of duplicate testing (e.g., both flow cytometry and IHC based cellular immunophenotyping).
Lymphoid lesions are commonly encountered in thoracic pathology specimens and largely fall into three of the six basic patterns of injury: acute injury, cellular infiltrates, and nodules. Diagnostic considerations depending on the main pathologic pattern are listed in Table 16.1 . There is extensive overlap between reactive and neoplastic lymphoproliferative lesions, and it is not unusual to report lesions as atypical.
|
This chapter is arranged in four main sections. The spectrum of special studies available for the work-up of hematopoietic lesions is reviewed first, followed by normal lymphoid tissue in the lung, reactive processes, and neoplastic processes.
The original work, authored by Dr. Madeleine Kraus, has been revised and updated following critical review and input from expert hematopathologist Dr. Dennis O’Malley (O’Malley Medical Consulting and Adjunct Associate Professor, Department of Hematopathology, MD Anderson Cancer Center).
Testing is performed to define the architecture of lymphoid neoplasms and their relationship to the tissue’s infrastructure (e.g., alveoli, bronchiolar epithelium, vessels), to correlate the phenotype of specific cell populations with morphologic findings, and to identify clinically significant phenotypic variants of certain lymphomas.
Well-fixed and thinly sectioned (i.e., 0.2- to 0.3-cm thick slices of excisional specimens) tissue that has been in formalin for at least 6 hours is required. Rush processing of core biopsies should be undertaken only if the protocol has been tested and is known to yield satisfactory cytomorphology and immunohistochemical results. Some immunohistochemical hematologic markers yield weak or nonreactive results in B5-, Bouin-, and Hollande-fixed tissue, even though the stain works well on formalin-fixed tissue from the same patient. Acidified zinc-formalin (AZF) preparations perform exceptionally well with most hematologic markers, and they may be the best choice for laboratories wishing to work without mercury and picrate.
Immunophenotypic analysis of tissue sections is an essential part of the evaluation of hematolymphoid proliferations in both nodal and extranodal sites. The number of antibodies available for paraffin-embedded material continues to expand, but most differential challenges can be resolved with a handful of markers. Table 16.2 lists markers that cover all of the entities described in this chapter. Boxes 16.1 and 16.2 describe panels and the manner in which they might be used. A few of the markers, including kappa and lambda light chains and Epstein–Barr virus (EBV), are best assessed with in situ hybridization (ISH).
| Marker | Description |
|---|---|
| B Cell Lineage Markers | |
| CD10 | Positive in follicle center cell non-Hodgkin lymphoma (not lineage-specific; also present on some epithelial and stromal tumors) |
| CD19 | Early B cell marker (also present on B-LBL; not present on plasma cells) |
| CD20 | Mature B cell marker (not present on B-LBL; most plasma cells negative) |
| CD23 | Activated B cells |
| CD79a | Immature and mature B cells (present on B-LBL as well as plasma cells) |
| PAX5 | Immature B cells, including lymphoblasts, mature B cells; negative in plasma cells; also positive in some neuroendocrine tumors, including small cell carcinoma |
| CD138 | Plasma cells, plasmablastic lymphoma; positive in many non-hematopoietic tumors |
| MUM1 | In the proper context, postfollicular B cells and plasma cells |
| IgD | Immunoglobulin heavy-chain delta, present in benign mantle cells, some lymphomas |
| κ, λ | Immunoglobulin light chains (cell surface expression assessed by flow; cytoplasmic expression by IHC or ISH preferably) |
| T Cell Lineage Markers | |
| CD1a | Some immature T cells (thymocytes), Langerhans cells |
| CD2 | Pan T cell marker; may also be present on natural killer cells by flow cytometry |
| CD3 | T cells |
| CD4 | Helper/suppressor T cells |
| CD5 | Preferential T cell marker (also positive in some B cell neoplasms) |
| CD7 | T cells, some natural killer cells |
| CD8 | Cytotoxic T cells |
| CD43 | Preferential T cell marker (also positive in some B cell neoplasms and granulocytic proliferations) |
| CD56 | Natural killer cells and some T cells; also positive in some neuroendocrine tumors |
| CD57 | Natural killer cells and some T cells; also expressed on some neuroendocrine tumors |
| Monocyte/Macrophage/Accessory Cell Markers | |
| CD1a | Langerhans cells, some T cells (thymocytes) |
| CD14 | Monocytes (paraffin markers available, not widely used) |
| CD15 | Granulocytes, also positive in Hodgkin lymphoma and adenocarcinoma |
| CD21 | Follicular dendritic cells, some B cells |
| CD31 | PECAM-1; marks vascular endothelium and monocytes, macrophages, and histiocytes |
| CD33 | Granulocytes (paraffin marker available, not widely used) |
| CD68 | Macrophages, monocytes (two clones; KP1 and PGM1 have slightly different specificities) |
| CD163 | Hemoglobin scavenger receptor; expressed on macrophages and histiocytes, including histiocytic malignancies |
| Langerin | Langerhans cells, both in Langerhans cell histiocytosis and Langerhans cell sarcoma |
| Miscellaneous Markers | |
| ALK1 | Positive in anaplastic large cell lymphoma, also in some inflammatory myofibroblastic tumors |
| bcl6 | Transcriptional regulator positive in germinal center B cells as well as some lymphoblasts; may be positive in the lesional cells of some T cell neoplasms |
| bcl2 | Anti-apoptosis protein positive in virtually all lymphoid proliferations except benign germinal center B cells and Burkitt lymphoma |
| Cyclin D1 | Cell cycle regulator positive in mantle cell lymphoma, myeloma, and rare cases of large B cell lymphoma |
| CD45 | Leukocyte common antigen present on lymphocytes, blasts, monocytes |
| Oct2 | Transcription factor in some B and T cells; also present in tumor cells of NLPHL |
| TdT | Terminal deoxynucleotidyl transferase, a marker of the blastic stage |
| EMA | Epithelial membrane antigen (positive in some large cell lymphomas and some plasmacytomas) |
| Ki67 | Proliferation marker that helps to identify proliferation centers in chronic lymphocytic lymphoma/small lymphocytic leukemia and is also useful in the multiparameter distinction of high-grade large B cell lymphoma and Burkitt lymphoma |
| EBER | Epstein-Barr virus (EBV) encoded RNA, EBV infection is associated with, and drives, many different lymphoproliferative processes, testing is performed via ISH |
Aberrant expression of CD5 in CD20+ B cells is the hallmark of chronic lymphocytic leukemia/small lymphocytic leukemia and mantle cell lymphoma
Aberrant expression of CD43 in CD20+ B cells is often seen in small lymphocytic leukemia and mantle cell lymphoma, occasionally in marginal zone lymphoma and seldom in other lymphomas (e.g., follicular lymphoma)
A profoundly skewed k :l ratio (e.g., 10:1 or 1:10) of kappa-positive or lambda-positive cells provides strong support for the presence of a monoclonal population of B cells (considered definitional of B cell neoplasia)
Absence of CD2, CD5, or CD7 on T cells is abnormal and is common in peripheral T cell lymphomas
Absence of both CD4 and CD8 is abnormal outside of the thymus and is commonly seen in T gamma-delta lymphomas
Presence of both CD4 and CD8 is abnormal outside of the thymus and may be seen in peripheral T cell lymphomas
Supports classification as a lymphoblastic lymphoma
Supports classification as mantle cell lymphoma (may also be seen in large B cell lymphoma and in plasma cell myeloma and should not be the sole basis for classification)
Supports classification as a lymphoma of follicle center cell origin
When it highlights a disrupted follicular dendritic cell meshwork indicative of colonization, it can assist in diagnosing marginal zone lymphomas of the lung
When other features suggest follicular colonization (bcl6–, bcl2+, CD20+ B cells inside a disrupted follicular dendritic cell meshwork), the presence of MUM1+ cells within such follicles strengthens the interpretation (i.e., an aid in excluding the possibility that the bcl2+, bcl6– cells are of B lineage and are not intrafollicular T cells)
Because benign cortical and medullary thymocytes are TdT+, great care should be taken in evaluating small biopsy specimens of hilar or midline intrathoracic masses. Should not be assessed without a corresponding keratin stain.
Small round blue cell proliferations with blastic nuclear features (fine or evenly dispersed chromatin, indistinct nucleoli, scant cytoplasm) for which the differential diagnosis may include LBL (B, T, or natural killer cell lineage), myeloid leukemia, small cell carcinoma, and Merkel cell carcinoma
TdT, CD34, PAX5, CD20, CD10, kappa and lambda, CD2, CD3, CD56, CD57, CD14, CD33, myeloperoxidase, lysozyme, cytokeratin, CK20, chromogranin, synaptophysin
Small lymphoid proliferations with a diffuse architecture in the lung for which the differential diagnosis may include CLL/SLL, MCL, FL, and MZL
CD3, CD20, CD5, CD10, LEF1, CD21, CD43, cyclin D1, cytokeratin; Ki67 and MUM1 may be informative in some cases
Small lymphoid proliferations with a nodular component in the lung for which the differential diagnosis may include MCL, FL, and MZL
CD3, CD20, CD5, CD10, CD23, CD21, CD43, bcl2, bcl6, cyclin D1, cytokeratin; IgD and MUM1 may be informative in some cases
Small lymphoid proliferations with any degree of plasmacytic differentiation in the lung for which the differential diagnosis may include CLL/SLL, LPL, MZL, WM, and plasma cell myeloma
CD3, CD20, CD5, CD10, CD23, CD21, CD138, cIg k/l, cytokeratin; IgD, EMA, and MUM1 may be informative in some cases.
Large lymphoid proliferations with or without plasmacytic differentiation for which the differential diagnosis may include LCL, plasma cell myeloma, progressed/transformed FL, and MZL
CD3, CD20, CD5, CD10, bcl2, bcl6, CD30, MYC, Ki67, MUM1; CD138, CD45, kappa, lambda, CD21 and CD23 may be helpful
High-grade nonblastic lymphomas for which the differential diagnosis may include high-grade BLCL and BL
CD3, CD20, CD10, bcl2, bcl6, CD138, Ki67, CD30, MYC, MUM1
Bimorphic small-cell and very large–cell populations (Hodgkin panel)
CD3, CD20, CD45, CD30, CD15, PAX5, MUM1; may add Oct2/BOB1, EBER, ALK1, CD21, CD2
The judicious and cost-effective use of immunostains is maximized if the pathologist puts each marker ordered to specific purpose. , In the work-up of hematolymphoid lesions of the lung, there are five principal goals:
Define the lineage. Is the abnormal cell population of T lineage? B lineage? Histiocytic? Myeloid?
Identify immunoarchitectural landmarks. Is the follicular dendritic meshwork in its normal compacted state, or are the edges frayed and the cell bodies more widely dispersed than usual? Is the mantle zone present or overrun? In the lymph nodes, are sinuses present but compressed, or are they entirely effaced? , ,
Document the aberrant phenotype that is indicative of neoplasia. Is the CD20+ cell population also positive for CD5 or CD43? Is there a restricted pattern of immunoglobulin light-chain expression (using kappa and lambda ISH)? Is there a double-positive or double-negative CD4/CD8 profile on the T cells?
Distinguish clinically different but morphologically similar entities. Is this a B lineage or a T lineage anaplastic lymphoma? Is this Burkitt lymphoma or a high-grade large cell lymphoma? Is this a cyclin D1+ large cell lymphoma or a mantle cell lymphoma? , , Does this T cell lymphoma express anaplastic lymphoma kinase (ALK) protein?
Identify the presence of pathogenic viral or other infectious agents . Is this immunoblast-rich process EBV driven (assessed with ISH for EBV)? Is HHV-6 or HHV-8 involved? Could necrotizing lymphoid infiltrates be due to herpes simplex virus (HSV)?
In working up hematologic tumors in the lung, the pathologist needs to be aware of certain pitfalls. It has long been recognized that CD138 (syndecan 1) marks both plasma cells and a range of epithelial tumors. More recently, PAX-5 has been shown in a broad range of neuroendocrine tumors, including small cell carcinoma and Merkel cell carcinoma. These studies show that, especially in the evaluation of lung tumors, no one marker should be the sole basis on which the B cell nature of a lung process is defined. Other pitfalls were reviewed recently by Yaziji and Barry.
Requesting that immunohistochemical stains be performed in a specific sequence on sequentially obtained serial sections can be of immeasurable help. Performing semiquantitative assessment of cytoplasmic light-chain expression is easier if the kappa and lambda stains are performed on two sequential sections with essentially the same population of plasma cells. Similarly, if an assessment for follicular colonization is the goal, requesting that the CD20, CD3, bcl2, bcl6, and CD21 stains be performed on sequential serial sections usually allows evaluation of these markers in the same follicle/germinal center to be made.
Flow cytometry is performed to define the lineage and cellular subsets within the lineage (e.g., B cells that are monotypic/restricted or polytypic/nonrestricted expression), to assess for aberrant and disease-defining coexpression or loss of expression of certain markers (e.g., CD5 expression on mantle cell lymphoma and loss of CD5 expression on a peripheral T cell lymphoma), or to identify and characterize maturational markers on myeloid and monocytic lineage cell populations.
Fresh (nonfixed) tissue is held in tissue culture media (e.g., RPMI) if not processed immediately. The quantity varies according to how tightly the lesional cells are held in a fibrotic or reticulin meshwork. In a cellular, nonfibrotic lymph node, as little as a 0.5-cm cube may suffice, but in sclerotic mediastinal tissue, a larger piece of tissue may be needed for full analysis. As a guide in individual cases, in general, if a touch preparation makes a richly cellular slide, then the lower limit may suffice, but if only few cells adhere to the slide, attempting flow cytometry may be a fruitless expenditure of tissue for flow cytometry when IHC would be more likely to yield the diagnosis.
An advantage of flow cytometry over tissue section immunophenotyping is that multiple markers can be evaluated simultaneously on specific small- and large-cell populations. Through this detailed multiparameter profile, many lymphoproliferative disorders can be classified more accurately. , However, flow cytometric phenotyping does not allow for visualization of the immune-architecture of the proliferation, which can be an important element of accurate classification of lymphomas, particularly in the lung, where marginal zone lymphoma (MZL) is so common. In tissue section-based immunophenotyping, the pathologist can readily identify situations in which the lesional foci have disappeared from the sections used for the stains. In flow cytometric immunophenotyping, however, the pathologist must ensure that the lesional cells are present on a cytospin made from the disaggregated cells.
With the rise of reference laboratories and the ease in sending out fresh tissue for flow cytometry, some pathologists without specialty training in hematopathology may be asked to interpret the histograms and data from such analyses. Attempting to simply “call it by the numbers” extracted from the histograms by the technician risks misrepresenting the data. The pathologist should first check to see that the cytospin contains the cells of interest and then determine from the histograms whether they have been appropriately selected (“gated”) for analysis. For instance, blasts, which are almost always very dimly CD45+, will not be present in the CD45-bright lymphocyte gate and will be missed if only the CD45-bright region data are analyzed. If the lesional cells are large, the low forward scatter CD45-bright small lymphocyte gate contains a polytypic population of B cells, and the only clue to clonality is present in a separately analyzed high forward scatter, CD45-bright large cell gate.
Flow cytometry yields continuous data that may be reported relative to the gated population (“32% of the gated CD45-dim+ cells are CD34+ blasts”) or relative to the total cellularity of the specimen (32% of the gated cells are CD34+ blasts, and the gate contains 2.4% of the total cellularity, so CD34+ blasts account for 0.6% of the total cellularity). If the pathologist is not personally extracting the numbers from the histograms, care should be taken to understand and clearly state which type of result is being presented. The difference between 32% blasts and 0.6% blasts in the example would, for instance, lead to different diagnoses.
Most reference laboratories performing flow cytometry have established panels for specific clinical scenarios (lymphoma panel, adult leukemia panel, pediatric leukemia panel, expanded T cell panel) that are tailored to efficiently identify the classic immunophenotypic profiles characteristic of common disease entities. The appropriate panel should be used to address the diagnostic question. Perhaps 10% to 15% of the time, however, a particular lymphoma or leukemia will have a variant phenotype. , Examples include a lack of CD10 in a lymphoma of follicular origin, acquisition of CD10 in hairy cell leukemia, CD5 expression in large cell lymphoma, CD19 expression in acute myeloid leukemia, apparent dim CD23 expression in mantle cell lymphoma, and a lack of CD23 expression in chronic lymphocytic leukemia. For this reason and because the immunoarchitecture is an important part of the disease definition for some lymphomas, both flow cytometry and IHC may need to be performed in some cases; if the report provides sufficient detail regarding the rationale, this may minimize the billing implications of such an approach.
Karyotype testing is indicated whenever preliminary assessment suggests a high-grade lymphoma (tumoral necrosis with a brisk mitotic rate), a blastic process, or a myeloid disorder, and for all pediatric biopsy specimens in which lymphoma or leukemic involvement is possible or likely.
Classic cytogenetic testing requires fresh (nonfixed) tissue taken sterilely (preferably in the operating room with a sterile scalpel and forceps) and transported in sterile tissue culture media to the laboratory performing the testing. Fluorescence in situ hybridization (FISH) can be performed on disaggregated cells left over from flow cytometry (if kept refrigerated) or on formalin-fixed, paraffin-embedded tissues.
Many leukemias and lymphomas have recurring chromosomal translocations that can be identified with FISH and classic cytogenetic testing. These tests are widely available at many referral laboratories, and the competition and automation have done well in bringing the price down on what used to be very expensive methodologies. Nonetheless, in an era in which patients have taken on greater responsibility for the cost of care through deductibles and coinsurance, it is important for the clinician and pathologist to work together to avoid a “shotgun” approach to test ordering while the sample is fresh. In circumstances in which there is sufficient tissue, keeping the material for cytogenetics on hold until the permanent hematoxylin and eosin (H&E) sections are reviewed will allow the pathologist to make an informed decision about whether karyotype or FISH is necessary, and if FISH is required, the most appropriate algorithm or panel must be determined.
One of the most important applications of cytogenetics is in distinguishing between morphologically similar tumors with widely different aggressiveness or treatment protocols. The distinction between small lymphocytic lymphoma and mantle cell lymphoma is an example of the first type, and the distinction between Burkitt lymphoma and high-grade large B cell lymphoma is an example of the second type. Although some translocations are characteristic of certain diseases, not all are as specific for a particular entity as once believed. For instance, c-myc –related translocations were once believed to be specific for Burkitt lymphoma, but they are now known to be present in some large B cell lymphomas and in some lymphomas that populate the new WHO category of B cell lymphoma unclassifiable with features intermediate between high-grade large B cell lymphoma and Burkitt lymphoma , (discussed later). The most common and clinically relevant karyotypic changes related to leukemias and lymphomas are enumerated in Table 16.3 .
| Disease | Abnormality | Implicated Loci | Percentage of Affected Cases |
|---|---|---|---|
| B cell lymphoblastic lymphoma |
|
|
|
| T cell lymphoblastic lymphoma |
|
|
|
| B cell chronic lymphocytic leukemia |
|
ATM |
|
| T cell prolymphocytic leukemia |
|
TCL1 | |
| Follicular lymphoma |
|
|
|
| Mantle cell lymphoma | t(11;14)(q13;q32) | bcl1-IgH | — |
| Mucosa-associated lymphoid tissue/marginal zone lymphoma |
|
|
|
| Large B cell lymphoma | t(3q27;V) | bcl6—variable partners | |
| Double-hit B-cell diffuse large cell lymphoma | (14;18)(q32;q21) with t(8;14)(q24;q32) | — | — |
| High-grade B large cell lymphoma, not otherwise classified | Complex karyotypes including c-myc translocations | See text | — |
| ALK+ diffuse large B cell lymphoma | t(2;17)(p23;q23) | ALK/clathrin | — |
| Burkitt lymphoma |
|
|
Almost always an isolated karyotypic abnormality |
| Tγ/δ hepatosplenic lymphoma | iso7q10 | — | — |
| T cell anaplastic large cell lymphoma |
|
NPM1-ALK (80% of cases) TPM3/ALK (10%–15% cases) |
— |
Molecular genetic testing is performed to document clonality in B or T lineage proliferations, to identify specific disease-defining rearrangement events (translocations), and to assess for genetic abnormalities that distinguish among chronic myeloproliferative disorders.
The use of formalin-fixed, paraffin-embedded material for polymerase chain reaction (PCR) is standard, although if peripheral blood or bone marrow is extensively involved, these tissues may be suitable alternatives. Some referral laboratories can also use fresh cells if they remain viable during transport and if the quality of DNA and RNA remains high. Some nonformalin fixatives are acceptable (e.g., Histochoice, Amresco, Solon, Ohio), but B5, Hollande, and Bouin fixatives denature the DNA and are not suitable for PCR analysis. Therefore, care should be taken during prosection to include sufficient tissue in formalin to allow for molecular studies if preliminary findings on touch preparations or frozen section suggest an hematolymphoid process.
PCR analysis may be used on formalin-fixed, paraffin-embedded material, or archived snap-frozen tissue to document clonality, define lineage, evaluate for certain translocation events, and assess for the presence of infectious agents. PCR may also be used to speciate mycobacterial organisms. Clonality is assessed by examination of areas of the immunoglobulin heavy- and light-chain loci or the T cell receptor α/β or γ loci that are rearranged during normal lymphoid development. Specially designed primer sets that flank certain gene loci are used for PCR-based evaluations for certain translocations, a helpful solution when there are no commercially available FISH probes or if tissue suitable for FISH is not available.
As “objective” as they are, molecular genetic tests do not replace the thought process of diagnosis. These test results are adjunctive data that point in one direction or another, but the final diagnosis must be made by the pathologist. The results of molecular studies must be integrated into the “big picture” painted by the clinical setting, the morphologic findings, and the immunophenotypic results. Although clonality is used to define neoplasia, lack of clonality does not prove that a lesion is reactive. The finding of an oligoclonal band is meaningless to the treating physician unless the pathologist puts the result into the context of the morphologic and clinical data. Low tumor cell numbers in Hodgkin lymphoma, lymphomatoid granulomatosis, and T cell–rich B cell lymphoma may yield nonclonal results because of the dilutional effect of reactive cells. Similarly, non–B, non–T cell malignancies, such as natural killer cell lymphomas and myeloid leukemias, yield a polyclonal smear because the lesional cells are of neither B cell nor T cell lineage.
Because of the timeliness of diagnosis and the completeness of classification of hematolymphoid proliferations, special handling of the tissue is required. Routinely handled formalin-fixed tissue is often all that is necessary, but occasionally, lack of fresh tissue for flow cytometry or cytogenetic testing can delay the diagnosis or even prevent classification. By chance or design, those affected are often the sickest patients, and delay in diagnosis introduced by a lack of tissue appropriate for ancillary testing can be frustrating to both the clinician and the pathologist. When the quantity of lesional tissue is limited, performing a frozen section with the idea of delivering a preliminary diagnosis can have the unintended consequence of delaying the case. The frozen tissue is not suitable for flow cytometry, and the embedding material may contain interfering substances that decrease the sensitivity of FISH and reduce the interpretability of immunohistochemical stains.
Under ideal circumstances, therefore, it is important to talk to the patient’s pulmonologist as well as the surgeon. If a patient with known or suspected hematologic disease is undergoing thoracoscopic or open-lung biopsy, tissue should be set aside sterilely in the operating room for culture, cytogenetic testing, or both, and the remainder should be sent for immediate evaluation of the quantitative adequacy and distribution of tissue for all appropriate ancillary studies. Touch imprints stained with H&E or Diff-Quik (Dade Behring, Newark, Delaware) can quickly discriminate among necrotizing, granulomatous, and neoplastic infiltrates, and they are fine means of addressing the difficult differential diagnosis of lymphoblastic, Burkitt, Burkitt-like, and high-grade large B cell lymphomas. Air-dried or alcohol-fixed imprints can be used for enzyme histochemistry (myeloperoxidase) as well as for FISH, and most clinical microbiology laboratories have established protocols for pneumocystis, fungal, and acid-fast stains on smears.
If the lesion is cellular and lymphoid, and if at least 1 cm of tissue is available, then half of the tissue should be sent to the flow laboratory for analysis and the other half fixed in formalin or AZF fixative. The flow laboratory should retain unstained, unfixed disaggregated cells in tissue culture media so that it can be sent for FISH analysis if initial studies warrant. If there is more than 1 cm 3 of lesional tissue, taking some for nonformalin fixation allows for assessment of nuanced cytologic detail.
Unless there is a compelling clinical question requiring resolution in the next 24 hours, frozen sections should be performed only when touch imprints do not provide sufficient information for triaging tissue. Important questions include: Of what value to the patient is this frozen section diagnosis likely to be? Am I ready to confidently diagnose or exclude lymphoma (or, for that matter, acute leukemia) from the diagnosis based on a frozen section? Can I distinguish MZL from follicular lymphoma with marginal zone differentiation or an unusual hyperplasia without the help of flow cytometry? Am I ready to make a definitive distinction among a necrotizing infection, lymphomatoid granulomatosis, and high-grade lymphoma? If not, then the frozen tissue has not advanced the patient’s care significantly, but it has rendered a portion of the specimen suboptimal for permanent section histology and IHC.
The lung contains an extensive lymphatic network that channels antigen-rich lymph centripetally toward the parenchymal, septal, hilar, and mediastinal lymph nodes. Organized lymphoid tissue in the periphery of the normal lung is limited to sparse submucosal aggregates of lymphocytes and intrapulmonary lymph nodes, but it can be more substantial centrally and along bronchiolar branch points. Inhaled particulate matter is trapped in the mucus layer of the proximal airways, and some passes across patches of specialized epithelium, where it initiates primary and secondary immune responses. Inhaled irritants stimulate a principally monocyte/macrophage response, whereas inhaled immunogens promote a lymphocytic (or lymphoplasmacytic) response. In practice, inhalational exposures are seldom purely one or the other, and so the tissue response tends to be mixed and is not infrequently masked by fibrinous exudates and actively phagocytic macrophages. The lymphoid tissue proliferates as a result of nonexogenous stimuli as well. In autoimmune conditions and immunodeficiency states, there is an intrinsic dysregulation of lymphoid proliferation, and the lymphoid hyperplasia— acquired mucosa-associated lymphoid tissue (MALT) or bronchus-associated lymphoid tissue , is less masked by acute-phase mucosal changes. Intrapulmonary lymph nodes, also a part of the pulmonary immune surveillance system, are uncommon, but when they are found, they are more often solitary, peripherally located, and located in the lower lung field. Their structure and immunoarchitectural compartments, and the diseases that affect them, are no different from those of extrapulmonary lymph nodes.
In addition, MALT has a close and specific relationship to the adjacent alveolar and bronchiolar epithelium, the lymphoepithelium , where antigen processing and presentation occur. Benign MALT has a distinctive immunoarchitecture with discrete compartments: the B cell-rich follicles, the mantle, and the T cell-rich interfollicular regions. In some cases and in some areas, a marginal zone of intermediate-sized cells with moderate to abundant amounts of cytoplasm may be interposed between the mantle and the interfollicular regions, although this marginal zone is never as well developed as it is where MALT was originally recognized in the spleen and Peyer patches ( Fig. 16.1 ). The lymphocytes in these structures shuttle between the mucosa and the circulation to provide ongoing immune surveillance and response to antigens that diffuse through the airways. Between follicles, lymphocytes range from small and resting to intermediate in size and somewhat activated. Plasma cells may be commingled, but none have Dutcher bodies. Where immunoblasts are present, their regular size, round nuclear shape, and smooth nuclear contour support a benign interpretation.
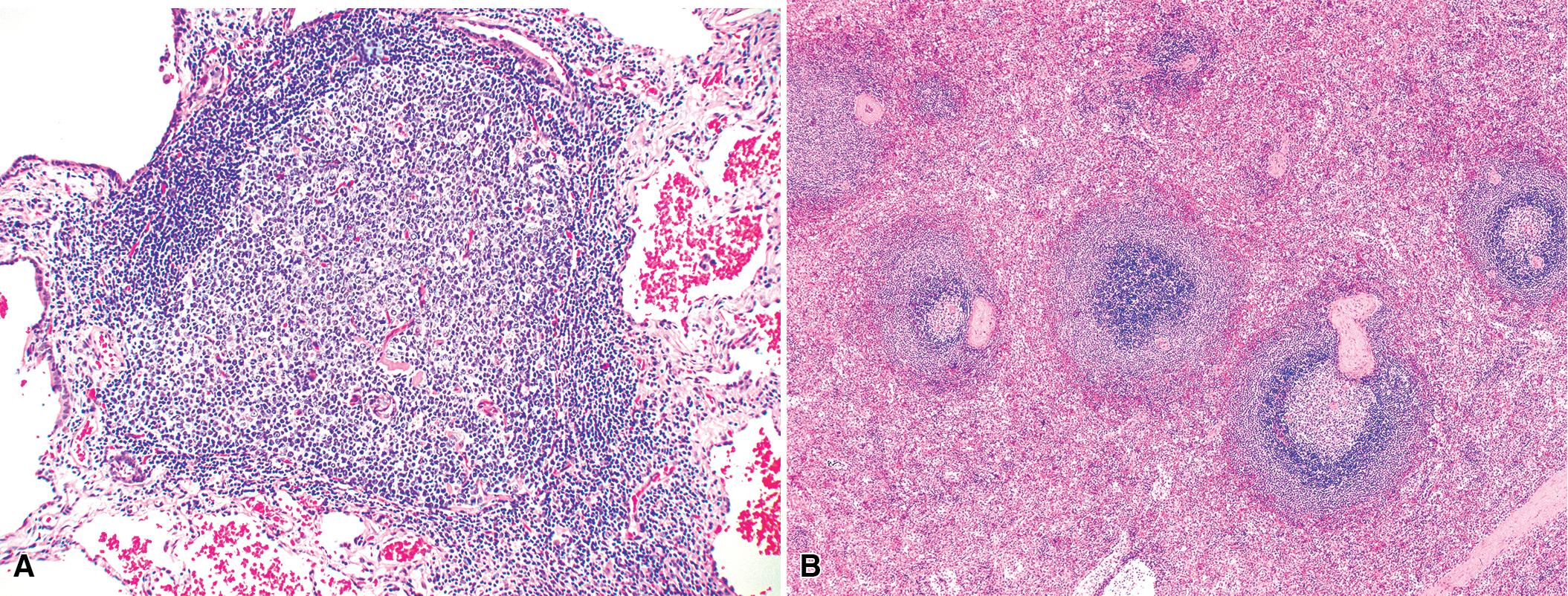
Very limited foci of lymphocytes likely have no clinical or radiologic correlate and may not require recognition with a diagnostic line in the report. However, lymphoid accumulations that either have a radiologic correlate or clearly associate with distortions of airways or air spaces should be mentioned ( Box 16.3 and Table 16.4 ).
Small discontinuous foci of lymphoid cells often near bronchiolar branch points
One or several germinal centers, with at least partial immunoglobulin D+ mantle, with light zone/dark zone polarization
Bcl6+, bcl2–, CD20+ B cells within but not between germinal centers
A tight meshwork of CD21+ follicular dendritic cells with sharp borders
Destruction of lung parenchyma, alveolar walls, and bronchiolar walls
Dutcher bodies
Follicular colonization
Monoclonal plasma cells
No aggregations of monocytoid or plasmacytoid cells within follicles
Compression of the bronchiolar lumen (which suggests follicular bronchiolitis)
Grossly identifiable nodules (which suggests non-Hodgkin lymphoma)
Significant extension into the alveolar walls (which suggests lymphoid interstitial pneumonia)
| Nodular Lymphoid Hyperplasia | Lymphoid Interstitial Pneumonia | Marginal Zone Lymphoma | |
|---|---|---|---|
| Clinical features | Adults > children ± altered immune state | Common in children, association with immunodeficiency | Adults > children, association with immunodeficiency |
| Location of infiltrates |
|
|
Masslike or patternless, typically unifocal destructive lymphoepithelial lesions are present |
| Architecture |
|
|
|
| Cellular composition |
|
|
|
| Immunophenotype | Polytypic | Polytypic |
|
Sustained hyperplasia of the MALT of the lung occurs in the setting of autoimmune or altered immune conditions (e.g., acquired immunodeficiency, connective tissue disorders, and congenital immunodeficiency). Biopsy is performed to distinguish among superimposed infection, treatment-related pneumotoxicity, and lymphoma.
The three main patterns of lymphoid hyperplasia: follicular bronchiolitis (FB), nodular lymphoid hyperplasia (NLH), and diffuse lymphoid hyperplasia (lymphoid interstitial pneumonia [LIP]), may be seen in isolation or may coexist in the same specimen. , Much of the work in identifying specific benign and malignant lymphoid proliferations requires the diagnostician to recognize the intactness or the patterned disruption of the morphologic and immunologic landmarks of normal MALT ( Fig. 16.2 ; see also Box 16.3 and Table 16.4 ).
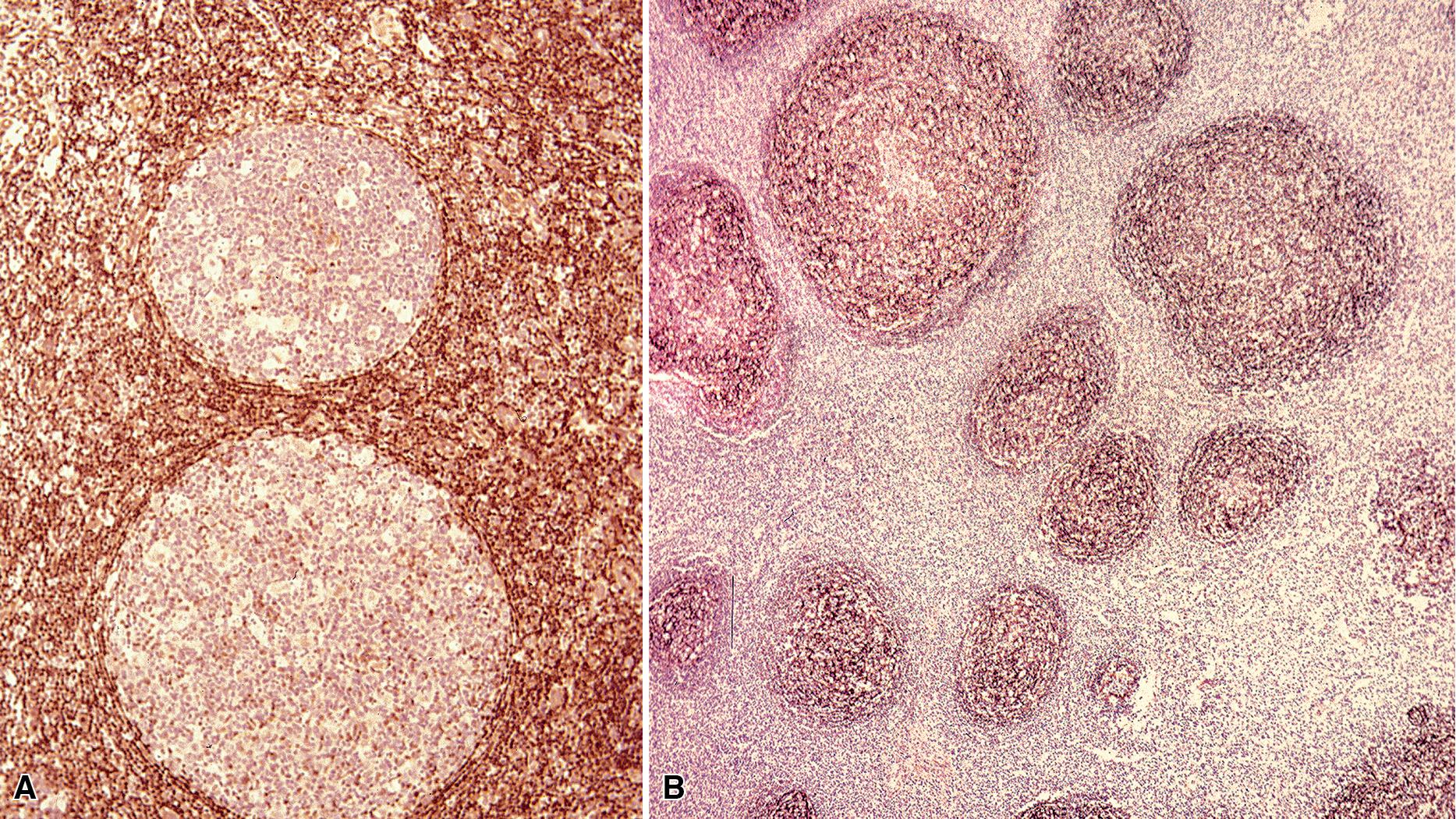
FB is slightly more common in males than in females, involves the lungs bilaterally, and produces a centrilobular reticulonodular pattern of involvement on radiologic studies. , In exceptional cases, opacities up to 1 cm in size may be seen. FB is most commonly seen in patients with congenital or acquired immunodeficiency (HIV, common variable immunodeficiency, or IgA deficiency), collagen vascular disease (especially rheumatoid arthritis), or chronic obstructive pulmonary disease, and it may also be seen at the periphery of localized infectious processes of the lung.
Microscopically, the key feature is multiple foci of eccentric peribronchiolar accumulations of lymphoid tissue that distort and may narrow the bronchiolar lumen ( Box 16.4 and Fig. 16.3 ). , Confluent nodule-forming infiltrates larger than 1 cm should raise concerns about lymphoma. The structure of benign MALT is preserved with bcl2- germinal centers that are crisply demarcated by an IgD–positive mantle zone and a polymorphous lymphocytic and histiocytic component at the interface with normal lung parenchyma. The proliferation may compress the airways, leading to postobstructive bronchiectasis in distal parenchyma. There is no interstitial involvement in the alveolar walls away from the bronchioles, and the air spaces are uninvolved ( Fig. 16.4 ), which is a feature that distinguishes FB from LIP. , ,
Germinal centers with crisp mantles located beside bronchioles along the pathway of bronchovascular bundles
At least partial immunoglobulin D+ mantle
Bcl2 negativity within follicles
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here