Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Lancefield group B β-hemolytic streptococci were first recorded as a cause of human infection in 1938, when Fry described three patients with fatal puerperal sepsis. Sporadic cases were reported until the 1970s, when a dramatic increase in the incidence of septicemia and meningitis in neonates caused by group B streptococci (GBS) was documented from geographically diverse regions. Emergence of GBS infections in neonates was accompanied by an increasing number of these infections in pregnant women and nonpregnant adults. In pregnant women, infection commonly manifested as localized uterine infection or chorioamnionitis, often with bacteremia, and had an almost uniformly good outcome with antimicrobial therapy. The incidence of perinatal infection associated with GBS remained stable through the early 1990s. Case-fatality rates had declined by then but remained substantial compared with case-fatality rates reported for other invasive bacterial infections in infants. The implementation of the consensus guidelines published in 2002 that are designed to prevent early-onset disease in neonates through universal antenatal culture-based screening at 35 to 37 weeks of gestation, and accompanying intrapartum antibiotic prophylaxis was associated with a substantial decline in the incidence of early-onset neonatal infection by 2008 and has reached a plateau since then.
In recent years, the genomes of several GBS types have been sequenced, opening new avenues for the identification of novel potential vaccine targets. The discovery that surface-associated pili are widely distributed among GBS and that a vaccine based on combinations of the three pilus-island variants protects mice against lethal challenge with a wide variety of GBS strains paves the way for the design of pilus-based and perhaps other putative surface protein vaccines for testing in humans.
Testing of glycoconjugate GBS candidate vaccines in healthy adults and in pregnant women is ongoing, offering promise that immunization to prevent maternal and infant invasive GBS disease could become a reality.
Streptococcus agalactiae is the species designation for streptococci belonging to Lancefield group B. This bacterium is a facultative gram-positive diplococcus with an ultrastructure similar to that of other gram-positive cocci. Before Lancefield’s classification of hemolytic streptococci in 1933, this microorganism was identified by its characteristic colonial morphology, its narrow zone of β-hemolysis surrounding colonies on blood agar plates, and its double zone of hemolysis that appeared when plates were refrigerated an additional 18 hours beyond the initial incubation. Occasional strains (1%-4%) are α-hemolytic or nonhemolytic. GBS are readily cultivated in various bacteriologic media. Isolation from respiratory, genital, and gastrointestinal tracts can be enhanced by use of selective enrichment broth containing antimicrobial agents that inhibit growth of other bacterial species indigenous to these sites.
Colonies of GBS grown on sheep blood agar are 3 to 4 mm in diameter, produce a narrow zone of β-hemolysis, are gray-white, and are flat and mucoid. β-Hemolysis for some strains is apparent only when colonies are removed from the agar.
Tests for presumptive identification include bacitracin and trimethoprim-sulfamethoxazole disk susceptibility testing (92%-98% of strains are resistant), hydrolysis of sodium hippurate broth (99% of strains are positive), hydrolysis of bile esculin agar (99%-100% of strains fail to react), pigment production during anaerobic growth on certain media (96%-98% of strains produce an orange pigment), and CAMP (Christie-Atkins-Munch-Petersen) testing (98%-100% of strains are CAMP positive). The CAMP factor is a thermostable extracellular protein that, in the presence of the β toxin of Staphylococcus aureus, produces synergistic hemolysis when grown on sheep blood agar. Hippurate hydrolysis is an accurate method for presumptive identification of GBS, but the incubation time required limits its usefulness. Definitive identification of GBS requires detection of the group B antigen common to all strains. Lancefield’s original method required acid treatment of broth-grown cells to extract the group B antigen from the cell wall. Supernatants brought to neutral pH were mixed with hyperimmune rabbit antiserum prepared by immunization with the group B–variant strain, and precipitins in capillary tubes were recorded. Less time-consuming techniques are now used. Conventional means for presumptive identification of isolates subcultured to blood-agar plates include use of the CAMP test or latex agglutination with GBS antisera. Chromogenic agars that undergo color change in the presence of β-hemolytic colonies of GBS have become available. Most of these do not detect the small percentage of strains that are nonhemolytic. In addition, more rapid techniques have been developed for identifying GBS directly from enrichment broth or after subculture. These include DNA probes and nucleic acid amplification tests (NAAT), such as polymerase chain reaction (PCR). The sensitivity of NAAT, when an enrichment step is included, ranges from 93% to 100%.
Group B streptococci were known to cause bovine mastitis before they were appreciated as pathogenic in humans. Modern veterinary practices have largely controlled epidemics of bovine mastitis, but sporadic cases still occur. Substantial biochemical, serologic, and molecular differences exist between human and bovine isolates. Among typeable bovine strains, patterns of distribution distinct from the patterns of human isolates are noted. Other distinguishing characteristics for bovine strains include their unique fermentation reactions, decreased frequency of pigment production, and susceptibility to bacitracin. Protein X, rarely found in human strains, is commonly present in pathogenic bovine isolates.
The relationship between GBS strains of human and bovine origin has been queried for years. There is no compelling evidence that cattle serve as a reservoir for human disease, and transmission from cows to humans is exceedingly rare. In addition, during the decades when GBS has been a dominant human pathogen, the risk of exposure through direct contact with dairy cattle or ingestion of unpasteurized milk has been unlikely. Application of molecular techniques to strains from bovine sources and those infecting human neonates supports the assertion that these lineages are unrelated. Phylogenetic lineage determination does indicate, however, that some clonal complexes of invasive or colonizing strains in humans are related to ancestral lineages of bovine GBS.
Lancefield defined two cell wall carbohydrate antigens by using hydrochloric acid–extracted cell supernatants and hyperimmune rabbit antisera: the group B–specific, or “C,” substance common to all strains and the type-specific, or “S,” substance that allowed classification into types—initially types I, II, and III. Strains designated as type I were later shown to have cross-reactive and antigenically distinct polysaccharides designated type Ia and type Ib. GBS historically designated type Ic possessed type Ia capsular polysaccharide (CPS) and a protein antigen common to type Ib, most type II, and rare type III strains. This protein now is designated C protein. Rabbit antibodies directed against CPS protected mice against lethal challenge with homologous, but not heterologous, GBS types, and cross-protection was also afforded when antibodies against C protein were tested.
Current nomenclature designates polysaccharide antigens as type antigens and protein antigens as additional markers for characterization. The former type Ic now is designated type Ia/c. Type IV was identified as a new type in 1982, when 62 strains were described that possessed type IV polysaccharide alone or with additional protein antigens. Antigenically distinct types, V through IX, now are characterized. Strains not expressing one of the CPS-specific antigens are designated as nontypeable by serologic methods but often can be assigned a GBS type by PCR-based methods.
C protein is composed of two unrelated components, the trypsin-resistant α C protein and the trypsin-sensitive β C protein. α C protein is expressed on many type Ia, Ib, and II strains. Strains expressing α C protein are more opsonoresistant than are α C–negative strains. α C protein consists of a series of tandem repeating units, and in naturally occurring strains, the repeat numbers can vary. The number of repeating units expressed alters antigenicity and influences the repertoires of antibodies elicited. The use of one or two repeat units of α C proteins elicits antibodies that bind all α C proteins with equal affinity, suggesting its potential as a vaccine candidate. β C protein is a single protein with a molecular mass of 124 to 134 kDa that is present in about 10% of isolates. β C protein binds the Fc region of human IgA. Strains bearing α and β C proteins possess increased resistance to opsonization in vitro.
GBS express numerous additional surface proteins. Designation of additional α-like repetitive proteins (Alp) numerically (e.g., Alp2 and Alp3) is being considered. Most strains have the gene for just one Alp family protein. Genes encoding Alp1 (also designated “epsilon”) are associated with type Ia, and genes encoding Alp3 are associated with type V strains. Alp also are referred to as R proteins, with R1 and R4 as the major ones found on clinical isolates. Rib protein, expressed by most type III strains, has an identical sequence to R4. The gene sequence of a protein initially designated R5 has been renamed group B protective surface protein (BPS). In one large collection, BPS was found in 3.5% of invasive or colonizing isolates, most often in type Ia, II, or V GBS and never in type III. Some GBS contain surface proteins designated as X antigens. The X and R antigens are immunologically cross-reactive. A laddering protein from type V GBS shares sequence homology with α C protein. A protein designated Sip (surface immunogenic protein) is distinct from other known surface proteins. It is produced by all GBS types and confers protection against experimental infection; its role in human infection is unknown.
Genome analysis has shown that GBS produce long pilus-like structures. These structures extend from the bacterial surface and beyond CPS ( Fig. 12-1 ). Formed by proteins with adhesive functions, these structures are implicated in host colonization, attachment, and invasion. The pilus-like structures are encoded in genomic pilus islands that have an organization similar to that of pathogenicity islands. Three types of pilus islands have been identified through genomic analysis; these are composed of partially homogeneous covalently linked proteins (pilus islands 1, 2a, and 2b). These pilus proteins are highly surface-expressed and are involved in paracellular translocation through epithelial cells. At least one pilus island is present on all GBS clinical strains tested to date.
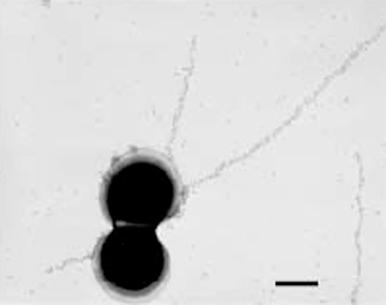
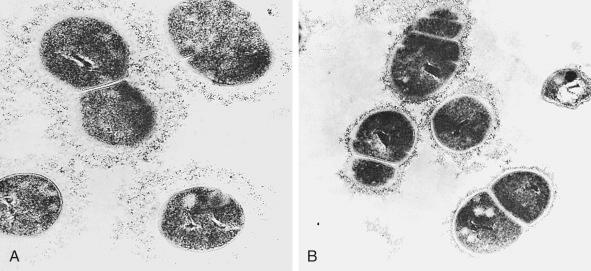
Early concepts suggested a thick, rigid peptidoglycan layer external to the cytoplasmic membrane surrounded by concentric layers of cell wall antigens. Evidence now supports a model in which the group B carbohydrate and the CPS are linked independently to cell wall peptidoglycan. Immunoelectron techniques reveal abundant capsule on Lancefield prototype strains Ia, II, and III, whereas less dense capsules are found on type Ib strains ( Fig. 12-2 ). Similarly, incubation of the reference strains with homologous type-specific antisera reveals a thick capsular layer on types IV, V, and VI. Ultrastructural studies show that the C protein also has a surface location. CPS capsule expression can be regulated by altering cell growth rate. Immunogold labeling and transmission electron microscopy show that the GBS pilus-like structures extend from the bacterial surface.
Lancefield’s initial serologic definition used hydrochloric acid and heat treatment, resulting in degraded antigens of small molecular mass. Gentler techniques isolated large molecular mass or “native” polysaccharides that contained sialic acid. Human immunity correlates with antibody to the sialic acid–containing type III structure. With the use of contemporary methods for determination, l -rhamnose, d -galactose, 2-acetamido-2-deoxy- d -glucose, and d -glucitol have been identified as the constituent monosaccharides of the group B antigen. It is composed of four different oligosaccharides, designated I though IV, and linked by a phosphodiester bond to form a complex, highly branched multiantennary structure.
The repeating unit structures of the type-specific CPSs are schematically represented in Figure 12-3 . Types Ia, Ib, and III have a five-sugar repeating unit containing galactose, glucose, N -acetylglucosamine, and sialic acid in a ratio of 2:1:1:1. Type II and type V have a seven-sugar repeating unit, type IV and type VII have six-sugar repeats, and type VIII polysaccharide has a four-sugar repeating unit. Molar ratios vary, but the component monosaccharides are the same among the polysaccharide types except that type VI lacks N -acetylglucosamine and type VIII contains rhamnose in the backbone structure.
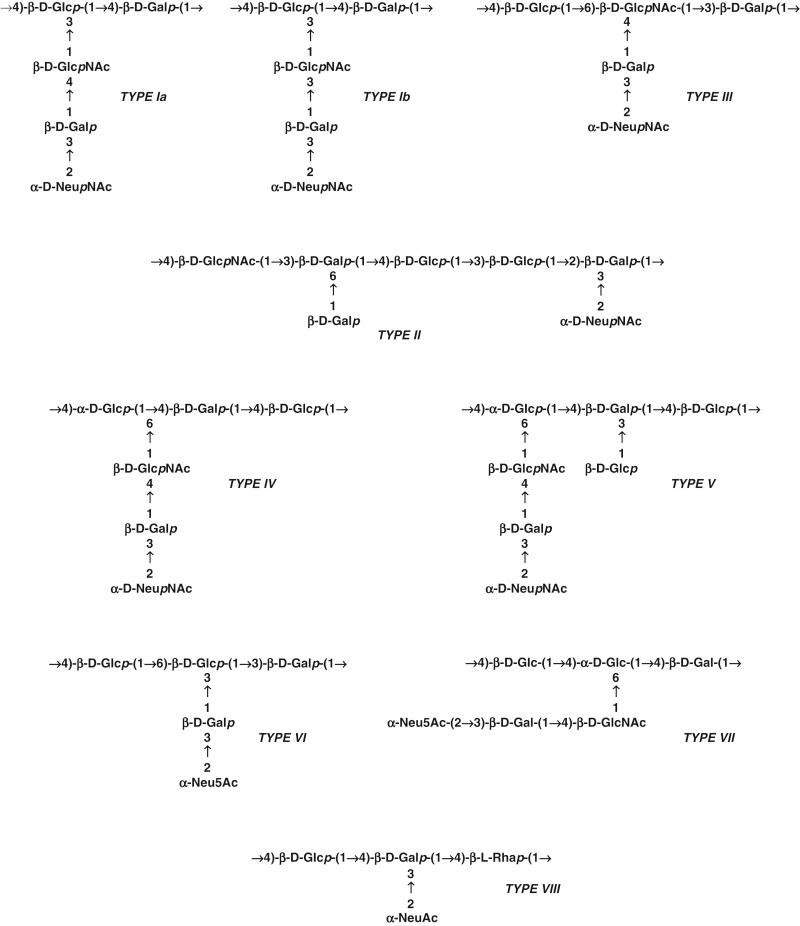
Each antigen has a backbone repeating unit of two (Ia, Ib), three (III, IV, V, VII, VIII), or four (II) monosaccharides to which one or two side chains are linked. Sialic acid is the exclusive terminal side chain sugar except for the type II polysaccharide, which also has a terminal galactose. The structures of the type Ia and type Ib polysaccharides differ only in a single side-chain linkage, although there are differences in the tertiary configuration of the molecules. These linkages are critical to immunologic specificity and explain the observed immunologic cross-reactivity. The desialylated type III polysaccharide is immunologically identical to that of type 14 Streptococcus pneumoniae . This observation stimulated investigations concerning the immunodeterminant specificity of human immunity to type III GBS and of antibody recognition of conformational epitopes as a facet of the host immune response. The type III polysaccharide also can form extended helices. The position of the conformational epitope along these helices is potentially important to binding site interactions.
Group B streptococci are quite homogeneous in their amino acid requirements during aerobic or anaerobic growth. A glucose-rich environment enhances the number of viable GBS during stationary phase and the amount of CPS elaborated. In a modified chemically defined medium, the expression of capsule during continuous growth is regulated by the growth rate. Invasiveness is enhanced by a fast growth rate and is optimal in the presence of at least 5% oxygen.
GBS elaborate many products during their growth. Among these is the hemolysin that produces the β-hemolysis surrounding colonies on blood agar. Hemolysin is a surface-associated toxin active against the erythrocytes from several mammalian species. The GBS hemolysin recently has been characterized as the ornithine rhamnolipid pigment and shown to function as a virulence factor, promoting invasion of placental cells. GBS can hydrolyze hippuric acid to benzoic acid and glycine. The hippuricase of GBS is cell associated and is trypsin and heat labile. It is antigenic in rabbits, but its relationship to bacterial virulence, if any, has not been studied.
Most strains of GBS have an enzyme that inactivates complement component C5a by cleaving a peptide at the carboxyl terminus. GBS C5a-ase seems to be a serine esterase; it is distinct from the C5a-cleaving enzyme produced by group A streptococci, although the genes that encode these enzymes are similar. C5a-ase contributes to pathogenesis by rapidly inactivating the neutrophil agonist C5a, preventing the accumulation of neutrophils at the site of infection.
Another group of enzymes elaborated by nearly all GBS are the extracellular nucleases. Three distinct nucleases have been physically and immunologically characterized. All are maximally activated by divalent cations of calcium plus manganese. These nucleases are immunogenic in animals, and neutralizing antibodies to them are detectable in sera from pregnant women known to be genital carriers of GBS. Their role in the pathogenesis of human infection is unknown.
An extracellular product that can contribute to virulence of GBS was originally defined as a neuraminidase and subsequently characterized as a hyaluronate lyase. Maximal levels are detected during late exponential growth in a chemically defined medium. Elaboration of large quantities can be a virulence factor for type III GBS. Musser and coworkers identified a high neuraminidase–producing subset of type III strains that were responsible for most serious GBS infections. Later studies indicated that these were from a single clonal complex, designated ST 17, that has been designated as “hypervirulent.” ST 17 is almost exclusively found in type III strains.
GBS synthesize acylated (lipoteichoic) and deacylated glycerol teichoic acids that are cell associated and can be readily extracted and purified. Strains from infants with early- or late-onset disease have higher levels of cell-associated and native deacylated lipoteichoic acid, and this product seems to contribute to attachment to human cells.
Group B streptococcal infection limited to mucous membrane sites is designated as asymptomatic infection, colonization, or carriage. Comparisons of the prevalence of colonization are related to differences in ascertainment techniques. Factors that influence the accuracy of colonization detection include density of colonization, choice of bacteriologic media, body sites sampled, number of culture specimens obtained, and time interval of study.
Isolation rates are higher with use of an enrichment broth, rather than solid agar media, and with enrichment broth containing substances inhibitory for normal flora (usually antimicrobials). Selective enrichment broths, include Todd-Hewitt broth supplemented either with gentamicin (8 μg/mL) and nalidixic acid (15 μg/mL; TransVag broth) or with colistin (10 μg/mL) and nalidixic acid (15 μg/mL; Lim broth). Addition of 5% sheep blood to TransVag broth or Lim broth can increase the recovery of GBS. Such media inhibit the growth of most gram-negative enteric bacilli and other normal flora that make isolation of streptococci from these sites difficult. Use of selective enrichment broth promotes detection of low numbers of organisms that escape detection when inoculation of swabs is directly onto solid agar.
Isolation rates also are influenced by body sites selected for culture. Female genital culture isolation rates double with progression from the cervical os to the vulva. In addition, culture sampling of lower genital tract and rectal sites increases GBS colonization rates 10% to 15% beyond that found if a single site is cultured. The urinary tract is an important site of infection, especially during pregnancy, which usually manifests as asymptomatic bacteriuria. To predict accurately the likelihood of neonatal exposure to GBS at delivery, maternal culture specimens from the lower vagina and rectum (not perianal area) should be collected.
The prevalence of GBS colonization is influenced by the number of cultures obtained from a site and the interval of sampling. Vaginal colonization patterns can be chronic, transient, or intermittent. A longitudinal cohort study of nonpregnant young women found that almost one half of those who are culture negative at enrollment acquired vaginal colonization during three 4-month intervals of assessment. The duration of colonization among college students is estimated to be 14 weeks for women and 9 weeks for men. The predictive value of a positive second trimester vaginal or rectal culture for colonization at delivery is only 67%. The predictive value of a positive prenatal culture result is highest (73%) in women with vaginal and rectal colonization and lowest (60%) in women with rectal colonization only. Cultures performed 1 to 5 weeks before delivery have a positive predictive value of 87% (95% confidence interval [CI], 83 to 92) for colonization status at delivery in term parturients. The negative predictive value is 96% (95% CI, 95 to 98). Culture specimens collected within this interval perform significantly better than specimens collected 6 or more weeks before delivery.
The primary reservoir for GBS is the lower gastrointestinal tract. The recovery of GBS from the rectum is three to five times more common than recovery from the vagina, and the rectal site predicts persistence or chronicity of carriage. GBS in the gastrointestinal tract is a risk factor for vaginal GBS. Additional support for the lower gastrointestinal tract as the primary reservoir is the association of GBS with infections resulting from gastrointestinal tract surgical procedures. Several factors influence genital carriage of GBS. Among healthy young men and women living in a college dormitory, sexually experienced subjects had colonization rates twice those of sexually inexperienced subjects. In a longitudinal cohort study of nonpregnant young women, African-American ethnicity, having multiple sex partners during a preceding 4-month interval, having frequent sexual intercourse within the same interval, and having sexual intercourse within the 5 days before a follow-up visit were independently associated with vaginal acquisition of GBS. These latter findings suggest either that the organism is sexually transmitted or that sexual activity alters the microenvironment to make it more permissive to colonization. In another study of college women, GBS were isolated significantly more often from sexually experienced women, women studied during the first half of the menstrual cycle, women with an intrauterine device, and women 20 years of age or younger. Colonization with GBS also occurs at a high rate in healthy college students and is associated with having engaged in sexual activity, tampon use, milk consumption, and hand washing done four times daily or less. Fish consumption increased the risk of acquiring some, but not all, capsular types.
A higher prevalence of colonization with GBS is found among pregnant diabetic patients than among nondiabetic controls. Carriage over a prolonged interval reportedly occurs more often in women who use tampons than women who do not. Colonization is more frequent among teenage women than among women 20 years of age or older and among women with three or fewer pregnancies than in women with more than three pregnancies. Hispanic women of Caribbean origin have a high rate of colonization, and African-American women have a higher rate of colonization at delivery than do other racial or ethnic groups. A large inoculum of vaginal GBS colonization also is more common among African-American than among Hispanic or non-Hispanic white women. Factors that do not influence the prevalence of genital colonization in nonpregnant women include use of oral contraceptives ; marital status; presence of vaginal discharge or other gynecologic signs or symptoms ; carriage of Chlamydia trachomatis, Ureaplasma urealyticum, Trichomonas vaginalis, or Mycoplasma hominis ; and infection with Neisseria gonorrhoeae .
Colonization with GBS can elicit an immune response. In a group of pregnant women evaluated at admission for delivery, vaginal or rectal colonization with types Ia, II, III, or V was associated with significantly higher serum concentrations of immunoglobulin (IgG) specific for the colonizing type compared with noncolonized women. Moderate concentrations of Ia, Ib, II, III, and V CPS-specific IgG also were found in association with colonization during pregnancy. Maternal colonization with type III was least likely to be associated with these CPS-specific antibodies. In contrast to infection with organisms such as N. gonorrhoeae or genital mycoplasmas, genital infection with GBS is not related to genital symptoms.
GBS have been isolated from vaginal or rectal sites or both in 15% to 40% of pregnant women. The range in colonization rates relates to intrinsic differences in populations (age, ethnicity, parity, socioeconomic status, geographic location) and to lack of standardization in culture methods used for ascertainment. True population differences account for some of the disparity in reported prevalence rates. When selective enrichment broth is used and vaginal and rectal sites are sampled, the prevalence of maternal colonization with GBS by region is 12% in India and Pakistan, 19% in Asia and the Pacific Islands, 19% in sub-Saharan Africa, 22% in the Middle East and North Africa, 14% in Central and South America, and 26% in the United States. The rates of colonization among pregnant women range from 20% to 29% in Eastern Europe, 11% to 21% in Western Europe, 21% to 36% in Scandinavia, and 7% to 32% in Southern Europe. The rate of recurrence of GBS colonization in a subsequent pregnancy is higher compared with women negative for colonization in their prior pregnancy. Pharyngeal carriage rates are low and are similar among pregnant and nonpregnant women and heterosexual men ; however, rates approach 20% in men who have sex with men. No definite relationship between isolation of GBS from throat cultures and symptoms of pharyngitis has been proved, but some investigators have suggested that these organisms can cause acute pharyngitis.
Cultures from the throat and rectum are the best sites for detection of GBS during childhood and until the start of sexual activity. In a study of 100 girls ranging in age from 2 months to 16 years, Hammerschlag and coworkers isolated GBS from pharyngeal, rectal or vaginal sites, or both, in 20% of children. The prevalence of positive pharyngeal cultures resembled the prevalence of adults in girls 11 years or older (5%) but approached the prevalence reported for neonates in younger girls (15%). Rectal colonization was detected frequently in girls younger than 3 or older than 10 years of age (about 25%), but was uncommon in girls 3 to 10 years of age. Mauer and colleagues isolated GBS from cultures of vaginal, anal, or pharyngeal specimens or all three in 11% of prepubertal boys and girls. Pharyngeal (5% each) and rectal (10% and 7%) isolation rates were similar for boys and for girls. Persson and coworkers detected fecal carriage of GBS in 4% of healthy boys and girls, and Cummings and Ross found that 2% of English schoolchildren had pharyngeal carriage. Genital colonization in girls is uncommon before puberty. Whether this relates to environmental influences in the prepubertal vagina or to lack of sexual experience before puberty, or both, awaits further study.
The presence of GBS in the maternal genital tract at delivery is the major determinant of colonization and infection in the neonate. Exposure of the neonate to the organism occurs by the ascending route in utero through translocation through intact membranes, through ruptured membranes, or by contamination during passage via the birth canal. Prospective studies have indicated vertical transmission rates of 29% to 85%, with a mean rate of approximately 50% among neonates born to women from whom GBS were isolated from cultures of vagina or rectum or both at delivery. Conversely, only about 5% of healthy infants delivered to culture-negative women become colonized at one or more sites during the first 48 hours of life.
The risk of a neonate acquiring colonization by the vertical route correlates directly with the density of colonization (inoculum size). Neonates born to heavily colonized women are more likely to acquire carriage at mucous membrane sites than neonates born to women with low colony counts of GBS in vaginal cultures at delivery. Boyer and associates found that rates of vertical transmission were substantially higher in women with heavy than in women with light colonization (65% vs. 17%) and that colonization at multiple sites and development of early-onset disease were more likely among infants born to heavily colonized mothers. The likelihood of colonization in a neonate born to a woman who is culture-positive at delivery is unrelated to maternal age, race, parity, or blood type or to duration of labor or method of delivery. It is unclear whether preterm or low-birth-weight neonates are at higher risk for colonization from maternal sources than term infants.
Most neonates exposed to GBS by their mothers have infection that is limited to surface or mucous membrane sites (colonization), which results from contamination of the oropharynx, gastric contents, or gastrointestinal tract by swallowing of infected amniotic fluid or maternal vaginal secretions. In neonates, external auditory canal cultures are more likely to yield GBS than cultures from anterior nares, throat, umbilicus, or rectum in first 24 hours of life, and isolation of organisms from the ear canal is a surrogate for the degree of contamination from amniotic fluid and vaginal secretions sequestered during the birth process. After the first 48 hours of life, throat and rectal sites are the best sources for detection of GBS, and positive cultures indicate true colonization (multiplication of organisms at mucous membrane sites), not just maternal exposure.
Other sources for acquisition of GBS in neonates have been established. Horizontal transmission from hospital or community sources is an important, albeit uncommonly proved, mode for transmission of infection. Acquisition can occur from hands of nursery personnel. In contrast to group A streptococci, which can cause epidemic disease in nurseries, GBS rarely exhibits this potential, and isolation of colonized neonates is not routinely indicated. An epidemic cluster of five infants with late-onset bacteremia infection caused by type Ib GBS occurred among very-low-birth-weight infants in a neonatal intensive care unit in the 1980s. None of the index cases was colonized at birth, establishing that acquisition during hospitalization had occurred. Epidemiologic analysis suggested infant-to-infant spread by means of the hands of personnel, although acquisition from two nurses colonized with the same phage type Ib strain was not excluded. The infection control measures instituted, including cohorting of culture-positive infants and strict hand hygiene, prevented additional cases. Community sources afford a likely potential for transmission of GBS to the neonate. Indirect evidence has suggested that this mode of infection is infrequent. Only 2 of 46 neonates culture negative for GBS when discharged from the newborn nursery acquired mucous membrane infection at 2 months of age. The mode of transmission likely is fecal-oral. Healthy infants colonized from a maternal source or postnatally show persistence of infection at mucous membrane sites for weeks or months.
The differentiation of GBS into CPS types has provided a valuable tool in defining the epidemiology of human infection. In the 1970s and 1980s, virtually all evaluations of GBS isolated from healthy neonates, children, or adults revealed an even distribution into types Ia or Ib, II, and III. This distribution also was reported for isolates from neonates with early-onset infection without meningitis and their mothers. In the 1990, types other than I, II, or III accounted for less than 5% of all isolates.
In the early 1990s, GBS type V emerged as a frequent cause of colonization and invasive disease in neonates and adults. Most type V isolates have one pulse-field gel electrophoresis pattern that has been present in the United States since 1975. Type V now causes a substantial proportion of cases of invasive early-onset disease and infection during pregnancy. Type Ia has increased in prevalence and a corresponding decline has occurred in type II strains causing perinatal disease. Type III strains account for about 70% of isolates from infants with meningitis and continue to be isolated from at least two thirds of infants with late-onset disease globally. Type IV, which accounts for occasional cases, could be emerging as a more important cause of early-onset infection. Types VI, VII, VIII, and IX rarely cause human disease in the United States or the United Kingdom, but types VI and VIII are the most common serotypes isolated from healthy Japanese women.
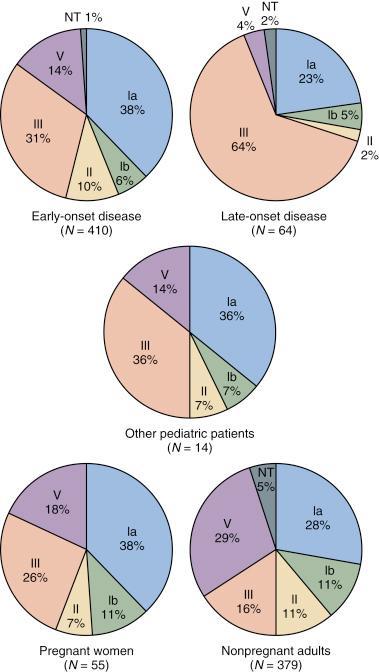
The contemporary CPS type distribution of GBS from different patient groups is shown in Figure 12-4 . Prospective population-based surveillance through the Active Bacterial Core Surveillance/Emerging Infections Program Network of the U.S. Centers for Disease Control and Prevention (CDC) defined the epidemiology of invasive GBS disease in the United States from 1999-2005. The GBS types represented in 528 early-onset disease cases were Ia (30%), III (28%), V (18%), and II (13%). The distribution for 172 pregnancy-associated cases was similar. The type distribution among 469 late-onset cases was Ia (24%), III (51%), and V (14%). Type V predominated among cases in nonpregnant adults, accounting for 31%, followed by Ia (24%), II (12%), and III (12%).
Tools such as multilocus enzyme electrophoresis, restriction-enzyme fragment-length polymorphism analysis, pulsed-field gel electrophoresis (PFGE), random-amplified polymorphic DNA assay and multiplex PCR have been used for molecular characterization of GBS isolates. Multilocus sequence typing (MLST) and PFGE are reportedly more appropriate than a semiautomated repetitive sequence-based PCR DiversiLab system (bioMérieux, Durham, NC) for determining the relatedness of invasive GBS strains. These molecular typing techniques have indicated that some geographically and epidemiologically distinct GBS isolates have identical patterns, suggesting dissemination of a limited number of clones in the United States. Molecular techniques also have confirmed the molecular relatedness of mother and infant strains, strains from twins and those from sexual partners. Multilocus sequence typing and capsular gene cluster ( cps ) genotyping have been used to investigate the dynamics of perinatal colonization. Changes in capsule expression and recolonization with antigenically distinct GBS clones were detected in culture-positive women over time by applying MLST.
Molecular characterization has been used to explore the role of virulence clones in contributing to invasive disease. Type III strains were classified into three major phylogenetic lineages, with most cases of invasive neonatal disease caused by strains with one restriction digest pattern (type III-3) on the basis of bacterial DNA restriction digest patterns. The genetic variation that distinguishes restriction digest pattern type III-3 strains seems to occur within localized areas of the genome that contain known or putative virulence genes. Using genomic subtractive hybridization to identify regions of the genome unique to virulent restriction fragment digest pattern type III-3 strains, a surface protein was identified that mediates epithelial cell invasion. Using MLST, 10 allelic profiles that converged into three groups on concatenation were identified among type III isolates recovered from neonates with invasive disease and from colonized pregnant women. One PFGE group bearing a gene from the capsular synthesis operon has been shown in type III strains causing neonatal meningitis, but not in type III colonizing strains. Clustering of most invasive neonatal isolates into major PFGE groups also has been noted. Among type III strains evaluated by MLST, a single clone, ST17, is reported to be hypervirulent. Additional studies are required to elucidate the differences in virulence among clones identified by these techniques. However, genetic analysis of GBS isolates from worldwide sources demonstrates that epidemic clones, such as clonal complex 17 (CC17) have adapted specifically to the human host. The finding that isolates with different capsular serotypes have the same sequence type suggests that capsular switching can occur. Bellais and colleagues have demonstrated that capsular switching from CPS type III to IV does occur within the highly homogeneous CC17 hypervirulent clone. Sequence analysis showed that this capsular switch was due to the exchange of a DNA fragment containing the whole cps operon.
Two clinical syndromes occur among young infants with GBS disease that are epidemiologically distinct and relate to age at onset. The attack rate for the first of these syndromes, designated early onset, because it occurs within the first 6 days of life (mean onset, 12-18 hours), ranged historically from 0.7 to 3.7 per 1000 live births. The attack rate for late-onset infection (onset 7-89 days of age) ranged from 0.5 to 1.8 per 1000 live births. The burden of early-onset disease is disproportionately high in African-American infants for reasons that are not well defined but might include higher maternal colonization rates, higher density of colonization, and higher rates of preterm deliveries compared with white women. There has been a dramatic decline in the incidence of early-onset disease in the United States in association with implementation of universal antenatal screening and use of intrapartum antibiotic prophylaxis (IAP). From 1993 to 1998, when risk-based and GBS culture-based methods were in use, incidence of early-onset disease declined by 65%, from 1.7 to 0.6 per 1000 live births. Comparison of the two approaches showed the superiority of a culture-based approach. The incidence of early-onset disease declined further in association with implementation of guidelines, published in 2002 and revised in 2010, that advocate a culture-based approach for prevention of early-onset disease. The national estimate of early-onset invasive disease in 2012 was 0.24 per 1000 live births, but this represented a plateau since 2008. Despite the decline in incidence, GBS remains the most commonly reported pathogen causing early-onset disease, accounting for approximately 40% of cases in the United States. In contrast to its impact on early-onset disease, IAP has had no impact on the incidence of late-onset disease, which has remained stable at approximately 0.3 per 1000 live births since 2002.
Globally, GBS is a leading cause of neonatal sepsis in developed countries, but the burden of disease in the developing world is less clear, and more high-quality studies are needed. The reported incidence of neonatal GBS disease in developing countries ranges from 0 to 3.1 per 1000 live births, with variation within and between geographic regions. Incidence rates are higher when automated culture methods are used. A systemic review and meta-analysis to examine the global burden reported a mean incidence of GBS in infants from birth to 89 days of age of 0.53 per 1000 live births. Substantial heterogeneity existed between studies. Studies that reported use of any intrapartum antibiotic prophylaxis were associated with a lower incidence of early-onset GBS (0.23/1000 live births [95% CI, 0.13 to 0.59]) than those in which prophylaxis was not used (0.75/1000 live births [95% CI, 0.58 to 0.089]).
The male-to-female ratio for early-onset and late-onset GBS disease is equal. Before 1996, 20% to 25% of all infants with GBS disease had onset after the first 6 days of life. In 2012, 57% of infants had disease with onset after 6 days of life. Infants born prematurely constitute approximately one fourth of the total with early-onset disease and one half of the total with late-onset disease.
The importance of GBS as a common pathogen for the perinatal period relates to the pregnant woman as well as her infant. The risk of intraamniotic infection is greater in women with heavy colonization. Implementation of IAP has been associated with a significant decline in the incidence of invasive disease in pregnant women, from 0.29 per 1000 live births in 1993 to 0.23 per 1000 live births in 1998, and a further decline to 0.12 per 1000 live births during 1999 to 2005. One half of these infections were associated with infection of the upper genital tract, placenta, or amniotic sac and resulted in fetal death. Among the other infections, bacteremia without a focus (31%), endometritis without fetal death (8%), and chorioamnionitis without fetal death (4%) were the most common manifestations.
The prevalence and severity of GBS diseases in neonates have stimulated intensive investigation to elucidate the pathogenesis of infection. The unique epidemiologic and clinical features of GBS disease pose several basic questions that provide a framework for hypothesis development and experimental testing: How does the organism colonize pregnant women and gain access to the infant before or during delivery? Why are newborns, especially infants born prematurely, uniquely susceptible to infection? What allows GBS to evade host innate immune defenses? How do these organisms gain entry to the bloodstream and then cross the blood-brain barrier to produce meningitis? What specific GBS factors injure host tissues or induce the sepsis syndrome?
Advances in knowledge of pathogenesis have been achieved through analysis of GBS behavior in cell-culture systems and animal models. Advanced molecular genetic techniques have yielded isogenic mutant strains varying solely in the production of a particular component (e.g., capsular polysaccharide). Such mutants are important in establishing the biologic relevance of a given trait and its requirement for virulence in vivo. The sequencing of numerous complete GBS genomes has provided additional context for interpretation of experimental data and comparison with other well-studied pathogens.
Although GBS have adapted well to asymptomatic colonization of healthy adults, they remain a potentially devastating pathogen to susceptible infants. This section reviews the current understanding of virulence mechanisms, many of which are revealed or magnified by the unique circumstances of the birth process and the deficiencies of neonatal immune defense. Many of the GBS virulence factors defined to date, with mode of action and proposed role in pathogenesis, are summarized in Table 12-1 . Key stages in the molecular, cellular, and immunologic pathogenesis of newborn infection are summarized schematically in Figure 12-5 .
| Virulence Factor | Molecular or Cellular Action(s) | Proposed Role in Pathogenesis |
|---|---|---|
| Host Cell Attachment/Invasion | ||
| C surface protein | Binds glycosaminoglycans | Epithelial/endothelial invasion |
| Fibrinogen receptor, FbsA | Binds fibrinogen in extracellular matrix | Epithelial cell attachment |
| Lipoteichoic acid | Binds host cell surfaces | Epithelial cell attachment |
| C5a peptidase, ScpB | Binds fibronectin in extracellular matrix | Epithelial cell adherence, invasion |
| Surface protein Lmb | Binds laminin in extracellular matrix | Epithelial cell attachment |
| Spb1 surface protein | Promotes epithelial cell uptake | Invasion of epithelial barriers |
| iagA gene | ?Alteration in bacterial cell surface | Blood-brain barrier invasion |
| Glycoprotein Srr1 | Binds fibronectin in extracellular matrix | Vaginal and brain capillary binding |
| CovRS two-component regulator | Global transcriptional regulator | Regulation of adherence factors |
| Surface protein HgvA | Endothelial cell binding/uptake | Promotes blood-brain barrier invasion |
| Pili | Promotes cell surface binding | Epithelial and endothelial cell invasion |
| Injury to Host Tissues | ||
| β-Hemolysin/cytolysin | Lyses epithelial and endothelial cells | Damage and spread through tissues causes neuronal apoptosis |
| Hyaluronate lyase | Cleaves hyaluronan or chondroitin sulfate | Promotes spread through host tissues |
| CAMP factor | Lyses host cells (co-hemolysin) | Direct tissue injury |
| Resistance to Immune Clearance | ||
| Exopolysaccharide capsule | Impairs C3 deposition/activation | Blocks opsonophagocytic clearance |
| C5a peptidase, ScpB | Cleaves and inactivates human C5a | Inhibits neutrophil recruitment |
| CAMP factor | Binds to Fc portion of IgG, IgM | Impairment of antibody function |
| Serine protease, CspA | Cleaves fibrinogen, coats GBS surface with fibrin | Blocks opsonophagocytosis |
| Fibrinogen receptor, FbsA | ? Steric interference with C3 function | Blocks opsonophagocytosis |
| C protein | Nonimmune binding of IgA | Blocks opsonophagocytosis |
| β-Hemolysin/cytolysin | Lyses neutrophils macrophages, proapoptotic | Impairment of phagocyte killing |
| Superoxide dismutase | Inactivates superoxide | Impairment of oxidative burst killing |
| Carotenoid pigment | Antioxidant effect blocks H 2 O 2 , singlet oxygen | Impairment of oxidative burst killing |
| dlt operon genes | Alanylation of lipoteichoic acid | Blocks antimicrobial peptides |
| Penicillin-binding protein 1a | Alteration in cell wall composition | Blocks antimicrobial peptides |
| Nuclease A | Degrades DNA | Escape neutrophil extracellular taps |
| CovRS two-component regulator | Global transcriptional regulator | Phagolysomal survival |
| Activation of Inflammatory Mediators | ||
| Cell wall lipoteichoic acid | Binds pattern recognition receptors (TLRs) | Cytokine activation |
| Cell wall peptidoglycan | Binds pattern recognition receptors (TLRs) | Cytokine activation |
| β-Hemolysin/cytolysin | Activation of host cell stress response pathways, inflammasome | iNOS, IL-10, IL-1β release |
| GAPDH | Triggers IL-10 release | Suppression of neutrophil migration |
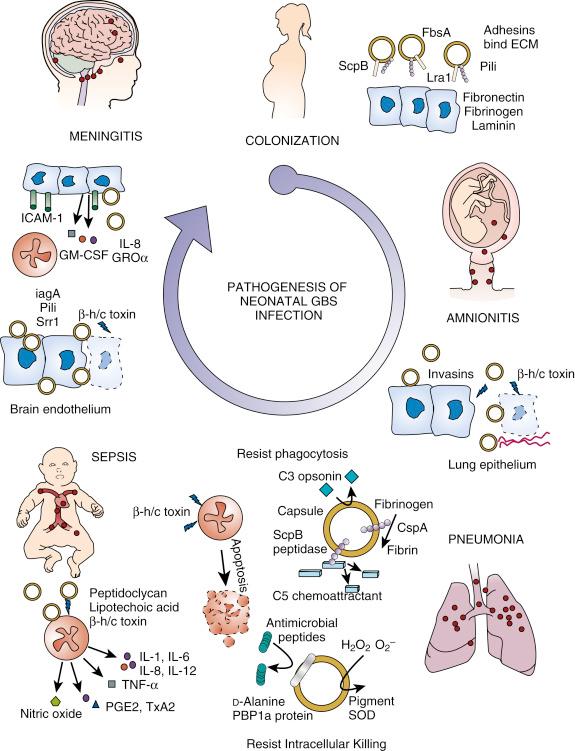
The presence of GBS in the genital tract of the mother at delivery determines whether or not a newborn is at risk for invasive disease. Among infants born to colonized women, the risk of early-onset disease is approximately 30-fold that for infants born to women with a negative result on prenatal cultures. A direct relationship exists between the degree (inoculum size) of GBS vaginal colonization, the risk of vertical transmission, and the likelihood of serious disease in the newborn. Consequently, a crucial step in the pathogenesis of invasive disease in the newborn caused by GBS is colonization of pregnant women.
To establish colonization of the female genital tract, GBS must adhere successfully to the vaginal epithelium. Compared with other microorganisms, GBS bind very efficiently to exfoliated human vaginal cells or vaginal tissue culture cells, with maximal biofilm formation at the acidic pH characteristic of vaginal mucosa. A low-affinity interaction with epithelial cells is mediated by its amphiphilic cell wall–associated lipoteichoic acid, whereas higher-affinity interactions with host cells are mediated by hydrophobic surface proteins. Soluble lipoteichoic acid competitively inhibits epithelial cell adherence and decreases vaginal colonization of pregnant mice.
High-affinity protein-mediated interactions of GBS with epithelium are mediated largely through extracellular matrix components, such as fibronectin, fibrinogen, and laminin, which interact with host cell–anchored proteins such as integrins. Binding occurs to immobilized but not soluble fibronectin, suggesting that this interaction requires close proximity of multiple fibronectin molecules and GBS adhesins. For example, a genome-wide phage display technique revealed a fibronectin-binding property associated with the surface-anchored GBS C5a peptidase ScpB, a dual functionality confirmed by decreased fibronectin binding of isogenic ScpB mutants and the direct interaction of recombinant ScpB with solid-phase fibronectin. Similar targeted mutagenesis studies showed that adherence of GBS to laminin involves a protein adhesin called Lmb, attachment to fibrinogen is mediated by repetitive motifs within the surface-anchored protein FbsA, and binding to human keratin 4 is carried out by the serine-rich repeat domain protein Srr-1.
Group B streptococci express filamentous cell surface appendages known as pili. Group B streptococcal pili contain three subunits: a backbone pilin protein (PilB), a pilus-associated adhesion (PilA) and a component anchoring the protein assembly to the cell wall (PilC); these components are encoded by two distinct loci in the genome, called pilus islands 1 and 2 (PI-1 and PI-2), the latter presenting two distinct variants, PI-2a and PI-2b. Epithelial cell adherence was reduced in isogenic GBS mutants lacking PilA or PilC, but not mutants lacking the PilB backbone. Elucidation of the crystal structure of PilC reveals a specific IgG-like fold domain (N2) required for epithelial cell binding. In a mouse model of GBS vaginal colonization, both pili and Srr-1 protein are required for efficient colonization. The regulation of these and other GBS adherence factors that determine vaginal epithelial cell and extracellular matrix binding is dynamically controlled by environmental pH and the two-component gene regulation system CovRS (CsrRS). Deletion of CovRS results in increased bacterial adherence but decreased invasion of vaginal epithelial cells. In the vaginal colonization model, the host mounts a more robust inflammatory response to the GBS CovRS mutant, accelerated clearance.
Group B streptococci can reach the fetus in utero through ascending infection of the placental membranes and amniotic fluid. Alternatively, the newborn may become contaminated with the organism on passage through the birth canal. Infection by the ascending route plays a pivotal role in early-onset disease. A direct relationship exists between the duration of membrane rupture before delivery and attack rate for early-onset disease, whereas an inverse relationship exists between the duration of membrane rupture and the age at which clinical signs of early-onset pneumonia and sepsis first appear. When the duration of membrane rupture was 18 hours or less, the attack rate was 0.7 per 1000 live births; when it was more than 30 hours, the attack rate increased to 18.3 per 1000, Histologic examination of placentas from women with GBS chorioamnionitis showed bacterial infiltration along a choriodecidual course, implying that ascending infection may be a primary trigger in many instances of premature rupture.
GBS may promote membrane rupture and premature delivery by several mechanisms. Isolated chorioamniotic membranes exposed to the organism have decreased tensile strength and elasticity and are prone to rupture. GBS also can modify the arachidonic acid metabolism of cultured human amnion cells, favoring production of prostaglandin E2, which is known to stimulate the onset of labor. Stimulation of placental release of macrophage inflammatory protein-lα (MIP-lα), interleukin-8 (IL-8), tumor necrosis factor-α (TNF-α), and matrix metalloproteases can recruit inflammatory cell and trigger alternative molecular signaling pathways that may provoke infection-associated preterm labor.
This GBS β-hemolysin/cytolysin (β-h/c) toxin is responsible for the characteristic β-hemolytic phenotype displayed by the organism when grown on sheep blood agar. Mutagenesis and heterologous expression studies have identified the cyl gene locus to encode β-h/c and production of an orange carotenoid pigment; a single gene within this locus, cylE , is necessary for GBS β-h/c expression and sufficient to confer β-hemolysis when cloned in E. coli , and one group has recently suggested that the pigment itself may function to form pores in target cell membranes. The β-h/c activity targets a wide variety of host epithelial, endothelial, and immune cell membranes, provoking cellular dysfunction and, ultimately, a necrotic or apoptotic cell death. GBS mutants lacking the CovRS regulator overexpress the toxin and penetrate isolated human chorioamniotic membranes more aggressively in a β-h/c–dependent manner, a finding which appears to correlate with increased hemolysis (and frequent CovRS mutation) among GBS isolated from women in preterm labor. Findings in a novel mouse model of GBS chorioamniotis confirm a critical role for the β-h/c in breaching maternal-fetal barriers to trigger preterm birth and intrauterine fetal demise.
GBS occasionally seem to penetrate into the amniotic cavity through intact membranes. Clinically, this mechanism of entry is suggested by reports of neonates with fulminant early-onset infection after cesarean section and no identifiable obstetric risk factors. Migration of the organism through freshly isolated chorioamniotic membranes has been documented by scanning and transmission electron microscopy, and GBS invade primary chorion cells efficiently in vitro and are capable of transcytosing through intact chorion cell monolayers without disruption of intracellular junctions. They also secrete an enzyme that degrades hyaluronic acid, an important component of the extracellular matrix that is abundant in placental tissues and may facilitate amniotic invasion. Placental expression of antimicrobial peptides such as human β-defensins and lactoferrins are induced by GBS and may constitute a key aspect of innate host defense against ascending infection by the pathogen.
Amniotic fluid supports the proliferation of GBS, such that when the organism gains access to the uterine cavity, a large inoculum can be delivered to the fetal lung; this results in a continuum of intrapartum (stillbirth) to early postpartum infant death. In utero infection probably accounts for the 40% to 60% of newborns with early-onset disease who have poor Apgar scores and in whom pulmonary signs develop within a few hours of birth because these infants almost invariably display clinical or histologic evidence of congenital pneumonia. Conversely, when GBS are encountered in the immediate peripartum period or on passage through the birth canal, a lesser inoculum is delivered to the neonate. Although a small but meaningful risk of subsequent invasive disease exists, most of these newborns have asymptomatic colonization limited to mucosal surfaces and remain healthy.
Fascinating new data collected in a nonhuman primate model of GBS choriodecidual inoculation suggest that the bacterium can induce fetal lung injury without ever crossing the placenta to produce intraamniotic infection or preterm delivery. Proinflammatory cytokines, including TNF-α and IL-8, elicited during transient choriodecidual infection appear to mediate this pathology, which included influx of neutrophils into the fetal intraalveolar space and thickening of the pulmonary interstitium. Analysis of the gene expression profile in these primate fetal lung tissues showed an upregulation in immune response genes but downregulation of key genes associated with cellular growth, angiogenesis, morphogenesis, and development.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here